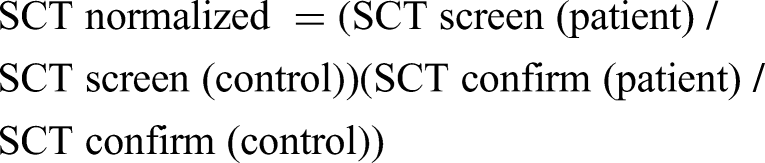Abstract
Background
Lupus anticoagulant (LA) prolongs phospholipid-dependent coagulation tests and can falsely reduce intrinsic coagulation factor activities in one-stage assays. The quantitative relationship between LA intensity and the degree of assay interference remains insufficiently defined.
Methods
One hundred patients with LA-positive antiphospholipid syndrome or systemic lupus erythematosus were studied. FVIII:C, FIX:C, and FXI:C were measured using one-stage assays, and a modified assay incorporating SCT-confirm reagent was applied to reduce LA interference. The pseudo-lowering degree (PLD) was calculated to quantify correction efficacy. Associations between LA characteristics and reduced factor activities were analyzed, and activity levels or PLD values across LA-intensity subgroups were compared.
Results
Using one-stage assays, reduced FVIII:C, FIX:C, and FXI:C were noted in 27%, 23%, and 26% of patients, respectively. LA intensity was unrelated to reduced FVIII:C but significantly associated with decreased FIX:C (P = .006) and FXI:C (P < .001). Moderate LA positivity showed higher FIX:C levels than weak positivity, whereas markedly lower FXI:C was seen only in strongly LA-positive patients. The SCT correction assay restored FVIII:C, FIX:C, and FXI:C to normal levels in approximately 66.7%, 82.6%, and 92.3% of affected cases, respectively. In strongly LA-positive patients, PLD values of FVIII:C, FIX:C, and FXI:C were significantly higher than in weak or moderate subgroups.
Conclusions
FVIII:C, FIX:C, and FXI:C exhibit differential susceptibility to LA interference. The SCT-based correction approach effectively mitigates this effect, especially for FXI and offers a practical improvement over conventional one-stage assays to avoid misclassification of intrinsic factor deficiencies in LA-positive patients.
Introduction
Lupus anticoagulant (LA) is a class of autoantibodies strongly associated with antiphospholipid syndrome (APS) and systemic lupus erythematosus (SLE). LA is not only linked to an increased risk of thrombosis but also to adverse obstetric outcomes, including recurrent pregnancy loss, fetal demise, and preeclampsia.1–3 LA is an immunoglobulin that binds to negatively charged phospholipids or phospholipid-protein complexes, and is an important component of the antiphospholipid antibody spectrum. The detection of LA is not performed via calibrated assays, but rather based on the functional behavior of its antibodies in a medley of coagulation assays. The combination of diluted Russell's viper venom time (dRVVT) and silica clotting time (SCT), is widely used and recommended. 4 However, the presence of LA in patients with APS/ SLE leads to a significant prolongation of APTT in laboratory tests, which easily creates a false impression of abnormal coagulation function. When APTT was persistent prolongation, an intuitive report indicating normal coagulation factor activity is usually a reassuring factor for surgeons before surgery.
