Abstract
Background
Replacing bioprosthetic valves is common but can cause coagulation issues. This study assesses the safety and efficacy of Direct oral anticoagulants (DOACs) versus vitamin K antagonists (VKAs) in bioprosthetic valve procedures.
Methods
PubMed, Cochrane Central, and ScienceDirect were searched till May 2025. The risk ratios (RRs) were pooled with 95% confidence intervals (CIs) under the random effects model using Review Manager software. The quality assessment was conducted using the Cochrane risk of bias (RoB 2.0) tool.
Results
Eight randomized controlled trials involving 3863 patients were included in the meta-analysis. DOACs were associated with a significant reduction in the risk of stroke or systemic embolism compared with VKAs (RR = 0.48; 95%CI: [0.26, 0.88]; p = .02). The rate of major bleeding was similar between the DOACs and VKAs (RR = 0.92; 95%CI: [0.49, 1.72]; p = .79). There was no significant difference in the incidence of any stroke between the two groups (RR = 0.65; 95%CI: [0.32, 1.31]; p = .23). The occurrence of clinically relevant non-major bleeding (RR = 1.17; 95%CI: [0.97, 1.40]; p = .10), all-cause mortality (RR = 0.94; 95%CI: [0.75, 1.17]; p = .57), and intracranial hemorrhage (ICH) (RR = 0.71; 95%CI: [0.40, 1.26]; p = .25) were comparable between the groups.
Conclusion
DOACs seem safe after bioprosthetic valve implantation, lowering the risk of stroke or systemic embolism. However, rates of major bleeding, clinically relevant non-major bleeding, any stroke, ICH, and all-cause mortality were comparable between DOACs and VKAs.
Keywords
Introduction
Bioprosthetic valve replacement is a common procedure for patients with structural heart disease requiring valve intervention. 1 Each year, approximately 300,000 heart valve surgeries are conducted globally, serving as the primary intervention for valvular diseases. 2 Bioprosthetic valves reduce the need for lifelong anticoagulation but are associated with several complications affecting long-term outcomes. One of the important complications of valve replacement is coagulation disturbance.3,4 Universal anticoagulation after bioprosthetic aortic valve replacement is debated due to unclear benefits and bleeding risks, leading to a Class IIb recommendation in ACC/AHA guidelines. 4 Other complications include structural valve deterioration (SVD), pannus formation, and several other hemodynamic changes, which can impact long-term valve function.5,6 However, despite the widespread use of bioprosthetic valve replacement, data on these thrombotic risks remain scarce, largely because such events appear to be infrequent. 7
Various pharmacological agents and techniques such as dual antiplatelet agents, vitamin K antagonists (VKAs), cerebral embolic protection devices have been used in patients with valve implantation or replacement.8,9 The use of direct oral anticoagulants (DOACs) in patients with bioprosthetic valve replacement is gaining attention as an alternative to VKAs. Traditionally, VKAs like warfarin have been the standard anticoagulants due to their established efficacy in preventing thromboembolic events. 10 However, they have a narrow therapeutic window and rigorous monitoring of international normalized ratio (INR), posing a high risk of bleeding. 11 DOACs, on the other hand, offer advantages such as predictable pharmacokinetics, fewer dietary restrictions, making them an attractive option.12,13
Extensive research has been conducted to confirm the safety of DOACs in post-op stroke and atrial fibrillation. Malik et al found a significant decrease in stroke and systemic embolism risk in the DOAC arm in patients with atrial fibrillation and valvular heart disease. 14 Cheema et al reported that the risk of stroke and systemic embolism with DOACs is comparable to that of left atrial appendage occlusion in atrial fibrillation patients. 15 Qamar et al demonstrated that DOACs significantly lower stroke risk in atrial fibrillation patients. 16 Bioprosthetic valve replacement can be performed using surgical or transcatheter methods, which differ in invasiveness and patient suitability. The risk of thrombosis varies by valve location, with a higher risk in mitral valves than in aortic valves. 17 Additionally, the risk of thromboembolism after bioprosthetic valve replacement is highest in the first 10 days and gradually decreases as the valvular surface endothelializes. 18
Limited evidence exists regarding the safety and effectiveness of DOACs after bio-prosthetic valve replacement. Meta-analyses by Bakr, Eikelboom, and Yokoyama found no significant stroke reduction, leaving their role unconfirmed.19–21 However, a 2025 trial showed that edoxaban improved stroke outcomes. 22 Therefore, we conducted an updated meta-analysis by integrating high-quality evidence from randomized controlled trials (RCTs) to generate a reliable pooled estimate of the efficacy and safety of DOACs versus VKAs in patients undergoing bioprosthetic valve replacement. The findings will provide insights into optimal anticoagulation strategies for this patient population, informing clinical decision-making and future guideline recommendations.
Methods
The systematic review and meta-analysis adhered rigorously to the established guidelines formulated by the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA).
