Abstract
Introduction
Carpal tunnel syndrome (CTS) is the most common entrapment neuropathy and rarely develops after drug therapy. This study describes the clinical, electrodiagnostic (EDX), and ultrasound (US) findings in seven patients who experienced CTS due to anti-cancer therapeutic agents.
Methods
All patients underwent EDX testing, and four patients had an US study.
Results
CTS occurred in four patients with aromatase inhibitors, two with immune checkpoint inhibitors, and one with a selective estrogen receptor modulator. The mean duration between initiation of the anti-cancer therapeutic agents and symptom onset was 6 weeks (range: 2–12 weeks). Decreased digit sensation was noted in all patients; wasting and weakness of the abductor pollicis brevis (APB) was observed in three (42.8%) patients. The compound muscle action potentials (CMAP) of the APB and sensory nerve action potentials of the second or third digit could not be recorded in two (28.5%) and four (57.1%) patients, respectively. The needle EMG detected fibrillations and positive sharp waves in the APB in two patients. The motor unit potentials of the APB were decreased with large polyphasics in three (42.8%) patients. Of the four patients who underwent US testing, all had increased cross-sectional area of the median nerve at the carpal tunnel inlet, three (75%) had thenar muscle atrophy, and two (50%) had a loss of fascicular pattern. Three (42.8%) patients underwent a CTR.
Conclusions
Physicians should be cognizant of the relationship between anti-cancer therapeutic agents and CTS. EDX studies and US play important roles in the diagnostic assessment of such patients.
Keywords
Introduction
With an incidence of 1–3 persons per 1000 per year and a prevalence of 50 per 1000, carpal tunnel syndrome (CTS) is the most frequent entrapment neuropathy resulting from compression of the median nerve by the transverse carpal ligament (TCL) as it travels through the carpal tunnel.1,2 Several factors are associated with an increased risk of developing CTS, including genetic predisposition, professions with repetitive wrist movements, obesity, endocrine abnormalities (hypothyroidism, Cushing's disease), autoimmune disorders, and pregnancy.2–5 Certain medications to treat breast, lung, and bladder cancers may also rarely cause CTS. The third-generation aromatase inhibitors (AIs) (nonsteroidal AIs anastrozole and letrozole, steroidal AI exemestane) serve as adjuvant treatment of postmenopausal women with hormone receptor-positive breast cancer.5–8 While these drugs have been shown to significantly lower recurrence rates of breast cancer compared with the selective estrogen receptor modulator tamoxifen,8–13 they are linked to an elevated risk of CTS compared to tamoxifen.5,7,12–14 Similarly, other musculoskeletal disorders such as arthralgia and myalgia occur significantly more often with third-generation AIs compared to tamoxifen and placebo.6,12,15 CTS usually develops within the first 6–8 weeks following AI initiation. 13 It has been reported that 68% of patients prematurely discontinue AIs due to associated musculoskeletal symptoms, placing their oncology treatment protocol in peril.12,13,16
Immune checkpoint inhibitors (ICIs) spur the antitumor immune response by interrupting coinhibitory signaling pathways and promoting immune-mediated elimination of tumor cells. 17 They act by releasing the inhibition of T-lymphocytes to restore antitumor immunity. 18 The ICIs pembrolizumab and nivolumab are humanized monoclonal antibodies that target programmed death-1 (PD-1) and are approved to treat melanoma, nonsmall cell lung carcinoma, and urothelial (bladder) cancer.17–20 Pembrolizumab and nivolumab bind to the PD-1 receptor and directly block the interaction between PD-1 and its ligands, thereby augmenting the ability of tumor-directed T-cells to mediate tumor destruction.19,20 While these anti-cancer therapeutic agents have shown favorable response rates, the incidence of neuropathy is 0.3–1.0% which often occurs 2–12 weeks after treatment commencement.19,20 CTS has been reported as a rare complication of ICIs.1,19,20
Other medications that have been reported to increase the risk of CTS include bisphosphonates, 21 fluoroquinolones,22,23 anticoagulant therapy,24–26 antiretroviral therapy, 27 and human growth hormone use. 28 Cases of acute CTS occurring in a patient with a tricyclic antidepressant overdose 29 and bilateral CTS following radioactive iodine-induced hypothyroidism have been reported. 30 The association between postmenopausal hormone replacement therapy and CTS has also been described. 31
We report seven patients who developed CTS shortly after treatment with anti-cancer therapeutic agents all of whom underwent EDX testing and four of whom had an ultrasound (US) study. The presenting symptoms, physical examination, EDX findings, and US are presented. The mechanism of the relationship between anti-cancer therapeutic agents and CTS as well as the significance of EDX and US studies in the diagnostic evaluation of upper limb neuropathy after treatment with anti-cancer therapeutic agents are discussed.
