Abstract
The primary purpose of this study was to assess the prevalence and the antimicrobial susceptibility rates of carbapenemase-producing Enterobacterales (CPE) in a German University Hospital during a 5-year period. From January 2020 to December 2024, all Enterobacterales detected were molecularly investigated for carbapenemases in every case of elevated minimum inhibitory concentration of ertapenem, meropenem, or imipenem. Subsequently, CPE were tested for susceptibility to reserve antibiotics. Overall, 101 CPE were identified. Most of the CPE strains harbored only one carbapenemase gene, such as blaOXA-48 (n = 32, 31.6%), blaNDM (n = 27, 26.7%), and blaVIM (n = 14, 13.8%). The annual number of CPE detected increased during the observation period (2020, n = 5; 2021, n = 5; 2022, n = 24; 2023, n = 29; 2024, n = 38). We also observed a progressive rise of the proportion of CPE harboring a metallo-β-lactamase gene such as blaNDM or blaVIM (2020, 0.0%; 2021, 0.0%; 2022, 50.0%; 2023, 55.2%; 2024, 65.8%). Regarding the antimicrobial susceptibility, only 3.3% of all CPE isolates tested showed resistance to aztreonam/avibactam. In contrast, the resistance rates for cefiderocol and ceftazidime/avibactam amounted respectively to 12.8% and 53.9%. The increasing annual number of CPE at our hospital is associated with a rise of the proportion of metallo-β-lactamase-producing strains. The newly available antibiotic aztreonam/avibactam showed promising in vitro activity against CPE.
Introduction
Enterobacterales are part of the normal flora of the human gastrointestinal tract but may also cause a variety of infections including urinary tract infections, respiratory tract infections, and bloodstream infections (BSIs). 1
Over the past years, the detection of carbapenem-resistant Enterobacterales has increased worldwide. 2 In Europe, the carbapenem resistance among Klebsiella pneumoniae strains is of particular concern. In 2021, ≥50% of all invasive isolates of K. pneumoniae from Belarus, Georgia, Greece, Moldova, Romania, Russia, Serbia, and Ukraine were resistant to carbapenems. Moreover, in 2023, the estimated incidence of BSIs caused by carbapenem-resistant K. pneumoniae in Europe increased from 2.52 in 2019 to 3.97 per 100,000 population, which corresponds to an increase of 57.5%. Even in Germany, where the percentage of carbapenem-resistant invasive isolates of K. pneumoniae amounted to ≤1% in 2021, the estimated incidence of BSIs per 100,000 population increased from 0.20 in 2019 to 0.25 in 2023, which corresponds to an increase of 25%.3,4
Resistance to carbapenems in Enterobacterales can result from (1) carbapenemases that degrade carbapenem antibiotics, (2) efflux pumps that remove carbapenems from the bacterial cell, and (3) the loss of porins, which impedes the uptake of carbapenems through the outer membrane. 5 Carbapenemases are β-lactamases that can be assigned to the Ambler classes A, B, or D, depending on their amino acid homology: class A and D include β-lactamases with a serine active site, while class B β-lactamases, also known as metallo-β-lactamases, require zinc at their active site. Carbapenemases are frequently encoded on mobile genetic elements that are transferred horizontally to different species by conjugation. 6
Infections with carbapenemase-producing Enterobacterales (CPE) represent a threat for health care systems and patients due to the limited therapeutic options and increased mortality.7,8 In the last years, the U.S. Food and Drug Administration and the European Medicines Agency (EMA) have approved new antimicrobials for use against multidrug-resistant Gram-negative bacteria (MDRGN) such as cefiderocol, ceftazidime/avibactam, and aztreonam/avibactam. As CPE may be acquired both in the hospital and the community, screening measures upon admission to hospital have to be taken into consideration to prevent nosocomial spread.9,10
The primary purpose of this study was (1) to assess the prevalence and distribution of carbapenemase genes among screening and clinical isolates of Enterobacterales from the University Hospital Erlangen (UHE), Germany, during a 5-year observational period, and (2) to determine the antimicrobial resistance rates of CPE in order to develop appropriate empirical treatment strategies in case of infection.
