Abstract
Background
Among damage-associated molecular patterns (DAMPs), the specific contributions of histone and high mobility group box 1 (HMGB1) levels to disseminated intravascular coagulation (DIC) and multiple organ dysfunction (MOD) remain unclear. In this study, we aimed to investigate the association between two DAMP markers, plasma histone H3 and HMGB1 levels, and concurrent DIC and Sequential Organ Failure Assessment (SOFA) score in critically ill patients.
Methods
Plasma levels of histone H3 and HMGB1 were prospectively quantified in 46 critically ill patients who demonstrated systemic inflammatory response syndrome, possible sepsis, and required intensive care unit (ICU) treatment. We analyzed two DAMP marker values, various plasma-inflammatory and non-inflammatory cytokine levels, acute DIC on ICU day 3, and the maximum SOFA score at 48 h after ICU admission.
Results
On ICU day 3, 25 patients had DIC while 21 did not. In multivariate logistic regression analysis, plasma histone H3 levels (odds ratio [95% confidence interval]: 1.17 [1.02- 1.49], p = 0.008) and TNF-α (1.006 [1.001- 1.014], p = 0.007) were significant independent factors of DIC pathogenesis. The Spearman's rank correlation coefficients for the maximum SOFA score were 0.664, 0.602, 0.348, and 0.221 for IL-8, IL-6, histone H3, and HMGB1, respectively.
Conclusion
In the early phase requiring intensive care, histone H3 levels exhibited a more positive association with the onset of DIC than HMGB1. Conversely, inflammatory cytokines may exert a more substantial influence on the pathogenesis of multiple organ failure in comparison to DAMPs.
Keywords
Introduction
Disseminated intravascular coagulation syndrome (DIC) is an acquired disorder characterized by the systemic activation of the coagulation cascade, resulting in microvascular damage and organ dysfunction. 1 This syndrome is triggered by infectious and non-infectious diseases, and it is frequently associated with the aggravation of multiple organ failure. The predominant causes of DIC include sepsis, solid tumors, and hematologic malignancies; however, in critical care settings, the most common precipitants are sepsis, trauma, and shock. 2
DIC is associated with a systemic inflammatory response, and the interaction between this acute inflammatory response and the coagulation-fibrinolytic pathways may play a pivotal role in the initiation of its pathogenesis. Pathogen-associated molecular patterns (PAMPs) from pathogenic microorganisms are recognized by pattern recognition receptors on the surfaces of immune cells, thereby initiating an innate immune response. Then, neutrophil extracellular traps (NETs) are released from neutrophils, inducing immunothrombosis, trapping and eliminating pathogens, and preventing their dissemination, thereby functioning as a biological defense mechanism.3,4 However, excess NET formation can be harmful to the host. In sepsis, NETosis, defined as the process of cell death by NET formation, results in the release of substantial amounts of damage-associated molecular patterns (DAMPs), such as nucleosomes and histones. Additionally, DAMPs are released from host tissues due to tissue hypoxia, oxidative stress, and direct injury. DAMPs, similarly to PAMPs, are identified by pattern recognition receptors such as TLR-4. This recognition can trigger non-infectious immune responses that can lead to systemic inflammation, organ damage, and ultimately death.5–8
DAMPs play a role in the formation of intravascular thrombus. They induce the expression of tissue factors on monocytes, enhancing their procoagulant activity and promoting platelet aggregation.9,10 This sequence of events contributes to the development of thrombi within blood vessels. Among DAMPs, histones and high-mobility group box 1 (HMGB1) are nuclear components. Both histones and HMGB1 exhibit proinflammatory actions and contribute to intravascular thrombus formation in their extracellular environment.9,10
Histone H3 and HMGB1 levels are emerging as potential prognostic markers for intensive care unit (ICU) patients.11,12 The severity of sepsis has been shown to correlate with DAMP levels, and elevated blood levels of histone H3 and HMGB1 are associated with poor prognosis11,13 and DIC. 7 However, very few studies have directly examined histone H3 and HMGB1 in the context of DIC and the critical illness severity.