Methods
Under an Institutional Review Board (IRB)-approved protocol and according to the Declaration of Helsinki, we performed a retrospective analysis of patients referred to our Neurodiagnostic Center for EDX studies to evaluate the presence of CTS following the initiation of anti-cancer therapeutic agents. The patients underwent our protocol of an initial neurological examination followed by nerve conduction and EMG studies. An US study was also conducted in four cases.
The EDX studies were performed in our American Association of Neuromuscular & Electrodiagnostic Medicine-accredited facility using a standard protocol of our laboratory. 32 Several EDX studies were performed including compound muscle action potentials (CMAP) (latency/amplitude) of the abductor pollicis brevis (APB), sensory nerve action potentials (SNAP) (latency/amplitude) of the second or third digit, presence of fibrillations/positive sharp waves in the APB, and motor unit recruitment in the APB. US studies were done according to our previously described protocol. 33
Informed consent was obtained from all patients. The IRB determined that our study was exempt according to 45 CFR 46.101(b) under Category 4. The IRB number is 22.0544.
Results
Clinical findings and neurological examination of patients with CTS following anti-cancer therapeutic agents
Seven patients were diagnosed with CTS based on presenting symptoms, neurological examination, as well as EDX and US findings (Table 1). Breast cancer was the diagnosis in five patients, urothelial cancer in one, and non-small cell lung cancer in one. Four patients were treated with AIs (2: exemestane; 2: letrozole), two with a PD-1 inhibitor (both pembrolizumab), and one with a selective estrogen receptor modulator (tamoxifen). The mean duration between initiation of anti-cancer therapeutic agents and symptom onset was 6 weeks (range: 2–12 weeks). The mean age was 66.3 years (range: 38–97 years), and 5 (71.4%) were female. Symptoms of CTS were bilateral in all cases.
Demographics, presenting symptoms, neurologic examination, and treatment/follow-up for patients who experienced carpal tunnel syndrome due to anti-cancer therapeutic agents evaluated at our neurodiagnostic center.
