Abstract
Introduction
Closed-system transfer devices (CSTDs) were introduced into clinical practice to protect healthcare practitioners (HCPs) from exposure to hazardous drugs. However, ambiguous guidelines have led to confusion as to when CSTD use is required, as institutes are instructed to maintain their own hazardous drug lists and determine the appropriate level of personal protective equipment for their staff. This study seeks to understand the current use of CSTDs by Canadian oncology HCPs, the influence of various stakeholders on their use and the challenges faced by HCPs surrounding the use of these medical devices.
Methods
The researchers compiled a set of questions to inform on the current use of CSTDs in clinical practice and administered an online survey to oncology HCPs across Canada.
Results
The results indicate that though CSTD use is common in Canadian oncology practice settings, there is variation in the extent of the use of these devices across provinces and with which products these devices are used. The survey results also show that the top challenges with the use of CSTDs include cost, lack of information on the compatibility of a CSTD with a drug product, and CSTD impact on drug quality. Many respondents are aligned that regulatory bodies are more likely to influence the use of CSTDs with specific drug products than drug manufacturers.
Conclusion
Guidelines for the application of CSTDs in clinical practice vary and are often ambiguous. Regulatory bodies are uniquely positioned to provide healthcare institutions with more clarity on when CSTD use is appropriate.
Introduction
Closed-system transfer device (CSTD) use is widely promoted as an effective method to reduce healthcare practitioner (HCP) exposure to hazardous drugs. Yet in recent years, the definition of hazardous drugs as used in clinical practice is becoming blurred, the use of these devices is becoming more ubiquitous, and patient outcomes may subsequently be compromised.
CSTDs are medical devices designed to prevent hazardous drugs from being released into the environment while simultaneously prohibiting the introduction of environmental contaminants into the system. 1 Though CSTD use was initially intended to protect HCPs from exposure during the preparation and administration of hazardous drugs, ambiguous guidelines have led to confusion as to when CSTD use is required. 2 The United States Pharmacopeia (USP) General Chapter <800> Hazardous Drugs—Handling in Healthcare Settings, mandates that entities that handle hazardous drugs, as defined by the National Institute of Occupational Safety and Health (NIOSH), 3 incorporate the standards of the chapter into their occupational safety plan, including the use of CSTDs when the dosage form allows. 4 Though USP <800> is currently informational, USP states the chapter is applicable “where there is a risk of exposure to patients, healthcare workers, and the environment”, 4 and encourages the widespread adoption of the recommendations in that chapter, including the use of CSTDs.
Despite numerous studies, the evidence that the use of CSTDs reduces exposure to hazardous drugs over traditional personal protective equipment (PPE) alone is inconclusive, 5 especially for biologics as they do not pose the same risk of occupational exposure to healthcare workers as traditional cytotoxic drugs. Moreover, there is a growing body of evidence that suggests the use of CSTDs with biologics has the potential to negatively impact product quality and patient outcomes.6–9
First, CSTDs can leave a larger residual volume in the various components of the device system relative to a traditional needle and syringe, ranging from 0.7 to 1.0 mL, making the delivery of the total intended dose from small vials a challenge.6,7,10–13 Furthermore, though CSTDs are purported to provide HCPs a protective barrier from hazardous drugs, a lack of impact on the quality of biologics is yet to be demonstrated and therefore cannot be negated.6,7 In addition, some CSTDs can introduce extrinsic particles into the drug product vials; particles resembling rubber fragments not visible to the naked eye upon CSTD use have been detected by micro-flow imaging. 13 The introduction of extrinsic particles into sterile injectable drug products that are subsequently administered to patients can lead to serious adverse events, 14 including fatal pulmonary emboli. 15 Another study found particles resembling silicone oil, a common lubricant used in CSTDs, 6 which may lead to protein aggregation and therefore immune responses to biologics ultimately minimizing response to treatment.16,17 Finally, there have been several reports that these devices can be used to extend the beyond-use-date (BUD) of single-dose vials which are based on studies demonstrating that CSTD use can reduce, or even prevent, microbial ingress into the vial.18–21
North America currently has the greatest global consumption of CSTDs by occupying 89.37% of the total market value; the worldwide market for CSTDs is predicted to almost double by 2026. 22 As rapid market growth continues, it is imperative that the benefits and risks of using these devices are understood and that the risks are mitigated, particularly those impacting patient safety.
This study seeks to understand the current use of CSTDs in Canada, the factors influencing their use and the challenges faced by HCPs surrounding the use of these medical devices. This study also seeks to identify the level of influence various stakeholders have on CSTD use within healthcare institutions across Canada, enabling targeted discussions to facilitate change.
Methods
The researchers compiled a set of questions that would provide information on the current use of CSTDs in clinical practice across Canada. The survey consisted of 6 screening questions to determine eligibility (participants were ineligible if they very infrequently or never used CSTDs when preparing or administering hazardous drugs), followed by up to 29 survey questions. These questions included both open-ended and closed-ended questions (including nominal, Likert scale, and rating scale).
Research ethics approval was obtained through the University of Toronto Health Sciences Research Ethics Board (REB). The survey questions were entered into Google Forms which was then individually piloted by a total of 5 pharmacists to ensure functionality of the online survey and to quality check the content of the questions to ensure clinical sensibility and face validity.
Upon REB approval, the survey questions were submitted to Sea to Sky Meeting and Association Management, who approved the contents of the survey and provided the survey to the Canadian Association of Pharmacy in Oncology’s (CAPhO) research committee. CAPhO’s research committee approved the survey for distribution to its members who had agreed to be contacted for research initiatives. Respondents were provided information about the study and were required to provide informed consent through the online form to be able to initiate the survey. Data collection took place between April 9, 2020 and April 27, 2020. A total of 22 responses were recorded of which 18 were included in the data analysis. Four responses were not included for analysis as 3 respondents did not meet the inclusion criteria of experience with CSTDs (i.e. rarely or never use CSTDs), and another was excluded as their responses were recorded twice.
The data were exported into a password-protected excel file and the analysis was performed using cross-tabulation to evaluate relationships between the variables.
Results
Data were collected and analyzed from 18 HCPs in oncology across 6 provinces in Canada. The respondents consisted of pharmacists and pharmacy technicians with varying degrees of experience from 4 practice settings (Table 1). The data collected and analyzed were grouped into 3 buckets: current use of CSTDs, factors influencing the use of CSTDs, and remaining challenges and unmet needs.
