Abstract
Background
The optimal timing for initiating oral anticoagulation (OAC) after acute ischemic stroke in patients with atrial fibrillation (AF) remains a critical clinical dilemma. Early anticoagulation may lower recurrent ischemic stroke risk but could raise hemorrhagic transformation risk, and current guidelines are conflicting. This meta-analysis examined the impact of early versus late initiation of anticoagulation on clinical outcomes—recurrent stroke, intracranial haemorrhage, Bleeding, mortality, transient ischemic attack (TIA), and thrombosis—in AF-related ischemic stroke.
Methods
Following PRISMA guidelines, we conducted a systematic review and meta-analysis of observational and experimental studies that compared early and late anticoagulation; seventeen studies met inclusion criteria.
Results
Early anticoagulation was associated with a significantly lower risk of recurrent ischemic stroke (OR = 0.72, 95% CI [0.55-0.96], P = .03). The incidence of intracranial hemorrhage did not differ significantly between groups (OR = 1.13, 95% CI [0.83-1.53], P = .44). No significant differences were observed for Bleeding (OR = 0.87, 95% CI [0.67-1.12], P = .27), mortality (OR = 0.94, 95% CI [0.72-1.24], P = .68), TIA (OR = 0.99, 95% CI [0.57-1.74], P = .98), or thrombosis (OR = 0.87, 95% CI [0.59-1.27], P = .47). Notable heterogeneity—likely due to differences in study design, anticoagulant type, and stroke severity—limits firm conclusions.
Conclusion
Early anticoagulation appears to reduce ischemic recurrence without significantly increasing major hemorrhagic events, but randomized trials are needed to define optimal OAC timing.
Keywords
Introduction
Atrial fibrillation (AF) is the most prevalent cardiac arrhythmia worldwide, contributing to 13%-26% of all acute ischemic stroke. 1 Without anticoagulation, the likelihood of early recurrence within the first 14 days after an ischemic stroke due to AF is estimated to be between 0.5% and 1.3% per day, based on historical observational data, including control groups in randomized controlled trials. 2
Risk factors for recurrent ischemic stroke in AF patients include atrial thrombus, left ventricular dysfunction, atrial enlargement, age, larger infarct volume, and an elevated CHA2DS2-VASc score.3,4
For decades, vitamin K antagonists (VKAs) have been the mainstay for stroke prevention in AF patients. Direct oral anticoagulants (DOACs), including dabigatran, apixaban, rivaroxaban, and edoxaban, have been increasingly used for primary and secondary stroke prevention in non-valvular AF patients in recent years. Compared to VKAs, DOACs notably reduce cerebral haemorrhage risk, even in patients with a previous stroke history. 5
In patients with AF, oral anticoagulation (OAC) is well-established and strongly recommended to prevent recurrent ischemic strokes.6,7 A significant and unresolved clinical challenge is determining the optimal time to initiate (OAC) therapy in patients with non-valvular AF. Regarding when and how to begin OAC following the development of an ischemic stroke caused by AF, the current guidelines are vague and contradictory. The European Heart Rhythm Association of the European Society of Cardiology (EHRA-ESC) 8 established the “1-3-6-12 days rule that talks about the timing of initiation (OAC) in AF patients in 2013, responding to data showing that massive infarcts, which result in severe stroke symptoms, are more likely than small infarcts to experience hemorrhagic transition. 9 This guideline has been adopted, with some modifications, by several associations (such as EHRA-ESC and the European Stroke Organization) and nations (such as Canada, Australia, Middle Eastern countries, and North African countries; panel), even though the timepoints and definitions of stroke severity are solely based on expert consensus.