23
This review also followed the Cochrane Handbook for Systematic Reviews of Interventions
24
and was registered on PROSPERO under the ID:
Data Sources and Search Strategy
Two authors (K.F. and A.N.) independently searched PubMed, Cochrane Central, and ScienceDirect from inception to May 2025 for studies that assessed the safety and efficacy of DOACs and VKAs following surgical or transcatheter bioprosthetic valve replacement or repair. Additionally, the bibliographies of the identified articles and previous meta-analyses were manually reviewed to identify relevant studies. The Mesh terms and keywords used were: “Direct-Acting Oral Anticoagulant,” “Factor Xa Inhibitors,” Edoxaban,” “Apixaban,” “Rivaroxaban,” “Dabigatran,” “Bioprosthetic valve implantation,” and “Bioprosthetic valve replacement.” The search strings used for each database are given in Supplementary Table 1.
Study Selection and Eligibility Criteria
All the articles were imported into EndNote Reference Library software, and duplicates were removed. Two authors (H.A.A.R. and Z.U.A.) independently screened the titles and abstracts of all the retrieved articles and removed those that did not fulfil the inclusion criteria. Full texts of the remaining articles were reviewed against the eligibility criteria. Disagreements regarding the study selection process, such as differing interpretations of eligibility criteria that the two authors could not resolve, were addressed through discussions with the third reviewer (M.H.W.).
We included studies in our systematic review and meta-analysis if they met the following eligibility criteria: (1) Study design: double-arm RCT; (2) Population: Adults (age > 18years) undergoing surgical or transcatheter bioprosthetic valve replacement or repair; (3) Intervention: DOACs administration following the procedure; (4) Comparison: VKAs administration following the procedure; (5) Primary outcomes: All-cause mortality, any stroke, stroke or systemic embolism, intracranial haemorrhage, and bleeding events (major, or clinically relevant non-major) as defined by the International Society on Thrombosis and Haemostasis.
We excluded the following studies: (1) Study design: Case reports, case series, editorials, systematic reviews, narrative reviews, observational studies, and animal studies; (2) Population: children or adolescents (<18 years), and patients with the presence of a mechanical valve.
No language restrictions were applied.
Data Extraction
Data regarding the baseline characteristics and outcomes of interest were extracted on an Excel spreadsheet. The information collected included various study details such as the first author's surname, publication year, study design, location, the procedure performed to insert bioprosthetic valve (surgical or transcatheter), the valve replaced or repaired in the procedure (aortic or mitral), mean follow-up period, demographic information about the participants such as age, gender; comorbidities like diabetes, hypertension, ischemic heart disease; medical history including history of any cerebrovascular stroke and mean CHADVasc scores.
The outcomes extracted were all-cause mortality, any stroke, stroke or systemic embolism, intracranial haemorrhage, and bleeding events (major, or clinically relevant non-major) as defined by the International Society on Thrombosis and Haemostasis. The data was extracted independently by two authors (A.H.C. and K.F.), and any discrepancies were resolved through discussion with a third author (M.H.W.).
Quality Assessment
We evaluated the quality of the included randomized controlled trials using the revised Cochrane risk of bias tool (RoB 2.0). 25 The RoB 2.0 tool evaluates trial quality across five domains: (1) bias from the randomization process, (2) bias from deviations in the intended intervention, (3) bias due to missing outcome data, (4) bias in outcome measurement, and (5) bias in selecting reported results. The risk of bias in the included studies was classified as high, low, or some concern. Studies were deemed low risk when their methodology was clear, including proper randomization and other aspects. They were labeled high risk if they had major methodological flaws, such as improper randomization or improper handling of bias in the other domains. Studies with unclear reporting on randomization or other domains of the RoB 2.0 scale received a rating of “some concerns.” Two authors (M.Z.T. and F.S.) independently assessed the risk of bias, and any disagreements about the quality assessment were resolved through discussion with a third author (M.H.W.).
Statistical Analysis
We conducted the statistical analysis on Review Manager (RevMan) software version 5.4.1. Clinical outcomes were evaluated using the Mantel-Haenszel random-effects model to pool risk ratios (RRs) with 95% confidence intervals (CIs). The pooled results were represented graphically as forest plots. The Chi-square test and the Higgins I2 statistics 26 were calculated to evaluate the statistical heterogeneity. Heterogeneity (I2) values < 50% were considered acceptable. Subgroup analyses based on the type of procedure (surgical valve replacement/transcatheter valve replacement/both), the type of DOACs (apixaban, edoxaban, rivaroxaban, and dabigatran), and the valve replacement sites (aortic valve, mitral valve, and combined) were performed. A p-value of <.05 was considered statistically significant. For outcomes with heterogeneity >50%, a leave-one-out sensitivity analysis was performed to investigate the cause of heterogeneity. Meta-regression was also performed to investigate the sources of heterogeneity. Publication bias was assessed visually using funnel plots and statistically using Egger's regression test in Comprehensive Meta Analysis V3. GRADE assessment was performed to assess the certainty of evidence using the GRADEpro GDT. 27
Results
Search Results
The initial search retrieved 371 articles. After removing 92 duplicates, 279 articles were screened by title and abstract, resulting in 69 articles. These were then examined in full text, leading to 8 articles22,28–34 included in the meta-analysis. The search and selection process is summarized in the PRISMA flow chart (Figure 1).