Study population demographics.
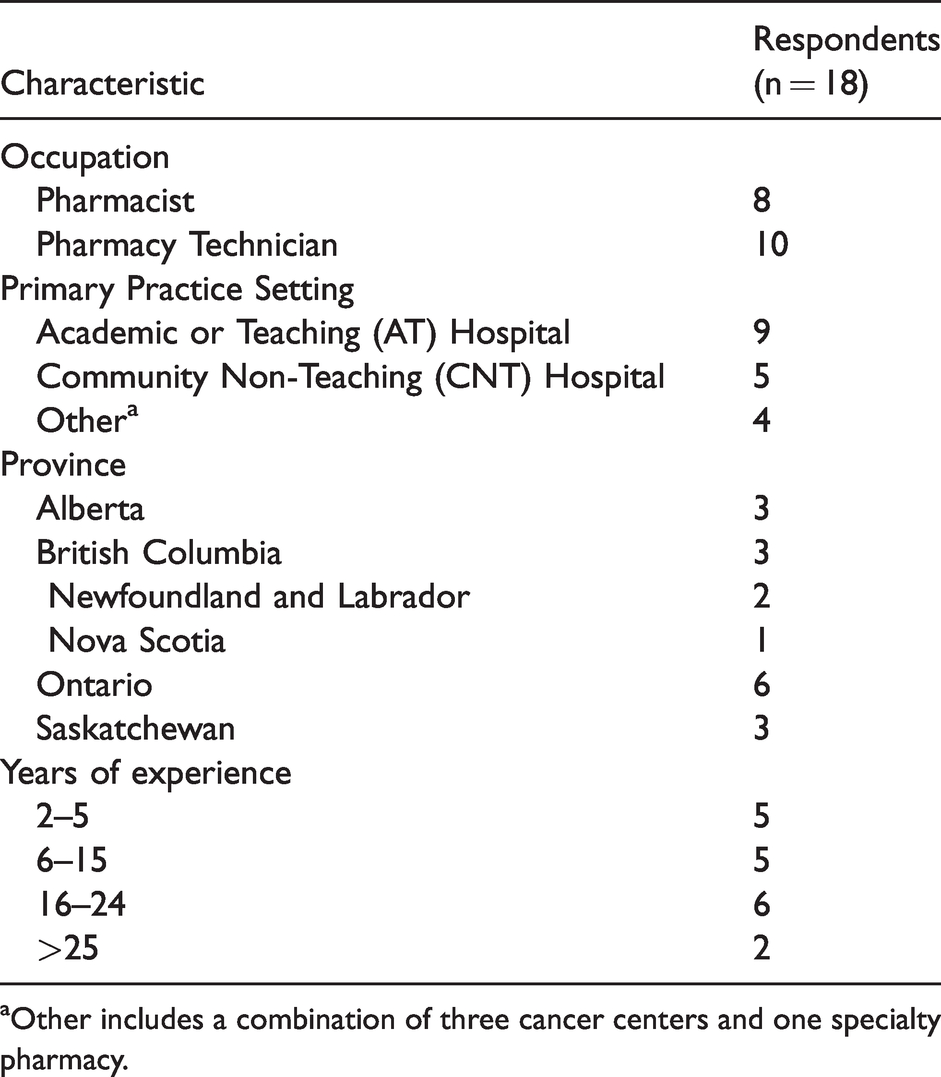
aOther includes a combination of three cancer centers and one specialty pharmacy.
Current use of CSTDs
All respondents included in the results were frequent users of CSTDs. More than three quarters of respondents used CSTDs multiple times per day, and all respondents used CSTDs at least once daily. All respondents stated that they use CSTDs with hazardous drugs either every time or the majority of the time. A broad overview of the current use of CSTDs is summarized in Table 2.
Current use of CSTDs.
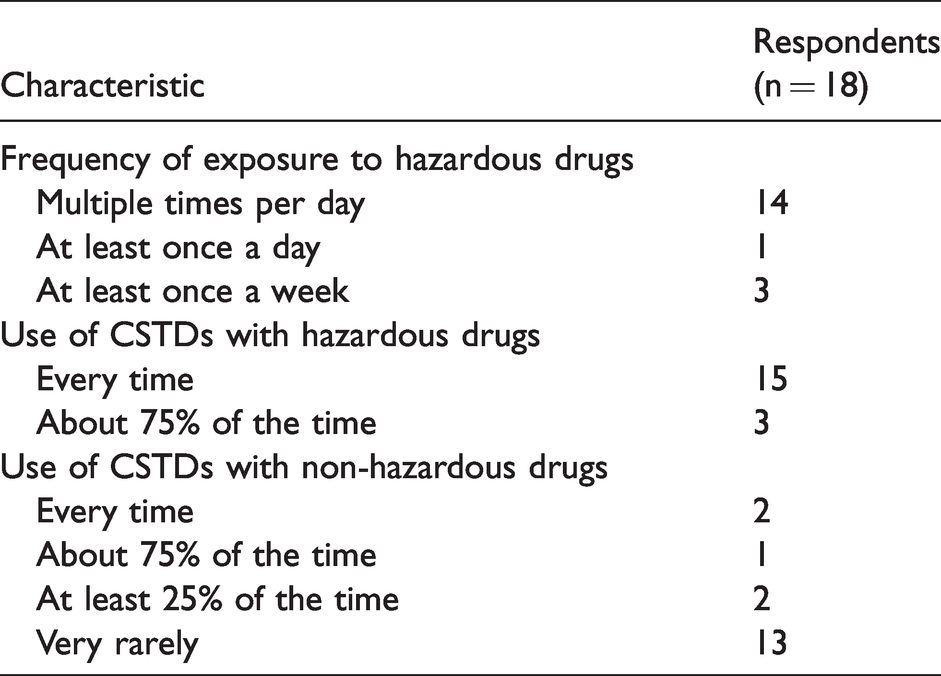
Though the majority of respondents stated that they use CSTDs with hazardous drugs either every time (83%) or the majority of the time (17%), a quarter of respondents stated that they use CSTDs with non-hazardous drugs at least some of the time. A total of 72% respondents reported very rarely using CSTDs with non-hazardous drugs. One third of respondents working in academic or teaching (AT) hospitals reported using CSTDs 75–100% of the time with non-hazardous drugs, and 40% of respondents working in community or non-teaching (CNT) hospitals reported using CSTDs with non-hazardous drugs at least 25% of the time. Respondents from other practice sites very rarely used CSTDs with non-hazardous drugs (Figure 1).
