Abstract
Introduction
The integration of clinical oncology pharmacists into multidisciplinary healthcare teams is not well-described in the community practice setting. This study aims to analyze the clinical and financial impact of a remote-based clinical oncology pharmacist in four community oncology practices within The US Oncology Network.
Methods
Oncology-trained clinical pharmacists electronically reviewed chemotherapy orders for clinical optimization and financial stewardship within four community oncology practices. Each pharmacist was appointed at 0.5 full-time equivalents per practice. Financial, clinical, and workload metrics were tracked to monitor the impact of pharmacist engagement.
Results
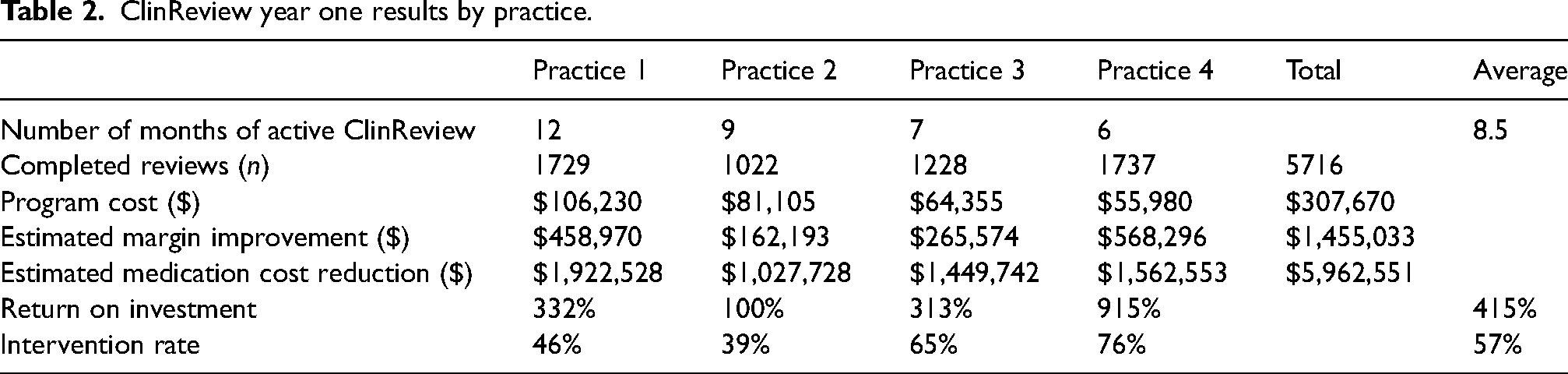
Over 12 months, 5716 order reviews were completed with an intervention rate of 57%. The most common interventions identified by the pharmacists were interventions with clinical impact on the patient (36%), followed by dose rounding (35%) and therapeutic interchange (30%). Overall, interventions improved the cumulative practice margins by $1,455,033 and reduced total medication costs by $5,962,551. The average program return on investment was 415% (range 100–915%).
Conclusion
Community oncology practices seek to provide high-value care in a lean, resource-constrained model. An oncology clinical pharmacist is a cost-effective and clinically invaluable care team member in community oncology practice. Pharmacists in this setting identified opportunities to improve medication safety and regimen optimization and demonstrated a significant tremendous financial impact on small-scale budgets in community oncology.
Introduction
There is well-established integration of on-site oncology pharmacists within multidisciplinary healthcare teams across the country in ambulatory and acute care settings, which has led to improved patient safety, financial stewardship, and access to new clinical pharmacy services.1–4 Although most cancer care is provided within the community practice setting, the impact and role of a remote clinical oncology pharmacist in this setting is not as well-described as in other settings, such as in academic medical centers. Oncology pharmacists have shown financial and clinical value in interprofessional activities (i.e. practice-related committees, patient education teams), however, there is limited literature describing pharmacist involvement in quality measurements and value-based reimbursement systems. 3 Additionally, the recent COVID-19 pandemic has increased the platform and availability of telehealth, providing remote-based opportunities across many healthcare settings. 5 Evaluation of remote pharmacist work and development of remote work policies in health care are lacking. 4
The US Oncology Network (The Network), supported by McKesson Corporation, is a network of community oncology practices treating over 1.2 million patients annually within approximately 500 cancer treatment centers across the United States. 6 Practices within The Network consist of one or more clinic sites collaborating to provide community-based oncology care. Many practices employ a pharmacy director who fills various operational roles to meet the day-to-day needs of the practice with little to no time explicitly dedicated to chemotherapy order review. Many smaller community practices may not support a full-time equivalent (FTE) of a clinical oncology pharmacist based on patient volumes. In contrast, larger practices often have fewer clinical pharmacists dedicated to chemotherapy order review than required to meet patient care needs and volumes. This implementation analysis aimed to evaluate the impact and value of integrating 0.5 FTE remote clinical oncology pharmacists within four community oncology practices in The Network.