Methods
Study design
The UHE is a tertiary-care hospital in Bavaria, Germany, with a capacity of approximately 1,400 beds. From January 2020 to December 2024, patients were screened for CPE by rectal swab upon admission to the UHE if they (1) had contact to other patients tested positive for CPE, (2) had contact to the health care system of a country with high prevalence of CPE within the last 12 months, and/or (3) were known to be colonized or infected with CPE. Moreover, all Enterobacterales isolated from screening and clinical samples during the above-mentioned observation period were molecularly investigated for carbapenemases in every case of elevated minimum inhibitory concentration (MIC) of ertapenem, meropenem, or imipenem (see the “Microbiological Procedures” section).
Microbiological procedures
Screening swabs were spread on screening agar plates (CHROMagar™ C3GR, MAST Diagnostica GmbH, Reinfeld, Germany). Enterobacterales isolated from screening and clinical samples were identified to the species level by matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry (MALDI Biotyper, Bruker Daltonik GmbH, Bremen, Germany). An automated antibiotic susceptibility testing (AST) was performed using the VITEK 2 with the cards AST-N289, AST-N430, or AST-N371 (bioMérieux SA, Marcy-l’Étoile, France). The results were interpreted according to European Committee on Antimicrobial Susceptibility Testing (EUCAST) breakpoints (eucast: Clinical breakpoints and dosing of antibiotics). In every case of elevated MIC of ertapenem (>0.125 mg/L), meropenem (>0.125 mg/L), or imipenem (>1 mg/mL), depending on the Enterobacterales species, a molecular biological test was carried out to identify possible underlying carbapenemases. Until June 2022, we used the commercially available and CE-labeled Xpert® Carba-R PCR test (Cepheid GmbH, Krefeld, Germany), which detects five carbapenemase groups (KPC, NDM, VIM, OXA-48, IMP). From June 2022 onward, the commercially available and CE-labeled eazyplex® SuperBug CRE (AmplexDiagnostics GmbH, Gars-Bahnhof, Germany) isothermal loop-mediated isothermal amplification test was used to confirm the occurrence of carbapenemase genes. This test detects the most frequent carbapenemase groups (KPC, NDM, OXA-48, OXA-181, VIM) and ESBL genes (CTX-M-1 and CTX-M-9 group) within 20 minutes. After detection of carbapenemases, CPE were tested for susceptibility to amikacin, aztreonam, ceftazidime/avibactam, ceftolozane/tazobactam, ciprofloxacin, colistin, cotrimoxazole, fosfomycin, gentamicin, levofloxacin, meropenem, and tobramycin by broth microdilution using the MICRONAUT-S MIC plate from March 2022 onward (MERLIN Diagnostika GmbH, Bornheim-Hersel, Germany). Moreover, susceptibility to further reserve antibiotics such as cefiderocol and aztreonam/avibactam was tested by Etest starting in March 2022 and January 2024, respectively (Bestbion dx GmbH, Köln, Germany).
Retrieval and analysis of clinical and demographic data
Clinical and demographic data of patients with CPE-detection were recorded using the Microsoft Excel-based Swisslab Statistics Module (NEXUS SWISSLAB GmbH, Berlin, Germany, Version 2.22.7.000016). Bacterial isolates belonging to the same species that showed the same pattern of carbapenemase genes were counted only once per patient.
Statistical analysis
We calculated annual prevalence rates as CPE-positive patients per 10,000 patients treated at UHE for the period 2020–2024 and estimated the annual percent change (APC) with 95% confidence intervals (CIs) over the natural log of the rate using Joinpoint regression, also known as segmented regression. 11 Regarding the high proportion of CPE-positive Ukrainian patients from 2022 onward, we compared the average annual prevalence rates of CPE-positive patients before the beginning of the war in Ukraine (years 2020 and 2021) and after (years 2022–2024), calculating the prevalence rate ratio with 95% CI using the Exact Poisson Method. 12 Statistical analyses were carried out using Joinpoint Regression Software version 5.3.0 (https://surveillance.cancer.gov/joinpoint) (APC) and Stata version 19 (https://www.stata.com/) (prevalence rate ratio).