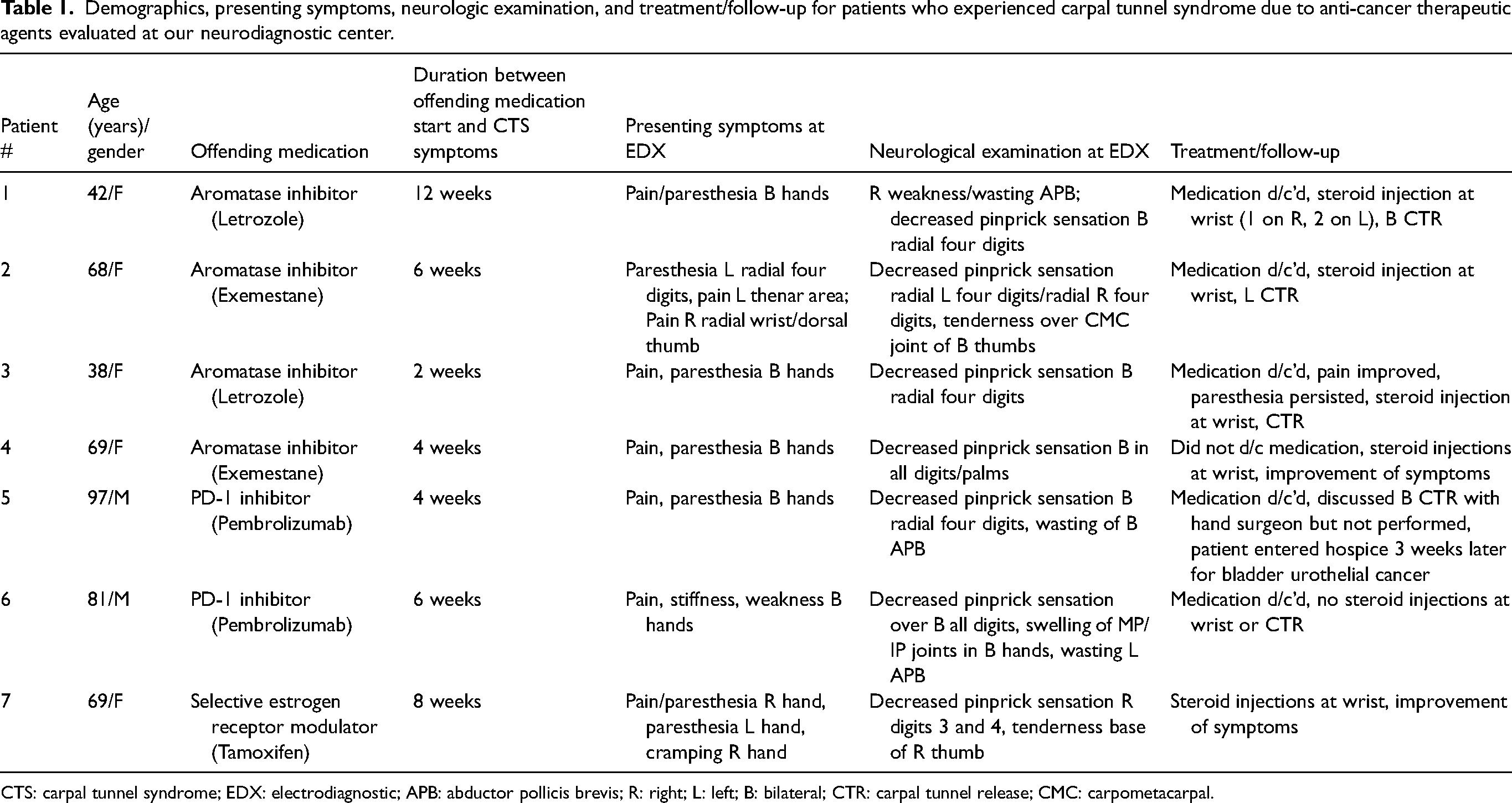
CTS: carpal tunnel syndrome; EDX: electrodiagnostic; APB: abductor pollicis brevis; R: right; L: left; B: bilateral; CTR: carpal tunnel release; CMC: carpometacarpal.
Upon presentation for EDX testing, all patients had experienced pain/paresthesia of the hands. One (14.3%) patient complained of hand weakness. On exam, decreased sensation of the digits was noted in all patients. Wasting of the APB was observed in three (42.8%) patients.