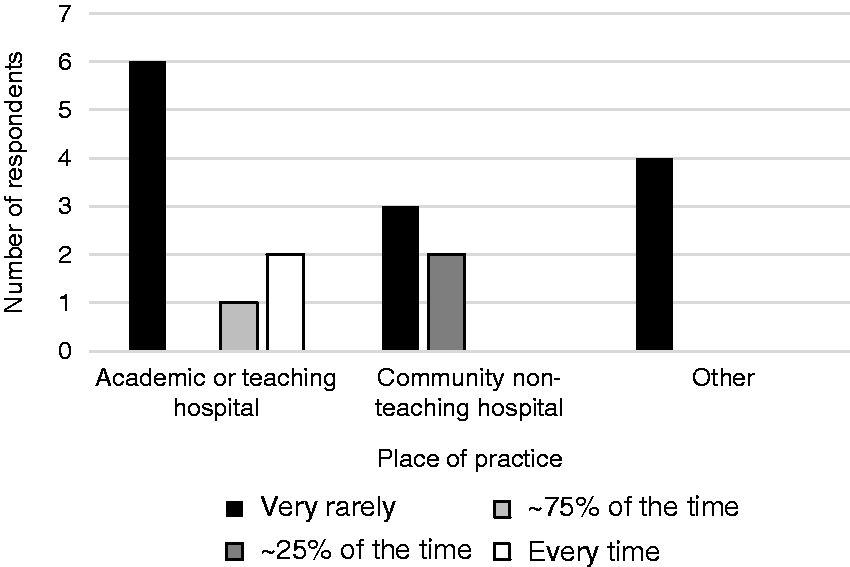
Frequency of CSTD use with non-hazardous drugs by practice setting. ‘Other’ includes a combination of three cancer centers and one specialty pharmacy.
The majority of respondents with 15 years or less of experience (60%) stated that not all oncology biologics require the use of CSTDs. Contrarily, only a minority of respondents with greater than 15 years of experience (25%) stated that not all oncology biologics require the use of CSTDs. When assessed by practice setting, a third of respondents from AT hospitals (33%) stated that not all oncology biologics require the use of CSTDs, while the majority of respondents from CNT hospitals (60%) and half of the respondents from other practice sites (50%) do not believe a CSTD is required for all oncology biologics. Furthermore, all respondents from AT hospitals stated that all hazardous drugs require the use of CSTDs, while the majority of respondents from CNT hospitals (80%) and other practice sites (75%) stated that all hazardous drugs required CSTDs. Both respondents who stated that all hazardous drugs do not require a CSTD practice in Ontario.
A total of 72% of respondents reported that they use a CSTD for investigational drugs in clinical trials. Of these, only 1 respondent confirmed that if a CSTD is used for preparation or administration, and this is not explicitly stated in the protocol or package insert, the drug manufacturer would always be notified (Figure 2). Furthermore, single respondents also reported that a drug manufacturer would rarely or sometimes be notified. The majority of respondents stated that either they did not know (31%) or that the drug manufacturers would never be notified if a CSTD was used (50%).
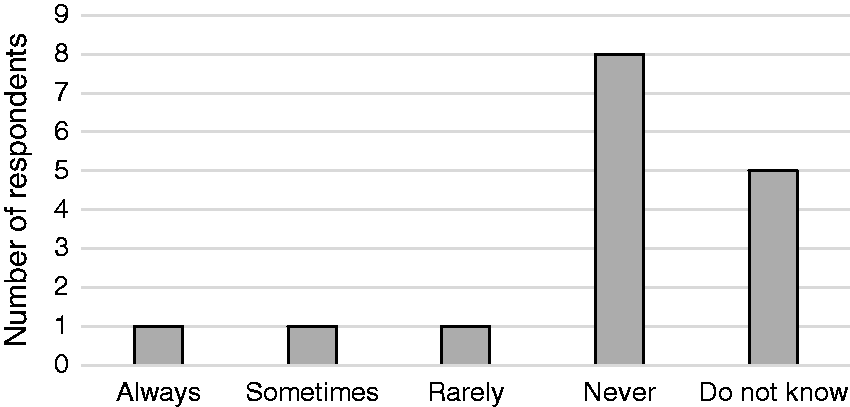
Survey responses evaluating if a drug manufacturer would be notified if a CSTD was used for drug preparation or administration if its use was not explicitly stated in the protocol or package insert.
Factors influencing the use of CSTDs
A majority of total respondents stated that their organization references the NIOSH List of Hazardous Drugs either directly or indirectly (67%) to classify drugs as hazardous; while others stated they used a combination of NIOSH and internal guidelines (11.1%), internal guidelines alone (11.1%) or provincial/other sources (5%). Of those, two thirds either stated their institute would certainly or probably cease using CSTDs with certain biologics if those biologics were to be removed from the NIOSH List of Hazardous Drugs (Figure 3).
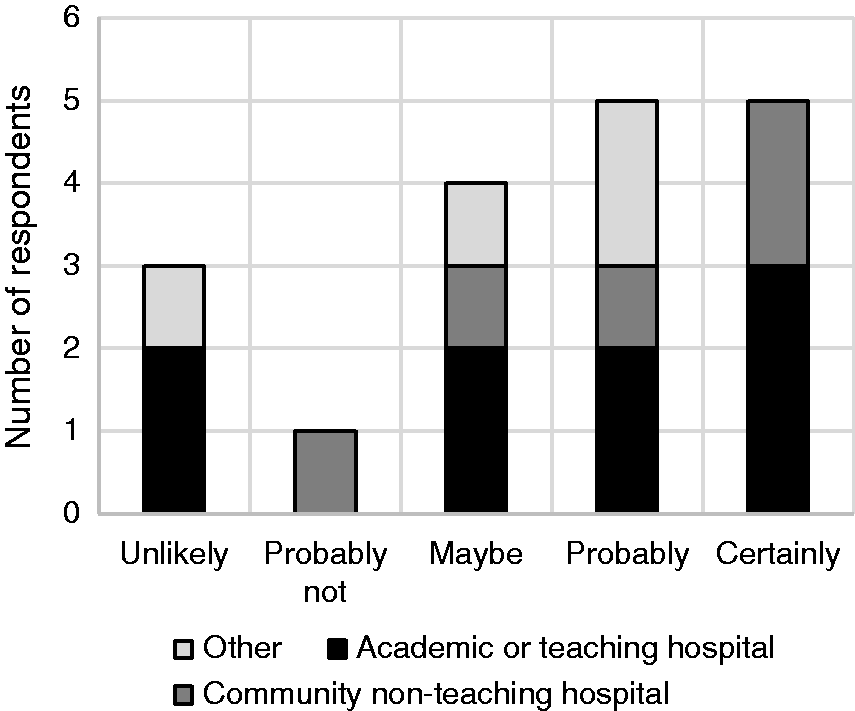
Likelihood of institutions ceasing to use CSTDs with specific biologics, if those biologics were removed from the NIOSH List of Hazardous Drugs.