This study assessed whether histone H3 or HMGB1 levels are strongly associated with the occurrence of DIC at 2 days after ICU admission. In most critically ill patients, organ damage and DIC often develop within the first few days after ICU admission. The worsening of these dysfunctions is a more reliable predictor of poor outcomes than their initial severity at admission.14,15 In addition, the Sequential Organ Failure Assessment (SOFA) scoring system, a tool used to evaluate organ failure, incorporates physiological parameters and laboratory values while being influenced by therapeutic intervention. For these reasons, the maximum SOFA score obtained over the first 48 h may better reflect sepsis severity compared to that obtained at admission. 14 Therefore, we investigated how the levels of histone H3 and HMGB1 at ICU admission relate to subsequent deterioration of critical illness within 48 h, specifically DIC occurrence and the worst SOFA scores.
Methods
Patients
This study was a single-center prospective observational study. Eligible patients admitted to the Sapporo Medical University Medical and Surgical ICU between October 2021 and January 2024 primarily included those with sepsis and those with systemic inflammatory response syndrome (SIRS), where sepsis could not be ruled out. SIRS-positive was defined as meeting at least two criteria: fever, tachycardia, abnormal leukocyte count, and tachypnea. Therefore, it also included critically ill patients who did not have sepsis. Exclusion criteria included age under 18 years, hematological malignancy or chemotherapy-induced bone marrow dysfunction, severe hepatic disease such as decompensated liver cirrhosis, persistent DIC due to identified causes (eg, solid tumor), and a severe cardiopulmonary failure requiring extracorporeal membrane oxygenation therapy or its imminent initiation.
Written informed consent for participation and sample collection was obtained from all patients or next of kin. Blood samples were collected within the first 6 h of admission (day 1), anonymized, and analyzed for histone H3 and HMGB1 levels. The following cytokine levels were also measured: tumor necrosis factor-α (TNF-α), interleukin-8 (IL-8), and interleukin-10 (IL-10). Our routine laboratory testing on admission of critically ill patients includes general tests and the following additional coagulation and inflammatory tests: thrombin-antithrombin complex (TAT), α2-plasmin inhibitor-plasmin complex (PIC), soluble fibrin monomer complex, and protein C antigen, protein S antigen, presepsin, interleukin-6 (IL-6), procalcitonin. Measurements of plasma levels of histone H3, HMGB1, and all cytokines were performed in a manner blinded to the clinical information of the patients.
This study gathered data on age, sex, body weight, chronic comorbidities, acute physiology, chronic health evaluation II (APACHE-II) and SOFA scores at admission. Clinical data, including vital signs, routine blood tests, and treatments required for the SOFA score, were collected up to day 3. For calculating the daily SOFA score, the essential values of the laboratory tests and various parameters were taken from the morning data, except for the day of ICU admission (day 1). The diagnosis of DIC was confirmed on day 3 using the criteria of the Japanese Association for Acute Medicine acute phase DIC diagnosis (JAAM-DIC) score. 16 Sepsis and septic shock were defined using the Sepsis-3 definition, 17 and patients were diagnosed with observation up to 24 h after ICU admission. The study complied with the Declaration of Helsinki and was approved by the Institutional Review Board of Sapporo Medical University Hospital (322–269).
Measurement of Histone H3 and HMGB1 Levels
Plasma histone H3 levels were assessed using a sensitive ELISA with specific antibodies targeted against histone H3 (Shino-Test Corporation, Sagamihara, Japan), as previously described by Ito et al11,18 This method has been validated for linearity, demonstrating consistent results up to a concentration of 250 ng/mL. The ELISA shows a high specificity for histone H3 and does not cross-react with other histone proteins, including H2A, H2B, and H4. 18
Plasma HMGB1 levels were determined using a sensitive ELISA with specific antibodies targeted against HMGB1 (Shino-Test Corporation, Sagamihara, Japan), as previously described.19,20 The assay has a lower detection limit of one ng/mL and demonstrated linearity up to 80 ng/mL. All test samples were analyzed in duplicate, and all relevant clinical data were blinded to the laboratory analysts.
Measurement of Cytokine Levels
We quantified the levels of various plasma cytokines using a commercial Bio-plex Pro Human Cytokine 8-plex assay (Bio-Rad Laboratories, Hercules, CA). Following the manufacturer's instructions, the cytokines measured included IL-2, IL-4, IL-6, IL-8, IL-10, GM-CSF, IFN-γ, and TNF-α. All test samples were analyzed in duplicate. The data were analyzed using the Bio-plex Manager Software Ver.6.2 (Bio-Rad Laboratories, Hercules, CA). In this study, IL-8, IL-10, and TNF-α were chosen as representative inflammatory and anti-inflammatory cytokines. IL-6 levels were obtained from the results of hospital laboratory tests. The measurement kit was adopted to minimize both reagent costs and blood sampling volume. In addition, IL-2, IL-4, and GM-CSF are not used to evaluate immune responses in the critically ill population. Consequently, the IL-2, IL-4, IFN-γ, and GM-CSF results were excluded from this study.