Results
Clinical epidemiology of CPE
From January 2020 to December 2024, 101 strains of CPE were isolated from 82 patients. Some of the patients were infected or colonized by more than one CPE (n = 21, 25.6%). Most of the patients were male (n = 56, 68.3%). Cardiovascular diseases (n = 35, 42.7%) followed by hematological malignancies (n = 22, 26.8%) and solid malignancies (n = 17, 20.7%) were the most prevalent underlying health conditions. Most of the CPE were found in samples from patients with signs of infection (n = 56, 55.4%), especially in urine samples (n = 27, 26.7%), blood cultures (n = 10, 9.9%), and wound swabs (n = 10, 9.9%); all other CPE isolates were derived from screening samples (n = 45, 44.6%; Table 1).
Demographic Data of the Patients and Materials Included in the Study

ICU, intensive care unit.
Most of the CPE strains (n = 101) harbored only one carbapenemase gene, such as blaOXA-48 (n = 32, 31.6%), blaNDM (n = 27, 26.7%), blaVIM (n = 14, 13.8%), blaKPC (n = 8, 7.9%), and blaOXA-181 (n = 5, 4.9%). Moreover, 15 CPE isolates carried two different carbapenemase genes, that is, blaOXA-48 and blaVIM (n = 11, 10.9%), blaNDM and blaOXA-48 (n = 2, 1.9%), blaNDM and blaVIM (n = 1, 0.9%), and blaNDM and blaOXA-181 (n = 1, 0.9%) (Fig. 1). K. pneumoniae was the CPE found most frequently (n = 46, 45.5%), followed by Escherichia (E.) coli (n = 20, 