Electrodiagnostic studies in patients with CTS following anti-cancer therapeutic agents
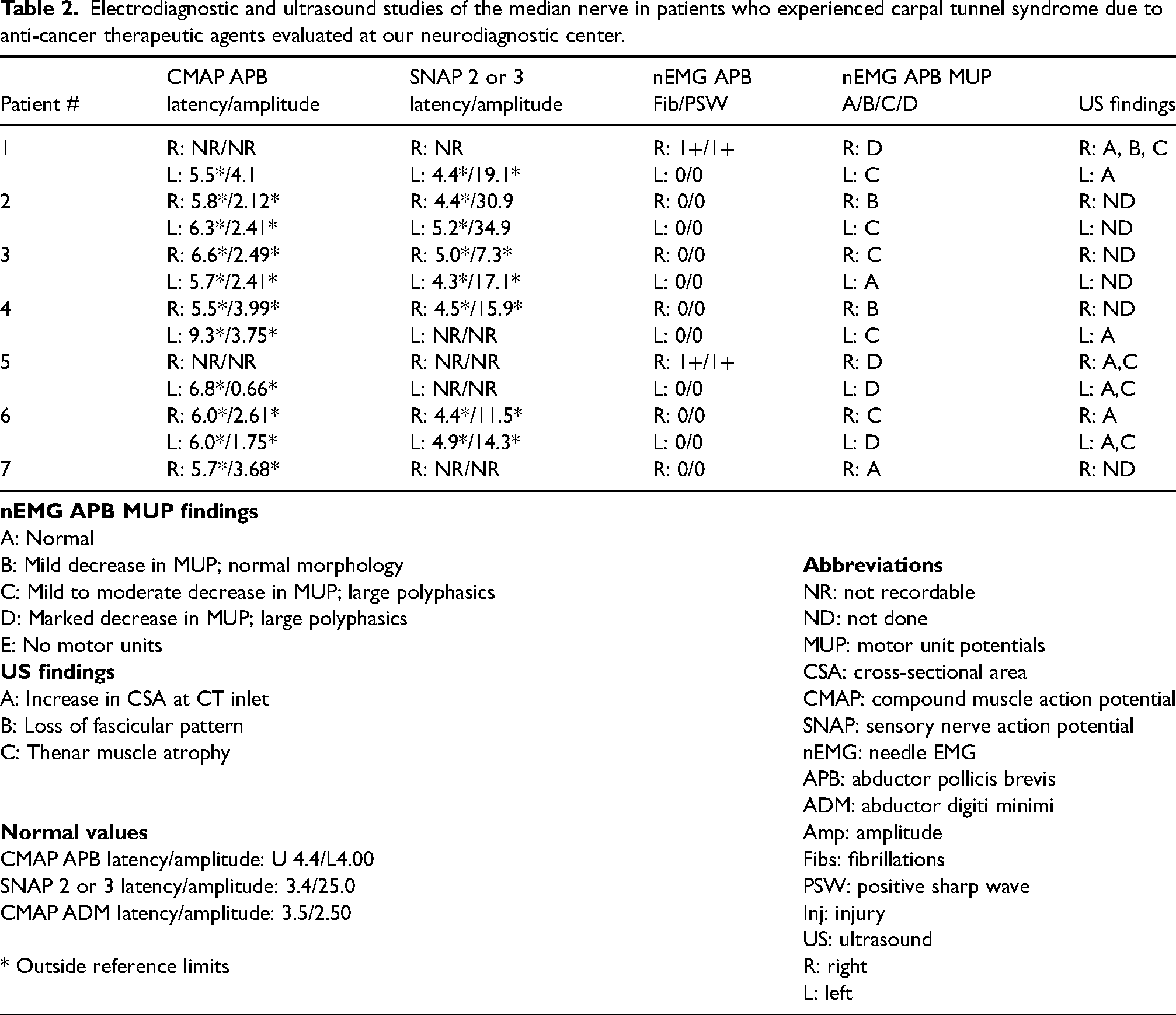
The EDX findings for patients with CTS following anti-cancer therapeutic agents are presented in Table 2. The CMAP of the APB and SNAP of the second or third digit could not be recorded in two and four patients, respectively (Figure 1). The needle EMG detected fibrillations and positive sharp waves unilaterally in the APB of two patients. The motor unit potentials of the APB were markedly decreased with large polyphasics in three patients.