Statistical Analysis
Plasma histone H3 and HMGB1 levels in patients diagnosed with DIC were assessed in comparison to those in non-DIC patients through the Mann-Whitney U test. Furthermore, receiver operating characteristic (ROC) curve analysis and calculations of the area under the curve (AUC) were performed to quantify the predictive performance of each parameter for JAAM-DIC positive findings.
Univariate and multivariate logistic regression analyses were performed to determine the relevant parameters, including DAMPs, inflammatory/anti-inflammatory cytokines, and inflammatory markers potentially involved in DIC pathogenesis. The stepwise method was performed to achieve the minimum Bayesian information criterion (BIC) value to extract related variables.
The correlation between two DAMP levels at ICU admission and the maximum SOFA scores during the first 48 h was evaluated.
The relationship between histone H3 or HMGB1 levels, the SOFA scores, and other coagulation and cytokine data was determined using Spearman's rank correlation coefficient (rho) to evaluate non-normally distributed variables.
Group comparisons for binary variables were performed with Fisher's exact test. Numerical data were compared between groups by the Student t-test for normally distributed data, and the Mann-Whitney U test for non-normally distributed data. All statistical analyses were performed using JMP Pro version 17.0 (SAS Institute Inc., Cary, NC, USA), and p values < 0.05 were considered statistically significant.
Results
Patient Characteristics
Table 1 shows the baseline characteristics of the study patients at ICU admission and their prognosis. Forty-six patients consented to participate in the study; 39 (84.8%) were diagnosed with sepsis, and seven (15.2%) were not. Twenty-six (56.5%) of the patients with sepsis met the criteria for septic shock. Among the seven patients without sepsis, three had severe acute pancreatitis, and one had AKI with anuria due to ischemic rhabdomyolysis of the lower extremities.
Baseline Characteristic of the Study Patients at ICU Admission and Their Prognosis.
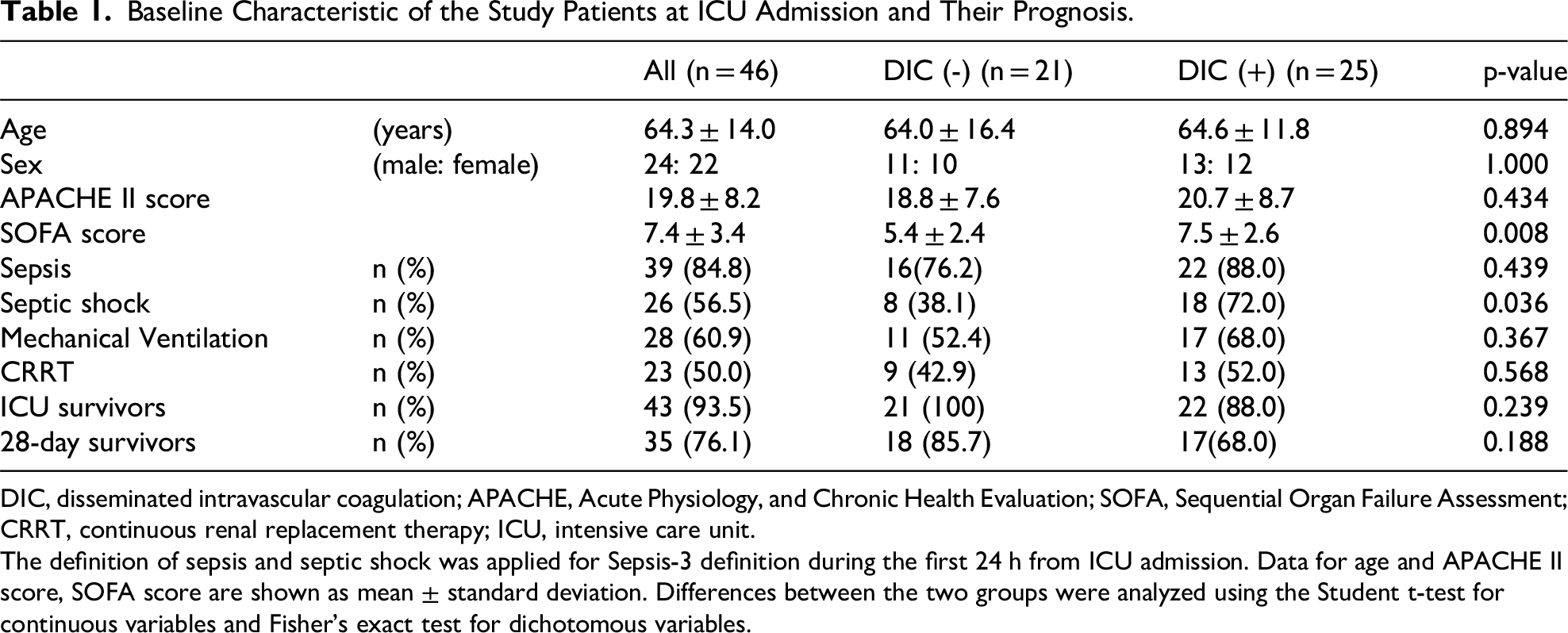
DIC, disseminated intravascular coagulation; APACHE, Acute Physiology, and Chronic Health Evaluation; SOFA, Sequential Organ Failure Assessment; CRRT, continuous renal replacement therapy; ICU, intensive care unit.
The definition of sepsis and septic shock was applied for Sepsis-3 definition during the first 24 h from ICU admission. Data for age and APACHE II score, SOFA score are shown as mean ± standard deviation. Differences between the two groups were analyzed using the Student t-test for continuous variables and Fisher's exact test for dichotomous variables.
