Abstract
Introduction
This observational study describes the real-world economic burden in patients with anaplastic lymphoma kinase (ALK) positive non-small cell lung cancer (NSCLC) receiving a first-line ALK inhibitor, and the economic impact of brain metastases (BM).
Methods
Administrative claims data (Truven Health MarketScan® Commercial Claims and Encounters database and Medicare Supplemental and Coordination of Benefits database; January 1, 2015–March 31, 2020) for adult patients with ALK+ NSCLC who received a first-line ALK inhibitor were retrospectively reviewed. Healthcare costs and resource utilization were calculated on a per-patient-per-month (PPPM) basis and stratified by the presence or absence of BM prior to first-line ALK inhibitor. Factors associated with costs were identified.
Results
A total of 496 patients were eligible for analysis. Mean PPPM total healthcare costs were $21,961 for all patients receiving up to 1 year of a first-line ALK inhibitor. Patients were significantly more likely to have higher mean PPPM total costs if they had BM prior to first-line ALK inhibitor (vs. no BM; odds ratio: 1.11; 95% confidence interval: 1.02, 1.21; p = 0.013). Mean PPPM days of hospital stay (p = 0.0056), and inpatient hospital visits (p = 0.0030) were significantly higher for patients with BM compared to no BM. The main cost drivers for non-inpatient procedures for all patients were medications, radiation therapy, and other diagnostic procedures.
Conclusions
The economic burden in patients with ALK+ NSCLC receiving a first-line ALK inhibitor was high. Patients with ALK+ NSCLC and BM had higher healthcare costs and resource utilization than patients without BM.
Keywords
Introduction
In the United States (US), in 2020, there were approximately 253,537 new lung cancer cases and 159,641 related deaths. 1 Non-small cell lung cancer (NSCLC) accounts for 85% of all lung cancers. 2 Anaplastic lymphoma kinase (ALK) gene rearrangements are found in 3–5% of NSCLC and define a distinct clinicopathologic subtype of NSCLC.3–5 Patients with ALK+ NSCLC are typically younger than the general population of patients with NSCLC and more likely to have never smoked or have a history of light smoking. 6 The NCCN Clinical Practice Guidelines In Oncology (NCCN Guidelines®) for ALK+ metastatic NSCLC recommend the next-generation ALK inhibitors alectinib (approved as first-line therapy by the US Food and Drug Administration [FDA] in 2017), 7 brigatinib (approved as first-line therapy by the US FDA in 2020), 8 or lorlatinib (approved as first-line therapy by the US FDA in 2021) 9 as preferred options, ceritinib (approved as first-line therapy by the US FDA in 2017) 10 as other recommended intervention, and crizotinib (first approved by the US FDA in 2011) 11 as useful in patients in certain circumstances. 12
Brain metastases (BM) are a common complication in ALK+ NSCLC. Approximately 22–33% of patients with ALK+ NSCLC have BM at initial diagnosis. 13 In addition to the symptoms of the primary tumor, patients with BM experience headaches, increased intracranial pressure, focal neurological signs (aphasia, ataxia, vision disturbances), epileptic seizures, or neurocognitive deficits that may impair health-related quality of life.14–16 BM in ALK+ NSCLC are typically treated with systemic therapy (ALK inhibitors and chemotherapy) and/or localized therapy, including whole-brain radiotherapy, stereotactic surgery, and surgical resection. 17 Next-generation ALK inhibitors are more effective against BM than the first-generation ALK inhibitor crizotinib.13,18–20 A retrospective analysis of the PROFILE 1005 or 1007 trials showed the central nervous system was the most common site of disease progression in 70% of patients with ALK+ NSCLC and BM during treatment with crizotinib. 21 In the ASCEND-7, ALEX, ALTA-1L, and CROWN trials, the intracranial objective response rate was 28.6–51.5%, 78.6–85.7%, 78%, and 82% for ceritinib, alectinib, brigatinib, and lorlatinib, respectively.22–25 In the ALEX trial, in patients with advanced ALK+ NSCLC and no prior systemic treatment for advanced NSCLC, median progression-free survival (PFS) was 27.7 months for alectinib and 7.4 months for crizotinib in the subgroup of patients with baseline BM. 26 In the ALTA-1L trial, in patients with tyrosine kinase inhibitor (TKI)-naive advanced ALK+ NSCLC, median intracranial PFS was 24.0 months for brigatinib and 5.5 months for crizotinib in the subgroup of patients with baseline BM. 24
Limited data describe healthcare utilization and costs in patients with ALK+ NSCLC, with and without BM, receiving ALK inhibitors. Among patients with ALK+ NSCLC treated with crizotinib as first- or later-line therapy, healthcare resource utilization (HCRU) increased after a diagnosis of BM, with monthly healthcare costs per patient rising from $,5983 per-patient-per-month (PPPM) to $22,645 PPPM in the pre- and post-BM-diagnosis periods, respectively. 27 In patients with ALK+ NSCLC receiving second-line treatment with ceritinib or alectinib post-crizotinib, higher mean PPPM total costs and PPPM HCRU were observed for patients with BM compared to no BM. 28 In a study of the real-world economic impact of BM among newly diagnosed patients with ALK+ NSCLC receiving ALK inhibitors (crizotinib, ceritinib, or alectinib), the total cost of BM was estimated at $6,029 PPPM. 29
As novel therapies and ALK inhibitor sequencing for ALK+ NSCLC with and without BM continue to emerge and overall survival improves, consideration of real-world healthcare costs, associated with treatment becomes important in medical decision-making. An understanding of healthcare costs in patients with ALK+ NSCLC, with and without BM, receiving first-line ALK inhibitors is needed. The objectives of the present study were to: (1) investigate the real-world economic burden in patients with ALK+ NSCLC receiving first-line ALK inhibitors; (2) evaluate the economic impact of BM in these patients; and (3) examine the cost drivers.
