Abstract
Introduction
The efficacy of antithrombin (AT) supplementation against septic disseminated intravascular coagulation (DIC) may depend on various pre-existing factors, particularly the AT dose and multiple organ dysfunction severity. This study aimed to identify the impactful factors for early DIC recovery.
Methods
Patients’ clinical records, including AT therapy and septic DIC data, were retrospectively extracted from January 2015 to December 2020. The patients were divided into those with early DIC recovery (n = 34) and those without (n = 37). Multivariate logistic regression analysis determined significant independent factors. Time-to-event analysis confirmed how these factors affected the DIC recovery time.
Results
The AT dose per patient body weight (odds ratio [95% confidence interval]: 2.879 [1.031-8.042], P = 0.044) and pre-existing organ dysfunction severity (0.333 [0.120-0.920], P = 0.034) were significant independent factors affecting early DIC recovery. A higher AT dose significantly shortened the DIC recovery time among patients with severe organ dysfunction (P < 0.01), but not among non-severe patients (P = 0.855).
Conclusion
The therapeutic efficacy of AT treatment for septic DIC might depend on the severity of pre-existing organ failure and the AT dose per patient body weight.
Keywords
Introduction
Sepsis causes an excessive inflammatory response due to immune system activation by infection. Excessive inflammation activates the host coagulation system. Amplification of the inflammation leads to uncontrolled coagulopathy and multiple organ dysfunctions (MODs).1,2 Among patients with sepsis, especially those with coagulation abnormalities, septic disseminated intravascular coagulation (DIC) is associated with worse outcomes. 3 However, the effectiveness of DIC pharmacotherapy in improving outcomes has not been consistent. Therefore, DIC treatment recommendations in the surviving sepsis campaign guidelines4,5 are controversial.
There are a few favorable clinical studies on the use of antithrombin (AT) supplements for septic coagulopathy.6,7 However, the AT dose varied considerably in these studies. Therefore, the appropriate AT dose for DIC remains to be determined. Previously, we reported that recombinant AT products, AT-gamma (ACOALAN®, Kyowa Hakko Kirin, Tokyo, Japan), which can be administered per patient's body weight (eg, 36 IU/kg/day), could more efficiently improve MODs and DIC scores than plasma-derived AT concentrates (pAT), which were administered at a fixed dose of 1500 IU/day. 8 However, the pAT dose per body weight was approximately 20% lower than that of the AT-gamma. After adjustment by titer, the pAT-equivalent dose in the AT-gamma group was significantly higher than that in the pAT group. The dose difference indicated that the amelioration of MODs might depend on the AT dose. Iba et al. revealed that twice the dose of pAT, eg, 3000 IU/day, might improve mortality compared to 1500 IU/day of pAT.9,10 However, no reports have described AT dose per patient body weight. Therefore, it is necessary to evaluate the AT dose per body weight therapeutic effect on DIC recovery.
Furthermore, Yamakawa et al. reported that the highly susceptible baseline sequential organ failure assessment (SOFA) score for anticoagulant therapy in sepsis was between 13 and 17. 11 In light of Yamakawa's report, it is suggested that the severity of MODs could affect the therapeutic efficacy of anticoagulants against DIC. However, the therapeutic effects of both the baseline severity of MODs and AT dose per body weight are uncertain. Therefore, this study aimed to identify the pre-existing or therapeutic factors that could affect DIC recovery and demonstrate how the identified factors affect clinical efficacy.
Methods
Ethics Statement
This study was approved by the Institutional Review Board of Sapporo Medical University Hospital (332-27), which waived the requirement for informed patient consent because of the anonymous nature of the data.
Patient Enrollment
This was a single-center retrospective observational study, and the facility was a medical and surgical intensive care unit (ICU) at Sapporo Medical University Hospital in Japan. Patients who received AT supplement therapy for both acquired antithrombin deficiency and sepsis-induced DIC during ICU stay from January 2015 to December 2020 were enrolled. We compared organ dysfunction improvement between pAT and AT-gamma in patients with septic DIC from January 2015 to October 2018. 8 The present study population overlaps with that of a past report 8 to an extent (2015−2018). Sepsis was diagnosed based on the Sepsis-3 criteria: organ dysfunction due to infection, as indicated by an increase in the SOFA score by ≥2 points in any organ. 12 We applied the Japanese Association for Acute Medicine formulated DIC scoring system (JAAM-DIC) to diagnose DIC and determine DIC recovery.