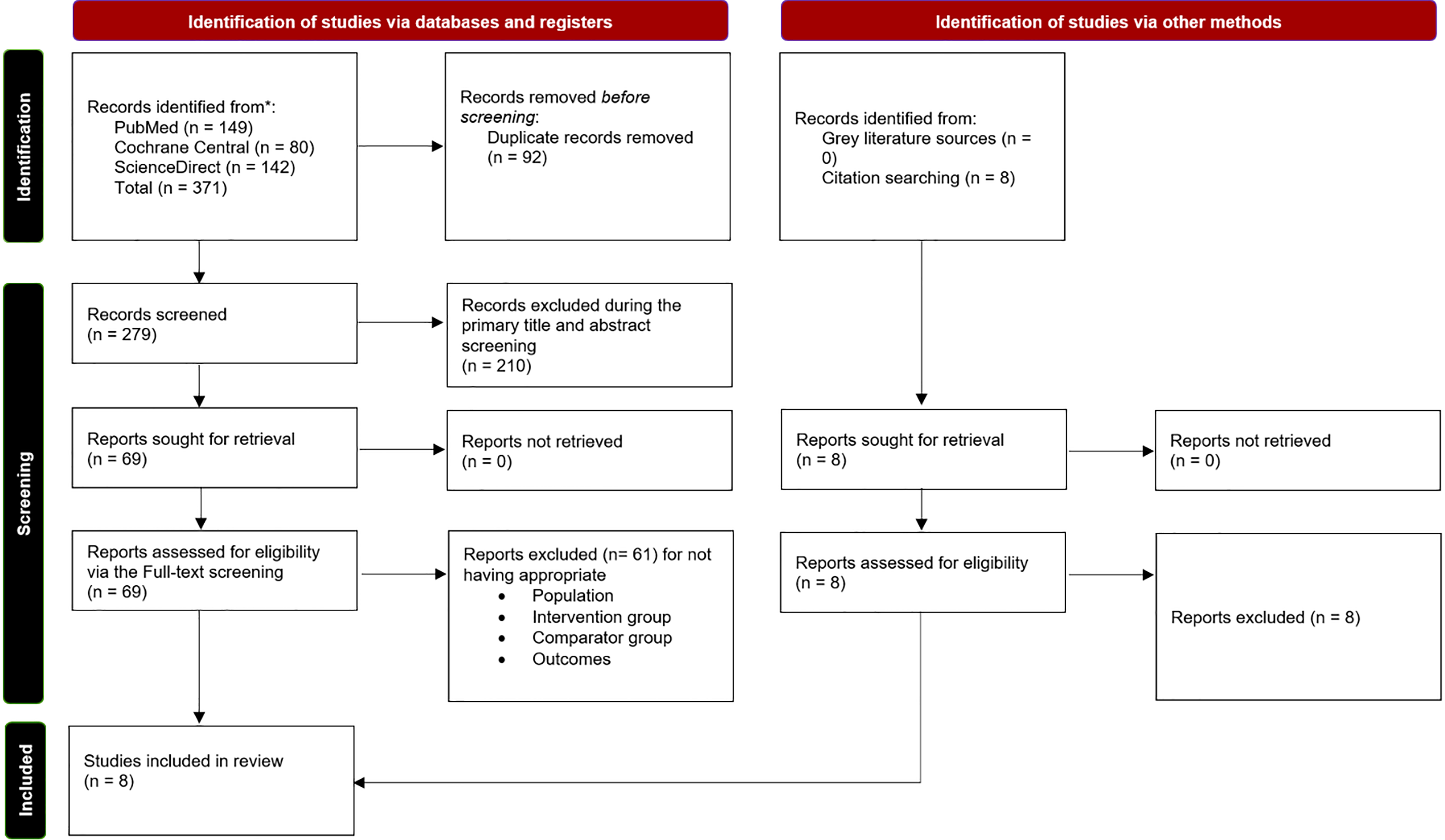
PRISMA flowchart of the study selection process.