The majority of total respondents (72%) agreed or strongly agreed that they follow the administration instructions in the drug product label. Pharmacy technicians (particularly those who have 5 or less years of experience) were the only respondents who strongly disagreed that they follow administration instructions in the label. The respondents were then asked if they would cease using CSTDs with a particular drug product if the drug manufacturer's label recommended the use of needle and syringe. In response, over 94% of respondents indicated they would not change the way they currently practice. However, when asked if they were likely to cease using CSTDs with certain drug products if advised directly by a Health Authority (e.g. Health Canada and FDA), many respondents indicated their institute may change its practice. The impact of various stakeholders and organizations influencing the use of CSTDs is summarized in Figure 4.
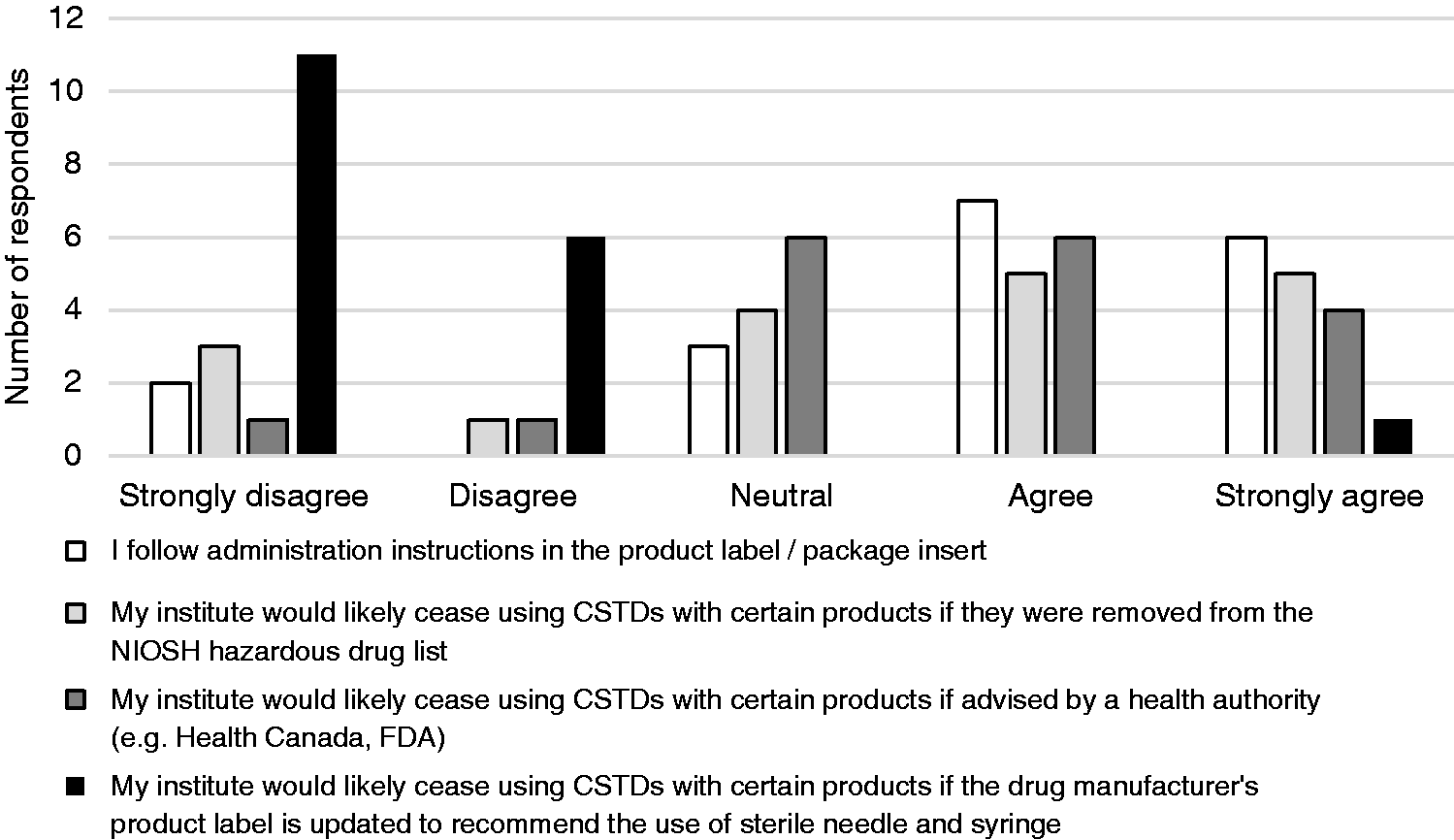
Stakeholder influence on CSTD use. Responses were mapped to comparable categories on a 5-point Likert scale to enable appropriate comparisons to be made between current practice and stakeholder influence.
Respondents were asked to select the top three drivers for their organization’s preference when selecting a CSTD. The top three drivers for choosing a specific CSTD were: ease of use (59%), reliable product (53%), and evidence supporting quality (e.g. lack of exposure) (41%). Only 3 respondents commented in the open-text field, of which 2 explicitly mentioned a particular CSTD by name. One stated “We have used [CSTD name] for 10 years and they were one of the first ones on the market” and the other stated “Stability [CSTD name] provides ([CSTD manufacturer] says the drugs are good for 28 days)”.