Methods
Program design
Following an internal review of the implementation time of The Network's pharmacy and therapeutics (P&T) committee's initiatives within individual practices, the ClinReview Program (CRP) was developed to address implementation barriers related to these initiatives by placing oncology clinical pharmacists remotely into practices. Pharmacists were licensed as required by law for each practice. Institutional Review Board (IRB) approval was unnecessary as this project is classified as a Quality Assurance and Quality Improvement Activity intended to improve clinical care delivery within The Network.
Practice eligibility and enrollment
Starting in March 2021, practices were eligible to participate in the CRP. Participation was voluntary, at the expense of the practice, and practices were enrolled following the execution of a mutually agreeable contract. The CRP established workflows, preferred communication practices, policies, and procedures with the practice. While the CRP adapted to practice-specific processes and preferences, repeatable onboarding processes were used. Standard policies and procedures were available to practices for customization and adoption if not already in existence. Practices that enrolled in the CRP before March 2022 were eligible for this analysis.
ClinReview pharmacist intervention
The impact of the CRP interventions was assessed retrospectively. Oncology-trained clinical pharmacists performed cycle 1 day 1 (C1D1) cancer treatment reviews within their assigned practice(s). CRP pharmacists were appointed to practices at 0.5 FTE or 20 h per week. Cross-training was available to provide limited coverage as needed. Regimens requiring a C1D1 review were identified using the practice's electronic medical record (EMR). Each CRP pharmacist reviewed medication orders and relevant clinical data. The clinical review was documented in the EMR and within an internal tracker. If an intervention was needed, the pharmacist advised changing the order per practice policy or discussed the case with the care team (Figure 1). Interventions were categorized at the pharmacist's discretion at time of documentation, including clinical impact. Pre-determined intervention categories included dose rounding, dose banding, product selection, and clinical change. Clinical changes were further categorized as follows: additional monitoring needed, anticancer drug dosing change, other medication dosing change, modification to supportive care made. The CRP pharmacist aimed to complete initial reviews within 1–2 business days of order placement to ensure timely collaboration with the business, authorization, and scheduling teams. In addition, the CRP pharmacist provided drug information assistance, collaboration surrounding drug shortages, assistance with complicated regimen entry, and participation in additional P&T committee initiatives.
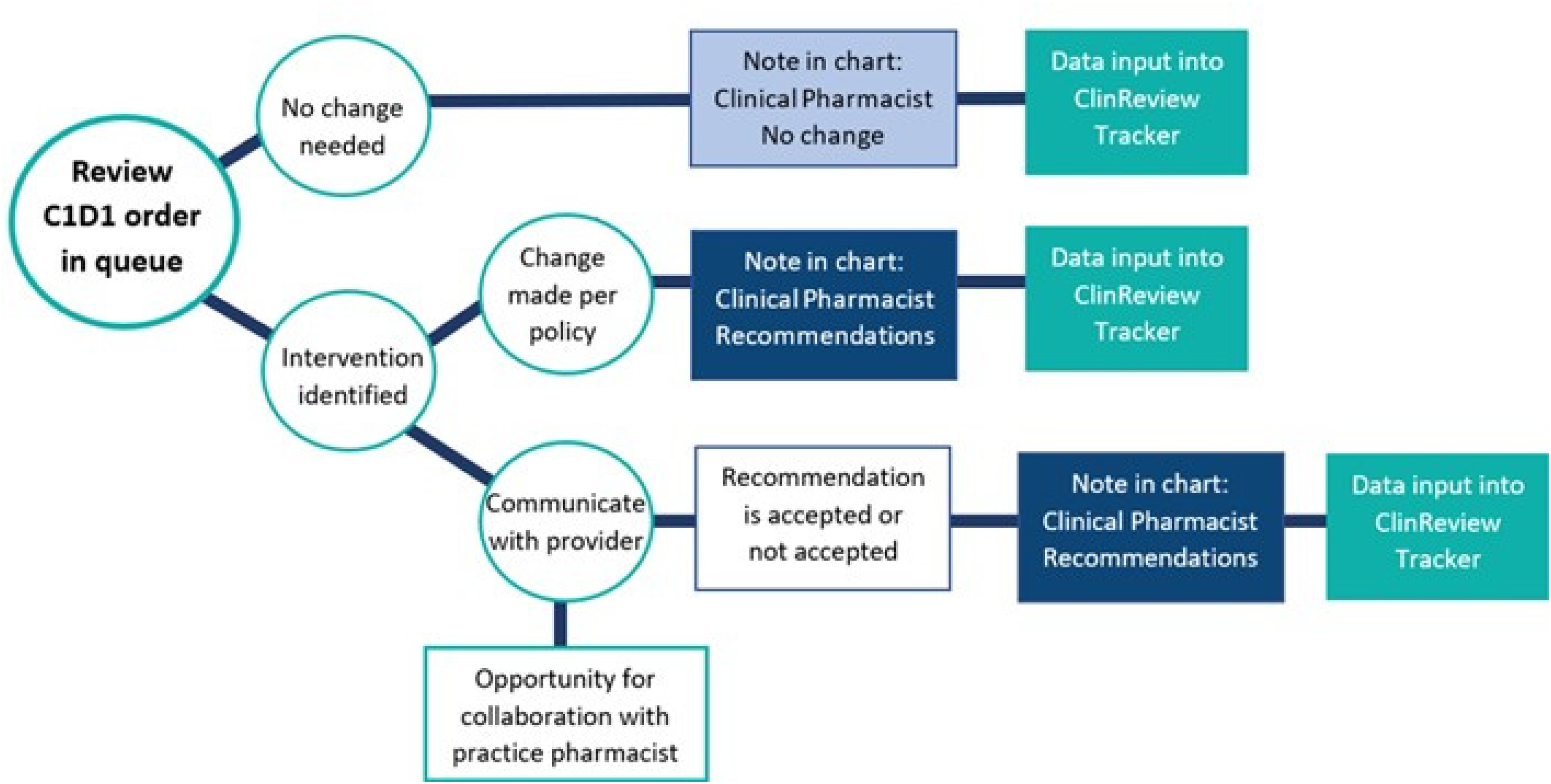
Steps for evaluation of interventions and documentation of C1D1 order review.
Program evaluation