Methods
Data source
This longitudinal, retrospective, observational study analyzed administrative claims data extracted from the Truven Health MarketScan® Commercial Claims and Encounters database and Medicare Supplemental and Coordination of Benefits database. The MarketScan database is a compilation of electronic claims data from approximately 350 unique carriers and over 273 million unique patients and contains 37 billion service records in the US. 30 Data were de-identified in compliance with the Health Insurance Portability and Accountability Act; therefore, reviews by an institutional review board were not required for this study.
Study design
The study design is shown in Figure 1. The index date was defined as the date of the first claim with an ALK inhibitor. Baseline period was defined as the 6 months prior to the index date. The follow-up period was defined as the 12 months following the index date. Patients were observed until the end of the 12-month follow-up period, end of eligibility, or the last date of data, whichever came first; the duration of follow-up was ≥6 months.
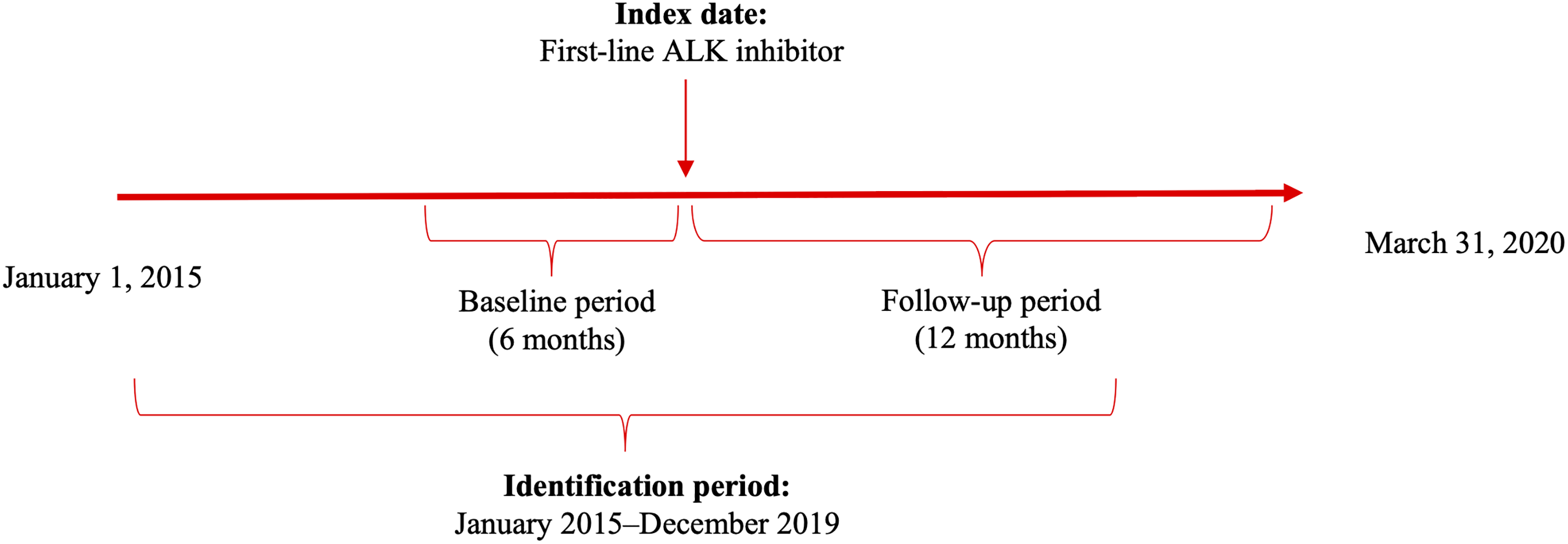
Study design. ALK, anaplastic lymphoma kinase.
Patient population
Adult patients with ALK+ NSCLC receiving a first-line ALK inhibitor (crizotinib, ceritinib, alectinib, brigatinib, and lorlatinib) between January 1, 2015, and March 31, 2020, were identified. Inclusion criteria were: (1) ≥1 inpatient claim or ≥2 outpatient claims ≥30 days apart within 1 year of a lung cancer diagnosis (International Classification of Diseases, Ninth Revision [ICD-9] codes: 162.x, 162.0x, 162.2x, 162.3x, 162.4x, 162.5x, 162.8x, 162.9x; Tenth Revision [ICD-10] C34.x, C34.0x, C34.1x, C34.2, C34.3x, C34.8x, C34.9x); (2) ≥6 months continuous enrollment prior to the index date and ≥6 months continuous enrollment post-index date; and (3) received a first-line ALK inhibitor approved as of 2020 (crizotinib, ceritinib, alectinib, brigatinib, and lorlatinib). Dispensing of ALK inhibitors served as a proxy for diagnosis of ALK+ NSCLC.
BM were identified based on a BM diagnosis (ICD-9: 198.3x, 198.4x; ICD-10: C79.31xx, C79.32xx, C79.4xxx; note: codes for “other central nervous system” are included) prior to the first-line ALK inhibitor.
Study measures
Patients’ demographic and clinical characteristics were assessed during the 6-month baseline period and included age, gender, region (defined based on US census definitions, Table 1), insurance type, and non-cancer-related Charlson Comorbidity Index (CCI).
Baseline characteristics.