Coagulation factor assays identify congenital or acquired deficiencies, assess replacement therapy pharmacokinetics, and determine coagulation factor concentrate potency. 5 Coagulation factor VIII (FVIII), factor IX (FIX), factor XI (FXI), and factor XII (FXII) are key components of the intrinsic coagulation pathway. These factors functional activities are measured via three principal methods: one-stage clotting, two-stage, and chromogenic substrate assays.6,7 The one-stage assay quantifies factor activity by correcting prolonged APTT in factor-deficient plasma. A calibration curve is generated by diluting reference plasma, mixing with factor-deficient plasma, and measuring APTT clotting times. Target factor activity is interpolated from this curve. Widely used clinically for its simplicity, cost-effectiveness, and correlation with in vivo hemostasis, 8 the one-stage assay is susceptible to heparin, direct oral anticoagulants, and endogenous inhibitors like LA. A global survey indicates that the one-stage clotting assay is currently the most widely used method for measuring factor activity. 9 LA interference in phospholipid-dependent APTT assays reduces measured clotting factor activity, potentially leading to misclassified disease severity, inaccurate bleeding phenotype estimation, and suboptimal therapy monitoring for coagulation factor replacement therapy. 10 What's more, the anticardiolipin antibodies (aCL), and anti-β2-glycoprotein I antibodies (anti-β₂GPI), as part of antiphospholipid antibodies (aPL), play a central role in its thrombotic manifestations. 11 However, the extent to which different aPL positivity patterns such as single, double, or triple positivity impact the accuracy of coagulation factor measurements remains unclear. Therefore, in this study, we evaluated the impact of different aPL profiles on the measurement of factor VIII, IX, and XI activities using the one-stage clotting assay.
This study characterized LA interference with FVIII activities (FVIII:C), FIX:C, and FXI:C measured by one-stage assay in a cohort of LA-positive APS/SLE patients. We compared difference across LA detection methods and evaluated associations between different aPL positivity patterns and reduced factor activities. To improve accuracy, we implemented an SCT-based correction method using SCT-confirm reagent (HemosIL, Spain), which counteracts LA-induced phospholipid consumption via phospholipid-supplemented APTT reagents (HemosIL, Spain), enabling reliable in vitro factor activity assessment. We further introduced a novel standardized metric, pseudo-reduction degree (PLD), to quantitatively assess the efficacy of this corrective approach.
Methods
Patients
This study enrolled APS/SLE patients in the Clinical Laboratory of Peking University First Hospital between January 2025 and July 2025. Inclusion criteria were as follows: Patients meeting established diagnostic criteria for either APS 12 or SLE. 13 Exclusion criteria included: (1) FVIII, FIX, and FXI deficiency; (2) Recent administration of fresh frozen plasma, cryoprecipitate, or recombinant coagulation factors; (3) Use of heparin or oral anticoagulants; (4) Patients lost to follow-up or those with incomplete medical records. All patients were carefully evaluated to ensure accurate classification and to minimize confounding factors that could influence laboratory results or clinical interpretation. This study was approved by the Ethics Committee of Peking University First Hospital. The study adhered to the ethical guidelines of the Declaration of Helsinki.
Sample Collection
The sample size was estimated using the maximum uncertainty principle. Based on the formula for estimating proportions, we assumed a 95% confidence level (Z = 1.96), a maximum uncertainty (P = .5), and a margin of error (d) of 0.1, resulting in a calculated sample size of approximately 96 cases. A total of 100 cases were ultimately enrolled in this study. Blood samples were collected by phlebotomy into buffered 3.2% sodium citrate blue-top vacutainer tubes. All samples were received and processed within 1 h of collection. Platelet poor plasma was prepared through double centrifugation at 2000×g for 15 min at 10-20 °C. The processed plasma was either analyzed within 4 h or stored at −80 °C. Frozen samples were rapidly thawed at 37 °C and immediately tested.
for LA, aCL and Anti-β2GPI
Lupus anticoagulant (LA) activity was assessed using dilute Russell's viper venom time (DRVVT) and silica clotting time (SCT) methods with Screen and Confirmatory reagents (Werfen, Spain). The results were quantified as normalized ratios. The Platelet Neutralization Procedure was not used in this study for LA testing. For DRVVT, LA positivity was stratified as follows: weakly positive (normalized ratio 1.2-1.49), moderately positive (1.5-1.99), and strongly positive (≥2.0). Similarly, SCT-based LA positivity was categorized into weakly positive (normalized ratio 1.16-1.49), moderately positive (1.5-1.99), and strongly positive (≥2.0).