Remaining challenges
The top remaining challenges identified by respondents surrounding the use of CSTDs included CSTD cost (75%), lack of information on the compatibility of a CSTD with the drug product (70%), CSTD impact on drug quality (62%) and that CSTDs are time-consuming to use (47%). A detailed list of remaining issues or unmet needs associated with the use of CSTDs, including a categorization of severity of each issue, is illustrated in Figure 5.
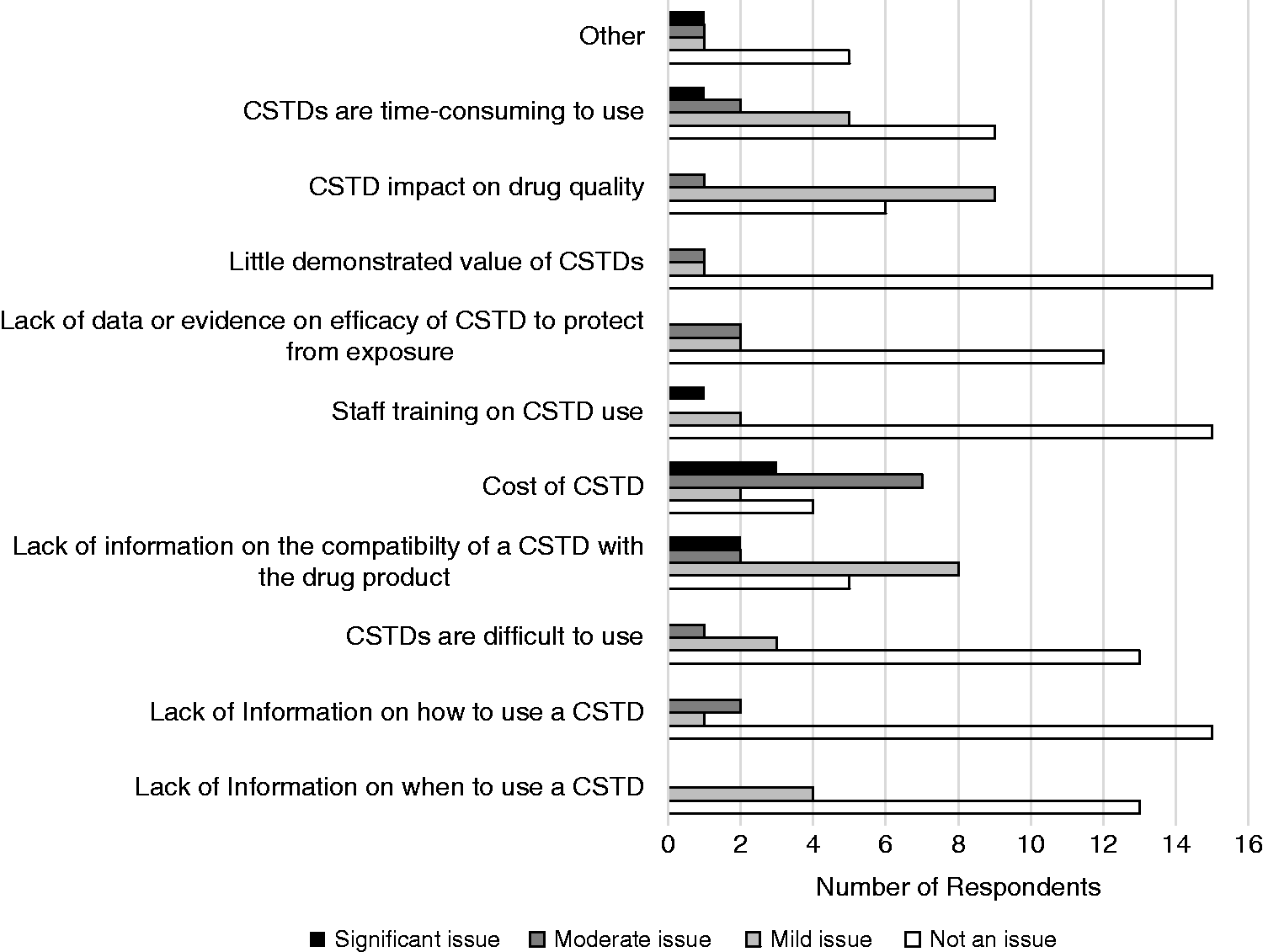
Remaining issues or unmet needs associated with the use of CSTDs.
Half of the respondents reported that their institute consistently verifies that the CSTD is compatible with that drug before using that CSTD. Only 28% checked device manufacturer specifications for compatibility and 17% checked with the drug manufacturer. The remainder either only verified compatibility for clinical trial drugs, were unsure if their institute verified compatibility, or admitted to not verifying compatibility with each drug they administered (Figure 6).