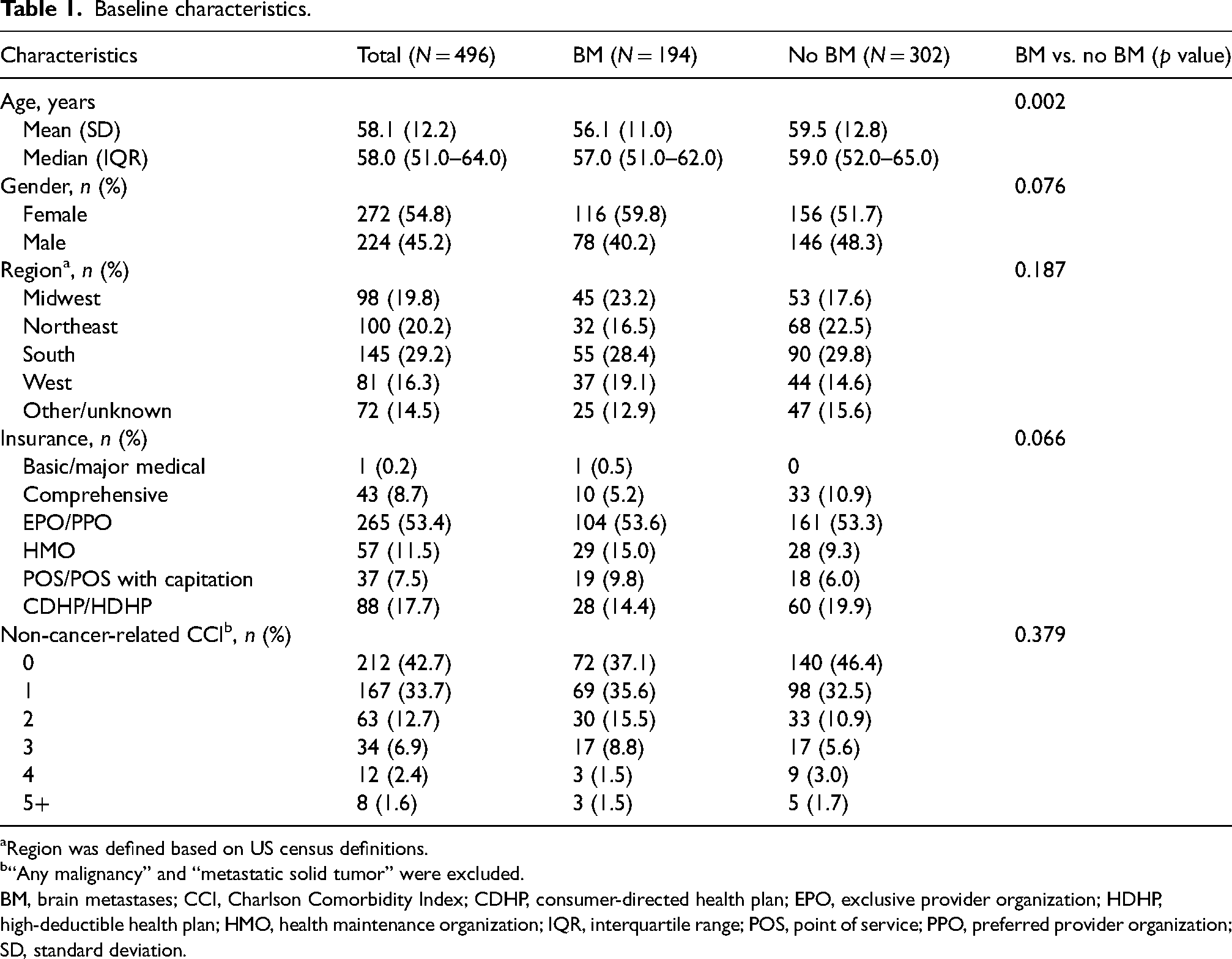
Region was defined based on US census definitions.
“Any malignancy” and “metastatic solid tumor” were excluded.
BM, brain metastases; CCI, Charlson Comorbidity Index; CDHP, consumer-directed health plan; EPO, exclusive provider organization; HDHP, high-deductible health plan; HMO, health maintenance organization; IQR, interquartile range; POS, point of service; PPO, preferred provider organization; SD, standard deviation.
Healthcare costs and HCRU were analyzed during the follow-up period. Total healthcare costs included medical and pharmacy costs. Medical costs included inpatient, emergency department, outpatient, office visit, and other (e.g., hospice, nursing home, etc.) costs. Pharmacy costs comprised ALK inhibitor costs and other pharmacy costs.
Non-inpatient cost drivers were determined by calculating procedure costs as a percentage of total non-inpatient costs. HCRU was summarized as the number of days of hospital stay, inpatient hospital visits, emergency department visits, outpatient visits, and office visits.
Data analysis
All analyses were performed using SAS Version 9.4 or later. 31 Descriptive analyses were conducted for baseline patient characteristics. Continuous measures were summarized as means, standard deviations (SD), and median/range, and categorical measures were summarized as numbers and percentages. Student t tests and chi-square tests were used to compare continuous variables and categorical variables, respectively; p < 0.05 was considered statistically significant.
Data including demographics and clinical characteristics, healthcare costs, and HCRU were assessed for all patients and stratified by the presence or absence of BM. Healthcare costs, calculated on a PPPM basis, represented paid amounts of adjudicated claims (i.e., insurer and health plan payments, copayments, coinsurance, and deductibles), and were adjusted to 2020 US dollars using the medical component of the Consumer Price Index. HCRU was calculated on a PPPM basis.
Factors associated with mean PPPM total costs were investigated in multivariate regression by fitting generalized linear models with gamma distribution and a log link adjusted for patient baseline characteristics (age, gender, region, and insurance type), BM prior to index date, and non-cancer-related CCI.
A sensitivity analysis to assess the impact of medicines prescribed was performed by adding first-line ALK inhibitors (alectinib, brigatinib, ceritinib, or lorlatinib vs. crizotinib) as covariates to the multivariate regression analysis.
A separate sensitivity analysis assessed the impact of BM development following first-line therapy by replacing the binary baseline BM variable with a category baseline BM variable (no BM [reference group]: no BM in both baseline and follow-up period; post-treatment BM: no BM in baseline period but developed BM within 1 year on or after index treatment; BM prior to baseline: BM in baseline period) to the multivariate regression analysis.
Non-inpatient procedure costs were estimated for all patients and patients stratified by the presence or absence of BM. The top 10 cost drivers were identified.