Characteristics of Included Studies
Most studies evaluated the effect of DOACs versus VKAs on bleeding following surgical or transcatheter valve replacement and repair. There were 3863 subjects from 8 studies, of whom 1963 (50.81%) received DOACs, and 1900 (49.18%) received VKAs. Baseline characteristics of the participants are shown in Table 1. The mean age of most of the patients was greater than 60. The incidence of diabetes and hypertension ranged from 0% to 37.9% and 46.7% to 92.8% respectively. The incidence of ischemic heart disease and prior cerebrovascular stroke ranged from 4.8% to 41.7% and 6% to 33.3% respectively. Most studies had comparable male and female populations. The mean CHADVasc score ranged from 2.1 to 4.7.
Baseline Characteristics of the Included Studies.

Median reported.
Abbreviations: RCT, randomised controlled trial; DOAC, direct oral anticoagulant; VKA, vitamin K antagonist; DM, diabetes mellitus; HTN, hypertension; IHD, ischemic heart disease; CVS, cerebrovascular stroke; TAVI, transcatheter aortic valve implantation.
Most studies focused on bioprosthetic valves implanted through surgery, except for Collet et al 34 and Van Mieghem et al, 33 which focused exclusively on Transcatheter Aortic Valve Implantation (TAVI). Carnicelli et al included patients who had either undergone surgery or transcatheter valve replacement. 28 Most trials studied the impact of DOACs versus VKAs on patients who had undergone bioprosthetic valve implantation in either the mitral or aortic valve location, except for Collet et al and Van Mieghem et al, which focused exclusively on the aortic valve.33,34 Guimaraes et al 30 only focused on the mitral valve. Two studies, Guimaraes et al 31 and Shim et al, 32 also examined valve repair in addition to valve replacement. The follow-up period ranged from 3 months to 2.8 years. Table 1 summarizes the baseline characteristics of the included studies. The inclusion and exclusion criteria for the studies included in this meta-analysis are listed in Supplementary Table 2.
Quality Assessment
We assessed study quality using the Cochrane risk of bias tool RoB (2.0) for the eligible RCTs. Most of the trials had a low risk of bias, except for Duraes et al 29 and Guimaraes et al, 30 which had some concerns due to deviation from the intended intervention. Overall, the studies had low risk of bias, except for some concerns related to deviations from the intended intervention. The quality assessment of the RCTs is depicted in the traffic plot (Figure 2).