OAC should be started 4-14 days following the onset of neurological symptoms, according to the 2018 American Heart Association/American Stroke Association (AHA/ASA) guidelines for the early care of patients with ischemic stroke. 9 Only 93 (12%) of the 1029 consecutive patients with acute ischemic stroke and known or newly diagnosed AF who were treated with DOACs were included in the Early Recurrence and Cerebral Bleeding in Patients with Acute Ischemic Stroke and AF (RAF) prospective observational study, which served as the basis for this recommendation. 10
The efficacy and safety of DOACs within 14 days following a stroke have not yet been established according to German society guidelines; 11 nevertheless, it offers no advice based on this assertion. While ESC, EHRA, and AHA/ASA guidelines are extensively utilized in India and Latin America, we only located guidelines for Japan (2011) that predate the adoption of DOACs. 12
Notwithstanding, the timing of OAC initiation remains largely subjective and lacks strong evidence-based guidance. Early initiation may reduce the risk of recurrent ischemic stroke but also increases the risk of hemorrhagic transformation (HT), a severe early complication. 12
In clinical practice, starting anticoagulation as soon as possible after the stroke event is preferable, given the high recurrence risk in the acute phase. Additionally, adherence rates are improved when inpatient care includes early preventive measures.13,14
The European Heart and Rhythm Association (EHRA) provides expert opinions suggesting a DOAC initiation window of 1-12 days post-stroke or transient ischemic attack (TIA), depending on infarct size. 15 According to a review by Paciaroni et al, the ideal window for starting anticoagulation to prevent recurrent strokes is within 4-14 days after the initial stroke. 16
Early initiation of anticoagulation post-ischemic stroke significantly lowers the long-term risk of recurrence. Still, it carries a risk of hemorrhagic transformation, making it challenging to determine the precise timing for therapy initiation. 17
In light of persisting discrepancies and a lack of consensus on the optimal timing for starting anticoagulation after ischemic stroke, this systematic review and meta-analysis were performed to answer this important clinical question.
Methods
Study Protocol
This meta-analysis was conducted in accordance with the Cochrane Handbook for Systematic Reviews and Meta-Analyses. 18 The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were followed. 19
Identification of Eligible Studies
Free text terms and medical subject headings (MeSH) terms were used in the searching process for the following search strategy: “Stroke” AND “Atrial fibrillation” AND “Anticoagulants” OR “Vitamin K antagonist” OR “VKA” OR “Direct oral anticoagulants” OR “New oral anticoagulants” OR “DOAC” OR “NOAC” OR “Warfarin” OR “Dabigatran” OR “Apixaban” OR “Edoxaban” OR “Rivaroxaban”. Filters for title and abstract were applied in PubMed. For Scopus, title, abstract, and keywords were used, while for Web of Science, the abstract was used only. The search strategy was developed and implemented in accordance with the specific criteria of each database. All studies from conception up to January 2025 were included.
Inclusion and Exclusion Criteria
For a study to be included, it must follow this PICOS criteria: population (patients with AF who developed an acute ischemic stroke), intervention (early anticoagulation), comparison (late anticoagulation), outcomes (reoccurrence of stroke, intracerebral hemorrhage [ICH], bleeding, mortality, TIA, or thrombosis), study design (observational or randomized controlled trials [RCTs]). Articles were excluded if they were: (1) animal studies, (2) abstracts, (3) not published in English, (4) of an inappropriate study design (reviews, case reports, editorials, guidelines, letters to an editor, or book chapters), or (5) duplicate studies.
Study Selection
After the identification of eligible studies and the removal of duplicates using EndNote. The title and abstract screening process was conducted by two authors who independently assessed each study in accordance with the inclusion and exclusion criteria. The full texts of the remaining articles were retrieved and subjected to a full-text screening. Any discrepancies in the two-stage screening process were identified and resolved by a senior author (OB).
Data Extraction
The following baseline criteria were extracted from each study: the last name of the first author, publication year, study design, the anticoagulants used, definition of early and late anticoagulation and study groups. For each group, we extracted the number of patients, the mean age with standard deviation (SD), the number and percentage of male participants, and the number and percentage of those with congestive heart failure or arterial hypertension. For study outcomes, we extracted the number of events of stroke reoccurrence, ICH, bleeding, TIA, thrombosis and mortality. The data extraction was conducted independently by two authors, and a senior author resolved any discrepancies.