Results
Baseline demographic and clinical characteristics
Patient selection is presented in Figure 2. A total of 496 patients with ALK+ NSCLC receiving a first-line ALK inhibitor were eligible for analysis. Among these, 309 patients received crizotinib (BM: n = 114; no BM: n = 195), 32 patients received ceritinib (BM: n = 16; no BM: n = 16), 149 patients received alectinib (BM: n = 60; no BM: n = 89), 4 patients received brigatinib (BM: n = 4; no BM: n = 0), and 2 patients received lorlatinib (BM: n = 0; no BM: n = 2). Baseline demographic and clinical characteristics of all patients and those stratified by presence or absence of BM are summarized in Table 1.
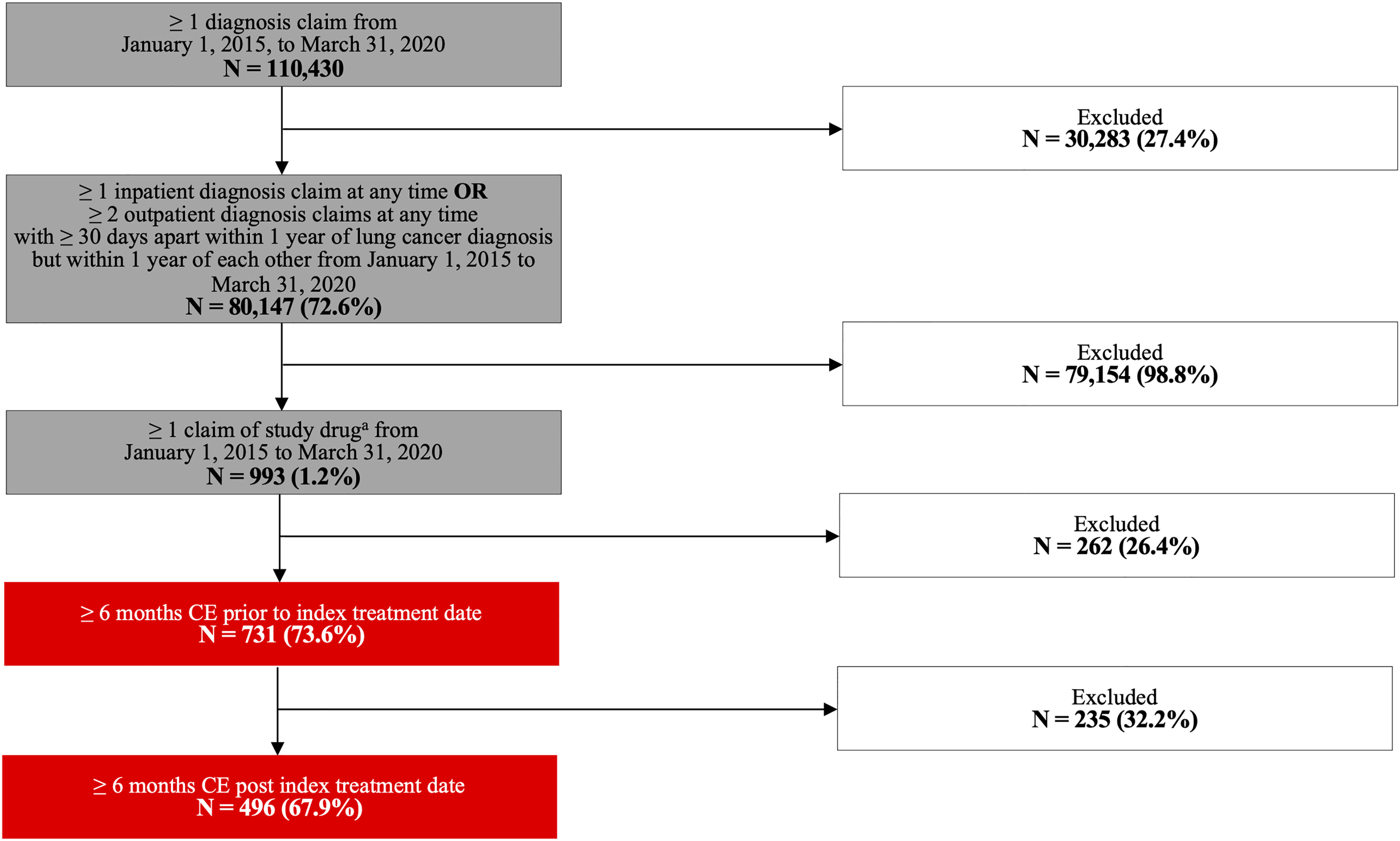
Sample attrition. aStudy drug: Crizotinib, ceritinib, alectinib, brigatinib, lorlatinib. CE, continuous enrollment.
For all patients, mean (± SD) age was 58.1 (± 12.2) years, and 54.8% were female. The majority of patients were from the South (29.2%), followed by the Northeast (20.2%), Midwest (19.8%), and West (16.3%). Most patients (76.4%) had a non-cancer-related CCI of ≤1. More than half (53.4%) of the patients had a preferred provider organization insurance plan, 11.5% had a health maintenance organization plan, and 17.7% had a consumer-directed health plan.
Of the 496 eligible patients, 194 patients had BM prior to baseline and 302 had no BM at baseline. Patients with BM were significantly younger than patients with no BM (mean [SD] 56.1 [11.0] years vs. 59.5 [12.8] years; p < 0.01). Sex, region, insurance type, and non-cancer-related CCI were not significantly different between patients with BM and with no BM (Table 1). Of the 302 patients without baseline BM, 61 (20%) developed BM within 1 year on or after index treatment.
Economic burden in patients with ALK+ NSCLC receiving first-line ALK inhibitors
Mean (SD) PPPM total healthcare costs were $21,961 (13,087) for all patients receiving first-line ALK inhibitors. Mean PPPM inpatient costs, emergency department costs, outpatient costs, office visit costs, pharmacy costs, and other costs were $2,662, $222, $5,342, $928, $12,604, and $204, respectively.