The DRVVT normalized ratio was calculated using the following formula:
The SCT normalized ratio was calculated using the following formula:
Serum levels of aCL and anti-β₂GPI, including both IgG and IgM isotypes, were measured using standardized enzyme-linked immunosorbent assay kits (Euroimmun, Germany), according to the manufacturer's instructions. Positive cutoff values were defined according to the criteria: aCL IgG/IgM ≥ 12 PL/ml and anti-β₂GPI IgG/IgM ≥ 20 RU/ml.
Coagulation Factor Activity Assay
APTT-based method: This assay was performed on a CN6000 analyzer (Sysmex, USA). The test system includes an appropriate concentration of phospholipids, Ca2+, specific factor-deficient plasmas (deficient in FVIII, FIX, and FXI), and ellagic acid. The clotting time is measured, and the activity level of the respective coagulation factor is determined by comparison with a reference plasma curve. Based on the reference interval lower limits determined by our laboratory, the cutoff values were: FVIII:C ≥ 78%, FIX:C ≥ 70%, FXI:C ≥ 62%.
SCT-based method: This method was performed using an ACL-TOP 700 instrument (Werfen, Spain). It employs an excess of phospholipids from LA-SCT-confirm regent, Ca2+, factor-deficient plasmas (specifically deficient in FVIII, FIX, and FXI), and a surface-activating agent to correct for potential interference and thereby restore the measured coagulation factor activity to a value approximating the true physiological level. Clotting time is recorded, and the activity of the coagulation factor is determined by interpolation from a calibrated standard curve generated using normal pool plasma.
PLD Calculation
The pseudo-lowering degree (PLD) of coagulation factor activity was defined as the relative difference between factor activity levels measured by the SCT-based method and those obtained by the APTT-based method, calculated according to the following formula:
A higher PLD value indicates a greater discrepancy between the two assays, suggesting a stronger interference of lupus anticoagulant on the one-stage (APTT-based) factor activity measurement.
Statistical Analysis
Statistical analysis was conducted using SPSS version 27.0, and figures were generated with GraphPad Prism version 9.0. The chi-square (χ2) test and Fisher's exact test were employed to evaluate the impact of LA detecting method and positivity levels on the measured FVIII:C, FIX:C, and FXI:C. Depending on normality and variance, one-way ANOVA with Tukey's test, Welch's ANOVA with Dunnett's T3, or Kruskal–Wallis test with Dunn's test was used to compare coagulation factor activities and PLD levels across LA positivity levels. All P values were two-tailed, and a P value < .05 was considered statistically significant.
Results
Characteristic of Patients
A total of 100 patients diagnosed with APS/SLE were enrolled in this study, including 20 males and 80 females. All participants underwent assay of FVIII:C, FIX:C, and FXI:C using the one-stage clotting assay. 27% patients exhibited FVIII:C below the lower limit of the normal reference range, 23% had reduced FIX:C, 26% demonstrated subnormal FXI:C. Notably, none of the patients presented with clinical evidence of bleeding diathesis. Moreover, all individuals tested positive for LA.
Effect of Different LA Testing Methodologies on Coagulation Factor Activity Assays
At first, we investigated the association between positive LA results, as determined by different testing methodologies, and reduced coagulation factor activity levels. LA test was evaluated using the DRVVT and SCT assays. Based o n these tests, patients were categorized into three groups:18 patients tested positive exclusively by DRVVT, 28 patients were positive only by SCT, and 54 patients were double-positive. As presented in Table 1, Fisher's exact test revealed no statistically significant associations between LA positivity (by either DRVVT or SCT) and decreased FVIII:C (P > .05) and FIX:C (P > .05). However, a significant association was observed with reduced FXI:C (χ2 = 9.094, P = .010). Post hoc pairwise comparisons with Bonferroni correction demonstrated that the double-positive group exhibited a significantly higher prevalence of decreased FXI:C (37.0%) compared to the SCT-only positive group (7.1%). In contrast, no significant differences were observed between the DRVVT-only and SCT-only groups, nor between the double-positive and DRVVT-only groups, in terms of FXI:C reduction.