Entries were made into Smartsheet software (Smartsheet Inc, Bellevue, WA) as an internal tracker of completed C1D1 reviews and associated interventions. Details were captured using pre-defined attributes to analyze interventions uniformly. The program's financial impact was assessed using program expense, margin improvement, and medication cost reduction. Program expenses included pharmacist salary and benefits. Margin improvements were calculated when the pharmacist implemented an order change, such as a therapeutic interchange (e.g. switching between two rituximab products). The margin improvement was calculated utilizing the difference between the estimated margin of the original provider order and the implemented pharmacist intervention. The margin was an approximation of the impact of medication changes only, calculated using the difference between estimates of average medication acquisition costs (internal data) and reimbursement estimates (CMS allowable, updated quarterly). Medication cost reductions were reported when a pharmacist implemented a change that resulted in an overall lower medication cost. These were calculated for dose rounding interventions when a medication dose was rounded down to the nearest whole vial size when appropriate and when a therapeutic interchange resulted in using a lower cost medication. The program's return on investment (ROI) was calculated by comparing the margin improvements to program expenses. The financial impact of recommendations related to non-medication aspects of care was not measured. Different categories and pharmacist-reported clinical significance analyzed documented clinical interventions.
Statistical analysis
Descriptive statistics were used to describe practice demographics, financial data, and clinical pharmacist interventions.
Results
Between March 2021 and March 2022, four US Oncology Network practices contracted with the CRP. During this period, 5716 order reviews were completed with an intervention rate of 57%. Intervention acceptance rate was 98% with 66 recommendations not accepted by providers for various clinical reasons. The most common interventions identified by the pharmacists involved recommendations with clinical impact (36%), followed by dose rounding (35%), and therapeutic interchange (30%). Clinical reviews were most often completed within two business days (71%). Reviews completed outside of this timeframe typically occurred due to pending tests results, awaiting completion of clinical note, or communication with the provider team and did not result in delay of treatment initiation.
Practice details
Practice demographics varied by geographic location, practice size, and pre-existing available on-site pharmacy services (Table 1). Three out of four practices had pharmacists dedicated to oral chemotherapy order review and dispensing. Maryland Oncology Hematology also had an on-site oncology clinical pharmacist who performed clinical reviews and various clinical leadership activities. The addition of a remote clinical pharmacist fully dedicated to regimen review allowed the on-site pharmacist to refocus time to clinical functions and objectives at the practice for which time previously did not allow.
Demographics for participating practices.