Compared to patients with no BM, patients with BM had statistically significantly higher mean [SD] PPPM total healthcare costs ($20,980 [13,383] vs. $23,490 [12,494]; p = 0.0343), mean [SD] PPPM inpatient costs ($1,925 [4,537] vs. $3,809 [7,918]; p = 0.0028) and mean [SD] PPPM office visit costs ($745 [1,435] vs. $1,212 [2,043]; p = 0.0059), and significantly lower mean [SD] PPPM emergency department costs ($267 [929] vs. $152 [287]; p = 0.0455). For patients with no BM, the mean (SD) PPPM outpatient, pharmacy, and other costs were $5,152 (11,074), $12,704 (5,110), and $186 (665), respectively. For patients with BM, the mean (SD) PPPM outpatient, pharmacy, and other costs were $5,637 (8,045), $12,448 (4,538), and $232 (550), respectively (Figure 3).
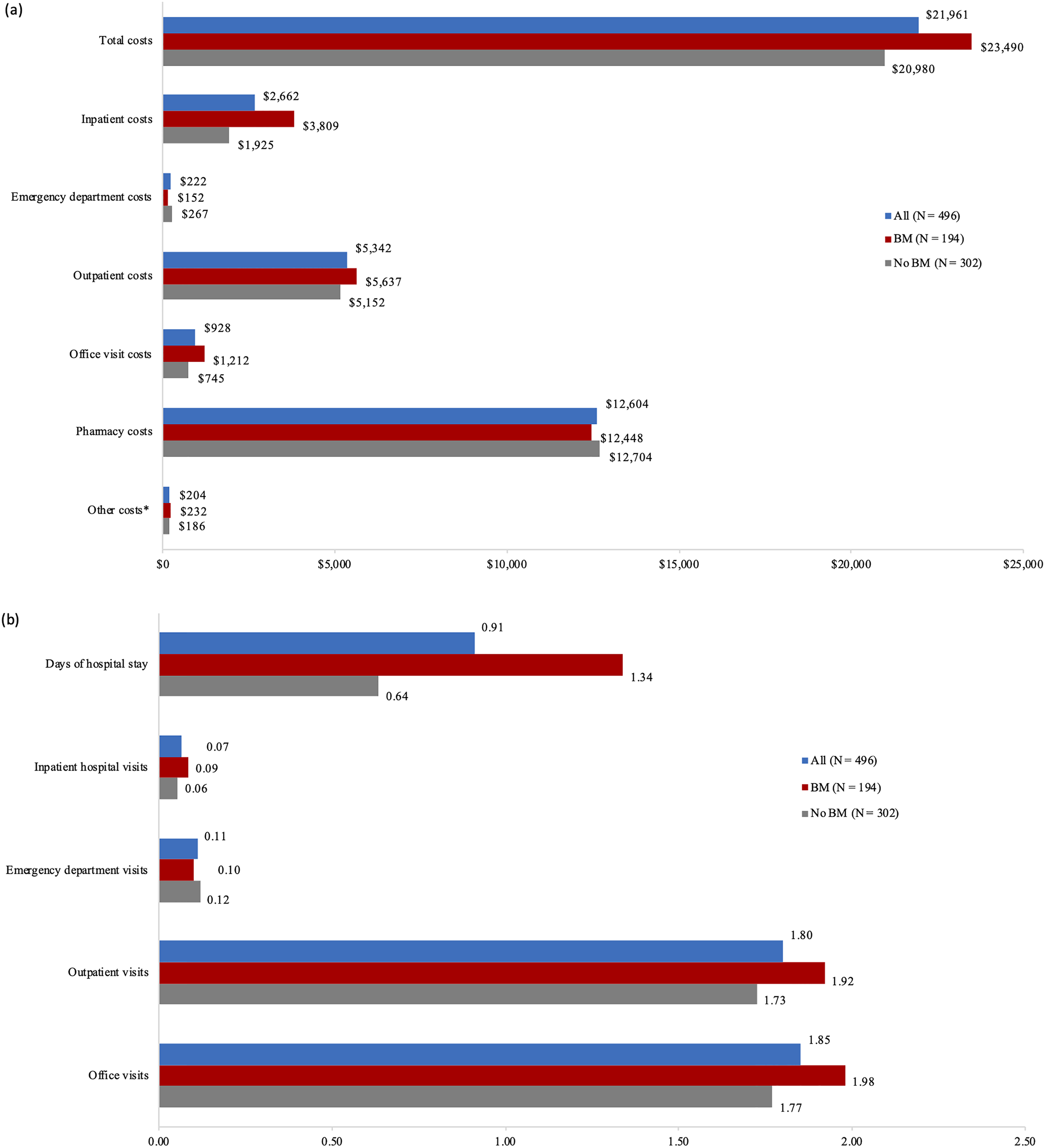
Economic burden associated with use of first-line ALK inhibitors within 12 months post-index date. (a) Unadjusted PPPM healthcare costs (mean, 2020 USD). (b) Unadjusted PPPM healthcare utilization (mean). *Other costs include all not listed miscellaneous services, such as hospice, nursing home, etc. ALK, anaplastic lymphoma kinase; BM, brain metastases; PPPM, per-patient-per-month; USD, United States dollar.
Mean (SD) PPPM days of hospital stay were 0.91 (2.37), inpatient hospital visits were 0.07 (0.11), emergency department visits were 0.11 (0.37), outpatient visits were 1.80 (1.37), and office visits were 1.85 (1.44) for all patients receiving first-line ALK inhibitors.
Compared to patients with no BM, patients with BM had statistically significantly higher mean [SD] PPPM days of hospital stay (0.64 [1.39] vs. 1.34 [3.33]; p = 0.0056) and mean [SD] PPPM inpatient hospital visits (0.06 [0.10] vs. 0.09 [0.12]; p = 0.0030). Patients with no BM had a mean (SD) PPPM 1.73 (1.37), 1.77 (1.40), and 0.12 (0.45) outpatient, office, and emergency department visits, respectively. Patients with BM had a mean (SD) PPPM 1.92 (1.37), 1.98 (1.48), and 0.10 (0.17) outpatient, office, and emergency department visits, respectively (Figure 3).