19.8%) and Enterobacter (E.) cloacae complex (n = 17, 16.8%). Notably, K. pneumoniae also carried the most diverse patterns of carbapenemases (n = 8). Some of the carbapenemase genes clustered in specific Enterobacterales species, such as blaKPC in K. pneumoniae or blaOXA-48 together with blaVIM in Citrobacter freundii (Fig. 1). Among the patients with CPE (n = 82), 13 were war-wounded refugees from Ukraine (15.8%), and a total of 16 CPE were found in this group from 2022 onward. K. pneumoniae was the most represented species (n = 9, 56.2%), and K. pneumoniae harboring blaNDM was the most frequent CPE-type (n = 4, 25.0%) (data not shown).
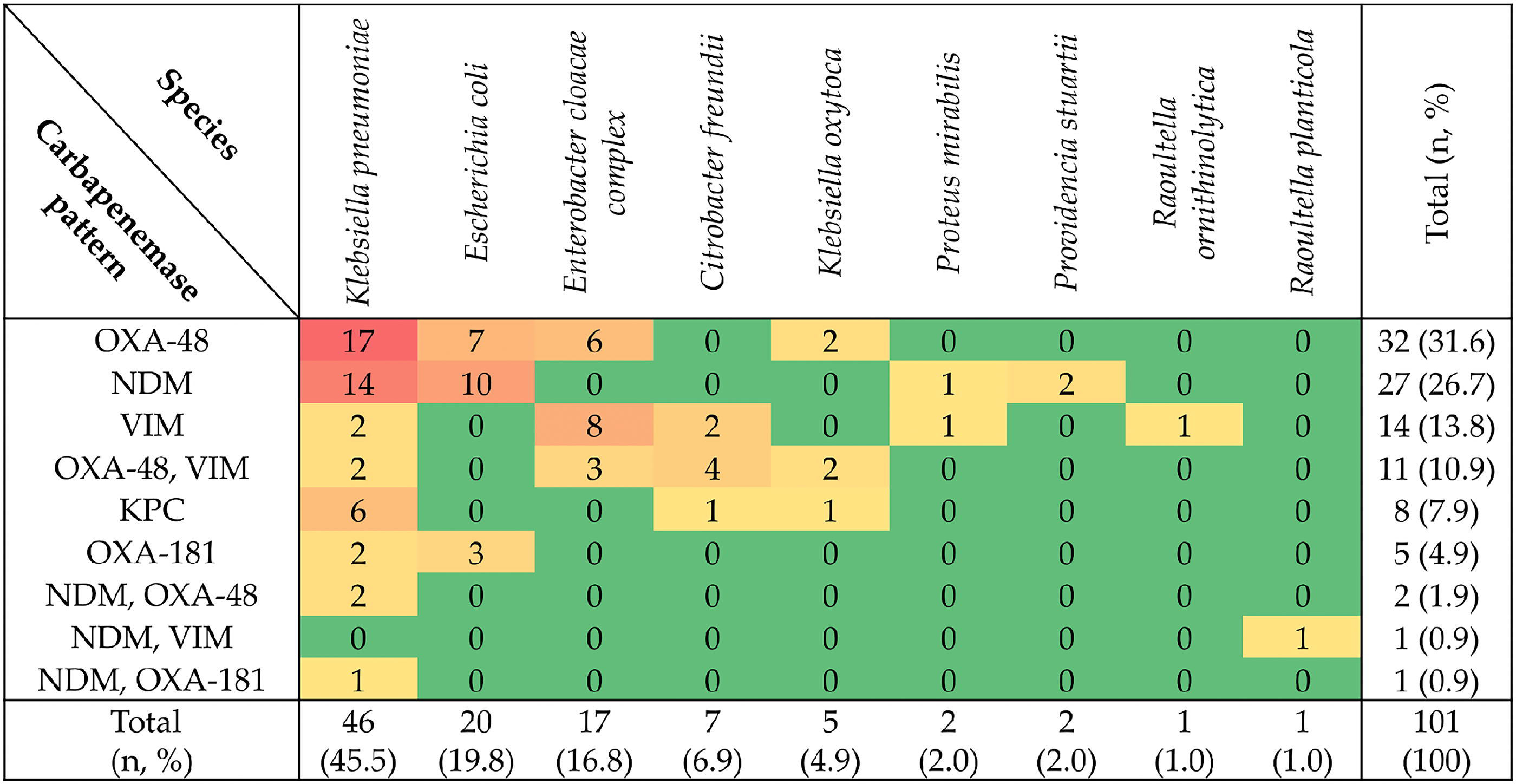
Distribution of the carbapenemase patterns in each Enterobacterales species of this study.
The annual number of CPE detected increased during the observation period (2020, n = 5; 2021, n = 5; 2022, n = 24; 2023, n = 29; 2024, n = 38), as did the number of carbapenemase patterns detected per isolate (2020, n = 2; 2021, n = 2; 2022, n = 4; 2023, n = 7; 2024, n = 8) (Fig. 2). Moreover, the proportion of CPE harboring only blaOXA-48 decreased (2020, 80.0%; 2021, 80.0%; 2022, 37.5%; 2023, 31.0%; 2024, 21.0%). In contrast, the proportion of CPE harboring a metallo-β-lactamase gene (blaVIM or blaNDM) increased during the same period (2020, 0.0%; 2021, 0.0%; 2022, 50.0%; 2023, 55.2%; 2024, 65.8%). In addition, Enterobacterales harboring more than one carbapenemase gene were isolated in 2023 for the first time (n = 5, 17.2%), with an increase in 2024 (n = 10, 26.3%) (Fig. 2).