There were no significant differences in age, sex, APACHE II score, sepsis prevalence, the use of mechanical ventilation and continuous renal replacement therapy, ICU survival rate, and 28-day survival rate between the two groups of DIC prevalence. However, the DIC group had a significantly higher SOFA score (p = 0.008) and a higher proportion of patients with septic shock (p < 0.001).
Comparison of two DAMP Markers by DIC Prevalence
Plasma histone H3 and HMGB1 levels at ICU admission in the DIC-positive and DIC-negative patients at ICU day 3 are depicted in Figure 1. HMGB1 levels exhibited no significant differences between two groups. Histone H3 levels were significantly higher in the DIC-positive group (median 3.31 ng/mL, interquartile range [IQR], 2.05-9.62) than in the DIC-negative group([median 1.60 ng/mL [IQR, 0.96-3.73], p = 0.036).

Comparison of plasma DAMPs levels (A) histone H3, (B) HMGB1 at ICU admission with prevalence of DIC. Box plots with error bars at 10% or 90%, boxes show 25% or 75%, and lines within them indicate median values. Black dots indicated outliers. Histone H3 levels increased with DIC incidence. HMGB1 levels did not show any significant effects. The Mann–Whitney U test was applied to compare the values between the two groups. HMGB-1, High-Mobility Group box-1; ICU, Intensive Care Unit; DIC, Disseminated Intravascular Coagulation.
The ROC curve for the histone H3 level and DIC showed a more up-left shift compared to that for HMGB1 (Figure 2); the corresponding optimal cut-off values were 2.00 ng/mL for histone H3 levels and 6.11 ng/mL for HMGB1 levels. The AUC showed discriminative powers for histone H3 and HMGB1 of 0.682 (95% CI: 0.509-0.816) and 0.605 (95% CI: 0.426-0.759) (p = 0.469), respectively.

Receiver operating characteristic curve for histone H3 and HMGB1 levels and DIC prevalence by 48 h from ICU admission. The two DIC-related AUC were comparable (p = 0.469). HMGB-1, High-Mobility Group box-1; AUC, Area Under the Curve; CI, Confidential Interval; DIC, Disseminated Intravascular Coagulation; ICU, Intensive Care Unit.
Humoral Mediators Involved in the Pathogenesis of DIC
Table 2 shows the results of univariate and multivariate logistic regression analyses. In the univariate analysis, histone H3, IL-6, IL-10, TNF-α, and procalcitonin levels were identified as significant variables. This study aimed to compare the impact of histone H3 and HMGB1 levels on DIC pathogenesis. Therefore, we used the stepwise method, minimizing the BIC value by forcibly incorporating histone H3 and HMGB1 values. Finally, histone H3 and TNF-α levels were identified as two potential variables, which were used in the multivariate logistic regression analysis; both markers were associated with DIC diagnosis on day 3 (histone H3, adjusted OR 1.147 [95% CI: 1.003-1.439]; TNF-α, adjusted OR 1.006 [95%CI: 1.001-1.014]).