Patients who met the following criteria were excluded from the study: (1) aged < 18 years; (2) hematopoietic disorders, such as acute leukemia, myelodysplastic syndrome, aplastic anemia, or having undergone hematopoietic stem cell transplantation; (3) liver failure secondary to extended hepatectomy; (4) patients who died within 48 h of the start of AT administration; and (5) end-stage patients with dilated cardiomyopathy.
Treatment of DIC
AT concentrates and recombinant human soluble thrombomodulin-alpha (rTM) (Recomodulin®, Asahi Kasei Pharma, Tokyo, Japan) were prescribed as therapeutic agents for DIC. AT concentrates were administered at approximately 50% or less of the plasma AT level, according to our institutional protocol. The administration of rTM was initiated when the platelet count decreased to approximately 50000/µL or less, or when the platelet count declined sharply by ≥ 50% compared to the previous day.
The choice of AT concentrate and dose were at the discretion of the individual ICU physicians. However, pAT was administered at 1500 IU/day, regardless of patient body weight, for three consecutive days because of the public health insurance consensus in Japan. In contrast, the AT-gamma dose is approved to be 36–72 IU/kg body weight per day for 3–5 days. A considerable number of patients were administered AT-gamma at 1800 IU/day without consideration of the patient's body weight. In some patients, AT administration was terminated in less than 3 days due to a remarkable improvement in organ damage and plasma AT levels. In principle, concomitant unfractionated heparin was avoided as a treatment for DIC; however, some patients were administered heparin for the treatment of comorbidities, such as venous thrombosis and chronic atrial fibrillation.
The principal care of sepsis was provided with reference to various surviving sepsis campaign guidelines at the time of treatment.
Data Collection
This study collected the following data: age, sex, body weight, chronic comorbidities, acute physiology and chronic health evaluation II (APACHE-II) score at admission, SOFA score at admission, source of sepsis infection, positive presence or absence of blood culture, type and dose of AT preparation administered, modified SOFA (mSOFA) score (subtracted the central nervous system score from the total SOFA score) for 7 consecutive days from the start of AT administration, daily DIC positivity or negativity depending on JAAM-DIC definition from the start of AT administration to 14 days later, concomitant administration of rTM and heparin, renal replacement therapy, polymyxin-immobilized direct hemoperfusion, and mechanical ventilation as combination therapy. Plasma AT values were collected from the AT administration initiation day to the fourth day. Hemorrhagic complications were also observed during the period from the initiation of AT administration to the seventh day. ICU and 28-day mortality rates were also recorded.
For missing data related to mSOFA and DIC score calculations, the last observed carried forward or the average value immediately before and after the missing value was substituted. The day of DIC recovery was defined as the first day when two consecutive days of JAAM-DIC negativity were observed. The mSOFA score was adopted because the central nervous system score of the SOFA was greatly affected by other treatments, such as tracheal intubation. Furthermore, an additional point for coagulation was added to the coagulation score of the SOFA on the next day when platelet concentrates were transfused.
Because the AT-gamma dose is 1.2 times higher than that of pAT, 13 the dose obtained by dividing the AT-gamma dose by 1.2 was used as the plasma-equivalent AT dose.
Statistical Methods
Data are expressed as mean ± standard deviation if they were normally distributed and as median (25-75% interquartile) if they did not have a normal Gaussian distribution. The Kolmogorov–Smirnov test was used to determine normality of the distribution of continuous variables. A P-value < 0.05 was considered statistically significant.
Since the number of DIC recovery patients was closest to half at 7 days after AT administration, the patients who recovered by Day 7 were allocated to the recovery group and those who did not recover by Day 7 were allocated to the non-recovery group. The background characteristics between the two groups were compared using the t-test, Mann–Whitney U test, and Fisher's exact test.
Univariate and multivariate logistic regression analyses were performed to determine the clinically important relevant factors involved in DIC recovery, such as concomitant DIC treatment, MOD severity, JAAM-DIC score at the start of AT treatment, and AT dose. To introduce the four independent variables in a multivariate logistic regression analysis, the sample size was calculated to be more than 32 in the smaller group. Platelet counts overlap both JAAM-DIC and mSOFA scores; therefore, another SOFA score for sepsis-induced coagulopathy (SIC-SOFA), 14 was applied to avoid multicollinearity. The SIC-SOFA score is a sum of the scores for four items: respiratory, cardiovascular, hepatic, and renal scores. Since the total number of patients was small (N = 71), the daily plasma AT-equivalent AT dose per body weight (pAT/BW), SIC-SOFA score, and JAAM-DIC score were divided into two groups: higher or lower than the median.