Comparison of the Detection Rates of Reduced Coagulation Factor Activity among LA Positive Cases Using Different Methodologies.

Comparison of the Detection Rates of Reduced Coagulation Factor Activity among LA Positive Cases Using Different Methodologies.

, the expected frequency was less than 5, and Fisher's exact test was employed. DRVVT, diluted Russell's viper venom time; SCT, silica clotting time. Bolded text indicates that P values are significantly different, P < .05. LA, Lupus Anticoagulant. “Decrease” indicates that the coagulation factor activity is below the normal reference range, whereas “Normal” denotes activity within the normal limits. The reference cutoff values were: FVIII:C ≥ 78%, FIX:C ≥ 70%, and FXI:C ≥ 62%.
The Interference of LA Combined with aCL and Anti-β2GPI for Coagulation Factor Activity
Based on the integrated results of the DRVVT and SCT assays, the final LA positivity level was assigned according to the higher of the two positivity grades obtained from either test. This classification yielded 40 patients with weak positivity, 18 with moderate positivity, and 42 with strong positivity. In patients positive for LA, approximately 27% exhibited reduced FVIII:C, with 12% of these cases classified as strongly LA-positive. Around 23% demonstrated decreased FIX:C, among which 14% are strongly LA-positive. Reduced FXI:C was observed in 26% of patients, with 21% of these being strongly LA-positive. Table 2 presented the analysis of the association between LA positivity levels and reductions in coagulation factor activity, evaluated using the chi-square test. No significant difference was observed in the proportion of patients exhibiting reduced FVIII:C across the different LA positivity levels (χ2 = 0.853, P = .66). However, statistically significant differences were found in FIX:C (χ2 = 9.774, P = .006), and FXI:C (χ2 = 22.968, P < .001). Additionally, we further evaluated the impact of single, double, and triple positivity within the antiphospholipid antibody profile on the measurement of coagulation factor activities based on LA positive. As shown in Table 3, no statistically significant differences were observed in the interference with FVIII:C (χ2 = 4.654, P = .18), FIX:C (χ2 = 3.182, P = .34), and FXI:C (χ2 = 6.833, P = .07) among samples with LA single positivity, double positivity, and triple positivity.
Impact of LA Positivity on the Measurement of Factor VIII, Factor IX, and Factor XI Activities Using the one-Stage Clotting Assay.

, the expected frequency was less than 5, and Fisher's exact test was employed. Bolded text indicates that P values are significantly different, P < .05. LA, Lupus Anticoagulant. Weakly positive, 1.20-1.49 (DRVVT) / 1.16-1.49 (SCT); Moderate, 1.50-1.99; Strong, ≥2.00. “Decrease” indicates that the coagulation factor activity is below the normal reference range, whereas “Normal” denotes activity within the normal limits. The reference cutoff values were: FVIII:C ≥ 78%, FIX:C ≥ 70%, and FXI:C ≥ 62%.
Impact of Lupus Anticoagulant Combined with aCL and Anti-β2GPI Positivity on the Measurement of Factor VIII, Factor IX, and Factor XI Activities Using the one-Stage Clotting Assay.

, the expected frequency was less than 5, and Fisher's exact test was employed. Bolded text indicates that P values are significantly different, P < .05. LA, Lupus Anticoagulant. LA positive, ≥1.20 (DRVVT) / 1.16 (SCT). “Decrease” indicates that the coagulation factor activity is below the normal reference range, whereas “Normal” denotes activity within the normal limits. The reference cutoff values were: FVIII:C ≥ 78%, FIX:C ≥ 70%, and FXI:C ≥ 62%.