Univariate and Multivariate Logistic Regression Analysis of Predictors of Disseminated Intravascular Coagulation.

DAMPs, damage-associated molecular patterns; HMGB1, high-mobility group box-1; IL, interleukin; TNF-α, tumor necrotizing factor-α; CRP, c-reactive protein.
Correlation Between Two DAMP Markers and Maximum SOFA Scores During the First 48 h
Associations between the levels of two DAMPs and the maximum SOFA scores during the first 48 h of ICU admission are shown in Figure 3. There was no significant correlation between HMGB1 levels and the maximum SOFA score (rho = 0.221, p = 0.150). Conversely, the histone H3 levels and maximum SOFA score showed a weak correlation (rho = 0.348, p = 0.021).
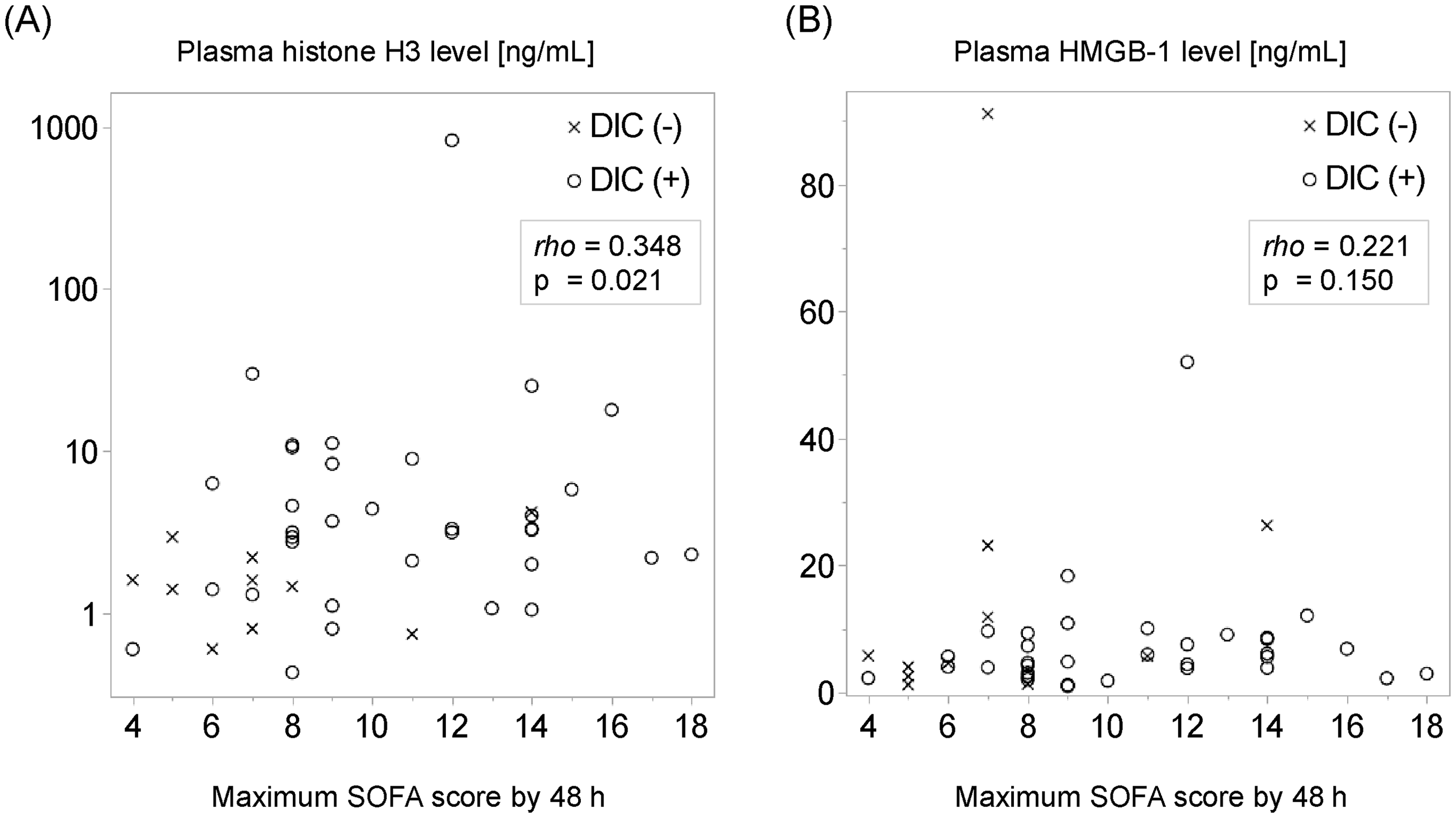
Correlation between two DAMPs and maximum SOFA score by 48 h from ICU admission. Scatter plots display the maximum SOFA scores recorded 48 h after ICU admission and plasma histone H3 levels (A) and plasma HMGB1 levels (B). The levels of histone H3 show a correlation with the maximum SOFA scores, whereas HMGB1 does not. Rho represents Spearman's rank correlation coefficient. DAMPs, Damage-Associated Molecular Patterns; SOFA, Sequential Organ Failure Assessment; HMGB-1, High-Mobility Group box-1; ICU, Intensive Care Unit.
