Abstract
Introduction
Hemophilia A and B are among the most common inherited bleeding disorders in humans with lifelong risk of spontaneous and recurrent bleeding. While prophylaxis with clotting factors has improved outcomes, challenges like inhibitor development and treatment burden persist. We aim for a comparative effectiveness between these agents and traditional therapy with this study.
Methods
A systematic review and meta-analysis of randomized controlled trials (RCTs) was conducted. Studies were identified through PubMed, Cochrane Central, and ClinicalTrials.gov up to May 30th, 2025. Eligible studies were double-arm RCTs comparing non-clotting factor prophylaxis (emicizumab, fitusiran, concizumab) and on-demand therapy in patients with hemophilia A or B.
Results
Non-clotting factor prophylaxis significantly reduced the annualized bleeding rate (ABR) for all treated bleeds [n = 399, (RR = 0.13; 95% CI: 0.09–0.19), I2 = 63.8%, p = 0.0107], spontaneous bleeds [RR = 0.08; 95% CI: (0.06, 0.11), I2 = 0.0%, p = 0.5933)] and joint bleeds [RR = 0.09; 95% CI: (0.06, 0.14), : I2 = 26.2%, p = 0.2468 ]. Haem-A-QoL total score improved with prophylaxis [MD = −11.08 [−16.34, −5.83], I2 = 57.5%, p = 0.0949]. Prophylaxis increased the likelihood of achieving zero treated bleeds [RR = 4.11; 95% CI: (1.48%, 11.45%), I2 = 88.5%, p < 0.0001]. An exploratory network meta-analysis comparing fitusiran, emicizumab, and concizumab reported no statistically significant difference in the ABR for all treated bleeds.
Conclusion
Compared to on-demand therapy, non-factor prophylactic therapies significantly reduce bleeding episodes, improve quality of life, and increase the likelihood of zero bleeds in patients with hemophilia.
Keywords
Introduction
Hemophilia A and B are among the most common inherited bleeding disorders in humans. Caused by mutations in the genes encoding coagulation factor VIII (FVIII) and factor IX (FIX), the X-linked recessive disease has high prevalence at one in 5000 male live births for Hemophilia A and one in 30,000 in Hemophilia B. 1 This leaves those affected with a lifelong risk of spontaneous and recurrent bleeding. Many patients experience bleeding without recognized trauma, often into joints (hemarthrosis) but also into muscles, soft tissues, and life-threatening locations like the central nervous system. This can lead to further complications such as hypertrophic synovitis, progressive cartilage degradation, and long-term hemophilic arthropathy, causing chronic pain and joint function impairment. 2
There are two main approaches to hemophilia management: on-demand treatment to control acute bleeding episodes and prophylactic therapy aimed at preventing bleeds, with prophylaxis being the preferred strategy for long-term disease control. The regular infusions may consist of either prophylaxis with clotting factors or prophylaxis with non-clotting factors. While prophylaxis with clotting factors has improved outcomes, challenges like inhibitor development and treatment burden persist. 3 Recently, non-clotting factor therapies have emerged as treatments that correct the hemostatic defect without replacing the missing protein (FVIII or FIX). With various alternative mechanisms, they include therapies like emicizumab, fitusiran, and concizumab. 4 They have each also been designed to allow for a lower treatment burden. 5 On-demand therapy is the second treatment option available for patients. In this therapy, clotting factor concentrates or other treatments are administered only at the time of a bleed. 6 This allows for a rapid and immediate stop to hemorrhages, reducing joint pain and restoring joint mobility, but does not prevent the beginning and subsequent development of hemophilic arthropathy, 7 and is therefore considered inferior to prophylactic strategies for long-term outcomes.