In multivariable analysis, controlling for patient characteristics (age, gender, region, insurance, CCI, and presence of BM prior to the first-line ALK inhibitor), patients were significantly more likely to have higher mean PPPM total costs if they had a CCI ≥ 2 (vs. 0; odds ratio [OR]: 1.13; 95% confidence interval [CI]: 1.01, 1.26; p = 0.031), and BM prior to the first-line ALK inhibitor (vs. no BM; OR: 1.11; 95% CI: 1.02, 1.21; p = 0.013). Patients were significantly less likely to have higher mean PPPM total costs if they were female (vs. male; OR: 0.91; 95% CI: 0.84, 0.99; p = 0.022), and resided in the South region of the US (vs. West; OR: 0.87; 95% CI: 0.77, 0.98; p = 0.028) (Figure 4).
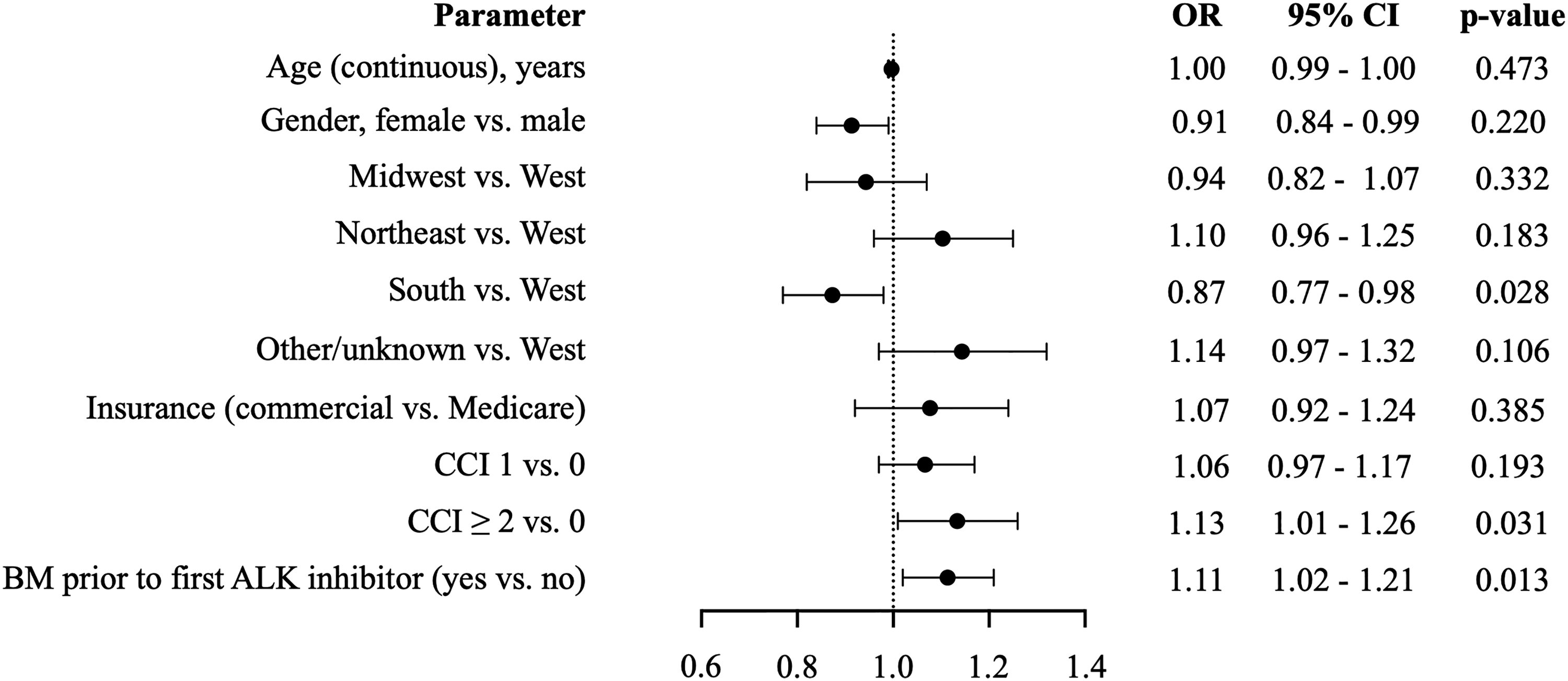
Generalized linear regression of per patient per month total cost. ALK, anaplastic lymphoma kinase; BM, brain metastases; CCI, Charlson Comorbidity Index; CI, confidence interval; OR, odds ratio.
In multivariable analysis, controlling for treatment vs. crizotinib, patients were significantly more likely to have higher PPPM costs if they had BM before treatment with ALK inhibitors (vs. no BM: OR: 1.13; 95% CI: 1.03, 1.23; p = 0.01) (Supplemental Table 1). Patients were significantly less likely to have higher mean PPPM total costs if they received brigatinib (vs. crizotinib; OR: 0.59; 95% CI: 0.37, 0.94; p = 0.03); no significant difference was observed for alectinib, ceritinib, or lorlatinib vs. crizotinib (Supplemental Table 1).
In multivariable analysis, controlling for the presence of BM post-treatment or prior to treatment (vs. no BM), patients were significantly more likely to have higher mean PPPM total costs if they developed BM post-treatment (vs. no BM; OR: 1.27; 95% CI: 1.12, 1.45; p < 0.001) or prior to treatment (vs. no BM; OR: 1.18; 95% CI: 1.08, 1.29; p < 0.001) (Supplemental Table 2).
The main drivers of non-inpatient procedure costs were medications (17.2%), radiation therapy (RT) (11.2%), and other diagnostic procedures (6.8%) for all patients receiving first-line ALK inhibitors. For patients with BM, the main cost drivers were medications (19.1%), RT (15.1%) and magnetic resonance imaging (10.2%). For patients with no BM, the main cost drivers were medications (15.8%), RT (8.4%), and other diagnostic procedures (6.8%). Data on cost drivers for non-inpatient procedures were missing for 28.3%, 20.8%, and 33.7% of all patients, patients with BM, and patients with no BM, respectively (Table 2).