Correlations Between Two DAMP Markers and Coagulation/Fibrinolysis Markers, Cytokines, and Inflammatory Markers
Correlations between two DAMP markers and various laboratory data are presented in Table 3. TAT was moderately correlated with histone H3 levels (rho = 0.489, p < 0.001), in contrast to HMGB1 levels (rho = - 0.004, p = 0.979). A similar correlation was observed for fibrin/fibrinogen degradation products (FDP) and D-dimer, with a decrease in their rho of histone H3 (rho = 0.394, p = 0.007 in FDP, rho = 0.420, p = 0.004 in D-dimer, respectively).
Correlations Between two DAMPs Markers, maximum SOFA Score and Coagulation/Fibrinolysis Markers, Cytokines, and Inflammatory Markers.

HMGB1, high-mobility group box-1; FDP, fibrin-degradation products; PT-INR, prothrombin time-international normalized ratio; AT III, antithrombin III: PIC, plasmin-α2 plasmin inhibitor complex; TAT, thrombin-antithrombin complex; SFMC, soluble fibrin monomer complex; IL, interleukin; TNF-α, tumor necrotizing factor-α; CRP, c-reactive protein. Rho is Spearman's rank correlation coefficient. *, p < 0.05; **, p < 0.01; N.S, non-significant
Finally, we assessed the correlation between maximum SOFA scores and all markers during the first 48 h after ICU admission. The maximum SOFA score significantly correlated with several biomarkers, particularly IL-8, IL-6, and procalcitonin levels, each demonstrating rho exceeding 0.50.
Discussion
This study aimed to compare histone H3 and HMGB1 levels as contributing factors to DIC in medical and surgical ICU setting. In this study, plasma histone H3 and TNF-α levels at ICU admission were significantly correlated with the manifestation of DIC 2 days later. The maximum SOFA scores within the first 48 h after ICU admission exhibited a weak correlation with histone H3 levels and a moderate correlation with IL-6, IL-8, and PCT. However, no significant differences or correlations were found for HMGB1 values.
Furthermore, we found a significant positive correlation between histone H3 levels and coagulopathy, while coagulation-related markers such as TAT levels showed a positive correlation. These results suggest that histone H3 may play a more critical role in coagulopathy than HMGB1 in the early days after ICU admission.
DAMPs for Suggestive Markers of Exacerbation
This study used the JAAM-DIC and maximum SOFA score during 48 h after ICU admission as assessment variables. This is because the severity of illness in patients admitted to the ICU varies considerably over time. Furthermore, the change in or maximum SOFA score observed within 4 days of ICU admission is a more accurate predictor of prognosis than that obtained at admission. 14 Therefore, the study aimed to assess whether various markers can predict the development of coagulopathy and the deterioration of organ failure for several days. The present study suggests that, constrained to DAMPs, histone H3 levels potentially exert a more substantial influence than HMGB1 as a contributor to deterioration around ICU admission.