Despite the increasing clinical utilization of and vast differences between non-clotting factor prophylaxis and on-demand therapy, data on their comparative effectiveness remains sparse. With the rising use of these agents and growing interest in cost-effectiveness and personalized care, we conduct a comprehensive systematic review and meta-analysis of randomized controlled trials to compare the efficacy and safety of non-clotting factor prophylactic therapies (specifically emicizumab, fitusiran, and concizumab) with traditional on-demand therapy in patients with hemophilia A or B. A network meta-analysis was then also conducted to indirectly compare non-factor agents. The goal of our findings is to further clarify the clinical value of non-clotting factor prophylaxis and inform treatment selection in hemophilia cases as treatments continually evolve.
Methodology
Study Design and Registration
This systematic review and meta-analysis was conducted according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
8
The protocol was registered in PROSPERO (ID:
Data Sources and Search Strategy
Eligible studies for analysis were identified through a comprehensive search of databases such as PubMed, Cochrane CENTRAL and ClinicalTrials.gov. A specific search strategy was used to narrow in on eligible studies until May 30th, 2025, were human studies, and that were written in English. Search terms included combinations of: “non-clotting factor prophylaxis”, “Fitusiran”, “Emicizumab”, “Concizumab”, “hemophilia”, and “on demand therapy”.
Study Selection
Upon retrieval, the collected articles were meticulously screened further for duplications and relevance to the topic using the Rayyan web version for the primary and secondary screening.
10
The predefined inclusion and exclusion criteria listed in Section 2.4 guided the reviewers in determining the final articles to be used in the meta-analysis. From an initial pool of 999 records, 168 duplicates were removed. The remaining 831 articles were then screened by title and abstract. Of these, 764 studies were excluded for not meeting the inclusion criteria. A total of 67 full-text articles were reviewed, and 61 were excluded either for not being a double-arm RCT or not reporting the outcomes of interest. Ultimately, 6 RCTs were included in this meta-analysis. Some of the included trials (eg, HAVEN 1, HAVEN 3) had multiple arms or sequential enrollment periods. These trials were parallel in design,
Inclusion and Exclusion Criteria
The inclusion criteria for article selection were defined as follows:
Only double-armed randomized controlled trials (RCTs) were considered for inclusion in this meta-analysis. These studies provided direct head-to-head comparison between Non-clotting factor prophylactic therapies and On-demand therapy. The selected studies featured a specific patient population, namely patients with hemophilia A or B regardless of inhibitor status. Although patients of varying disease severity were eligible, most enrolled participants in the included trials had severe hemophilia (factor activity <1%), reflecting the design of the randomized studies. Within the outcomes of the selected studies, at least one of the following must be reported: ABR (for all treated bleeds or spontaneous or joint bleeds), Haem-a-QoL total score, TEAE and TSAE, proportion of zero and ≤3 bleeds. The language for any of the selected studies was English.
The exclusion criteria for article selection were defined as follows:
Non-comparative designs (eg, single-arm studies, cross-over designed studies) were not considered. Publications in the forms of case reports, editorials, conference abstracts, observational studies and short communications were not considered. Publications with a lack of extractable outcome data were not considered.
Data Extraction
Data were extracted independently by two reviewers using a standardized form. To ensure precision and agreement, discrepancies were resolved by consensus. The variables of interest included study design, sample size, publication year, patient demographics and baseline characteristics, and outcome data for both intervention and control arms at baseline and longest follow-up.