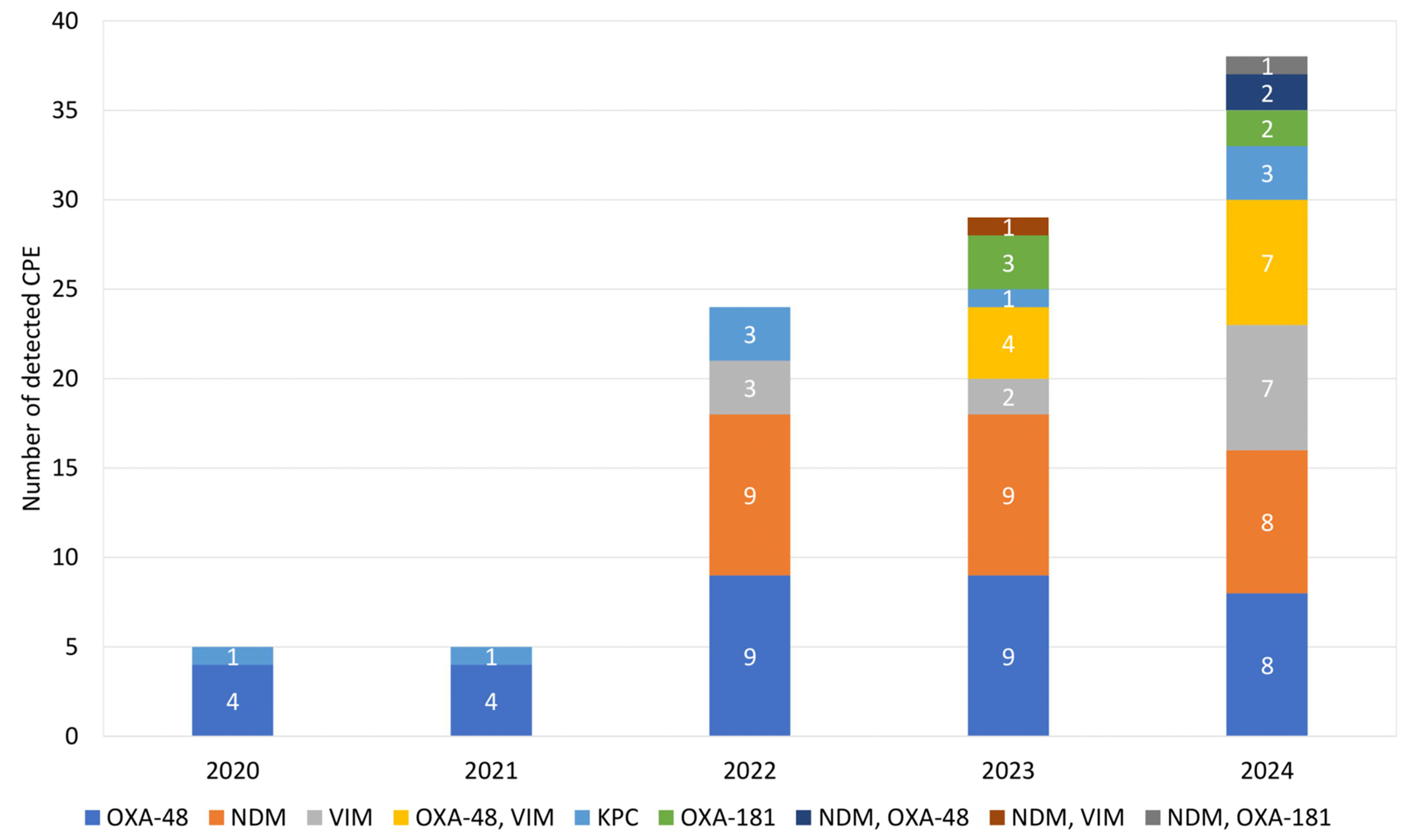
Annual numbers of carbapenemase-producing Enterobacterales (CPE) detected during the observational period 2020–2024.
The annual number of CPE-positive patients among all patients treated at the UHE increased throughout the study period (2020, 4 of 59.986; 2021, 5 of 59.732; 2022, 21 of 59.820; 2023, 21 of 61.314; 2024, 31 of 61.454). The APC of the prevalence rate of CPE-positive patients over the study period was 52.8% (95% CI: 23.6–109.4%), and the comparison of the prevalence rate for 2020–2021 with 2022–2024 gave a prevalence rate ratio of 5.3 (95% CI: 2.7–12.1).
Antimicrobial susceptibility
Regarding the antimicrobial susceptibility, only 1 of 30 CPE isolates tested showed resistance to aztreonam/avibactam (3.3%). In contrast, the resistance rates for cefiderocol and ceftazidime/avibactam amounted respectively to 12.8% and 53.9% (Table 2). In some cases, CPE strains resulted susceptible to meropenem in vitro according to the EUCAST breakpoints, despite the molecular detection of carbapenemase genes. In detail, 16 of 22 (72.7%) CPE strains carrying only blaOXA-48 showed MIC values for meropenem ≤8 mg/L (data not shown).