Risk of bias traffic plot for quality assessment of the randomized controlled trials.
Outcomes
The summary of the meta-analysis is provided in Table 2, while the main endpoints reported in the individual studies, along with their results, are listed in Supplementary Table 3. The differences between our meta-analysis and the previous one are systematically shown in Supplementary Table 4.
Summary of Meta-Analysis.

Abbreviations: RR, risk ratio; CI, confidence interval; df, degree of freedom.
Major Bleeding
Major bleeding was reported by 6 of the included studies, pooling a total of 3455 (1725 DOACs vs 1730 VKAs). Our results show that following surgical or transcatheter valve replacement, DOACs do not reduce the incidence of major bleeding significantly as compared to VKAs (RR = 0.92; 95%CI: [0.49, 1.72]; p = .79; I2 = 60%) (Figure 3A).
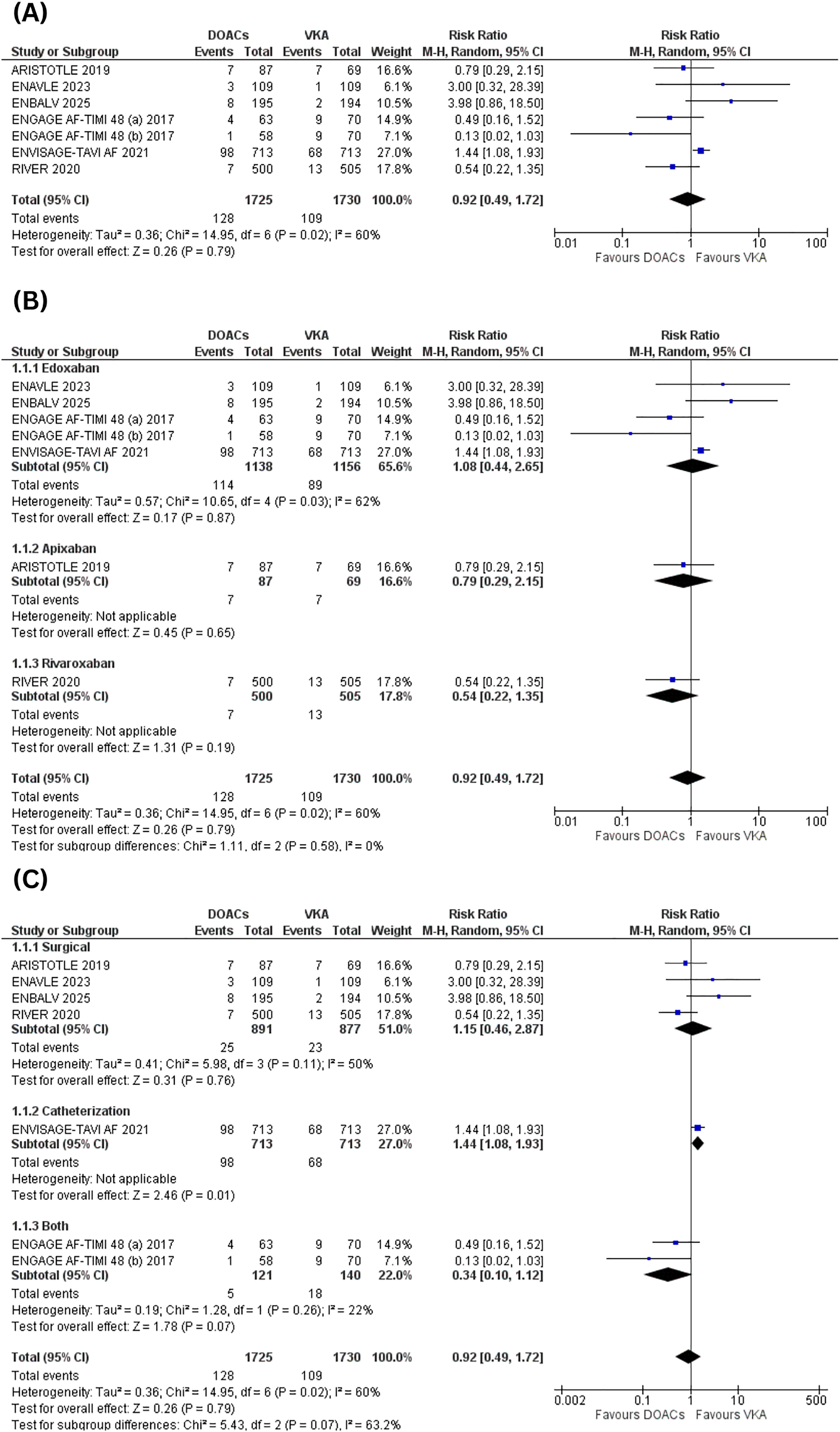
Major bleeding (A) forest plot, (B) subgroup analysis based on the DOACs subtype, (C) subgroup analysis based on the route of valve implantation.
Stroke or Systemic Embolism
This outcome was reported by five of the included studies, pooling a total of 1838 (918 DOACs vs 920 VKAs). Compared to VKAs, DOACs were associated with a significant reduction in stroke or systemic embolism (RR = 0.48; 95%CI: [0.26, 0.88]; p = .02; I2 = 0%) (Figure 4A).

Stroke or systemic embolism (A) forest plot, (B) subgroup analysis based on the DOACs subtype, (C) subgroup analysis based on the route of valve implantation.
Clinically Relevant Non-Major Bleeding:
Four of the included studies reported clinically relevant non-major bleeding, pooling a total of 3038 (1517 DOACs vs 1521 VKAs). Our results showed that there was no significant difference between the two groups, showing no benefit of DOACs over VKAs (RR = 1.17; 95%CI: [0.97, 1.40]; p = .10; I2 = 0%) (Figure 5A).

Clinically relevant non-major bleeding (A) forest plot, (B) subgroup analysis based on the DOACs subtype, (C) subgroup analysis based on the route of valve implantation.
All-Cause Mortality
All-cause mortality was reported by 7 of the studies included, pooling a total of 3672 (1842 DOACs vs 1830 VKAs). Our results show that there is no significant difference between DOACs and VKAs in terms of all-cause mortality (RR = 0.94: 95%CI: [0.75, 1.17]; p = .57; I2 = 0%) (Figure 6A).