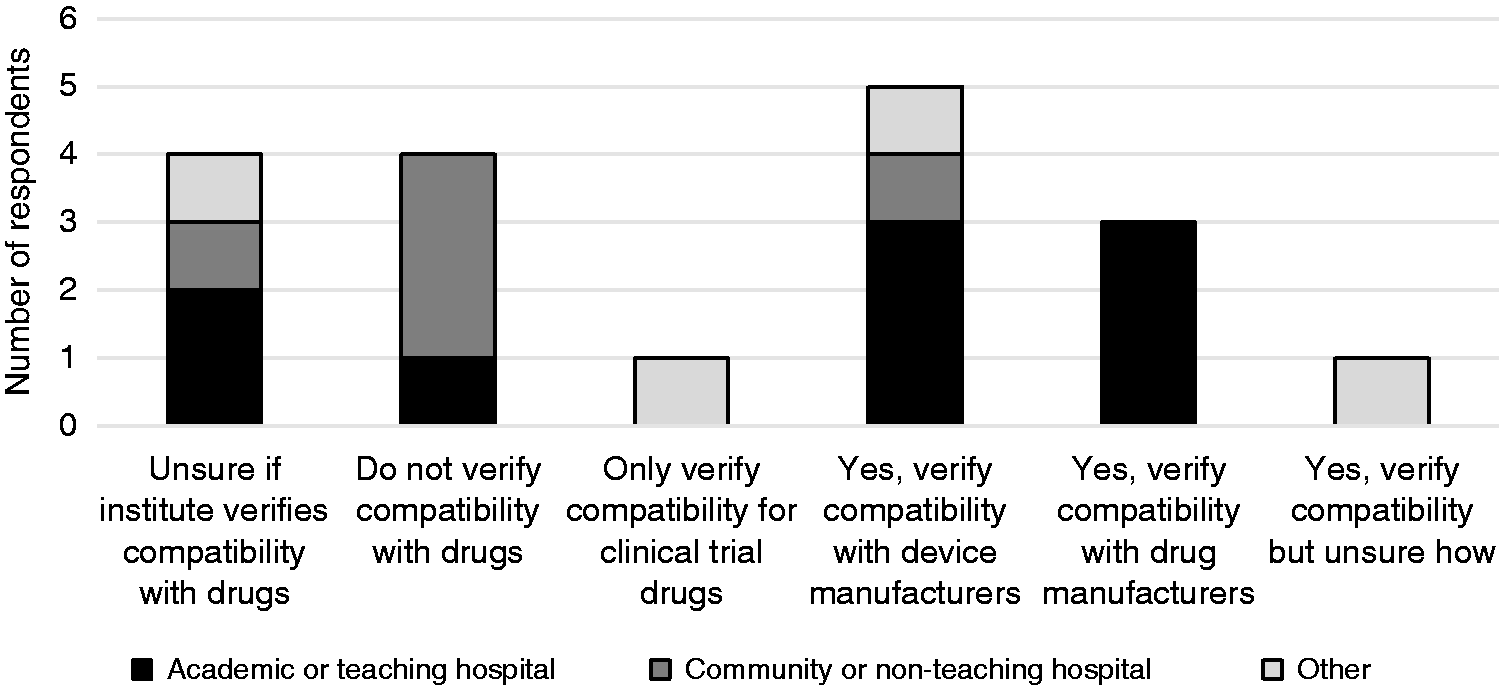
Healthcare institution verification of CSTD compatibility with drugs products by practice setting. ‘Other’ includes a combination of 3 cancer centers and 1 specialty pharmacy.
Fifty-six percent of HCPs from AT hospitals stated they would check for compatibility data, whereas only one respondent (20%) from CNT hospitals stated they would verify for compatibility information. All respondents from the Western provinces (British Columbia and Alberta) checked either device or drug manufacturer references to verify compatibility, whereas only one respondent (17%) from Ontario confirmed that they checked for drug compatibility with CSTDs. All respondents from Saskatchewan and the majority of respondents from Ontario (83%) admitted to not verifying CSTD compatibility with each drug they either administer, or were unsure if their institute verified compatibility.
Finally, a strong majority of respondents (89%) either agreed or strongly agreed that device manufacturers should be accountable for ensuring compatibility with drug products. Only two-thirds of respondents (67%) either agreed or strongly agreed that drug manufacturers should be accountable for ensuring device compatibility with drug products (Figure 7).
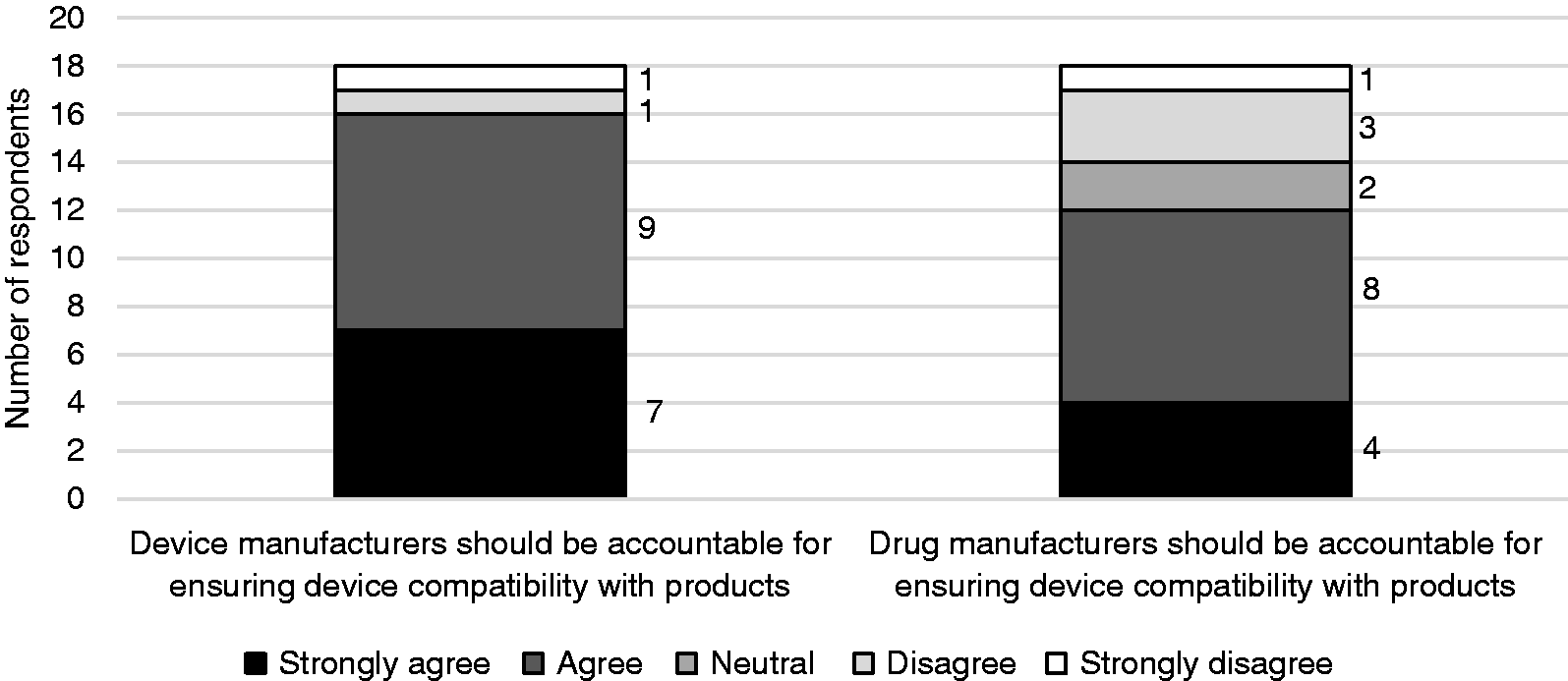
Perception of healthcare workers on the accountability of device and drug manufacturers to ensure device compatibility with products.