Antimicrobial Resistance Rates of the Carbapenemase-Producing Enterobacterales Isolates of This Study. Number of Isolates Tested Resistant/Total Number of Isolates Tested (%)
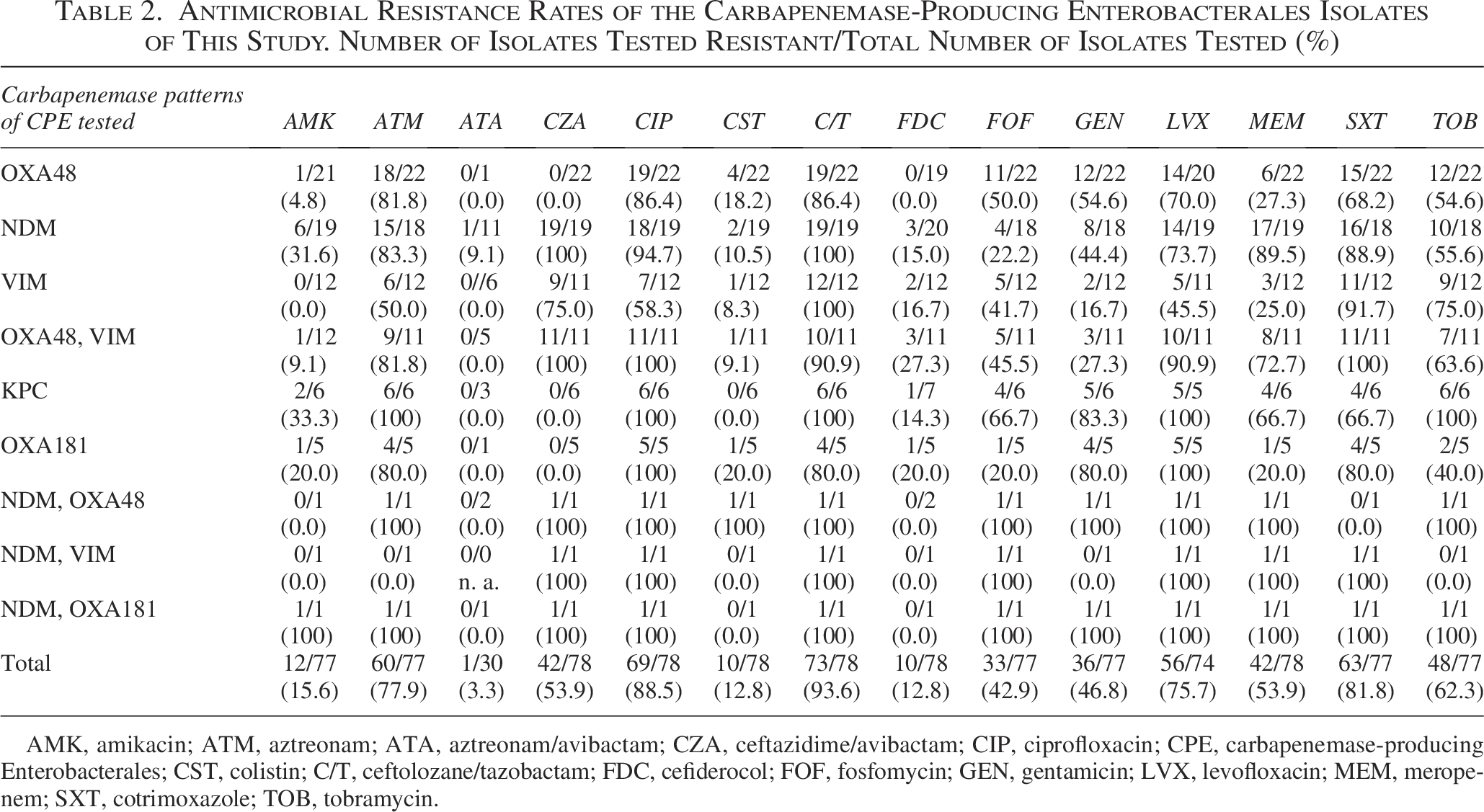
AMK, amikacin; ATM, aztreonam; ATA, aztreonam/avibactam; CZA, ceftazidime/avibactam; CIP, ciprofloxacin; CPE, carbapenemase-producing Enterobacterales; CST, colistin; C/T, ceftolozane/tazobactam; FDC, cefiderocol; FOF, fosfomycin; GEN, gentamicin; LVX, levofloxacin; MEM, meropenem; SXT, cotrimoxazole; TOB, tobramycin.
Discussion
In this study, we evaluated the clinical epidemiology of CPE at a German tertiary-care University Hospital during a 5-year period. From January 2020 to December 2024, we observed an increase in the annual number of (1) CPE detected and (2) CPE-positive patients, as well as a rise of the annual number of carbapenemase patterns detected. Moreover, the increasing trend of the annual number of CPE-positive patients was statistically significant (APC: 52.8%).