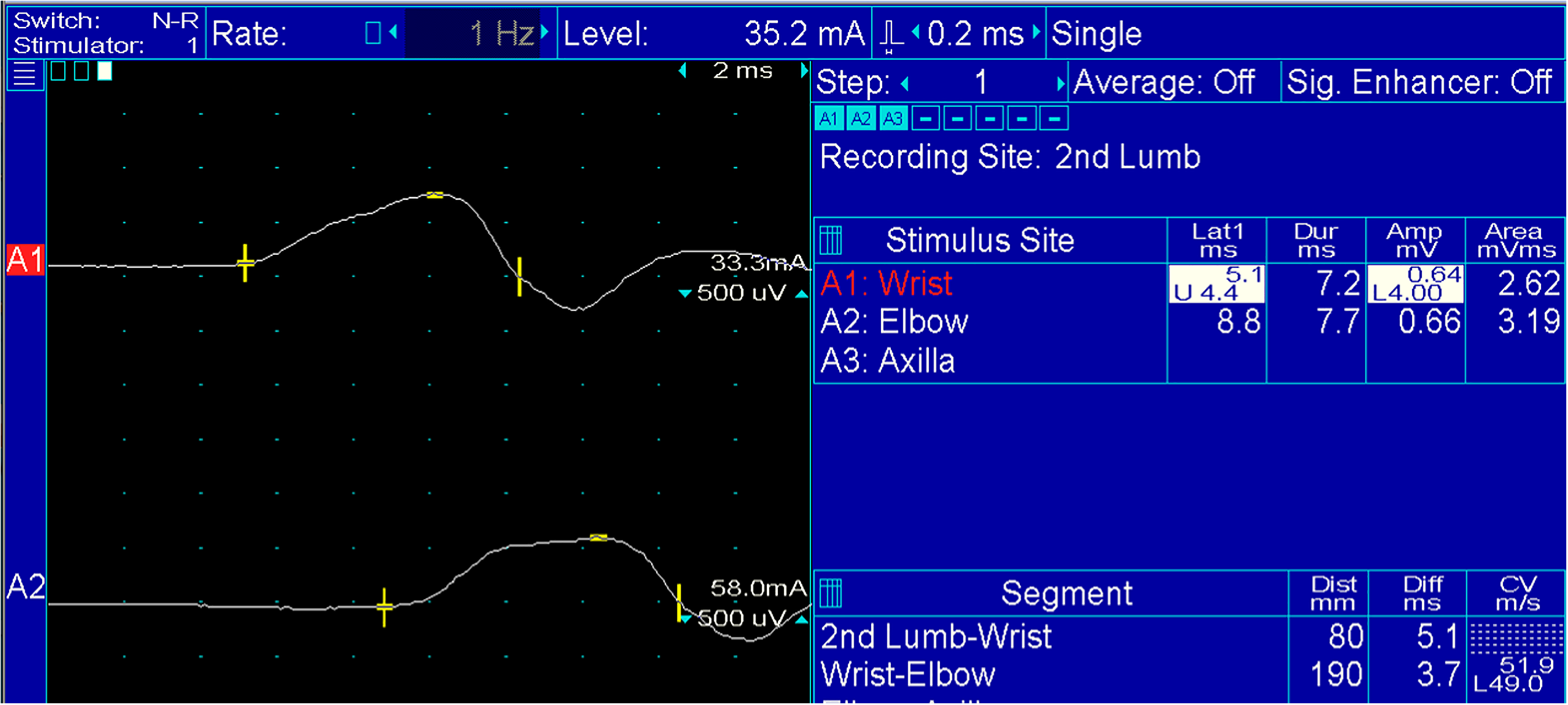
Median nerve motor conduction with recording electrode over second lumbrical (no compound muscle action potentials [CMAP] over the abductor pollicis brevis [APB]). Note the prolonged distal latency and low amplitude of CMAP.
Electrodiagnostic and ultrasound studies of the median nerve in patients who experienced carpal tunnel syndrome due to anti-cancer therapeutic agents evaluated at our neurodiagnostic center.