Discussion
Although CSTDs were developed to protect HCPs from exposure to hazardous drugs, the results of this survey indicate that some institutes, at least some of the time, are using these devices with non-hazardous drugs. This study also indicates that there is no uniform classification of hazardous drugs across institutes, as many resources are referenced when creating institutional hazardous drug lists, including the NIOSH List of Hazardous Drugs, internal guidelines or provincial/other sources; nor is there a consistent method of identifying which drugs require the use of CSTDs. These findings are consistent with Mathias et al., 2019, which showed that though many provinces reference NIOSH in the guidance for handling of hazardous drugs, there is variability across the country. 23
The use of biologics in oncology gained traction a few decades ago and continues to evolve. Biologics are becoming increasingly common in pharmacy practice as well as in pharmacy school curriculums as their mechanisms and utility are becoming better understood. HCPs who have graduated more recently may therefore have a greater understanding of the inherent properties of these large molecules, which in general are minimally hazardous through routine occupational exposures due to negligible bioavailability through oral inhalation or dermal absorption.24,25 This may also be why they are less likely to use CSTDs with all oncology biologics than their counterparts with more than 15 years of experience.
Cancer centers appear to be less likely to use CSTDs with non-hazardous drugs compared to both CNT hospitals and AT hospitals, though respondents from CNT hospitals were less likely to state that all oncology biologics require the use of CSTDs than AT hospitals. This may indicate that AT hospitals may be more likely to employ overly precautionary measures that are disproportionate to the inherent risks posed by biologics to protect healthcare workers, or that cancer centers better understand the risks of occupational exposures from oncology biologics as they specialize in treatment regimens using both traditional chemotherapy agents and oncology biologics.
Regardless of the reason for the use of CSTDs with oncology biologics, drug manufacturers are often unaware that these devices are being used with their products. This is especially concerning when these devices are used during clinical trials, as their potential to impact product quality and patient safety may ultimately impact study outcomes. Only one respondent stated that a drug manufacturer would always be notified if CSTDs are used in clinical trials, whereas over three quarters either responded that drug manufacturers would either rarely or never be notified if CSTDs are used. Therefore, if patients enrolled in clinical trials experience adverse effects and the trial sponsor is not aware that CSTDs were used, the adverse events would be attributed to the drug product, without considering the potential risk associated with CSTDs, such as the introduction of extrinsic particles which are then inadvertently administered to patients. Similarly, if the sponsor is not aware that a CSTD is being used, a lack of efficacy due to patient under-dosing as a result of large residual volumes in the device, may be overlooked. For example, if an oncology biologic product’s maintenance dose is 420 mg (420 mg/16 mL vial) every three weeks and residual volumes of up to 1 mL can be lost within select CSTDs,6,7,10–12 CSTD use could result in a loss of up to 26 mg of drug per dose at each visit. Therefore, patients treated with this regimen may experience progression of their disease earlier if they were treated at institutes that use CSTDs to prepare this product compared to those treated in institutes that use a traditional needle and syringe.
This survey demonstrates many stakeholders play a role in influencing the use of CSTDs across healthcare institutions, which may ultimately affect patient safety and drug product quality. Although almost three quarters of respondents strongly agreed or agreed that they follow the instructions in drug product labels, when asked if they would cease using CSTDs with a particular drug product if the label changed to specify the use of needle and syringe, all but one respondent were unlikely to modify their practice. However, when asked if they would cease using CSTDs with a particular drug product if advised by NIOSH or a Health Authority, respondents were much more likely to indicate that they would change the way they use CSTDs. At the time this survey was distributed, the NIOSH List of Hazardous Drugs included monoclonal antibody antineoplastics; however, NIOSH has since released an updated List of Hazardous Drugs for public comment. This update removed the monoclonal antibody pertuzumab from the NIOSH List of Hazardous Drugs, and removed trastuzumab and bevacizumab for consideration from the list because their molecular properties limit the potential for adverse effects through occupational exposures. 26 It remains unclear whether these amendments to the NIOSH List of Hazardous Drugs will reduce the use of CSTDs with these monoclonal antibodies in clinical practice, or if the preparation of drugs for conditions treated by the monoclonal antibodies removed from the NIOSH List of Hazardous Drugs will transition from using CSTDs to using sterile needle and syringe. Although respondents generally responded favorably to limiting the use of CSTDs with drugs removed from the NIOSH List of Hazardous Drugs, this alone is unlikely to facilitate the appropriate use of CSTDs with biologics across Canada. NAPRA encourages institutions to develop their own lists of hazardous drugs by referencing the NIOSH List of Hazardous Drugs, 27 which does not guarantee that institutions will mirror these changes in their own lists. However, respondents also responded favorably to changing the way they use CSTDs if advised by Health Authorities or NAPRA. Health Canada and NAPRA are key stakeholders within Canada to enable provincial agencies to mirror these changes and align in their guidance documents to facilitate the appropriate use of CSTDs with oncology biologics in clinical practice at an institutional level. This is because many provincial cancer organizations and associations (e.g. Cancer Care Ontario, 28 Alberta Health Service, 29 British Columbia Cancer, 30 etc.) rely on and make reference to USP, NIOSH, and NAPRA to establish their own definitions and criteria for hazardous drugs and to establish when CSTD use is required. NAPRA itself references USP and NIOSH throughout its Model Standards for Pharmacy Compounding of Hazardous Sterile Preparations. 27 USP, NIOSH, and NAPRA are therefore uniquely positioned to influence the appropriate use of CSTDs across North America through collaborations with manufacturers, Health Authorities, as well as state and provincial bodies.