Risk of Bias Assessment
In order to decrease risk of bias in studies chosen for meta-analysis, the quality of chosen studies was assessed using the Cochrane RoB tool 2.0 for randomized controlled trials. 11 (Supplementary S2)
Data Analysis
The statistical analysis for this meta-analysis was conducted using Version 5.4.1 of the Rstudio software. The meta, netmeta, metafor, and dmetar packages were utilized. A random-effects model (REML) was used to pool outcomes as mean differences (MD) or risk ratios (RR) with 95% confidence intervals (CIs). This accounted for potential variability across studies. In order to ascertain the degree of heterogeneity among the selected studies, the Higgins I2 test, Cochran's Q test, and τ2 were utilized. Significant heterogeneity was considered at I2 > 50%. P-value was used as an indicator of statistical significance with a value of less than 0.05 suggesting a significant difference. The primary outcome of the study, Annualized bleeding rate (ABR) is a rate-based outcome. While individual trials reported ABR as mean values with confidence intervals, differences in how ABR was calculated made pooling absolute ABR values inappropriate. Therefore, ABR outcomes were synthesized using pooled risk ratios comparing non-clotting factor prophylaxis with on-demand treatment. In addition, an exploratory
Results
The review includes 6 randomized controlled trials involving 403 patients with congenital hemophilia (A/B) and 3 therapeutic regimens, including Emicizumab, Concizumab and Fitusiran. The following randomized trials were included in this meta-analysis: HAVEN 1, 12 HAVEN 3, 13 ATLAS-INH, 14 ATLAS-A/B, 15 Explorer7, 16 and Explorer8. 