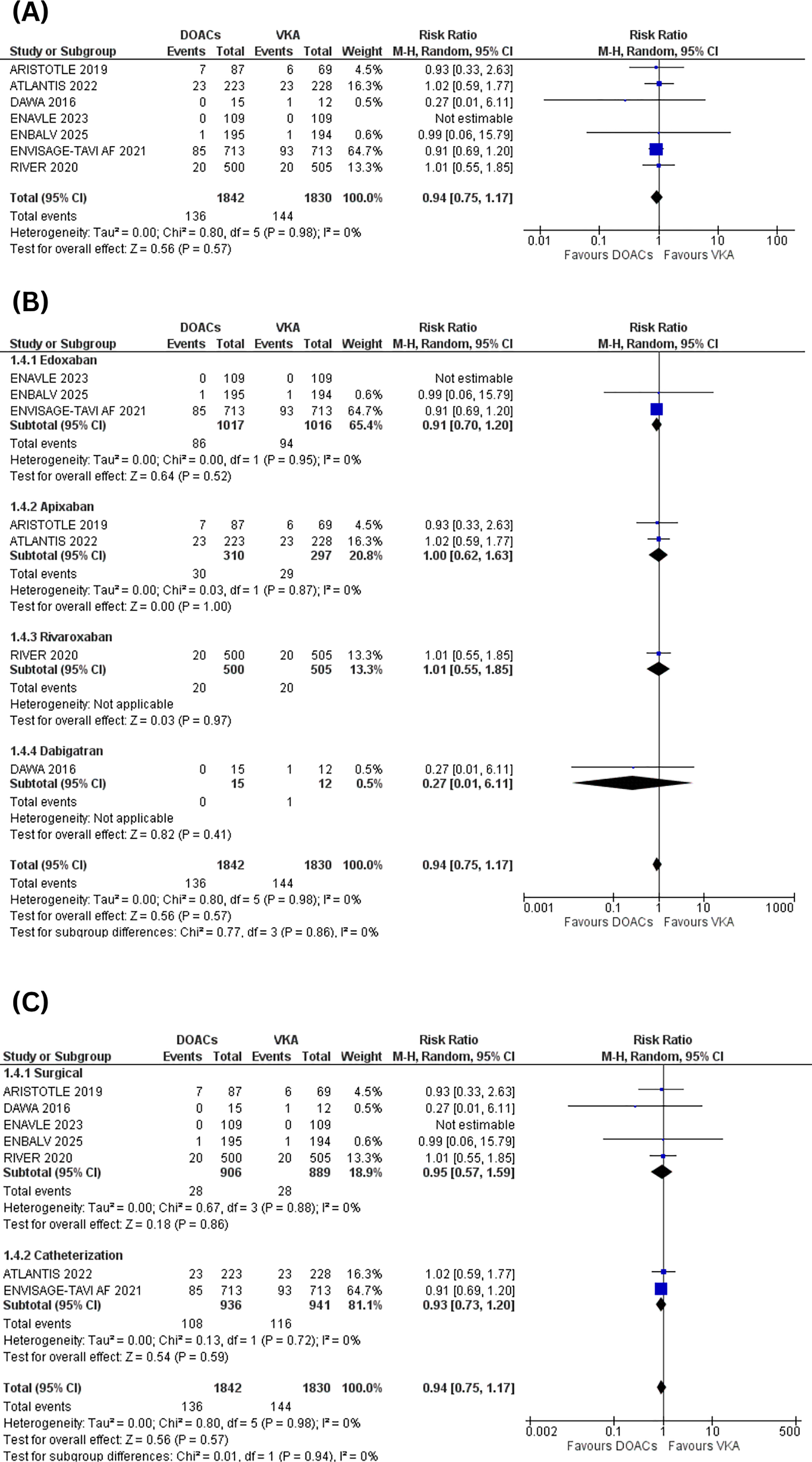
All-cause mortality (A) forest plot, (B) subgroup analysis based on the DOACs subtype, (C) subgroup analysis based on the route of valve implantation.
Any Stroke
The incidence of any stroke was reported by four studies, pooling a total of 2976 (1495 DOACs vs 1481 VKAs). DOACs do not lead to a significant reduction in risk of any stroke as compared to VKAs (RR = 0.65: 95%CI: [0.32, 1.31]; p = .23; I2 = 31%) (Figure 7A).

Any stroke (A) forest plot, (B) subgroup analysis based on the DOACs subtype, (C) subgroup analysis based on the route of valve implantation.
Intracranial Hemorrhage
Six of the included studies reported the incidence of intracranial hemorrhage, pooling a total of 3645 (1827 DOACs vs 1818 VKAs). Our results show that following surgical or transcatheter valve replacement, the incidence of intracranial hemorrhage is not reduced significantly in DOACs as compared to VKAs (RR = 0.71: 95%CI: [0.40, 1.26]; p = .25; I2 = 0%) (Figure 8A). The results of this meta-analysis are shown as a central illustration in Figure 9.

Intracranial hemorrhage (A) forest plot, (B) subgroup analysis based on the DOACs subtype, (C) subgroup analysis based on the route of valve implantation.

Central illustration of the Meta-analysis.
Subgroup Analysis
Subgroup analyses were performed based on the type of procedure (surgical valve replacement/transcatheter valve replacement/both), the type of DOACs (apixaban, edoxaban, rivaroxaban, and dabigatran), and the valve replacement sites (aortic valve, mitral valve, and combined).
In subgroup analysis by type of DOACs, the results are consistent across all outcomes (Figures 3B, 5–6B, 8B). However, the reduction in stroke or systemic embolism is significant only in the Rivaroxaban subgroup (Figure 4B). Additionally, the pooled results for any stroke that were insignificant became significant solely within the Rivaroxaban group (Figure 7B). Since these results are supported by only one study, they should be interpreted with caution.
Regarding subgrouping by procedure type, the results were consistent across all endpoints (Figures 5–8C). However, the risk of major bleeding significantly increased in the transcatheter valve replacement subgroup (Figure 3C). Additionally, the previously significant pooled incidence of stroke or systemic embolism became insignificant when looking at the surgical and combined techniques subgroups (Figure 4C).