Beyond stakeholder influence, there are also characteristics of CSTDs that HCPs take into consideration when selecting a particular CSTD and employing it in clinical practice. These include ease of use, reliability of the product, and evidence of quality (i.e. lack of exposure). Furthermore, one respondent mentioned stability as a factor for choosing a specific CSTD, stating “Stability [CSTD name] provides ([CSTD manufacturer] says the drugs are good for 28 days)”. This is in line with reports that these devices can be used to extend the beyond-use-date (BUD) of single-dose vials.18–21 United States Pharmacopeia (USP) General Chapter <797> states that a single-use vial may be used up to 6 hours after initial needle puncture. 31 This practice is echoed by NAPRA, 27 which references USP <797> in support of this claim. This practice of use within 6 hours exists as compounded medications are at high risk of bacterial contamination; the rate of bacterial growth is thought to increase considerably 6 hours after contamination onset. 27 Many healthcare institutions leverage the claims made by these studies that CSTDs can extend the BUD of single dose vials if maintained by facility level sterility testing as a cost saving measure.32,33 Unlike stability testing performed by drug manufacturers which may extend the in-use stability claim, studies that only test for sterility do not take into account the potential for this practice to negatively impact the quality of these biologics. 18 Furthermore, the majority of the products in these studies are not truly deemed hazardous by NIOSH (i.e. alemtuzumab, bevacizumab, and infliximab) which may lead HCPs to conclude that these products are hazardous and should be prepared with CSTDs despite minimal risk of clinically meaningful exposures during preparation.18,32,34–36 If the stability of a drug is limited, the ability to extend the BUD will be limited as well, 21 regardless of whether sterility is maintained.
The lack of compatibility information was identified as one of the largest remaining challenges associated with the use of CSTDs. However, these results beg the question – What compatibility parameters are and should be verified? Despite the compatibility claims made by device manufacturers, compatibility testing criteria has not been defined, nor is it required by regulators approving these devices for sale. To be approved for use in Canada, CSTDs must be issued an ISO 13485 certificate. 37 This certificate serves as evidence of Quality Management System compliance, yet does not require CSTD manufacturers to demonstrate compatibility with biologics through device performance testing. Despite this, some CSTD manufacturers still claim compatibility with a variety of drug products. For example, one CSTD manufacturer claims compatibility with 108 drug products. 38 However, compatibility testing was only performed on slightly over 10% of the drug products with which compatibility was claimed, 39 and the compatibility testing suite only covered three parameters: functional integrity of the device, limited stability and a measure of plastic migration into the drug product; none of which are a comprehensive evaluation (e.g. stability indicating methods, hold up volume assessment) of the potential impact these devices are having on biologics. Device compatibility with each drug product should be established prior to use in clinical practice in order to confidently treat patients without safety or product quality concerns. Though respondents were almost unanimous that the onus should be on the device manufacturer to ensure device compatibility with drug products, the majority also thought that drug manufacturers should share the responsibility. Currently there is a regulatory gap making it unclear who is responsible for compatibility testing of these devices with biologics and it is more than likely that since their use is recommended by regulatory bodies, that consumers assume they have been adequately tested. Furthermore, the onus should not be on the HCP or institutes to verify compatibility, as this evidence should be readily available for CSTDs that claim compatibility with specific drug products to the institutes treating patients. Despite relying on manufacturers to provide compatibility data, institutes are faced with uncertainty due to ill-defined testing criteria and extrapolations made by device manufacturers.
Although CSTDs are becoming increasingly popular, the use of these devices is also becoming increasingly controversial given that USP < 797> and USP < 800> may not be applicable to most oncology practices that prepare and administer oncology drugs, as many of these products are reconstituted, not compounded. 2 The results of this survey demonstrate the lack of confidence HCPs have in CSTD compatibility with drug products due to a scarcity of reliable evidence. The perception of clinicians in this study is consistent with the results of a CADTH report and Cochrane review stating that the evidence for CSTDs that currently exists is limited, conflicting, inconclusive, and of high risk of bias.33,40 Overall, more high quality research is needed in order to understand how these devices are used in clinical practice in order to prompt stakeholders to align and provide institutions with consistent and explicit messaging on which drugs require the use of CSTDs based on comprehensive risk-benefit analyses.
Limitations
This study has some potential limitations. Only surveying oncology HCPs that are members of CAPhO may have introduced selection bias and the individuals who responded to the survey may not be representative of a random sample. Although response bias is also possible, the researchers attempted to mitigate this by restricting the amount of information provided to respondents prior to their participation. Furthermore, the sample size of the study is small and limited to Canadian healthcare workers in oncology, and therefore may not be generalizable to the general population. The small sample size also limits the ability to identify any significant relationships from the data collected, yet provides a foundation for future research into the topic. Despite these potential limitations, this study forms the basis for understanding stakeholder influences and HCP perceptions of the challenges associated with CSTD use.
Conclusion
The definition of hazardous drugs and the use of CSTDs not only varies across regions, but also within regions based on practice setting, years of experience, and stakeholder influence. The data from this survey suggests there is not only a lack of clarity on CSTD compatibility with oncology biologics, but also a lack of control drug manufacturers have as a single stakeholder to protect the quality of their products and the safety of the patients who use them. Multiple stakeholders including drug manufacturers, device manufacturers, FDA, Health Canada, NIOSH, USP, and NAPRA must come together to optimize outcomes for cancer patients across Canada and the United States, and to set the stage for best practices globally. Health Authorities are encouraged to seek more high quality evidence from manufacturers of CSTDs by implementing more stringent compatibility testing criteria and establishing performance standards to mitigate the risks to patient safety and product quality, prior to granting regulatory approval and integrating these devices into clinical practice. Other stakeholders such as NIOSH, USP, and NAPRA are uniquely positioned to facilitate change, and should aim to provide institutes with more clarity on identifying which drugs do and do not require the use of CSTDs, as well as leveraging evidence-based approaches to establish appropriate practices for BUDs for drugs when used with CSTDs. While protecting HCPs is a common goal of industry partners and regulatory authorities, it should be accomplished using evidence-based practice and not at the expense of patient outcomes.
Footnotes
Acknowledgements
The authors would like to thank Kathy Hanley, Ivy Chang, and Camellia Zamiri of Genentech for their contributions to the original survey and/or feedback on the adapted survey used in this study; Dr. David Dubins from the University of Toronto, Leslie Dan Faculty of Pharmacy for his support in gaining Research Ethics Board approval to conduct the survey and for his feedback on the survey and manuscript; and Tom McFarlane, Chair of the CAPhO research committee in facilitating the distribution of the survey to CAPhO members. In addition, the authors would like to thank Hoffmann-La Roche Pharmacy Residents Kiana Gozda, Jekaterina Davydova, Katie Lee, Arash Rezaeian, and Munaza Saleem for their review and feedback of the survey prior to its distribution. Finally, the authors would like to thank Camellia Zamiri, Bea Lavery, Lisa Chartrand, and Chi-Sing Nip from Genentech/Hoffmann-La Roche for their comments and feedback on the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