The results were then stratified according to the derived parameters of the background characteristics from the multivariate logistic regression analysis. The effects of the AT dose on the DIC recovery day and changes in the mSOFA score from Days 0–7 were compared. The log-rank test was used for time-to-event analysis for the DIC recovery day, and changes in the mSOFA score were adopted by two-way repeated analysis of variance. IBM SPSS Statistics, version 25 (IBM Corp., Armonk, NY, USA) was used for statistical analyses.
Results
A flowchart of patient enrollment in this study is shown in Figure 1. Of the 3823 patients admitted to the ICU in 6 years, 119 were treated with AT. Forty-eight patients were excluded based on the exclusion criteria. Finally, 71 patients were enrolled for analysis. The patients were divided into two groups based on DIC recovery status on Day 7: non-recovery (n = 37) and recovery (n = 34) groups.

Selection schema of the enrolled patients. ICU, intensive care unit; DIC, disseminated intravascular coagulation; AT, antithrombin.
Patient Characteristics
Patient backgrounds are shown in Table 1. Both groups were statistically similar in terms of age, sex, body weight, and APACHE II and SOFA scores at ICU admission. Similarly, there were no statistical differences in primary infection sites and percentages of positive blood cultures between the two groups. Furthermore, there were no statistically significant differences in concomitant treatments between the groups. However, the percentage of renal replacement therapy in the recovery group (61.8%) tended to be lower than that in the non-recovery group (83.8%; P = 0.059). Furthermore, the SIC-SOFA score in the recovery group (7 [IQR 4-8]) was significantly smaller than that in the non-recovery group (9 [IQR 6-11]; P = 0.010).
Baseline Characteristics of the Enrolled Patients.
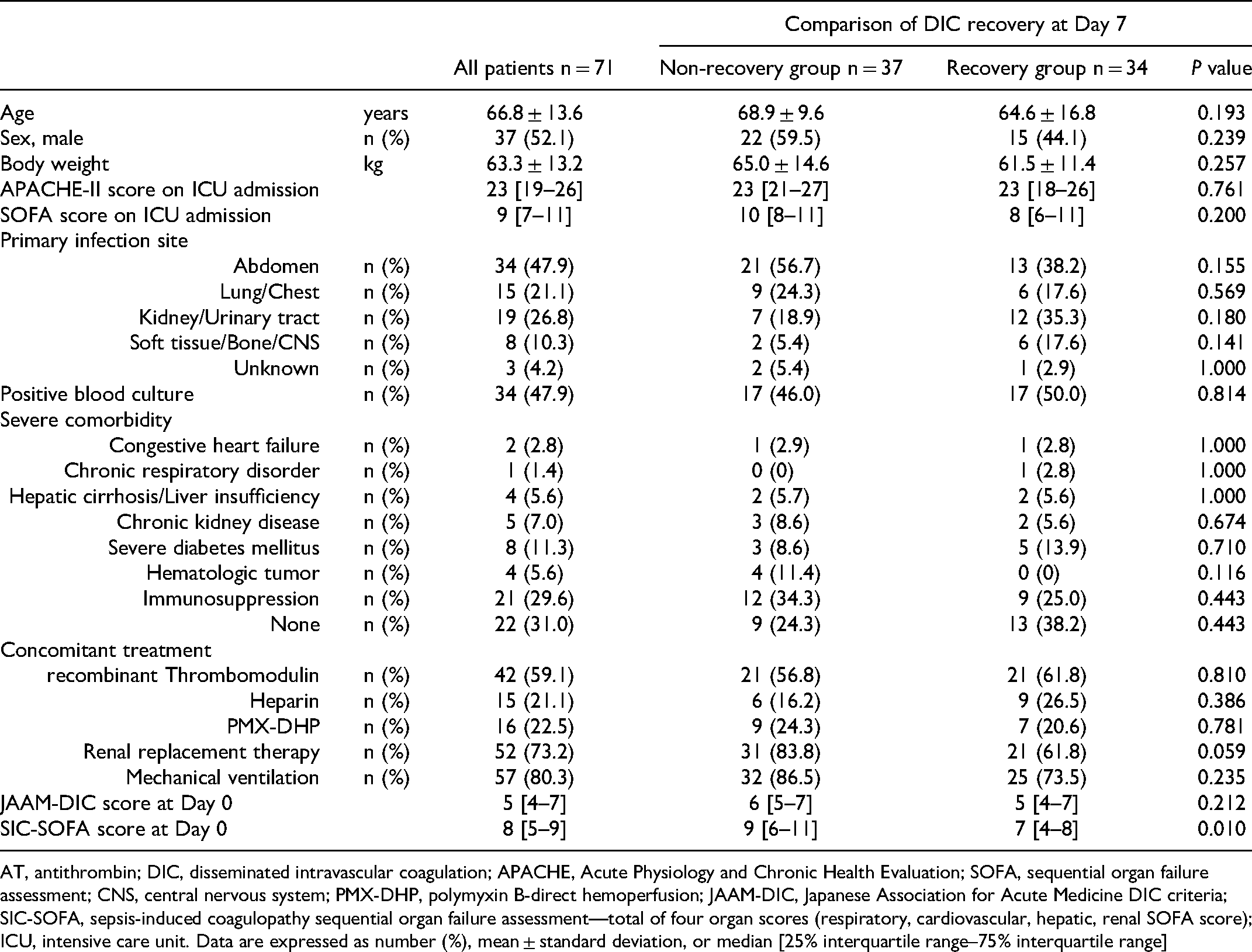