On subgrouping based on the site of valve replacement, the results were consistent with the overall results across all endpoints (Supplementary Figures 1-3). However, the risk of major bleeding significantly increased in the aortic valve subgroup (Supplementary Figure 4). Also, the risk of stroke or systemic embolism became insignificant in the combined aortic and mitral valves subgroup (Supplementary Figure 5), whereas the risk of any stroke became significantly decreased in the mitral valve subgroup (Supplementary Figure 6).
Sensitivity Analysis and Meta-Regression
For outcomes having heterogeneity >50% a leave-one-out sensitivity analysis was performed to investigate the cause of heterogeneity. Removing the ENVISAGE-TAVI AF trial 33 decreased heterogeneity from 60% to 49% in the major bleeding outcome. This likely reflects the trial's inclusion of patients undergoing transcatheter valve replacement, who are typically at higher bleeding risk. This could have introduced variability due to differences in bioprosthetic valve replacement procedures (surgical or transcatheter) (Supplementary Figure 7).
Meta-regression analyzing the outcome of major bleeding with age and CHADVasc score as covariates did not show any significant influence, as shown in the Supplementary Figures 8–13.
Publication Bias
The publication bias was assessed visually through funnel plots, which showed no asymmetry. Although the results of Egger's regression test may be unreliable for fewer than 10 studies, it further supported the absence of publication bias (Supplementary Figures 14–25).
GRADE Assessment
The pooled effect size of stroke and systemic embolism showed High GRADE certainty of evidence. The pooled results for the remaining endpoints, including clinically relevant non-major bleeding, all-cause mortality, any stroke, and intracranial hemorrhage, showed Moderate GRADE certainty of evidence, except for major bleeding, which showed Low GRADE certainty. The GRADE summary of findings is presented in Table 3.
GRADE Summary of Findings Table.

The heterogeneity is above 60%.
The 95% confidence interval crosses 1.
Abbreviations: CI, confidence interval; RR, risk ratio.
Discussion
This meta-analysis evaluated 8 RCTs assessing the safety and efficacy of DOACs versus VKAs in patients who underwent bioprosthetic valve implantation or repair, either by surgery or by catheterization. A total of 3863 patients were included, with nearly equal distributions of DOAC recipients (n = 1963) and VKA recipients (n = 1900). Our findings indicated that DOACs showed no significant differences regarding major bleeding, clinically significant non-major bleeding, all-cause mortality, intracranial hemorrhage, and the risk of any stroke compared to VKA. DOACs were associated with a significantly reduced risk of stroke or systemic embolism compared to VKAs, with the Rivaroxaban subgroup demonstrating the most pronounced benefits. Based on the implantation method, subgroup analysis suggested that DOACs significantly raised the risk of major bleeding in patients undergoing transcatheter valve replacement. However, this result should be viewed with caution, as only one study supports this finding. Compared to the previous meta-analyses by Bakr et al, Eikelboom et al, and Yokoyama et al,19–21 our study demonstrated significant reductions in the risk of stroke and systemic embolism, and integrated high-quality evidence from RCTs to provide reliable pooled estimates.
Our findings align with the growing body of research suggesting that DOACs may be a superior option to VKAs for some patients with bioprosthetic valves. VKAs have historically served as the principal means of avoiding thromboembolism post-valve replacement due to their extensive usage in clinical practice and endorsement by guidelines.11,35,36 VKAs possess various drawbacks, including restricted therapeutic windows, the necessity for frequent INR monitoring, and numerous food-drug interactions.37,38 There is interest in utilizing DOACs in valve recipients due to their ease of use, decreased interactions, and more predictable pharmacokinetics.39,40
Outcomes from small-scale trials and prior observational research were often constrained by multiple factors and lacked consistency. The RIVER study by Guimaraes et al assessed mitral bioprostheses in relation to mortality, severe bleeding, and significant cardiovascular events, concluding that rivaroxaban was not inferior to warfarin. 30 Nevertheless, the studies revealed no evidence indicating that any outcomes, such as hemorrhage or cerebrovascular accident, were superior. Our findings corroborated that rivaroxaban appears to reduce the occurrence of embolic events; nevertheless, there was no notable decrease in severe bleeding overall.
As demonstrated by the ENVISAGE-TAVI AF study, unlike VKAs, edoxaban increased the risk of bleeding after transcatheter aortic valve implantation (TAVI). 33 Our research indicated that this trial introduced variability in the major bleeding outcome, which was significantly diminished following its exclusion in a sensitivity analysis. This shows potential variations in DOAC efficacy by drug and population.