Risk of Bias Assessment
The revised Cochrane risk-of-bias tool (ROB2) was used for assessing the quality of RCTs. 20 This tool evaluates the quality of each study across five domains: randomization procedures, deviation from the intended intervention, missing data, measurement of outcomes, and selection of reported outcomes. The Newcastle-Ottawa Scale (NOS) was used for assessing the quality of observational studies. 21 This tool assesses the quality of each study across questions grouped under the selection, comparability, and outcomes domains, with scores of 4, 2, and 3 for each domain, respectively. The quality assessment was conducted independently by two authors, with conflicts resolved by the senior author.
Statistical Analysis
The statistical analysis was conducted using Cochrane Review Manager (RevMan) software. We used the odds ratio (OR) by pooling the events and the total. The random effects model was used to account for heterogeneity among the included studies, with 95% confidence intervals (CI) and a P-value of ≤.05 considered statistically significant.
Results
Search Results and Study Selection
The search process resulted in 2845 articles, including 1271 duplicates. Therefore, this yielded 1574 articles for title and abstract screening. According to this process, 22 articles were eligible for full-text screening, resulting in 17 studies being included in the current systematic review and meta-analysis. (Figure 1)
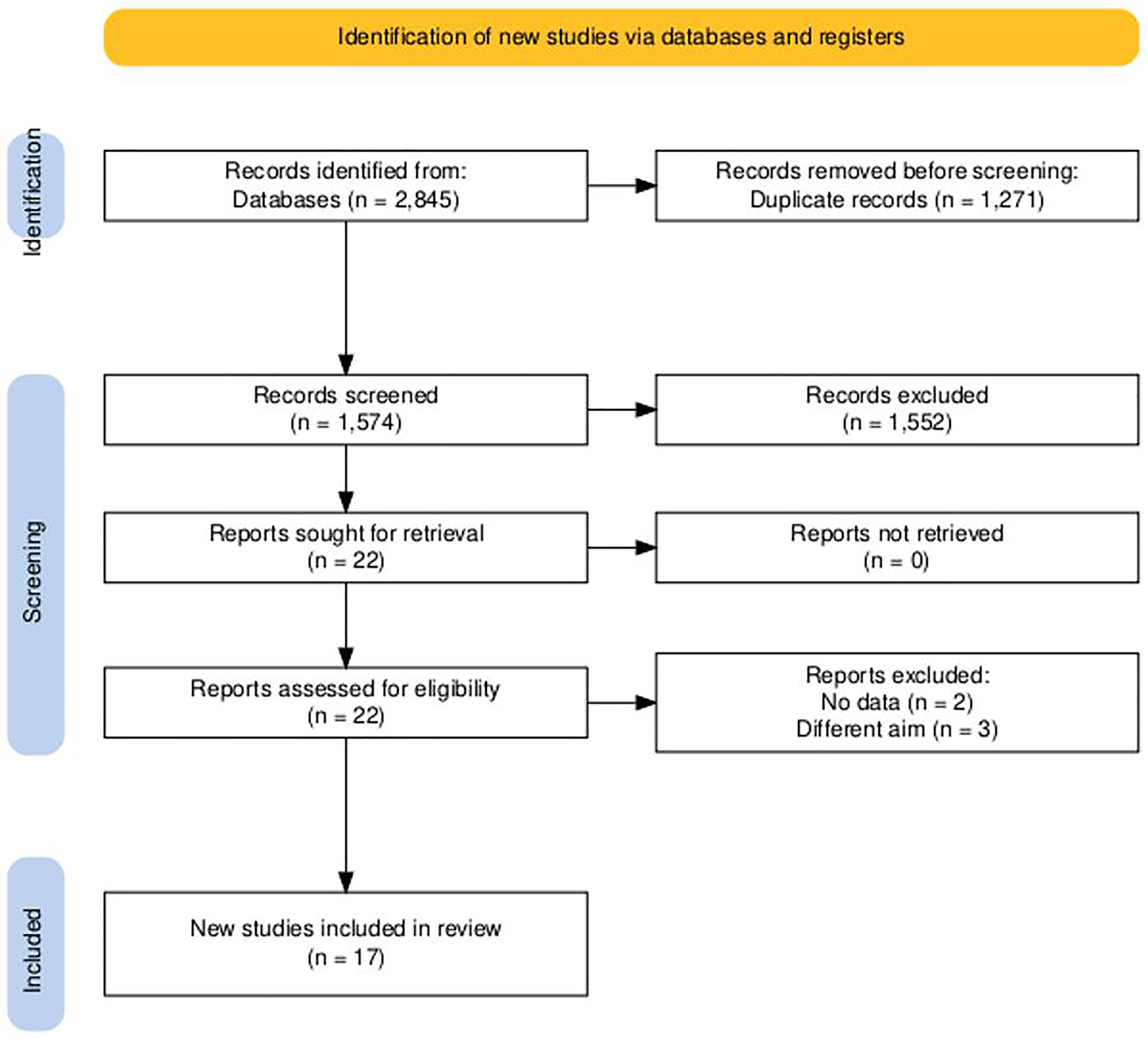
PRISMA Flow Diagram of Searching and Screening.
Characteristics of the Included Studies