17 A detailed PRISMA 2020 flow diagram is presented in Figure 1, that walks through the complete selection process. Across the six included randomized controlled trials, 320 participants (79.4%) had hemophilia A and 83 participants (20.6%) had hemophilia B. The age of the patients ranged from 12–75 years old. Participants in the included studies had congenital hemophilia A or B, with or without the presence of factor VIII or IX inhibitors. The number of White patients was (n = 95/243, 39%) across 3 studies, while Asian patients were (n = 139/243, 57.2%) across the same studies. Hispanic or Latino patients were (n = 7/183, 3.8%) and Non-Hispanic/Latino patients were (n = 169/183, 92.3%), both reported in 2 studies. The number of bleeding events in the past 6 months before screening ranged from 7 to 25, as reported in three studies. The median follow-up duration of the included studies ranged from 24 weeks to approximately 13 months. For the quality assessment, ATLAS–A/B, HAVEN 1, HAVEN 3, Explorer 8 showed some concerns overall, EXPLORER 7 and ATLAS–INH demonstrated low risk across all domains (Supplementary S2).
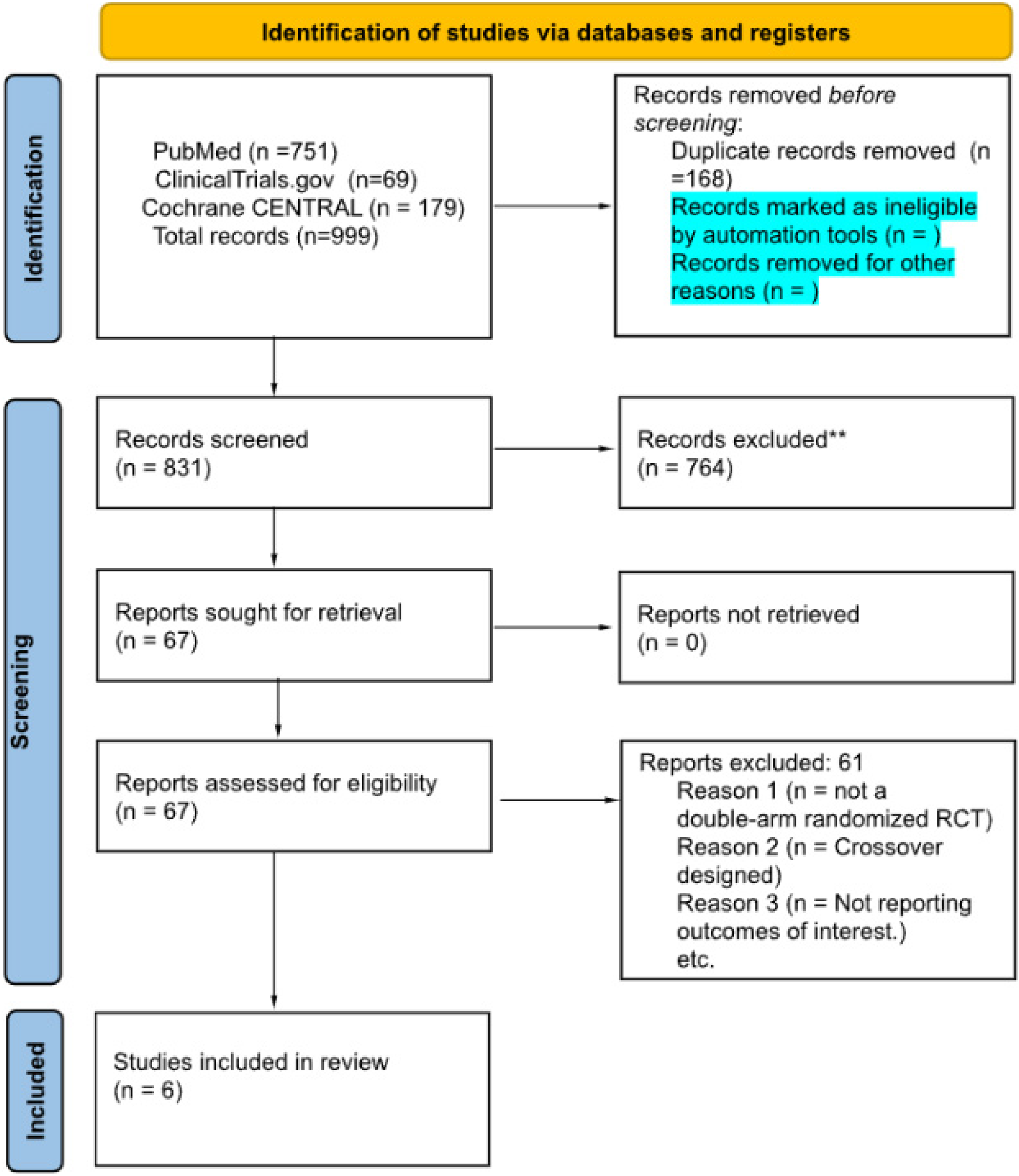
PRISMA 2020 flow diagram of study selection: flow diagram showing identification, screening, eligibility assessment, and inclusion of studies in the meta-analysis, with reasons for exclusion at each stage.
A full description of the baseline characteristics of the included studies is presented in Table 1.

Values are reported as median (interquartile range), mean ± standard deviation, or number (percentage), as provided by the original studies. NR indicates not reported.
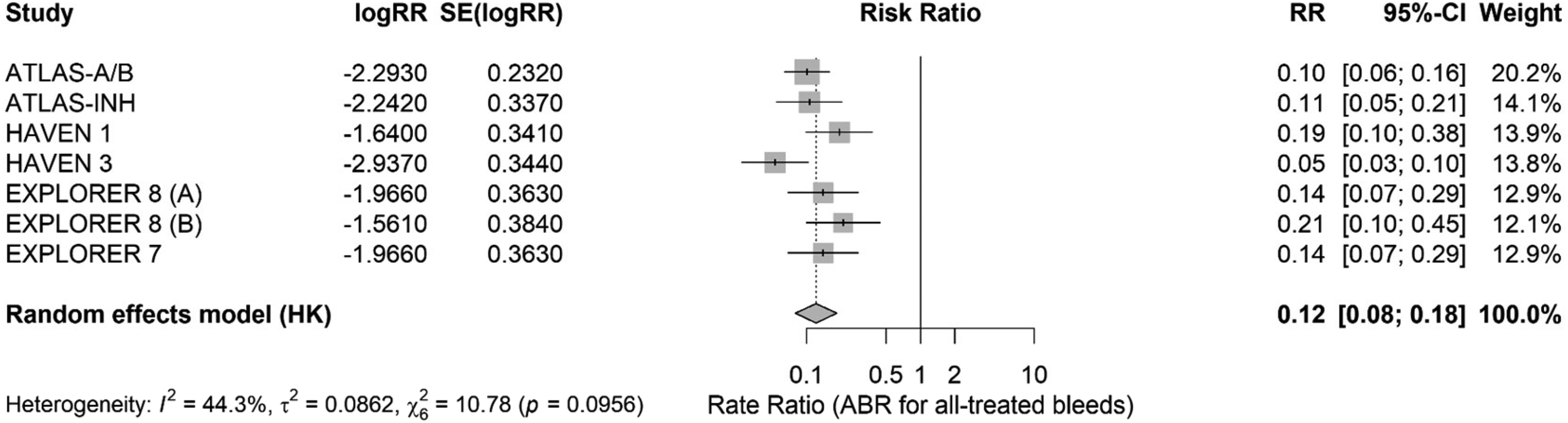
Forest plot of annualized bleeding rate (ABR) for all treated bleeds: individual study risk ratios (RRs) with 95% confidence intervals are shown, pooled using a random-effects model. The diamond represents the overall pooled effect, and study weights are indicated by box size.