AT, antithrombin; DIC, disseminated intravascular coagulation; APACHE, Acute Physiology and Chronic Health Evaluation; SOFA, sequential organ failure assessment; CNS, central nervous system; PMX-DHP, polymyxin B-direct hemoperfusion; JAAM-DIC, Japanese Association for Acute Medicine DIC criteria; SIC-SOFA, sepsis-induced coagulopathy sequential organ failure assessment—total of four organ scores (respiratory, cardiovascular, hepatic, renal SOFA score); ICU, intensive care unit. Data are expressed as number (%), mean ± standard deviation, or median [25% interquartile range–75% interquartile range]
Comparison of AT Related Variables in Both Groups
Table 2 shows various AT-related variables and outcomes, such as the maximum plasma AT level, pAT/BW, accumulated plasma AT, AT administration duration, total AT dose during the administration period, and ICU survival. There were no significant differences in the baseline plasma AT levels, pAT/BW, percentage of plasma AT products, AT administration duration, total pAT/BW, and hemorrhagic complications. However, the AT administration duration in the recovery group (3 [IQR 2-3]) tended to be shorter than that in the non-recovery group (3 [IQR 3-3]; P = 0.056). Maximum plasma AT levels in the recovery group (92.9 ± 21.1) were significantly higher than those in the non-recovery group (82.1 ± 13.9; P = 0.015). Additionally, the area under the curve of plasma AT levels during days 0–4 was significantly different between the groups (recovery group, 348.3 ± 60.1 vs non-recovery group, 317.7 ± 54.6; P = 0.028). The recovery group had significantly more 28-day ICU-free days (recovery group, 20 [IQR 16-22]; non-recovery group, 14 [IQR 0-20]; P < 0.001), a higher ICU discharge rate (recovery group, 100%; non-recovery group, 78.4%; P = 0.005), and a higher 28-day survival rate after ICU admission (recovery group, 97.1%; non-recovery group, 78.4%; P = 0.029).
Antithrombin related Variables and Outcomes.