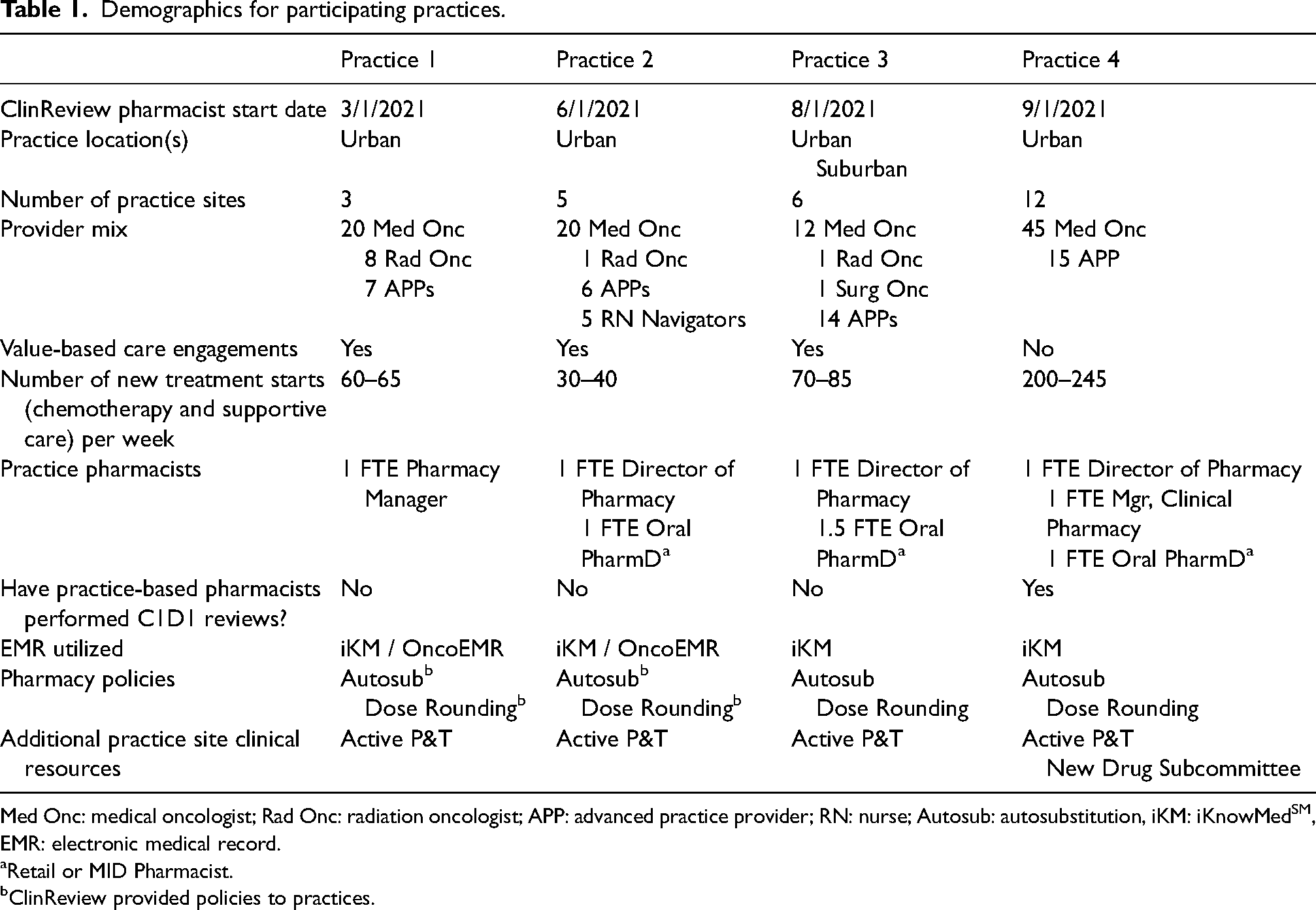
Med Onc: medical oncologist; Rad Onc: radiation oncologist; APP: advanced practice provider; RN: nurse; Autosub: autosubstitution, iKM: iKnowMedSM, EMR: electronic medical record.
Retail or MID Pharmacist.
ClinReview provided policies to practices.
ClinReview year one results by practice.
CRP pharmacist details
During the evaluation period, three oncology clinical pharmacists provided services to the four CRP practices. The pharmacists completed residency training (PGY1, PGY2 oncology, or both), had experience in academic medical centers and community practice sites, were certified by the Board of Pharmacy Specialties (BCPS or BCOP), and had practiced in oncology for an average of 11 years.