Ultrasound findings in patients with CTS following anti-cancer therapeutic agents
Of the four patients who underwent US testing, all had an increased cross-sectional area at the carpal tunnel inlet, three had thenar muscle atrophy, and two had a loss of fascicular pattern (Table 2; Figure 2(a) and (b)).
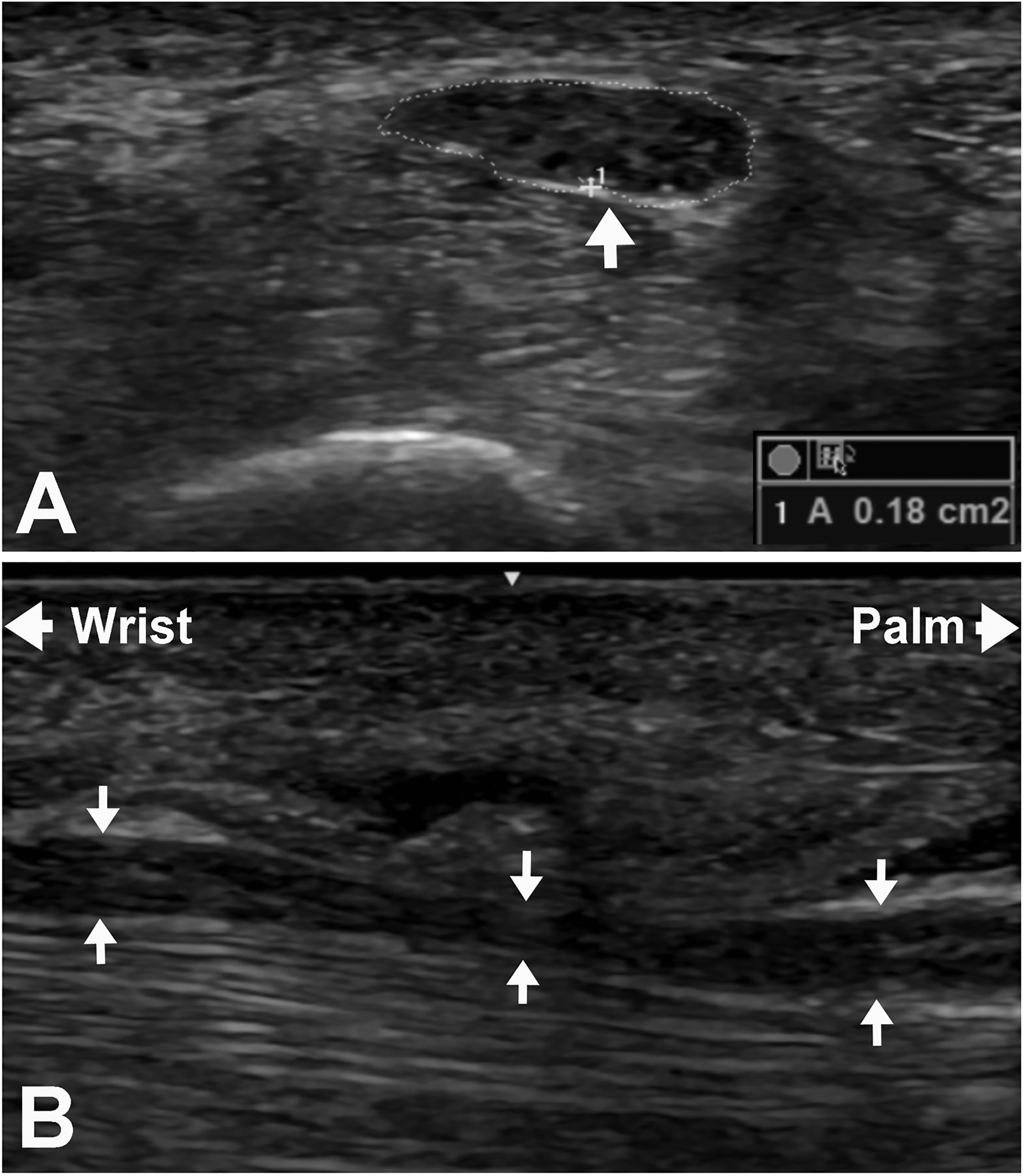
Ultrasound demonstrating (a) the short axis view of the median nerve (arrow) at the distal wrist crease showing an increase in cross-sectional area (0.18 cm2 compared to normal of 0.12 cm2 or less) and (b) the long axis view of the median nerve (arrows) at the carpal tunnel showing “hourglass” appearance.