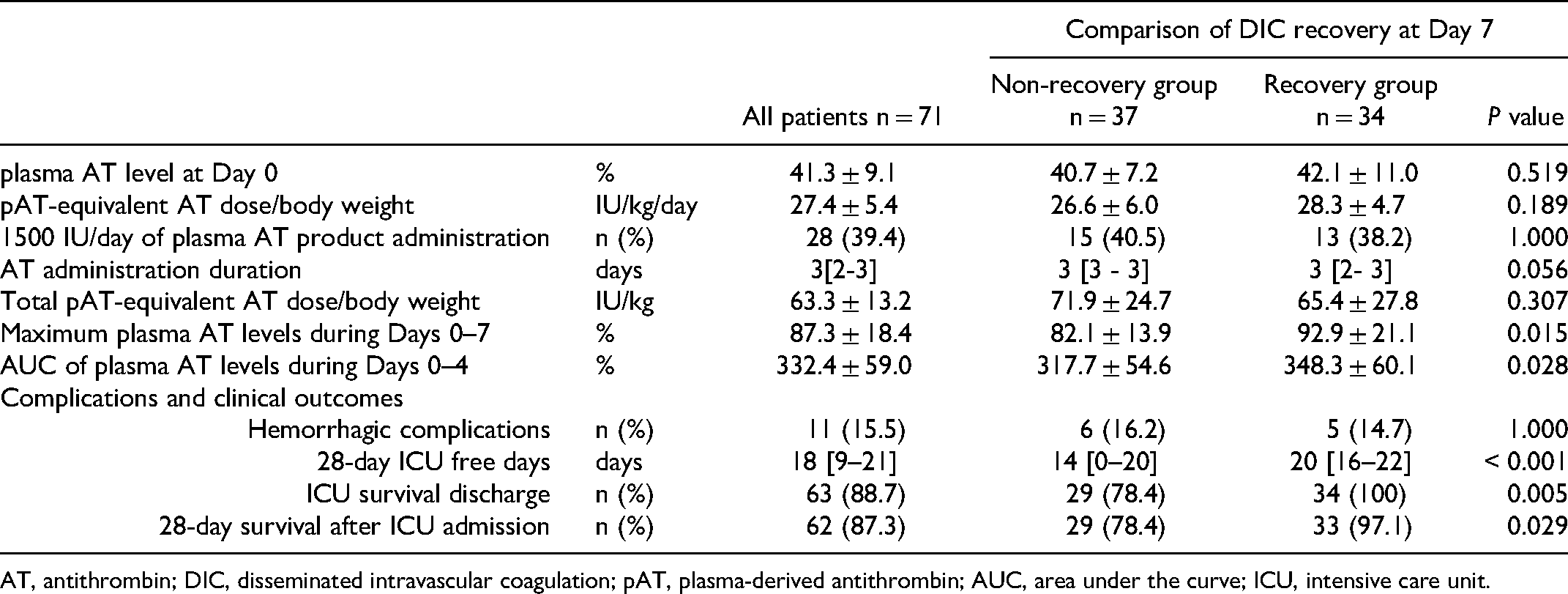
AT, antithrombin; DIC, disseminated intravascular coagulation; pAT, plasma-derived antithrombin; AUC, area under the curve; ICU, intensive care unit.
Risk Factors Associated with Recovery from DIC
Table 3 shows the results of logistic regression analyses based on four clinically relevant parameters: pAT/BW, JAAM-DIC score, SIC-SOFA score, and concomitant rTM supplement. According to the median pAT/BW (27.73 IU/kg), patients were divided into lower- and higher-dose groups. The pAT/BW in the lower- and higher-dose groups were 23.28 ± 3.32 and 31.43 ± 3.82 IU/kg, respectively. Similarly, the median JAAM-DIC and SIC-SOFA scores were 5 and 8, respectively.
Risk Factors Associated with DIC Recovery at Day 7.
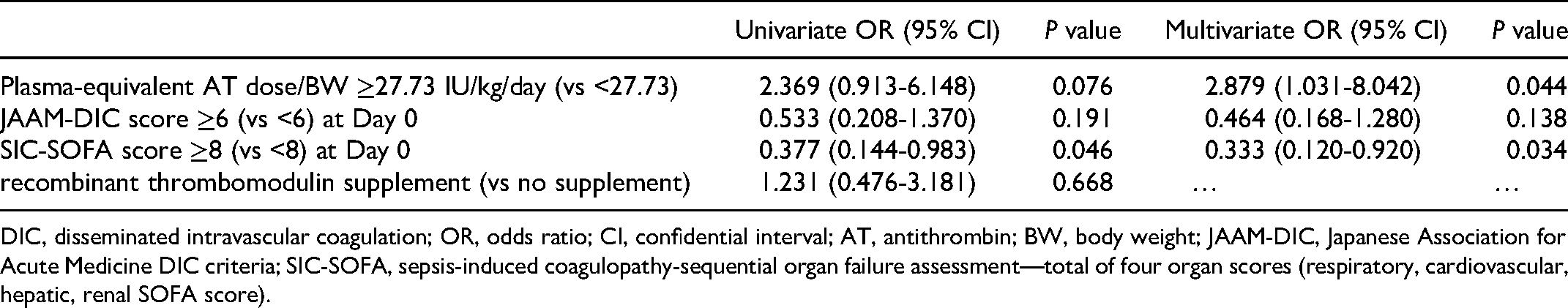
DIC, disseminated intravascular coagulation; OR, odds ratio; CI, confidential interval; AT, antithrombin; BW, body weight; JAAM-DIC, Japanese Association for Acute Medicine DIC criteria; SIC-SOFA, sepsis-induced coagulopathy-sequential organ failure assessment—total of four organ scores (respiratory, cardiovascular, hepatic, renal SOFA score).