Subsequently, coagulation factor activity levels were assessed using both APTT-based and SCT-based assays. Table 4 presented the in vitro activities of intrinsic pathway coagulation factors, as measured by APTT- and SCT-based methods, in patients stratified according to the degree of LA positivity. The results indicated that FIX:C and FXI:C activities, determined by the APTT-based method, were significantly correlated with the extent of LA positivity (P = .0004 and P < .0001, respectively), whereas FVIII:C levels show no significant association with LA positivity (P = .13).
The Levels of Intrinsic Coagulation Factors Activities in Vitro Measured by APTT and SCT Based Methods in Patients with Varying Degrees of LA Positivity.

Bolded text indicates that P values are significantly different, P < .05. LA, lupus anticoagulant; CI, confidence interval. LA-Weakly positive, 1.20-1.49 (DRVVT) / 1.16-1.49 (SCT); Moderate, 1.50-1.99; Strong, ≥2.00.
Figure 1 presented a comparison of coagulation factor activity levels across different LA positivity groups. Figure 1A-C displayed the distribution of FVIII:C, FIX:C, and FXI:C, respectively, as measured by APTT-based assays. No significant differences in FVIII:C levels were observed among the three LA positivity groups (Figure 1A). In contrast, individuals with weakly LA-positive status exhibited significantly higher FIX:C levels (109.8% ± 25.1%) compared to those who were moderately LA-positive (84.6% ± 39.4%, P = .03) or strongly LA-positive (80.5% ± 39.2%, P = .0004) (Figure 1B). Furthermore, patients who were strongly LA-positive demonstrated markedly reduced FXI:C activity (65.0% ± 36.2%) relative to both weakly LA-positive (102.0% ± 25.6%, P < .0001) and moderately LA-positive (96.5% ± 32.9%, P = .002) individuals (Figure 1C).

The difference of coagulation Factor VIII (A and D), Factor IX (B and E), and Factor XI (C and F) activities in patients with varying degrees of LA positivity by APTT- and SCT-based methods. LA, Lupus Anticoagulant. Weakly positive, 1.20-1.49 (DRVVT) / 1.16-1.49 (SCT); Moderate, 1.50-1.99; Strong, ≥2.00. ****, P < .0001; ***, P < .001; **, P < .01; ns, P > .05.
Given that LA primarily interferes with coagulation assays through the inhibition of phospholipid-dependent reactions—either due to antibody-mediated disruption of phospholipid surfaces or excessive consumption of phospholipids—the study ensured accurate assessment of coagulation factor activity by incorporating an excess of exogenous phospholipids in the SCT-method assay, thereby neutralizing LA interference. The results of coagulation factor activity assays measured by the SCT method revealed no significant association between LA positivity and the activities of factors VIII, IX, and XI, as shown in Table 3. Figures 1D-1F illustrated the distribution of FVIII:C, FIX:C, and FXI:C levels, as determined by the SCT method, across patient groups stratified by the intensity of LA positivity. No statistically significant differences in factor activity levels were observed among the different LA intensity categories, suggesting that the interference caused by LA status in the measurement of these coagulation factor activities is effectively mitigated when the SCT method was employed.
In samples with reduced coagulation factor activities as measured by the APTT assay, Figure 2 visually illustrated the differences between FVIII:C, FIX:C, and FXI:C as determined by the APTT method versus the SCT method. The results indicated that the activities of all three coagulation factors were significantly higher when measured by the SCT method compared to the APTT method (P = .0004, P < .0001, and P < .0001, respectively), suggesting improved detection sensitivity of the SCT assay in these deficient samples.

The differences in FVIII:C (A), FIX:C (B), and FXI:C (C) levels measured by the APTT-based one-stage assay versus the SCT-based correction assay in samples with lupus anticoagulant–mediated interference causing falsely reduced coagulation factor activities. ****, P < .0001; ***, P < .001.