Follow-up of patients with CTS following anti-cancer therapeutic agents
Six patients discontinued the offending anti-cancer therapeutic agent following the development of CTS symptoms (Table 1). Five (71.4%) patients were treated with steroid injections at the wrist, and three underwent a CTR.
Discussion
Several mechanisms have been proposed to explain the relationship between specific anti-cancer therapeutic agents and CTS. Women are three times more likely to develop CTS at menopause which implies the influence of hormonal factors in CTS. 15 Estrogen depletion is the mechanism of action for antitumor effects, 12 and estrogen deprivation is posited to be a primary explanation for the development of CTS and other musculoskeletal symptoms. 5 AIs greatly decrease plasma estrogen levels in postmenopausal women by inhibiting the peripheral conversion of androgenic precursors into estrogen (peripheral testosterone and androgens to estradiol) by the aromatase enzyme.5–7,12,15,34 Anastrozole and letrozole bind reversibly to aromatase, while exemestane binds irreversibly. Tamoxifen acts by blocking the binding of estrogen to its receptor which influences several downstream signaling mechanisms. 34
An enhanced pain stimulus is observed with complete estrogen depletion. 35 AIs reduce the protective antinociceptive effect of estrogen and decrease the threshold for pain stimuli, making patients susceptible to musculoskeletal disorders.6,12,15,34 It has also been reported that patients treated with anastrozole have a significant decrease in grip strength as well as an increase in tenosynovial changes and intra-articular fluid compared with tamoxifen.6,8 This fluid accumulation around the digital flexor tendons and thickening of the tendon sheaths of the wrist may result in compression of the median nerve with ensuing CTS. Additionally, estrogen and progesterone receptors (ER and PR, respectively) have been identified in the TCL, suggesting that female sex steroid hormones may impact TCL metabolism leading to CTS.6,12,36 In their immunohistochemical study of the expression of ERs and PRs in CTS versus control, Toesca and colleagues reported that the number of ER- and PR-positive cells in the TCL in non-CTS patients was significantly lower than in patients with CTS. 36
The association between tamoxifen and CTS has rarely been reported in the literature. In Mieog and colleagues’ Intergroup Exemestane Study of postmenopausal women with early breast cancer treated with exemestane (2319 patients) or tamoxifen (2338 patients) after 2–3 years of tamoxifen, CTS was diagnosed in 66 (2.8%) patients treated with exemestane and 13 (0.6%) patients with tamoxifen (p < 0.0001). 12 Tamoxifen selectively binds to estrogen receptors and produces both estrogenic and anti-estrogen effects. 37 Our single case of tamoxifen-associated CTS contributes to the few cases previously reported about this extraordinarily uncommon interaction.
EDX studies play an important role in the diagnostic evaluation of patients with CTS symptoms induced by specific anti-cancer therapeutic agents. Few studies have reported the EDX findings of patients treated with hormone therapy for breast cancer.6,13,34 In Shin and colleagues’ study of patients with breast cancer who developed CTS, 15 (88.2%) of the 17 patients treated with AIs underwent EDX testing.
13
The CTS results were reported as normal, mild, moderate, and severe. Eight (53.3%) of the 15 patients had severe CTS, and a CTR was performed on seven (41.2%) patients. Fourteen (82.4%) patients had bilateral CTS. In Nishihori and colleagues’ study of six patients who experienced CTS after adjuvants AIs, four underwent NCV/EMG testing, all with varying levels of median mononeuropathy in the affected wrist.