DAMPs and Their Deteriorating Mechanisms
DAMPs can activate the procoagulant cascade9,10 and are recognized as the predominant trigger of DIC. 21 In this study, three patients with severe acute pancreatitis and one patient with ischemic lower extremity rhabdomyolysis were included among the seven patients without sepsis. These diseases can cause extensive tissue damage, which can lead to the release of DAMPs, and which is known to trigger DIC. Of the four patients in this study, three developed DIC by the ICU day 3 with histone H3 levels of 10.9, 10.5 and 3.3 ng/mL; the patient who never developed DIC had a histone H3 level of < 0.1 ng/mL. Extensive tissue damage could also increase the DAMP levels and DIC onset risk.22,23 Furthermore, a thorough examination of DAMPs in conjunction with coagulation markers demonstrated that TAT and histone H3 exhibited the most substantial rho values. Meanwhile, histone H3 exhibited no correlation with PIC, which is an indicator of fibrinolytic upregulation. The present finding suggests that the histone-mediated pathophysiological mechanism of DIC involves hypercoagulation due to thrombin production.
Although histone H3 and HMGB1 are grouped as DAMPs, their different mechanisms can affect organ function differently. For example, Ito et al demonstrated that the combination of HMGB1 and thrombin enhanced fibrin deposition within the lungs and kidneys, increasing mortality risk, which was not observed for these factors separately. The authors suggested that the lethal activity of HMGB1 may depend on other synergistic factors, such as thrombin. 24 In addition, HMGB1 release is observed not only in instances of severe acute inflammation, such as sepsis, severe trauma, and acute pancreatitis, but also in chronic inflammatory disorders, including type 2 diabetes mellitus, chronic heart failure, and autoimmune diseases. 25 The observations indicate that exposure to low or transient high levels of HMGB1 may not increase the risk of mortality. Conversely, in sepsis, elevated levels of HMGB1 have been observed. Persistently elevated HMGB1 levels and delayed peaks have been associated with an unfavorable outcome. 25 This finding leads to the hypothesis that the early-stage increase in HMGB1 level upon ICU admission may have minimal impact on coagulation abnormalities or organ dysfunction.
Numerous studies have demonstrated the involvement of histones in the platelet aggregation and coagulation processes.26–28 Histones derived from NETs appear to promote coagulation and thrombin formation by activating platelets, 28 downregulating anticoagulants such as thrombomodulin, 27 and directly activating the intrinsic coagulation pathway. It is noteworthy that excessive NET release induces thrombin generation via Factor XII activation, thereby contributing to thrombosis. 28 Nucleosomes, NET-associated histones, and free histones disrupt the endothelial barrier, thereby instigating and perpetuating the extrinsic coagulation pathway by inducing tissue factor expression in endothelial cells. This helps illustrate the manner in which NETs link innate immunity and coagulation in the context of immune thrombosis. The deleterious effects of histones and HMGB1 on organ damage result from disparate mechanisms.
Differences Between Histone H3 and HMGB1 as Markers of the DIC Criteria and Sepsis Severity
Clinical studies have shown that levels of HMGB1 are elevated in DIC.8,29,30 However, in patients with sepsis, HMGB1 values in patients with ISTH-overt DIC were higher than in those without DIC, although this difference lacked statistical significance. A study of patients with sepsis revealed no statistically significant differences in HMGB1 and histone H3 levels between the presence and absence of ISTH overt DIC. 31 Our findings were based on the JAAM-DIC diagnostic criteria, which help predict multiple organ dysfunction and poor prognosis in patients with sepsis. 15 This difference in diagnostic criteria may have contributed to the discrepancy in results. However, two studies applied JAAM-DIC criteria and demonstrated a positive association between histone H3 and coagulopathy,11,32 which agrees with this study.
Four clinical studies have simultaneously measured HMGB1 and histone H3 levels.19,31,33,34 Although a study of a patient with acute leukemia was excluded, 33 the levels of histone H3 and HMGB1 were similar between patients with and without ISTH-overt DIC. 31 Although Mori et al 34 suggested that plasma HMGB1 levels predicted 28-day mortality in ISTH overt DIC patients, compared to histone H3 levels, approximately 50% of patients in the study had hematopoietic disorders, accounting for half of the non-survivors. Conversely, in this study, nearly 80% of patients had sepsis, and hematopoietic disorders were excluded. Therefore, it is reasonable that our findings diverge from those reported by Mori et al 34 Furthermore, Sawada et al 19 compared HMGB1 with histone H3 levels in postoperative ICU patients; the authors’ results exhibited a marked similarity to those of the present study, wherein histone H3 values were correlated with SOFA scores, whereas the HMGB1 values were not.