The SIC-SOFA score at Day 0 was identified as a significant variable affecting DIC recovery on Day 7 (SIC-SOFA score, P = 0.046) in a univariate logistic regression model. A multivariate logistic regression model was used to select the following variables: pAT/BW above or below the median and SIC-SOFA score above or below the median. Based on the multivariate logistic regression analysis, pAT/BW above or below the median and SIC-SOFA score above or below 8 were independently and significantly associated with DIC recovery on Day 7 (pAT/BW, [95% confidence interval: 1.031-8.042]; SIC-SOFA, [0.120-0.920]).
Relationship of AT Dose with DIC Recovery Time
The time-to-event analysis of DIC recovery is shown in Figure 2. In the severe group, which included patients whose SIC-SOFA scores were > 8, the DIC recovery time in the higher-dose group was significantly shorter than that in the lower-dose group (log-rank test, P < 0.001). In contrast, pAT/BW did not affect DIC recovery time in the non-severe group (P = 0.855).

Kaplan–Mayer curves of DIC recovery according to pAT/BW. Probability of DIC recovery curves after AT product administration are shown in the two SIC-SOFA groups. (A) Non-severe group, SIC-SOFA score <8; (B) Severe group, SIC-SOFA score ≥8. DIC, disseminated intravascular coagulation; AT, antithrombin; pAT/BW, plasma AT-equivalent AT dose per body weight per day; SIC-SOFA, sepsis-induced coagulopathy associated sequential organ failure assessment score.
Effect of AT Dose on Changes in mSOFA Score
Changes in the mSOFA score following AT administration are shown in Figure 3. The higher AT doses tended to decrease mSOFA scores faster than did the lower doses; however, the difference was not significant (P = 0.067) in the severe group (SIC-SOFA score ≥ 8). In contrast, in the non-severe group (SIC-SOFA score < 8), mSOFA scores showed a similar decrease between the two AT dose groups (P = 0.783).

Changes in mSOFA scores according to pAT/BW. Changes in mSOFA scores after AT product administration are shown in the two SIC-SOFA groups. (A) Non-severe group, SIC-SOFA score <8 at day 0; (B) Severe group, SIC-SOFA score ≥8 at Day 0. mSOFA, modified sequential organ failure assessment, which is a sum of five organ scores (lung, coagulation, hepatic, cardiovascular, and renal scores); SIC-SOFA, sepsis-induced coagulopathy associated SOFA score, which is a sum of four organ scores (lung, hepatic, cardiovascular, and renal scores); AT, antithrombin; pAT/BW, plasma AT-equivalent AT dose per body weight per day.
Discussion
The present study aimed to evaluate the importance of the titrated AT dose per patient body weight in early DIC recovery. We identified two relevant factors associated with early DIC recovery. DIC recovery by Day 7 was significantly related to the AT dose per body weight and SIC-SOFA score on Day 0. Time-to-event analysis revealed that compared with a lower AT dose, a higher AT dose significantly shortened the DIC recovery time in the severe group. In contrast, no difference in DIC recovery time was observed in the non-severe group. Similarly, in the severe group, a higher AT dose decreased the mSOFA score faster than a lower AT dose; however, the difference was not significant. In contrast, in the non-severe group, the DIC recovery time and the transition of the SOFA score were not significantly different between the two AT groups. In brief, a sufficient AT dose adjusted for patient body weight could provide more remarkable therapeutic efficiency for DIC recovery in septic DIC patients with severe organ failure.