34
Four patients were switched to tamoxifen, and one underwent a CTR. In Dizdar and colleagues’ study of postmenopausal patients with breast cancer treated with adjuvant AIs (
Few studies have described the association between ICIs and CTS.1,18,20 In Lechevalier and colleagues’ study of 11 patients with melanoma treated with pembrolizumab or nivolumab, most cases were severe, bilateral (82%), and involved a motor deficit (54%). 18 The median duration was 3 months (range: 2–24 months) between medication initiation and symptom onset. An EMG confirmed axonal damage in nine (82%) patients, and four (36%) underwent a CTR. In Shalata and colleagues’ series of three patients (lung adenocarcinoma, renal cell carcinoma, and melanoma) treated with ICIs who experienced CTS, all attained complete symptomatic relief with corticosteroids or IVIG after discontinuing the offending anti-cancer therapeutic agent. 20 One patient whose EMG demonstrated prolonged distal sensory and motor latency, low SNAPs, and borderline CMAP and f-waves of the right median nerve as well as prolonged distal sensory and latency of the left median nerve underwent a CTR without improvement. Eisenbud and colleagues reported two patients (one with clear cell renal cell carcinoma treated with pembrolizumab; another with melanoma treated with nivolumab), both of whom developed CTS within four cycles of chemotherapy. 1 The ICIs were discontinued in both patients, and corticosteroids were initiated with subsequent symptom resolution.
While the immune-mediated mechanism of ICIs is unclear, peripheral T-cell dysregulation has been proposed. 20 It has also been suggested that the immune-mediated neurological side effects induced by ICIs may be due to inflammation of the vessels and perineural edema around the nerve. 18 The immune systemic activation by anti-cancer therapeutic agents may trigger the production of antibodies which may damage myelinated fibers causing CTS, as depicted in chronic inflammatory demyelinating polyneuropathies. 18
The strength of the present study is a large number of patients with varied cancers treated with anti-cancer therapeutic agents who shortly afterward developed CTS and underwent EDX and US testing. We based our conclusion that the anti-cancer therapeutic agent was the cause of the CTS on the following: (1) onset of symptoms within 2–12 weeks of starting the medication; (2) bilateral occurrence of CTS; (3) lack of other predisposing causes; and (4) objective documentation of abnormal nerve function at the carpal tunnel. While few previous studies have reported the use of EDX studies in evaluating upper limb neuropathy after these medications,6,13,34 none of these merged the presenting symptoms, neurological examination, and detailed EDX evaluation to form a comprehensive evaluation of the patient. Additionally, no study in the literature except that of Dizdar and colleagues reported the value of US in the diagnostic assessment of CTS during treatment with anti-cancer therapeutic agents. Our study adds to the existing literature on CTS due to anti-cancer therapeutic agents and highlights the need for a prospective study of patients with symptoms of CTS developing after the initiation of anti-cancer therapeutic agents. The limitation of this study is its retrospective nature.
In conclusion, our study highlights the importance of incorporating EDX and US studies to the clinical features in the evaluation of patients who develop symptoms of CTS after initiation of anti-cancer therapeutic agents. A high index of suspicion of CTS is warranted after patients start AIs or ICIs. Early recognition may avert the self-discontinuation of these agents and prompt treatment to avoid irreversible motor and sensory deficits from severe entrapment of the median nerve at the carpal tunnel.
Footnotes
Acknowledgments
We acknowledge Norton Healthcare for its continued support.
Author contributions
VI designed the study. LS and VI collected the data, performed the analysis, and interpreted the results. LS drafted the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Informed consent was obtained from all patients. The University of Louisville IRB determined that our study was exempt according to 45 CFR 46.101(b) under Category 4. The IRB number is 22.0544.
Lisa BE Shields, Vasudeva G Iyer, Yi Ping Zhang Christopher B Shields
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