However, the heterogeneity analysis confirmed the robustness of the results, with the effect estimates ranging from 0.12 to 0.14, with no single study altering the statistical significance. Egger's test showed no significant publication bias (p = 0.9828), further supported by symmetry on funnel plot inspection.
Subgroup analyses were conducted to explore sources of heterogeneity across intervention type, hemophilia subtype, and inhibitor status. No statistically significant difference in treatment effects was observed between intervention subgroups overall (p = 0.0510); however, fitusiran (RR = 0.13, 95% CI 0.09-0.19) and concizumab (RR = 0.22, 95% CI 0.14-0.37) showed no heterogeneity (I2 = 0%), while emicizumab demonstrated substantial variability across studies (RR = 0.10, 95% CI 0.07-0.15; I2 = 86.1%, τ2 = 0.7247, p = 0.0074). When stratified by hemophilia subtype, prophylaxis produced consistent reductions in all-treated bleeds among patients with mixed hemophilia (RR = 0.11), hemophilia A (RR = 0.11), and hemophilia B (RR = 0.21), with no significant subgroup interaction (p = 0.26). Treatment effects were similarly stable across inhibitor status, with pooled RRs of 0.11 for inhibitor-negative and 0.14 for inhibitor-positive patients with no statistically significant subgroup interaction (p = 0.43) (Supplementary S3). Collectively, these results indicate that non–clotting factor prophylaxis provides robust and consistent bleeding protection across drug classes and clinical subgroups, regardless of hemophilia type or inhibitor status.

Forest plot of annualized bleeding rate (ABR) for spontaneous bleeds: this plot displays study-specific and pooled risk ratios with 95% confidence intervals derived from a random-effects model, summarizing the effect on spontaneous bleeding episodes.
The evaluation of ABR for spontaneous bleeds, conducted across five studies involving 336 patients, revealed that non-clotting factor prophylactic therapy had a significantly lower ABR compared to on-demand therapy [RR = 0.08; 95% CI: (0.06, 0.11), I2 = 0.0%, p = 0.5933)] (Figure 4). The heterogeneity analysis confirmed the robustness of the results, with no single study significantly altering the effect estimates. Egger's test showed no significant publication bias (p = 0.8418).

Forest plot of Haem-A-QoL total score: mean differences (MDs) with 95% confidence intervals are shown for individual studies comparing prophylactic non-clotting factor therapy with on-demand treatment. Estimates are pooled using a random-effects model. Negative values indicate improvement in health-related quality of life as measured by the Haem-A-QoL total score. The diamond represents the overall pooled effect, and box sizes correspond to study weights.
All the remaining forest plots, sensitivity analyses, and funnel plots are present in the Supplementary S3

Risk ratios (RR) are reported for bleeding and safety outcomes, and mean differences (MD) are reported for health-related quality of life.
Discussion
This is the first comprehensive systematic review and meta-analysis that presents strong evidence in favour of non-clotting factor prophylactic therapies compared predominantly with traditional on-demand treatment in hemophilia patients, while recognizing that factor-replacement prophylaxis remains the standard of care in many clinical settings. Previously, most of the studies have focused on single agents in isolation. For instance, A systematic review on non-clotting factor prophylactic therapies by Olasupo et al (2024) lacked pooled estimates or a network meta-analytic framework. These prophylactic agents (emicizumab, fitusiran, and concizumab) have shown a consistent and statistically significant reduction in the bleeding episodes, as reported by the annualized bleeding rates (ABR) for all treated, spontaneous and joint bleeds. This is in line with the findings from the pivotal RCTs such as HAVEN 1, HAVEN 3, ATLAS-A/B, and EXPLORER 8, which each independently reported significant efficacy for their respective agents in reducing the bleeding rates.12–15,17 Although these results are clinically meaningful, they also indicate a transformation shift in the therapeutic landscape of hemophilia, particularly for cases complicated by the presence of inhibitors or those that seek alternatives to intravenous factor replacement.