Top non-inpatient procedure cost drivers in patients receiving up to 1 year of post-first-line ALK inhibitor.
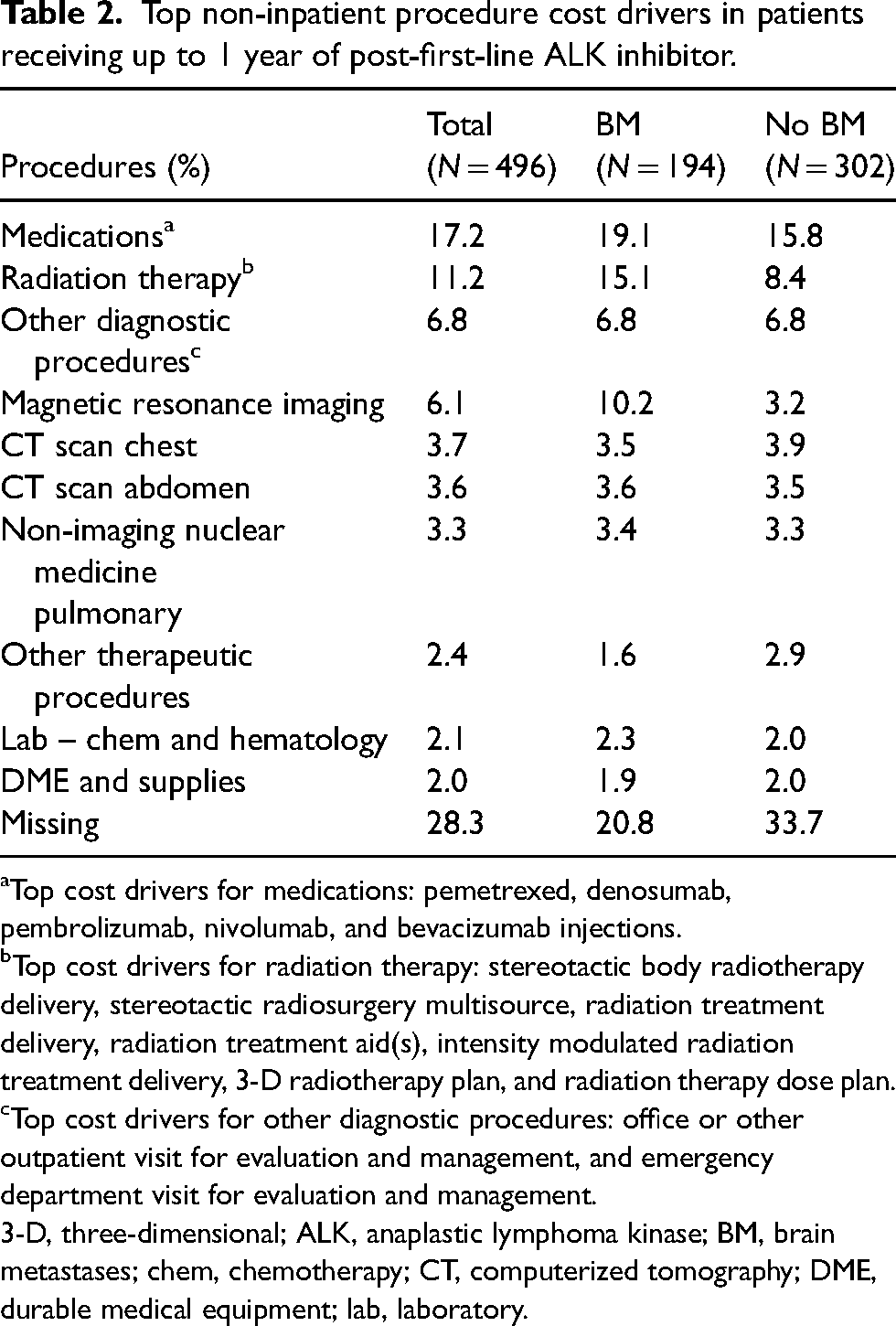
Top cost drivers for medications: pemetrexed, denosumab, pembrolizumab, nivolumab, and bevacizumab injections.
Top cost drivers for radiation therapy: stereotactic body radiotherapy delivery, stereotactic radiosurgery multisource, radiation treatment delivery, radiation treatment aid(s), intensity modulated radiation treatment delivery, 3-D radiotherapy plan, and radiation therapy dose plan.
Top cost drivers for other diagnostic procedures: office or other outpatient visit for evaluation and management, and emergency department visit for evaluation and management. 3-D, three-dimensional; ALK, anaplastic lymphoma kinase; BM, brain metastases; chem, chemotherapy; CT, computerized tomography; DME, durable medical equipment; lab, laboratory.
Discussion
This real-world study showed that the economic burden in patients with ALK+ NSCLC receiving first-line ALK inhibitors is high. Patients with BM prior to the first-line ALK inhibitor were significantly more likely to have higher PPPM total costs than patients with no BM. Patients with BM had greater HCRU than patients with no BM, including more inpatient visits and a longer length of hospital stay.
In the present study, the unadjusted PPPM total cost for all patients with ALK+ NSCLC receiving first-line ALK inhibitors reached nearly $22,000. These costs were similar to the high costs observed in a prior study that evaluated treatment costs regardless of ALK inhibitor use in patients with and without BM (mean PPPM cost: $29,497 and $22,791, respectively). 29 Previously, a lung cancer disease model built using the US adjudicated claims data from October 2017 to September 2018 to evaluate costs associated with the use of ALK inhibitors demonstrated that PPPM all-cause costs for patients with ALK+ NSCLC receiving any-line alectinib (59% of patients) or crizotinib (33% of patients) were $18,541 and $19,078, respectively. 32 Retrospective analyses conducted between January 2001 and September 2017 in the US reported that overall costs associated with the use of ALK inhibitors at any time following a diagnosis of NSCLC,33,34 including the second-line setting, 28 ranged from $111,468 per-patient-per-six-months to $23,984 PPPM.