In Figure 3, we presented the results of APTT-based assays showing reduced activities of coagulation factors VIII, IX, and XI alongside corresponding findings obtained using the SCT method in the same patient cohort. Among 27 patients with initially low FVIII:C levels, approximately 66.7% (18/27) demonstrated normalized factor activity within the established reference range following SCT correction (Figure 3A). Of 23 patients with reduced FIX:C, about 82.6% (19/23) exhibited activity levels restored to the normal range with the SCT method (Figure 3B). In the cohort of 26 patients with decreased FXI:C, roughly 92.3% (24/26) achieved factor activity within the reference interval upon correction with SCT (Figure 3C).

The distribution of levels of FVIII:C (A), FIX:C (B), and FXI:C (C) measured by the APTT method versus the SCT method in samples with la-mediated interference causing falsely reduced coagulation factor activities. LA, Lupus Anticoagulant.
The PLD was calculated to quantitatively evaluate the correction capability of the SCT method. When weak and moderate LA-positive samples were grouped together, the mean PLD values were as follows: 37.4% ± 5.6% (95% CI: 33.6-41.2%) for FVIII, 44.3% ± 6.3% (95% CI: 37.7-50.9%) for FIX, and 35.7% ± 12.8% (95% CI: 19.4-51.3%) for factor XI (FXI). In patients with strongly positive LA, the corresponding PLD values were significantly higher, with means of 62.1% ± 21.8% (95% CI: 41.9-82.3%) for FVIII, 71.7% ± 13.2% (95% CI: 63.7-79.6%) for FIX, and 66.7% ± 15.2% (95% CI: 59.3-74.0%) for FXI. Figure 4 indicated that patients with strong positive LA exhibited significantly higher PLD levels for FVIII (P = .02) (Figure 4A), FIX (P = .0002) (Figure 4B), and FXI (P = .0004) (Figure 4C).
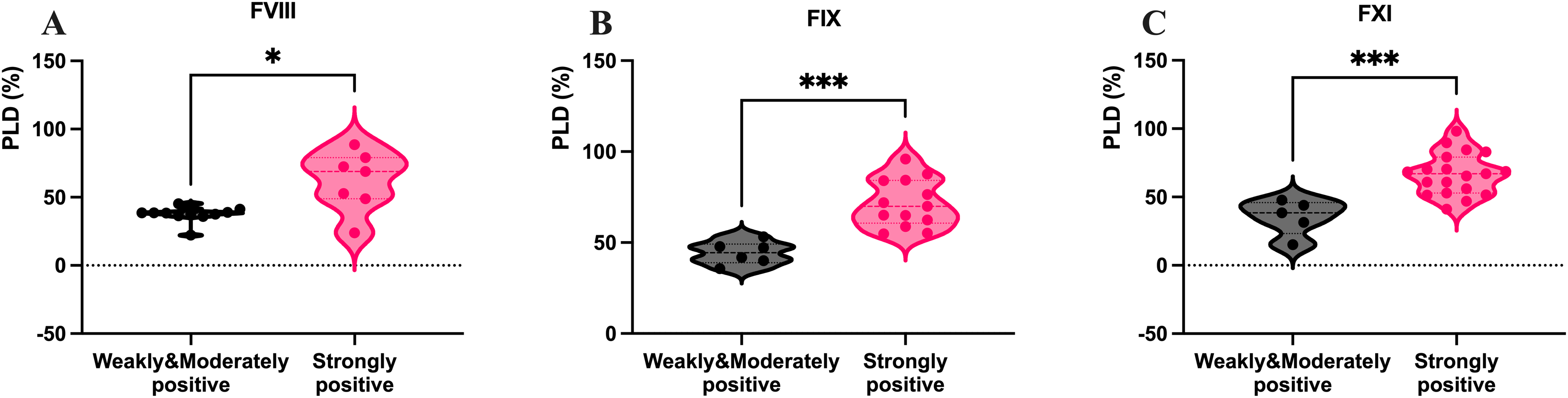
Correlation between the intensity of LA positivity and PLD of coagulation factor VIII (A), factor IX (B), and factor XI (C) activities. LA, Lupus Anticoagulant; PLD, pseudo-lowering degree; Weakly positive, 1.20-1.49 (DRVVT) / 1.16-1.49 (SCT); Moderate, 1.50-1.99; Strong, ≥2.00. ****, P < .0001; ***, P < .001; *, P < .05.