Other than the clinical benefits in reduction of bleeding episodes, the use of non-clotting factor prophylaxis also results in a significant advancement in the living experience of hemophilia patients. As captured through validated measures like the Haem-A-QoL total score, improvements have been recorded in the health-related quality of life (HRQoL). 18 However, substantial heterogeneity was observed for Haem-A-QoL and zero-treated bleed outcomes, and evidence of publication bias was detected for one outcome; therefore, these results should be interpreted cautiously. This reveals that these therapies also help alleviate the emotional, psychological and social discomfort that accompanies the bleeding episodes. Above all, its mode of subcutaneous administration, particularly with agents like emicizumab, supports consistent adherence by the patients, all of which contribute to a better quality of life. 19
Clinical Implications
The shift from on-demand treatment to prophylactic therapy in hemophilia patients has gained recognition, driven by the fact that preventing bleeding episodes, particularly those involving the major joints is critically important in slowing the progression of chronic, irreversible complications i.e hemophilic arthropathy, chronic pain or functional disability. Our study strongly addresses this shift by demonstrating that, apart from reducing the frequency of bleeds, non-clotting factor prophylactic agents do so across multiple subtypes of bleeding with high statistical and clinical significance.
These agents act through distinct mechanisms to restore thrombin generation without direct factor replacement, allowing for personalized treatment approaches in patients with inhibitors, poor venous access, or adherence challenges.
Non-clotting factor therapies offer an important advantage by bypassing limitations of conventional factor VIII or IX replacement, particularly the development of neutralizing inhibitors. 12 These agents act through distinct mechanisms to restore thrombin generation without direct factor replacement, allowing for personalized treatment approaches in patients with inhibitors, poor venous access, or adherence challenges.20–22
Beyond exceptional control of bleeding, the reported improvement in the health-related quality of life (HRQoL), based on the Haem-A-Qol total scores, highlights the other effects of these treatments. In contrast with on-demand regimens that result in frequent interruptions in the daily routine activities, missing school or work days, and imposed limitations on physical activity, prophylactic drugs provide a more stable course of the disease which aligns with the HAVEN-3 trial findings. 13 This fastens the functional recovery process and assures more involvement in physical, educational and social activities and helps bring down the psychological burden and anxiety caused by the fear of spontaneous bleeding.
Safety Considerations
Although no statistically significant difference in the overall rate of treatment-emergent serious adverse events (TESAEs) was observed between prophylaxis and control groups, the higher frequency of TEAEs in the prophylaxis groups requires careful interpretation. TEAEs encompass a wide spectrum of events, and many reported in the included trials were mild such as injection-site reactions, mild constitutional symptoms, or minor laboratory abnormalities rather than clinically meaningful safety concerns. Only a small proportion represented moderate or severe events, and discontinuations due to adverse effects were uncommon. Therefore, while prophylaxis is associated with a greater number of reported treatment-emergent adverse events, these appear to be predominantly low-grade events, with no corresponding increase in serious adverse events.
Comparison Among Non-Clotting Factor Agents
The network meta-analysis did not reveal significant differences in the ABR among emicizumab, fitusiran, and concizumab. Therefore, it suggests that all three agents may provide comparable clinical benefits; however, the network meta-analysis is exploratory in nature and characterized by a star-shaped structure that lacks closed loops and reduces confidence in indirect comparisons between active agents. Consequently, the ability to establish equivalence or hierarchical ordering among non-clotting factor agents is limited, and these estimates should be interpreted with caution due to the absence of trials providing direct comparisons and the practical limitations of conducting such studies in a rare disease such as hemophilia.