It is well known that inflammation and coagulation play important reciprocal roles in sepsis.1,2 AT has an anticoagulant effect by regulating thrombin, factor Xa, and factor XIa. Moreover it has an anti-inflammatory effect via several mechanisms. AT binds heparan sulfate on vascular endothelial cells, which is followed by the production of prostacyclin (PGI2).15,16 This local increase in prostacyclin suppresses the adhesion of activated platelets and neutrophils to vascular endothelial cells and produces inflammatory cytokines. Additionally, AT reduces neutrophil migration, inflammatory cytokine production, and chemokine receptor expression, resulting in anti-inflammatory action. The anti-inflammatory and anticoagulant effects suppress hypercoagulopathy. These complementary features can promote DIC recovery and halt the progression of severe organ failure. On the other hand, since the fibrinolysis data were not collected in this study, the triadic relationship between AT, fibrinolysis, and attenuation of organ damage remains unknown.
Regarding the relationship between AT dose and DIC recovery, Iba et al. showed that the DIC recovery rate on the seventh day in the 3000 IU/day group was better than that in the 1500 IU/day group. 10 Nonetheless, there are no reports focusing on the relationship between AT dose per body weight and DIC recovery. In a previous study comparing pAT and AT-gamma, AT-gamma provided a consecutive improvement in JAAM-DIC and mSOFA scores. 8 However, in comparison with pAT, AT-gamma exhibited a significantly higher plasma-equivalent AT dose per body weight. These findings led to the hypothesis that a sufficient amount of AT, considering patient body weight, might mitigate septic DIC, particularly in obese or well-built patients. The present study also demonstrated that the AT dose considering patient body weight could affect the DIC recovery time.
The current study demonstrated that the two AT dose groups did not significantly differ in DIC recovery time in the non-severe MOD group. The severe MOD group showed that a higher AT dose significantly shortened DIC duration and possibly improved the mSOFA score faster than a lower dose. These findings indicate that the efficacy of AT treatment for septic DIC might vary with the severity of organ failure. Non-severe patients had less hypercoagulation and/or hyperinflammation. Thus, AT supplementation might not greatly affect DIC recovery and the improvement of organ failure. There have been several reports on the SOFA score at the start of DIC treatment. Yamakawa et al. reported that anticoagulation therapy against DIC significantly improved the outcome in the 13–17 points SOFA score group. In contrast, the other score groups did not show a significant difference in survival benefit. 11 Additionally, Kawano et al. revealed that the survival benefit of rTM for infectious DIC deteriorated as the SOFA score increased. Furthermore, according to ROC curve analysis, the best SOFA score for predicting therapeutic effect was 10. 17 These reports nearly coincide with our result that a SIC-SOFA score ≥ 8 is an independent factor for early recovery from DIC. In other words, the SOFA score may be an important determinant of the therapeutic effectiveness of DIC treatment. In the future, appropriate severity stratification, such as the SOFA score, might be required to assess the therapeutic efficacy of septic DIC treatment.
During sepsis, intracellular proteins, such as high-mobility group box 1 (HMGB-1), histones, and nucleosomes, known as damage-associated molecular patterns (DAMPs), are released extracellularly. These cause neutrophil extracellular traps. Thus, DAMPs activate immune cells, and the consequent inflammatory response is self-enhanced. 18 DAMPs also have a procoagulant effect. 19 Therefore, DAMPs play a critical role in the pathogenesis of DIC. 20 In previous studies, the blood HMGB-1 concentration correlated with the SOFA score and DIC occurrence. 21 Furthermore, Yokoyama et al. reported an association between blood histone H3 levels and multiple organ failure in ICU patients. 22 Therefore, DAMPs play a critical role in organ failure associated with sepsis. Thus, it has been suggested that modulation of DAMPs may be the key to manage MODs caused by sepsis. However, little is known about whether AT can regulate DAMPs. In animal studies, high AT doses have been reported to neutralize HMGB-1. 23 Additionally, Iba et al. demonstrated that AT-gamma protects against histone-induced damage of vascular endothelial cells. 24 It has been reported that AT and rTM combination therapy regulates leukocyte necrosis and reduces DAMPs, such as histone H3 and cf-DNA, in the blood in lipopolysaccharide-induced rat sepsis models. 25 Thus, AT supplementation might be a potential option to counteract the deleterious effects of DAMPs. Future studies should investigate whether AT supplementation can reduce DAMP toxicity in septic DIC.