Discussion
In our study, we first elucidated the interference of LA on the measurement of coagulation factor activities within the intrinsic coagulation pathway. We further evaluated and demonstrated that there was no significant difference between the SCT method and the DRVVT method in assessing the extent of LA positivity with regard to their interfering effects on coagulation factor assays. Secondly, we provided novel evidence showing that FVIII activity levels did not significantly differ among patients with weakly positive, moderately positive, or strongly positive LA status. In contrast, the measured activities of FIX and FXI exhibited varying degrees of sensitivity to LA interference. Notably, FIX:C activity was already markedly affected in patients with moderate LA positivity, whereas significant reduction in FXI:C was observed only in those with strong LA positivity. We employed a corrective approach using SCT-confirm reagents in combination with APTT reagents (HemosIL, Spain) to mitigate the in vitro interference caused by antiphospholipid antibodies, thereby enabling more accurate assessment of true coagulation factor activities. Given the inherent inter-individual variability in coagulation factor levels, PLD were utilized to evaluate the degree of LA-mediated interference. Our findings conclusively demonstrate that higher LA positivity correlates with progressively greater interference in the measured activities of FVIII, FIX, and FXI.
The 2020 International Society on Thrombosis and Haemostasis Scientific and Standardization Committee (ISTH-SSC) guidelines recommend the SCT assay as a screening test for LA, primarily due to its methodological dependence on phospholipid content rather than specific activators. The DRVVT assay is another guideline-recommended method for detection. 14 In our study, we observed no significant difference in the levels of FVIII:C and FIX:C activity between patients who tested positive for LA by either the DRVVT or SCT method. However, among patients who were double-positive by both assays, a higher proportion exhibited reduced FXI:C. This finding may be attributable to the high prevalence (80.7%) of strongly positive LA results in this subgroup, which could plausibly explain the observed association.
The one-stage assay for coagulation factor activity is performed in the presence of exogenous phospholipids. It is well established that LA can interfere with this assay by consuming phospholipids, leading to a falsely decreased measurement of coagulation factor activity in vitro. The chromogenic substrate assays demonstrate superior analytical specificity in such scenarios. By relying on synthetic substrates and direct measurement of enzymatic activity rather than clot formation, these assays are largely independent of phospholipids and do not involve contact activation or the prothrombinase complex. This design minimizes interference from LA, thereby providing more accurate and reliable quantification of FVIII and FIX activities. The reduced susceptibility to LA interference is attributed to several factors, including higher sample dilution, decreased dependence on phospholipid surfaces, and the bypass of physiological coagulation pathways that are vulnerable to LA-mediated inhibition. Despite these advantages, the clinical adoption of chromogenic assays is limited by practical constraints. These include higher reagent and instrumentation costs, limited commercial availability for certain factors (eg, FXI), and challenges in rapid implementation during emergency settings or off-hours, where immediate turnaround is critical. Currently, validated chromogenic methods are primarily established for FVIII and FIX, leaving a gap in standardized alternatives for other coagulation factors. 15 An increasing number of studies have been exploring novel methodologies, such as the use of liquid chromatography–tandem mass spectrometry to quantify human FVIII plasma concentrations. Linear regression analysis has been employed to correlate these measured plasma concentrations with FVIII:C levels determined by one-stage clotting assays. However, this approach is significantly compromised in the presence of anti-FVIII antibodies or exogenous FVIII products, which can markedly interfere with the accuracy and reliability of the measurements. 16
In our study, no significant differences were found in the degree of interference with coagulation factor activity measurements among different aPL positivity patterns. The number of positive aPL subtypes may not directly correlate with the extent of assay interference in one-stage clotting-based factor activity testing. Notably, a trend toward greater interference with FXI:C (P = .07) was observed in triple-positive samples, which may warrant further investigation in studies with larger sample sizes. The lack of statistical significance may be partly attributed to the limited number of triple-positive cases and inter-individual variability in aPL titers and avidity.