Strengths and Limitations
The notable strengths of this study include: (1) It is the first one to combine all currently available RCTs comparing non-clotting factor prophylaxis to on-demand therapy, while also indirectly comparing the most relevant prophylactic agents via a network meta-analysis. (2) Methodological rigor was maintained through adherence to PRISMA guidelines, controlled and supervised data extraction, and use of up-to-date risk-of-bias tools.
However, limitations do also exist: (1) The total number of included RCTs and patients remains small or modest, particularly for new agents like concizumab and fitusiran, which have limited long-term follow-up data. (2) Second, heterogeneity was reported in some outcomes, such as Haem-A-QoL and ABR for spontaneous bleeds, which most likely are due to differences in patient characteristics, and follow-up duration. (3) The methodologic quality of the included studies was suboptimal, with the majority having a moderate risk of bias. (4) Generalisability is limited, as most included trials enrolled younger, trial-compliant patients; real-world adherence and outcomes in older or more complex populations may differ. (5) Although this analysis focused on comparisons between non-clotting factor prophylaxis and on-demand therapy, factor replacement prophylaxis remains the standard comparator in many clinical settings due to cost-effectiveness. In addition, non-clotting factor therapies are associated with substantial costs, which may influence treatment selection even in resource-rich healthcare systems.
Conclusion
Compared to on-demand therapy, non-factor prophylactic therapies significantly reduce bleeding episodes, improve quality of life, and increase the likelihood of zero bleeds in patients with hemophilia. Although associated with a higher rate of adverse events, the therapies themselves did not increase the risk of serious adverse events. These findings support the potential clinical utilization of non-factor prophylaxis in managing hemophilia A and B, regardless of inhibitor status, while acknowledging the limitations of indirect comparisons and outcome heterogeneity.
Supplemental Material
sj-docx-2-cat-10.1177_10760296261425039 - Supplemental material for Comparative Efficacy and Safety of Non-Clotting Factor Prophylaxis Versus. on-Demand Therapy in Hemophilia: A Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-2-cat-10.1177_10760296261425039 for Comparative Efficacy and Safety of Non-Clotting Factor Prophylaxis Versus. on-Demand Therapy in Hemophilia: A Meta-Analysis of Randomized Controlled Trials by Rehan Ishaque, Abdul Subhan Talpur, Huda Memon, Laksh Ahuja, Fnu Reya, Amna Ikram, Muhammad Ahsan Siddiqui, Manahil Ali, Hamza Danish, M Raafe Ali Khan, Raghad El joujou, Hamza Janjua, Hira Janjua, Aswanth Reddy and Supratik Rayamajhi in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-csv-3-cat-10.1177_10760296261425039 - Supplemental material for Comparative Efficacy and Safety of Non-Clotting Factor Prophylaxis Versus. on-Demand Therapy in Hemophilia: A Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-csv-3-cat-10.1177_10760296261425039 for Comparative Efficacy and Safety of Non-Clotting Factor Prophylaxis Versus. on-Demand Therapy in Hemophilia: A Meta-Analysis of Randomized Controlled Trials by Rehan Ishaque, Abdul Subhan Talpur, Huda Memon, Laksh Ahuja, Fnu Reya, Amna Ikram, Muhammad Ahsan Siddiqui, Manahil Ali, Hamza Danish, M Raafe Ali Khan, Raghad El joujou, Hamza Janjua, Hira Janjua, Aswanth Reddy and Supratik Rayamajhi in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviations
Acknowledgment
None.
ORCID iDs
Author Contribution
RI was involved in conceptualization, supervision, writing the original draft, and manuscript review. AST was responsible for data curation, interpretation of results, manuscript writing, editing, and revision. HM was responsible for the literature search and data curation. LA and HJ curated data and performed statistical analysis. HD, AST, HJ curated data and generated tables. LKA and HJ were responsible for reviewing and editing the final draft.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data extracted and analyzed is presented in the manuscript. For additional information, please reach out to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