Association Between DAMP Levels and SOFA Score
The present study observed a weak correlation between histone H3 levels and the SOFA scores. This observation could be attributed to the elevated histone H3 levels observed in the DIC-positive group, which also encompassed a higher proportion of septic shock patients.
Firstly, as DIC is characterized by thrombocytopenia, it concomitantly elevates the coagulation point. In short, elevated histone H3 levels may play a significant role in the escalation of the coagulation point through the DIC pathological process.
Secondly, as norepinephrine administration is a component of the diagnostic criteria for septic shock; patients diagnosed with septic shock are assigned a circulation point score of 3 or 4. In this dataset, the histone H3 values for septic shock patients were in the IQR range of 1.825 to 8.490, while those for non-septic shock patients ranged from 0.800 to 3.232 (p = 0.047). This finding suggests that elevated histone H3 levels may be indicative of a patient population with a high prevalence of septic shock-induced circulatory point elevation. These two factors could contribute to the weak correlation between histone H3 levels and SOFA scores.
Moreover, IL-6, IL-8, and procalcitonin levels correlated more strongly with the SOFA score than with histone H3 levels. This finding can be attributed to excessive proinflammatory cytokines that provoke tissue and organ damage. 35 Some evidence suggests that histones and inflammatory cytokines interact; however, weak correlation was observed between them in this study. As inflammatory cytokine levels were more strongly related to the SOFA scores than to histone levels, non-histone-induced inflammation may increase the risk of organ dysfunction.
The present study has some limitations. This a single-center, prospective observational study, resulting in a relatively small number of patients in each group. First, the sample size in this study was small, creating some risk of type II error. Larger studies are recommended to replicate and validate the present findings. Second, the present study was conducted at a university hospital's medical and surgical ICU. Patients had already undergone some therapeutic procedures prior to their admission to the ICU. Consequently, data collected from patients admitted to the ICU via the emergency department may yield different results. Third, approximately 85% of the patients had sepsis. Further validation is needed to determine whether the results of this study apply to DIC caused by other factors, such as hematologic malignancies and trauma.
Increased levels of histone H3 upon admission to the ICU are associated with DIC development and the severity of multiple organ dysfunction up to 48 h of ICU admission, in contrast to those of HMGB1. This finding indicates that histone H3 plays a crucial role in the early pathogenic potential of DIC in medical surgical ICU setting.
Footnotes
Acknowledgments
The authors thank the ICU medical and nursing teams at Sapporo Medical University Hospital.
Ethics Approval and Consent to Participate,and for Publication
This study was approved by the Institutional Review Board of Sapporo Medical University Hospital (322–269) on March 11th, 2021.
Consent to Participate
Prior to participating in this study, all participants or their legal representatives provided written consent to participate, including the collection of blood samples, the use of anonymized medical data, and the publication of study results.
Consent for Publication
All participant consent forms are stored in a secure and accessible manner.
Author Contributions
HK designed the study and experimental protocol, analyzed and interpreted the data, and wrote the manuscript. SY participated in laboratory experiments to measure plasma samples and interpret the results. SS collected blood samples and patients’ clinical data and interpreted the results. SK, HT, and YM critically appraised the manuscript. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was mainly funded by JSPS KAKENHI Grant Number 21K09051, and partial financial support was received from Japan Blood Products Organization (Tokyo, Japan). The funders had no role in the conceptualization, design, data collection, analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The ELISA for HMGB1 and histone H3 is a product developed by Shino-Test Corporation, where SY is employed. YM received grants from JIMRO Co. Ltd (Takasaki, Japan), personal fees from MSD K.K. (Tokyo, Japan), personal fees from Asahi Kasei Pharma Co. Ltd (Tokyo, Japan), and personal fees from Japan Blood Products Organization (Tokyo, Japan), outside the submitted work. HT received lecture fees from Otsuka Pharmaceutical Factory, Inc. (Naruto, Japan). SK received grants from Asahi Kasei Pharma Co. Ltd (Tokyo, Japan), and from Japan Blood Products Co. Ltd (Tokyo, Japan), outside the submitted work. The other authors declare that they have no conflicts of interest.
Availability of Data and Materials
The datasets used and analyzed during this study are available from the corresponding author upon reasonable request.
Past Presentations
Some of the results of this study were presented at the 52nd Annual Meeting of Japanese Association for Acute Medicine in Sendai on Oct 13, 2024.