Financial impact
Over 12 months, the CRP pharmacist interventions improved the cumulative practice margin, the total margin improvement of all four practices, by $1,455,033 and achieved a total medication cost reduction of $5,962,551 across all four practices combined (Table 2). Both margin improvement and medication cost reductions were fairly consistent over time (Figure 2). Slight variations in monthly margin by practice were observed as drug initiatives, prescribing patterns, patient volumes, and regimen utilization varied over time. The pharmacist reviewed orders written up to 3 months before the program start date at practice 1. The average program ROI was 415% (range 100–915%). Therapeutic interchange with biosimilars (rituximab, bevacizumab, pegfilgrastim) as well as neurokinin 1 (NK-1) receptor antagonists and immune checkpoint inhibitors most frequently contributed to margin improvement (Figure 3).
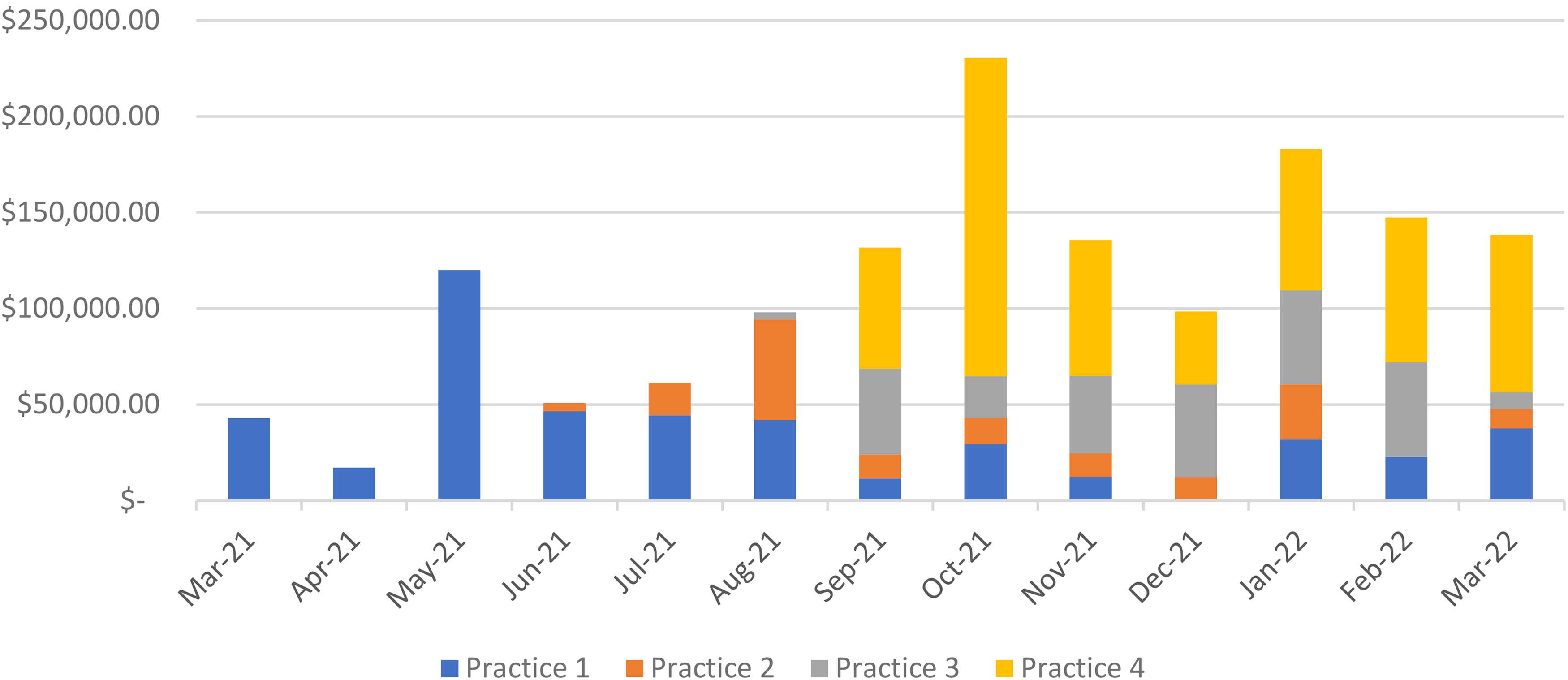
Estimated monthly financial margin improvement per practice.