Table 1 shows the characteristics of the studies included in this review. A total of 17 studies were included.22–38 Regarding study design, 13 studies were observational, and four were interventional. Among the interventional studies, only one study was non-randomized. 35 The sample size within each study arm ranged from 4 to 1362 patients. Variations in mean age and percentage of male participants were observed across all studies. Arterial hypertension was a common comorbidity, with at least 63% of participants in each arm of the studies reporting the prevalence of this comorbidity. Only seven studies provided prevalence data of coexisting congestive heart failure in these patients, with a much lower prevalence than that of arterial hypertension. The anticoagulants used included DOAC, VKA, Rivaroxaban, warfarin, and heparin. There were variations in the definitions of early and late anticoagulation across the studies, with some studies stratifying the definition per stroke severity. In the included studies, early anticoagulation was defined variably, generally corresponding to initiation of OAC within ≤3-7 days of the index ischemic event. Some studies adopted a stratified definition based on stroke severity, initiating within 1-3 days for TIA or minor stroke, 4-6 days for moderate stroke, and 7-14 days for severe stroke, consistent with the EHRA “1-3-6-12-day rule.” Late anticoagulation was defined as initiation after 7-14 days or beyond the timeframe recommended by current guidelines (AHA/ASA 2018, 2021). (Table 1)
Characteristics of the Included Studies.