The rapid surge of CPE detected since 2022 is in accordance with previous observations in Germany.13,14 In particular, strains of K. pneumoniae producing carbapenemases of NDM-type and cultured from Ukrainian patients contributed to this process, as recently reported by the German National Reference Center for MDRGN. 13 The latter finding most likely reflects the high prevalence of multidrug-resistant organisms in Ukraine and among Ukrainian patients who were admitted to German hospitals.15–21 In our study, the prevalence rate of CPE-positive patients after the beginning of the war in Ukraine (2022–2024) was about five times higher than before (2020–2021). We expect the prevalence rate to remain at this level rather than to increase as described by the above-mentioned APC.
From January 2020 to December 2024, we observed a progressive decrease of the proportion of CPE harboring only blaOXA-48 and a rise of the proportion of CPE harboring a metallo-β-lactamase gene. In addition, we also observed that since 2023, Enterobacterales harboring more than one carbapenemase gene. The above-mentioned findings pose a threat to empirical treatment. In fact, novel antibiotics (e.g., ceftazidime/avibactam), which are now widely used for the therapy of infections caused by CPE, could definitely lose their effectiveness in the next years because of their lack of efficacy against metallo-β-lactamase-producing strains. In this study, more than half of the CPE tested were resistant against ceftazidime/avibactam. In contrast, the newly available reserve antibiotic aztreonam/avibactam, approved by the EMA in 2024, showed an overall promising in vitro activity. Prior studies also documented good in vitro and in vivo activity of aztreonam/avibactam. 22 Only one isolate of E. coli harboring blaNDM was found to be resistant against aztreonam/avibactam, most likely due to carbapenemase-unrelated modifications known to occur in E. coli. 23 Although cefiderocol is clearly a therapeutic option for CPE infections,24,25 in our study, the rate of resistance against cefiderocol was higher than the rate of resistance against aztreonam/avibactam (Table 2). Notably, for some of our CPE isolates, the MIC determined for meropenem indicated in vitro activity, especially when only blaOXA-48 was detected. Therefore, meropenem accompanied by therapeutic drug monitoring may pose a therapeutic option in some cases. 26
The main limitation of our study is that not all CPE strains (n = 101) could be tested for susceptibility to reserve antibiotics. The reason for this limitation is that the MICRONAUT-S MIC plate (MERLIN Diagnostika GmbH, Bornheim-Hersel, Germany) and the Etests for aztreonam/avibactam and cefiderocol (Bestbion dx GmbH, Köln, Germany) were not immediately available from January 2020. In detail, the MICRONAUT-S MIC plate and the cefiderocol Etest were introduced in our laboratory in March 2022. Therefore, only 78 of the 101 CPE isolates of this study were tested using the above-mentioned diagnostic tools. Furthermore, the aztreonam/avibactam Etest has only been used in our laboratory since January 2024, so that only 30 isolates have been tested using this methodology. Moreover, not all strains have been stored for later examinations. Therefore, retrospective antimicrobial susceptibility testing was not possible.
Conclusions
The increasing incidence of CPE at our hospital in the last years is associated with (1) a rise in the proportion of the metallo-β-lactamase-producing strains and (2) the occurrence of Enterobacterales harboring more than one carbapenemase gene since 2023. Moreover, the newly available antibiotic aztreonam/avibactam showed promising in vitro activity against CPE.
Authors’ Contributions
Conceptualization: G.V. and J.E. Methodology: G.V. Software: J.E. Validation: G.V., J.E., and J.S. Formal analysis: G.V., J.H., and C.B. Investigation: G.V., J.E., J.S., and S.V. Statistical analysis: S.V. Resources: G.V., J.H., and C.B. Data curation: J.E. and J.S. Writing—original draft preparation: J.S., J.E., and G.V. Writing—review and editing: J.H. and C.B. Visualization: J.E. Supervision: G.V. and C.B. Project administration: G.V. All authors have read and agreed to the published version of the article.
Footnotes
Acknowledgments
The authors would like to thank the diagnostic staff members of the Institute of Clinical Microbiology, Immunology, and Hygiene of the University Hospital Erlangen for their technical assistance.
Funding Information
This study has been supported by the Bayerisches Zentrum für Präventive Infektionsmedizin (BZI).
Ethical Approval
Ethical approval was not required as the reported analysis was regarded as part of the routine surveillance and infection control measures at UHE in accordance with the German “Protection against Infection Act” (§ 23 IfSG).
Disclosure Statement
The authors have no conflicts of interest to disclose.