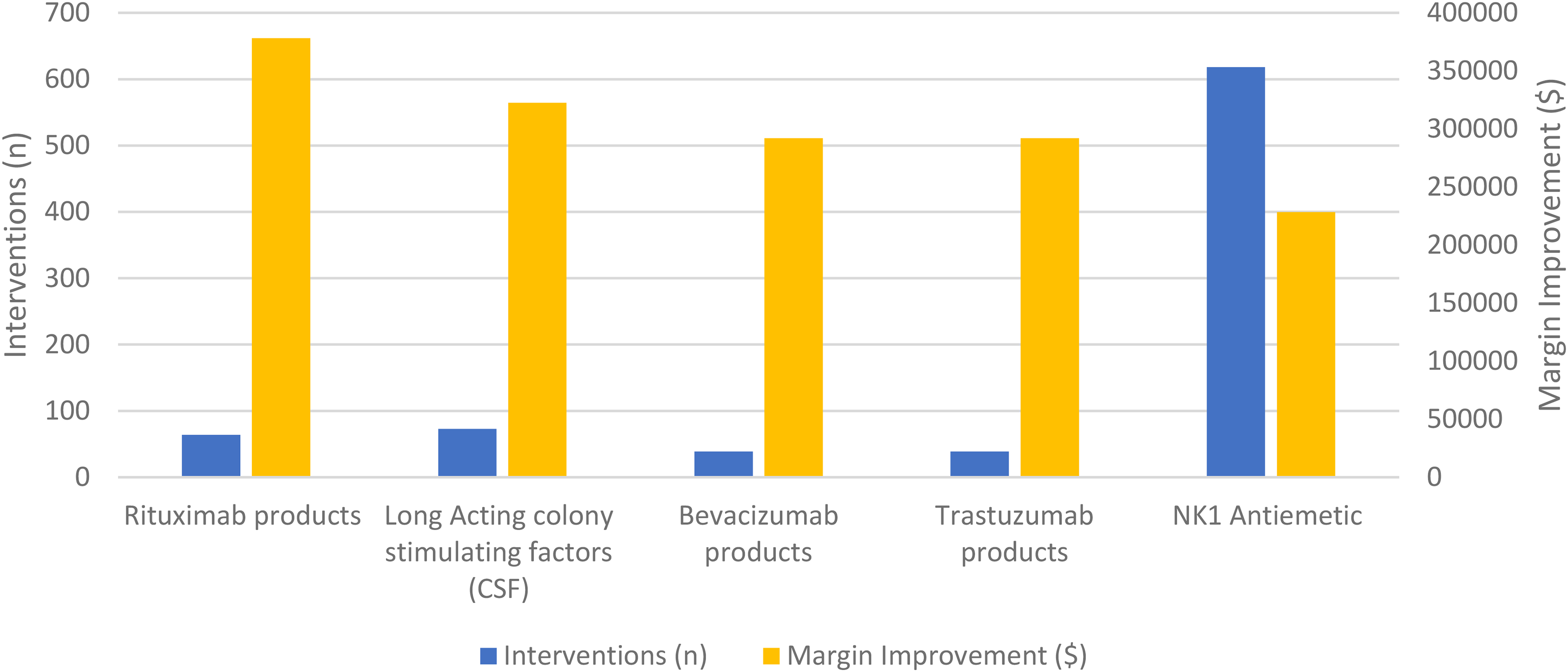
Top 5 margin improvement for therapeutic interchanges by class.
Clinical impact
Of the 5716 orders reviewed, 1165 pharmacist-led interventions with clinical patient impact were documented. These clinical interventions frequently included recommendations for adding additional imaging or laboratory monitoring specific to the chemotherapy regimen (39%), changes to anticancer drug dosing or schedule (31%), and changes within supportive care regimens (26%). These data were further delineated according to specific interventions (Figure 4). Of the 1165 clinical interventions, 794 (68%) were considered clinically significant at the pharmacist's discretion during the documentation process, improving patient care quality and/or safety. Common clinically significant interventions related to safety included growth factor support adjustments, renal and hepatic dose changes, and modifications to antiemetic or home prophylactic medications. Efficacy-related clinically significant interactions included regimen schedule or cycle length modifications, treatment optimization through dose and drug changes based on disease state and genetic testing results, and identification of treatment-altering drug–drug interactions. More detailed examples of clinically impactful interventions are included in Table 3.