Whether combination treatment with AT and rTM is viable, remains controversial. AT and rTM exhibit similar anti-inflammatory effects and anti-coagulation features via thrombin conjugation.16,26 However, rTM-thrombin complex activates protein C, and inactivation of Va and VIIIa inhibits further thrombin production, 27 which is separate from the inhibition of Xa and Xia caused by AT. Moreover, it was demonstrated that activated protein C can degrade histones H3 and H4, 28 therefore, rTM can reduce the damage induced by histones. 29 Therefore, we anticipated that the simultaneous administration of AT and rTM would exhibit additive and synergistic effects. In the present study, however, contrary to the theoretical understanding, combination treatment with rTM did not effectively contribute to early DIC recovery. Similarly, using data extracted from the Japanese Diagnosis Procedure Combination database, Umegaki et al. retrospectively compared the efficacy of AT alone and AT + rTM in septic DIC patients who were ventilated. They concluded that AT + rTM combination therapy did not improve the prognosis more than AT monotherapy. 30 In a similar retrospective study using the DPC database, Suzuki et al. assessed rTM alone versus rTM + AT in patients with DIC due to severe community-acquired pneumonia and found no benefit of combination therapy. 31 Furthermore, Umemura et al. found equivalent efficacy and similar in-hospital mortalities between monotherapy and combination therapy. They performed a propensity score-adjusted comparison using nationwide retrospective cohort data. 32 In contrast, Iba et al. 33 obtained favorable findings for combination therapy, using propensity score matching from multi-institutional post-marketing surveillance data. The pharmacological rationale for combination therapy has been demonstrated in various animal experiments mentioned above. However, the discrepancies between these results have not been elucidated. The present results cannot reveal the difference in efficacy between AT and rTM. That is because the administrating principles for each drug are different. A possible explanation for the rTM ineffectiveness could be that rTM was administered to patients with a markedly low platelet count; therefore, severe thrombocytopenia at intervention could have delayed DIC score improvement.
This study had several limitations. First, two groups of AT administered patients, in which either the plasma-derived AT product or the rAT product was administered, were enrolled. A phase 3 trial by Endo et al. compared the therapeutic efficacy between both AT products under strictly controlled equivalent doses. Their efficacies were quite similar; 34 therefore, the present study was conducted using mixed AT products. In the future, prospective observational studies on the AT dose using AT-gamma, which can be set in the range of 36–72 IU/kg/ of body weight, are warranted. Second, this was a single-center, retrospective observational study, and the major problems were selection bias and the small number of enrolled patients. Third, the DIC treatment policy at our institution may have had a strong impact on the results. In this study, rTM administration did not reduce the duration of DIC. However, the enrolled patients were AT-administered patients whose AT levels had decreased to approximately 50%. Furthermore, patients treated with rTM alone were excluded from the study. Therefore, the present results should be limited to low antithrombin activity in patients with septic DIC, and we can consider that with concurrent rTM treatment there may be some influence on the progression of septic DIC. It would be better to conceive that rTM does not necessarily indicate that it is ineffective for DIC. Fourth, based on the results of the present study, we could not clarify the mechanisms of DIC recovery and organ failure improvement from the perspective of the fibrinolytic function. Therefore, fibrinolytic function should be investigated in a future clinical study.
Conclusion
Both the organ failure score and an appropriate AT dose considering patient body weight could be independent factors influencing early DIC recovery in patients with sepsis. Additionally, it was demonstrated that DIC recovery differed between the two AT dose groups with severe MODs. Thus, the efficacy of AT supplementation against septic DIC might depend on the severity of MODs and an appropriate AT dose. In other words, the AT dose for septic patients with DIC who present with severe MOD should be carefully determined in clinical practice.
However, this was a single-center retrospective study; therefore, it has insufficient statistical robustness. Multicenter randomized trials are required to confirm the AT dose benefit stratified by organ failure severity.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
YM received grants from JIMRO Co. Ltd (Takasaki, Japan), personal fees from MSD K.K. (Tokyo, Japan), personal fees from Asahi Kasei Pharma Co. Ltd (Tokyo, Japan), and personal fees from Japan Blood Products Co. Ltd (Tokyo, Japan), outside the submitted work. HT received lecture fees from TSUMURA & Co. The other authors declare that they have no conflicts of interest.
Funding
Credits and grant information: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Research Ethics and Patient Consent
This study was approved by the Institutional Review Board of Sapporo Medical University Hospital (332-27), which waived the requirement for informed patient consent because of the anonymous nature of the data.
Author Contributions
HK and YM contributed to the conception and design of the study and drafted the manuscript. HK extracted the clinical data, performed the statistical analysis, and interpreted the results. HT created the patient database system and completed the clinical data. TS supervised the statistical methodology and provided statistical instruction. All authors approved the final manuscript.