In our analysis of subgroups categorized by DOAC subtype, only rivaroxaban consistently showed favorable outcomes in reducing both stroke/systemic embolism and overall stroke incidence. The reduction in the risk of stroke or systemic embolism seen in the rivaroxaban subgroup is probably due to its direct inhibition of factor Xa and high bioavailability, which prevent thrombus formation and lower the risk of stroke or embolism, improving its effectiveness in people at higher risk for thrombosis.41,42Additionally, the once-daily dosing of rivaroxaban helps ensure patients adhere to their medication and maintain a steady anticoagulant effect, which may have contributed to the lower risk. Edoxaban and apixaban possessed analogous structures; nevertheless, they did not provide any statistically meaningful benefits. This may be due to insufficient study power or differing study designs.43–45
Our statistics on all-cause mortality indicated that neither anticoagulation method was superior. This aligns with findings from other studies and meta-analyses: the selection of anticoagulants typically does not influence mortality in the short to medium term following bioprosthetic valve implantation. In these individuals, mortality is primarily attributed to preexisting health conditions and the deterioration of the structural valve, rather than solely to thromboembolic or hemorrhagic events. 46
Our pooled analysis, including all subgroups, did not reveal a significant difference in the risk of intracranial hemorrhage between DOACs and VKAs. This data is intriguing since it contradicts the prevailing belief that DOACs reduce the risk of intracranial hemorrhage compared to VKAs in individuals with atrial fibrillation.47,48 The insignificance observed may be attributed to the infrequency of such instances or the variation in follow-up durations. In summary, our meta-analysis indicates that DOACs may be a safe and effective alternative to VKAs for individuals with bioprosthetic valves, with a lower risk of stroke or systemic embolism. When making determinations regarding anticoagulation, it is essential to consider the specific DOAC selected, the location of the valve, and the method of its implantation.
Strengths and Limitations
This study is more robust because it includes only RCTs, which provide substantial data and reduce bias, although the newly added RCTs have small sample sizes. The comprehensive subgroup analyses enhance our understanding of the impacts of various DOAC medications, implantation techniques and implantation sites on individual outcomes. Furthermore, the GRADE assessment also showed high to moderate certainty of the generated evidence.
Nonetheless, a few limitations are worth mentioning. The results may lack generalizability due to variations in patient cohorts and follow-up durations. The subgroup analyses which considered the procedure type (surgical valve replacement, transcatheter valve replacement, or both), the type of DOACs (apixaban, edoxaban, rivaroxaban, and dabigatran), and the valve replacement sites (aortic valve, mitral valve, and combined) were based on a limited number of studies and might not have enough statistical power to draw definitive conclusions. Additionally, due to limited data, the subgroup analysis based on early versus late valve implantation could not be conducted. Some studies have reported using DOACs for atrial fibrillation in patients with concurrent valve implantation, which might carry a higher thrombotic risk and could contribute to heterogeneity. Our study also did not compare DOACs with other anticoagulation strategies, like dual antiplatelet therapy, or evaluate different DOAC dosages and the timing of DOAC administration after valve replacement.
Future Implications
Our results indicate the necessity for larger, more robust randomized controlled trials that directly compare individual direct oral anticoagulants with vitamin K antagonists in specific bioprosthetic valve contexts. Future research should investigate the optimal timing for initiating anticoagulation, particularly during the perioperative and early post-TAVI phases.
Conclusion
DOACs appear to be a safe and effective alternative to VKAs for patients undergoing surgical or catheter-based bioprosthetic valve implantation or repair. They are associated with a reduced risk of stroke or systemic embolism without significantly increasing bleeding risk. These findings are encouraging; however, physicians must base their decisions on an individual patient's risk assessment and the specifics of the treatment. Larger-scale RCTs are needed to improve and tailor anticoagulant therapies for bioprosthetic valve implantation.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251415375 - Supplemental material for Comparing the Direct Oral Anticoagulants versus Vitamin K Antagonists in Bioprosthetic Valves: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Supplemental material, sj-docx-1-cat-10.1177_10760296251415375 for Comparing the Direct Oral Anticoagulants versus Vitamin K Antagonists in Bioprosthetic Valves: A Systematic Review and Meta-Analysis of Randomized Controlled Trials by Muhammad Hassan Waseem, Zain ul Abideen, Ameer Haider Cheema, Kanza Farhan, Hafsa Arshad Azam Raja, Muhammad Zubair Tahir, Fahad Saleem, Amna Nadeem and Pawan Kumar Thada in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Ethical Approval
Not applicable.
Patient Consent
Not applicable.
Authors’ Contributions CRediT Roles
Study concept and design: MHW and ZUA; acquisition of data: AHC and KF; analysis and interpretation of data: ZUA and HAAR; drafting of the manuscript: MZT, FS, AN, PKT, and AHC; critical revision of the manuscript: MHW and AHC.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be made available upon reasonable request to the authors.
Status
This manuscript has not been published previously and is not under consideration for publication elsewhere.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