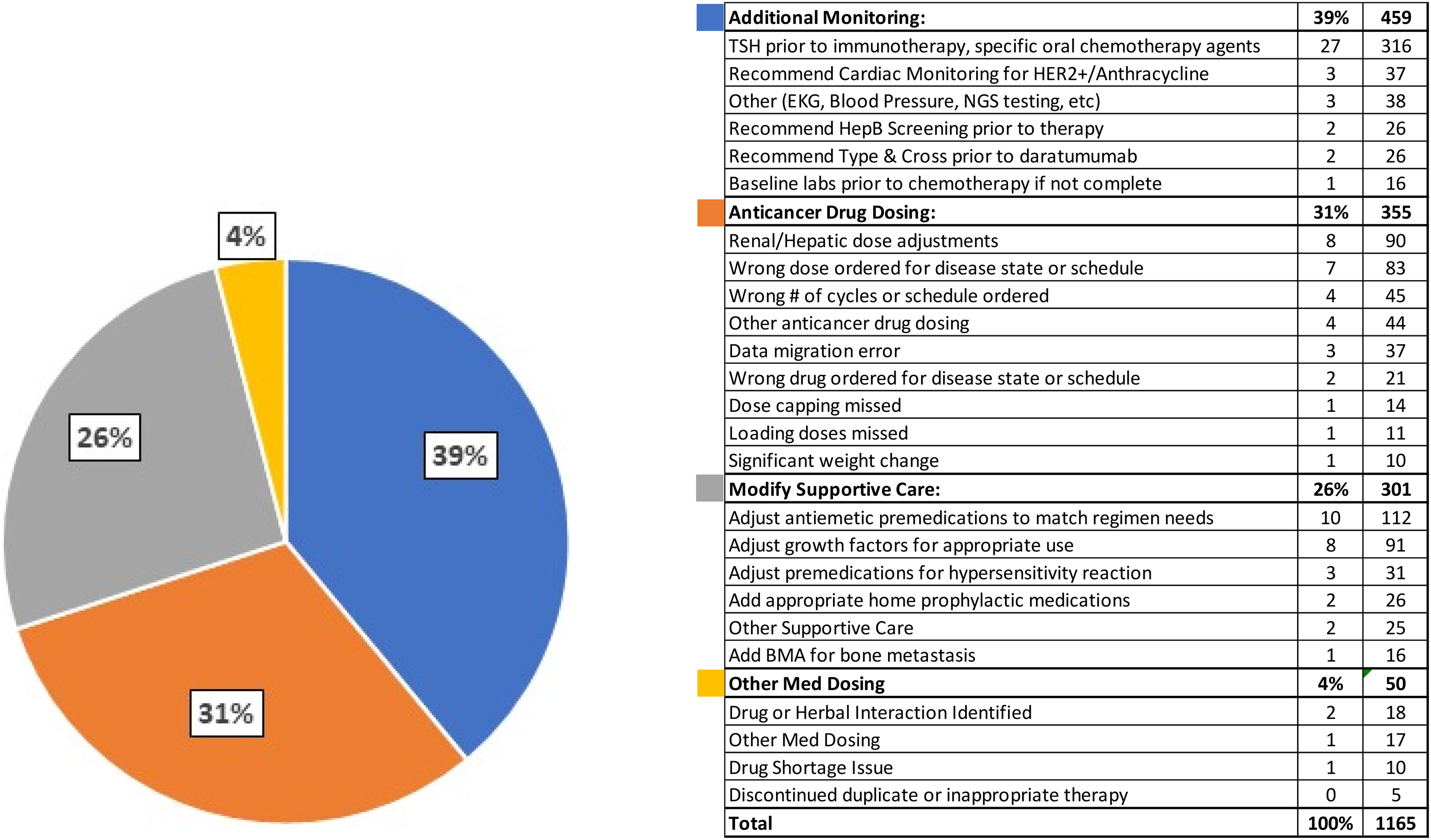
Breakdown of intervention types with clinical impact.
Examples of significant clinical interventions completed by oncology pharmacists.

In addition to these clinical interventions, pharmacists spent 80 h on consultations and drug information assistance, as well as 57 h on regimen entry. These additional inquiries of the pharmacist team from practice providers were a rising trend as pharmacist tenure within the practice increased.
Discussion
Community oncology pharmacist responsibilities are complex and can vary widely between practices. The traditional role of a pharmacist in this setting previously encompassed tasks related to compounding and dispensing. However, the pharmacist's role has evolved as cancer care has changed. 7 A pharmacist with a strong knowledge of oncology medications, regulatory requirements, and the intricacy of insurance reimbursement policies is vital to a successful practice. This evolving role created gaps in many oncology practices as clinical and operational tasks have overwhelmed the current pharmacist staffing model.
Although staffing challenges extend beyond healthcare sectors in the post-pandemic era, community oncology practices experience unique challenges in recruiting and retaining clinical pharmacists. Community oncology practices are commonly found outside the urban, metropolitan areas where pharmacy schools are aligned with academic medical centers and training programs. They are also often smaller and private, physician-owned practices with limited operating budgets, hampering recruitment of oncology-trained clinical pharmacists to these areas. The remote/work-from-home model of the CRP can overcome this geographic barrier, allowing practices to hire the best skill match regardless of a pharmacist's physical location, and can also benefit when on-site staffing is reduced or limited, such as during surges in COVID-19. The opportunity to work remotely can also increase the overall job satisfaction of the pharmacist, minimizing the downstream effects of pharmacist turnover and re-training. The flexibility of the CRP to provide 0.5 FTE of an oncology-trained clinical pharmacist for smaller community practices is also relevant and conducive to the introduction and buy-in of the oncology clinical pharmacist into practices on a smaller scale. As practice needs and volumes grow, the CRP can evolve to provide more pharmacist support. In addition to two of the original four practices increasing from 0.5 to 1.0 FTE, the program has added eight new practices with a total of 4.5 FTE.