To the authors’ knowledge, this is the first study to examine the economic burden in patients with ALK+ NSCLC, with and without BM, receiving first-line ALK inhibitors. Findings showed that PPPM costs for patients with BM prior to the first-line ALK inhibitor were 1.11 times as high as those for patients with no BM. This analysis is more representative of the current treatment landscape than older studies, as the time period selected (2015–2020) encompasses the recent approvals of next-generation ALK inhibitors for the first-line treatment of ALK+ NSCLC.27–29,34
A sensitivity analysis that assessed the impact of medicines prescribed (vs. crizotinib) showed patients were significantly more likely to have higher PPPM costs if they had BM before treatment with ALK inhibitors (OR 1.13, similar to the base case, OR 1.11), and patients were significantly less likely to have higher mean PPPM total costs if they received brigatinib vs. crizotinib; no significant differences were noted for the other ALK inhibitors. A sensitivity analysis that controlled for the presence of BM post-treatment or prior to treatment (vs. no BM) showed patients were significantly more likely to have higher mean PPPM total costs if they developed BM post-treatment or prior to treatment.
The availability of more efficacious and tolerable next generations of ALK inhibitors with better intracranial activity may alleviate some of the economic burden associated with BM. In particular, early use of next-generation ALK inhibitors may be beneficial, as the economic burden of BM can increase in later lines of therapy if patients discontinue initial treatment due to disease progression and/or toxicity, potentially resulting in additional HCRU and costs. A previous report of ALK inhibitor (crizotinib) use in any-line setting showed that monthly costs and HCRU were 4- and 6-fold higher, respectively, for patients with NSCLC with BM compared to those with no BM. 27 Among newly diagnosed patients with ALK+ NSCLC who received an ALK inhibitor (crizotinib, ceritinib, or alectinib) at any time following diagnosis, PPPM costs were 1.3-fold higher, with more frequent inpatient stays and radiology imaging procedures for patients with BM compared to no BM. 29 An analysis of post-crizotinib use of ALK inhibitors ceritinib or alectinib in the second-line setting demonstrated that mean PPPM total costs for patients with BM were 1.37 times as high as those for patients without BM, and mean PPPM outpatients visits and inpatient hospitalization stays were higher for patients with BM than with no BM. 28
Information on the drivers of costs in ALK+ NSCLC is scarce. The present study identified medications (15.8%), radiation therapy (8.4%) and other diagnostic procedures (6.8%) as the main cost drivers among patients with no BM, and medications (19.1%), radiation therapy (15.1%) and magnetic resonance imaging (10.2%) as the main cost drivers among patients with BM. Similar findings were reported in a retrospective analysis of adult patients with ALK+ NSCLC treated with ceritinib or alectinib as second-line ALK inhibitors (Table 2). Costs for patients with no BM were driven by medications (11.8%), diagnostic radiology (7.1%) and radiation therapy (6.0%), and costs for patients with BM were driven by radiation therapy (16.9%), medications (11.2%) and radiographic procedures (12.5%). 28 In a US claims database study of patients with ALK+ NSCLC, outpatient services, including radiation therapy costs and radiology imaging costs, were 3.7 and 1.5 times higher, respectively, for patients with BM compared to patients with no BM, with approximately one-third of the difference in radiology imaging costs attributable to head- and brain-related radiology imaging. 29 An understanding of factors that drive healthcare costs may guide more efficient resource allocation and offers an opportunity to effectively manage costs in ALK+ NSCLC.
Limitations
This study was subject to limitations common to retrospective database studies, including the possibility of inaccurate recording of patients’ diagnoses, incomplete records on clinical outcomes, lack of information on ALK rearrangement status, underestimation of comorbidities, and inaccurate recording of drug use. Patients who developed BM after the index date may have been misclassified to the no BM group, potentially attenuating the cost burden of BM. The study included a relatively large and racially diverse population of patients with commercial healthcare coverage, such that the findings may not be generalizable to patients with other or no health insurance coverage. Consistent with other reports on the economic burden and outcomes among patients with ALK+ NSCLC, the present study used ICD-9 or ICD-10 diagnosis codes for lung cancer with a prescription fill for an ALK inhibitor to identify ALK+ NSCLC.28,29,33 This may have resulted in the misclassification of patients with ALK+ NSCLC, as no ICD-9 or ICD-10 diagnosis codes exist to directly identify patients with ALK rearrangements.
Conclusions
The findings of the present study show that BM posed a substantial economic burden for patients with ALK+ NSCLC who received first-line ALK inhibitors. PPPM total costs for patients with BM were 11% higher than those for patients with no BM. Cost drivers among patients with BM included medications, radiation therapy, and magnetic resonance imaging. Patients with BM had greater HCRU than patients with no BM. Early treatment of patients with BM using therapies with intracranial efficacy that have the potential to improve patient outcomes may reduce the economic burden of ALK+ NSCLC.
Supplemental Material
sj-docx-1-opp-10.1177_10781552221126174 - Supplemental material for Economic burden in patients with anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC), with or without brain metastases, receiving first-line ALK inhibitors
Supplemental material, sj-docx-1-opp-10.1177_10781552221126174 for Economic burden in patients with anaplastic lymphoma kinase (ALK)-positive non-small cell lung cancer (NSCLC), with or without brain metastases, receiving first-line ALK inhibitors by Yanyu Wu, Kaili Ren, Yin Wan and Huamao M Lin in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgments
Medical writing support was provided by Jane Kondejewski, PhD of SNELL Medical Communication, Inc. and funded by Takeda Development Center Americas, Inc.
Authors’ Note
Conceptualization: Yanyu Wu, Mark Lin; Data curation: Kaili Ren; Formal analysis: All; Validation: Yanyu Wu, Kaili Ren; and Writing – review & editing: All.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yanyu Wu, Kaili Ren, Yin Wan, and Mark Lin are employees of Takeda Development Center Americas, Inc. and may own stock.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Takeda Development Center Americas, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