A previous study validated an approach to mitigate the interference of LA on coagulation factor activity assays by combining the use of the SCT-C reagent with the APTT-based assay. In LA-positive samples, the utilization of the SCT-C reagent effectively neutralized the inhibitory effects of strong lupus anticoagulants, thereby enabling more accurate measurement of clotting factor activities. 17 Therefore, this approach was employed to correct the interference caused by LA on one-stage clot-based assays for coagulation factor activity, thereby enabling more accurate assessment of true factor levels in our study. The method demonstrated effective correction for FIX:C and FXI:C activities. However, correction was only partial for FVIII:C, with correction rate of 66.7%.
FVIII is known to be relatively labile in vitro; delays in centrifugation or failure to promptly freeze plasma samples after processing may result in partial degradation of FVIII, leading to an underestimation of its activity. A study has demonstrated that FVIII exhibits a rapid decline in stability when stored at room temperature, with activity losses ranging from 23% to 26.9% observed after 24 h. In contrast, FIX and FXI have been shown to remain stable for up to 24 to 48 h under similar storage conditions. 18 In this study, samples for coagulation factor activity assays were collected and frozen approximately 4 to 8 h after completion of LA testing. Therefore, the lack of a consistent room temperature storage duration prior to freezing may have influenced the assessment of LA-mediated interference on FVIII:C and affected the interpretation of the SCT-based correction. These methodological limitations highlight the need for standardized pre-analytical procedures and more robust assay designs when evaluating contact pathway factors in the context of LA. Incomplete correction of coagulation factor levels to the normal range observed in this study may be attributed to factors such as residual antiphospholipid antibody interference and coagulation factor inhibitors. First, suboptimal phospholipid concentrations may have been insufficient to fully neutralize the inhibitory effects of antiphospholipid antibodies. In patients with strongly positive lupus anticoagulant (LA), higher antibody titers or increased antibody avidity may lead to more pronounced interference in phospholipid-dependent coagulation assays. When the exogenously added phospholipid concentration is inadequate, this interference cannot be completely overcome, resulting in persistently reduced coagulation factor activity despite supplementation. Second, the presence of specific coagulation factor inhibitors cannot be ruled out as a contributing factor to diminished factor activity. Such inhibitors may independently impair clotting function and confound the interpretation of factor assays.
This study has several limitations. First, the modest sample size and single-center design may limit the generalizability of the findings. Second, samples in which SCT failed to correct intrinsic coagulation factor activities were not subjected to further confirmatory or mechanistic evaluation. Third, factors related to the extrinsic pathway were not assessed, which restricted a more comprehensive characterization of LA-related assay interference. Finally, the use of a chromogenic assay may have provided a more accurate measurement of FVIII:C.
Conclusions
Lupus anticoagulant markedly interferes with one-stage clotting assays, often leading to underestimation of intrinsic coagulation factor activities. This analytical limitation highlights the need for more accurate evaluation strategies in LA-positive patients. Although the one-stage assay is widely used in clinical laboratories, a simple methodological modification using the SCT-based correction assay substantially reduces LA-mediated interference and provides more reliable factor activity measurements. Incorporating SCT-based testing into routine practice may therefore improve the accuracy of intrinsic pathway factor assessment and support more appropriate clinical decision-making in patients with LA.
Footnotes
Acknowledgements
All people who contributed to this work are listed among co-authors.
Informed Consent and Patient Details
This study was approved by the Medical Ethics Committee of Peking University First Hospital (No.2025-1163). Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Statement
All relevant data are included in the article. Further inquiries can be directed to the corresponding author.