The CRP sought to provide oncology clinical pharmacists to community oncology practices to achieve practice goals and improve the P&T initiative rollouts. Justifying a pharmacist's expenses to a practice executive team can be a barrier to implementation. Many executive teams are acutely aware of the difference between hard and soft dollars in justifying additional FTEs. Pharmacists have traditionally contributed to the bottom line by generating revenue for dispensing prescriptions and collecting payments from an insurance pharmacy benefits manager. Establishing the additional value of “soft” dollars from correct drug selection, formulary, medication management, and adequate patient and provider education become more complex. For these reasons, the Smartsheet internal tracker system was utilized. These real-time metrics were used to communicate progress and share program impact regularly.
The CRP also expanded the clinical capacity of practices with existing clinical pharmacy services. The bandwidth of the practice-based clinical pharmacists was limited to therapeutic initiatives during C1D1 reviews and completing administrative tasks due to practice size and order volume. Incorporating the CRP increased total pharmacist FTE hours, allowing for a more comprehensive review of C1D1 regimens and expedited implementation of new therapeutic initiatives within the organization. The CRP team also increased access to a larger pharmacy community for sharing knowledge and resources amongst practices.
Implementation of the CRP required conquering challenges. Remote work was novel to most practices, and integration into daily workflow required multidisciplinary collaboration between site administration staff, nurses, medical assistants, physicians, advanced practice providers, and the authorization teams. The development of policies, procedures, and clinical note templates was needed to support these changes. Once policies and workflows were established, the next challenge became building relationships and rapport remotely with clinical staff. Each practice had to establish its preferred communication methods to maximize the CRP pharmacist's efficiency. Some practices utilized different EMRs and had varying preferences in receiving clinical recommendations (e.g., email messaging, EMR messages and notes, direct instant messaging). Navigating more than one EMR proved challenging when finding pertinent information to complete clinical reviews and increased training and onboarding time. Creating pharmacist note templates increased productivity and practice standardization, allowing providers to quickly find recommendations and improve coordination with both on- and off-site pharmacy staff.
The financial impact of the CRP is based on self-documented interventions associated with projected margin improvement and medication cost reductions. Because this is not associated with a direct revenue source (e.g. a pharmacist billing for services), it can be challenging to justify the intangible financially yet clinically impactful interventions that pharmacists can make to leadership teams.
Adding financial measurements of CRP interventions is a strength of the program. However, rather than exact figures for the practice's financial benefit, these data are forecasts with clearly defined assumptions as stated in the methods. In addition, the pharmacist enters the number of doses planned or prescribed at the order review time. In the current state, there is not a retrospective review to validate these with delivered treatments. This analysis is planned as a future direction of the program.
Looking forward, the role and scope of the ClinReview pharmacist could expand through several avenues, requiring growth beyond a 0.5 FTE appointment. Opportunities include clinical reviews beyond C1D1, establishing collaborative practice agreements for improved workflow efficiency, expanding the P&T committee role, payer-focused approaches for individual drug classes, oral chemotherapy reviews, and maintenance of chemotherapy order sets for EMRs needing manual updates. This expansion can be tailored to practice goals while maintaining consistent care improvement across The Network as ideas and initiatives are shared and expanded.
Conclusion
Implementing the CRP within four community oncology practices in The US Oncology Network proved cost-effective and clinically significant. Within the first year, the CRP generated a 415% ROI, demonstrating tremendous financial impact on small-scale community oncology practice budgets. This program also demonstrated the ability of geographically remote-based clinical oncology pharmacists to effectively identify opportunities to improve medication safety and regimen optimization, even at a 0.5 FTE staffing appointment. Although remote-based pharmacists can pose some logistical challenges, the opportunity for collaboration between a remote-based oncology pharmacist and the practice-based pharmacist can optimize both roles, providing more comprehensive support for complex and budget-conscious community oncology practices.
Footnotes
Author Contributions
MC, EK, JC, AR, MK, and SH researched literature and MC, EK, JC, AR, MK, SH, TP, VP, TM, and LY wrote the first draft of the manuscript. All authors conceived the study, reviewed and edited the manuscript and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