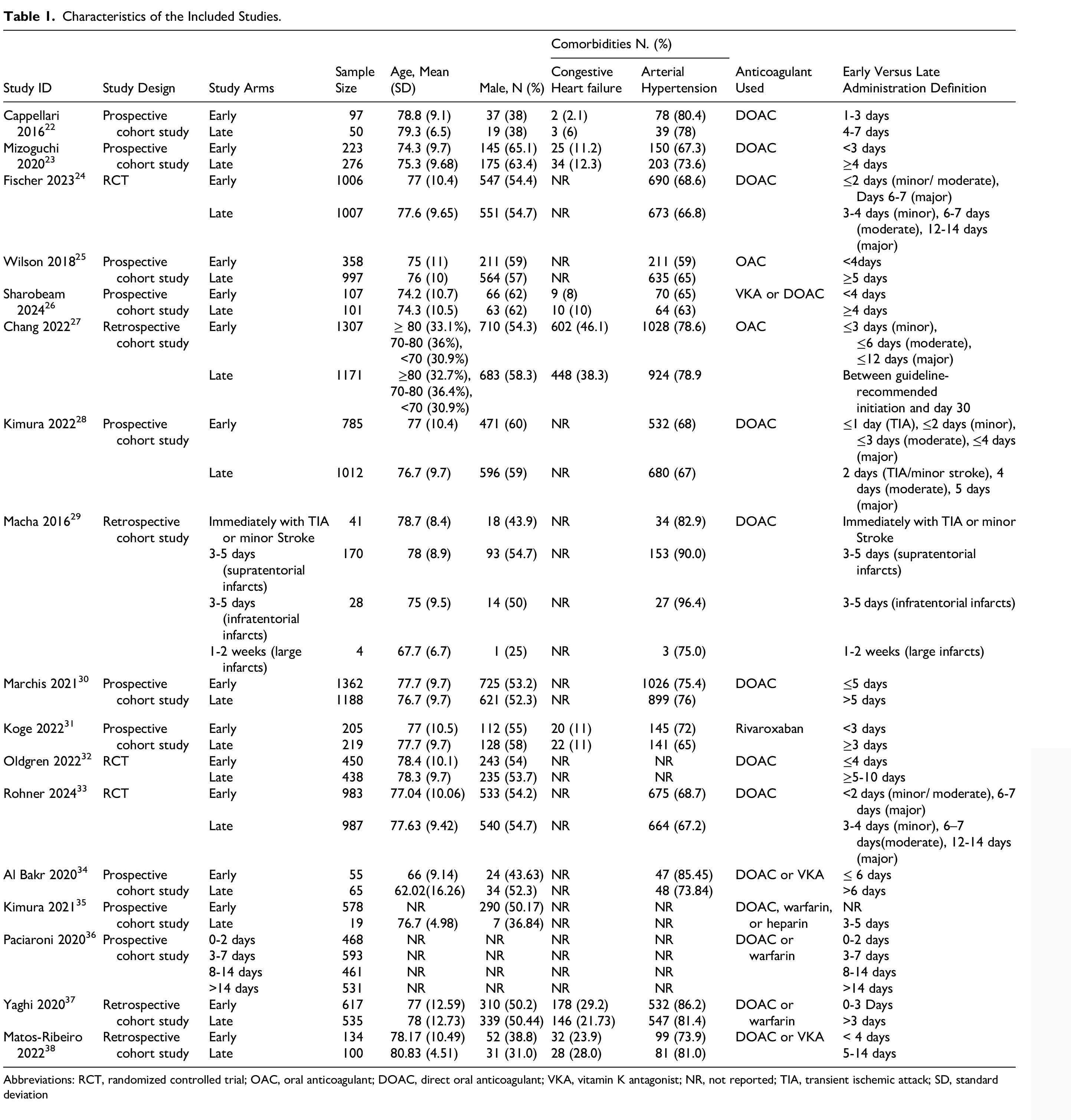
Abbreviations: RCT, randomized controlled trial; OAC, oral anticoagulant; DOAC, direct oral anticoagulant; VKA, vitamin K antagonist; NR, not reported; TIA, transient ischemic attack; SD, standard deviation
Quality Assessment
According to NOS, the 14 included cohort studies were considered high quality. (Supplementary Table 1) The risk of bias assessment of the RCTs revealed that the three studies were of low risk of bias. (Supplementary Figure 1)
Outcomes
Stroke
Twelve studies reported the incidence of recurrent stroke with a total of 576 patients, 199 of whom were in the early group, and 377 were in the late group. The data revealed significant results favoring the early introduction of anticoagulants over their late introduction (OR = 0.72, 95% CI [0.55 to 0.96], P = .03). However, there was significant heterogeneity among the included studies (P = .03, I2 = 47%). (Figure 2) Minimal publication bias was observed. (Supplementary Figure 2)

Comparison Between Early Anticoagulation and Late Anticoagulation in AF Patients Regarding the Risk of any Stroke.
Intracerebral Hemorrhage
Eleven studies reported the incidence of ICH after the index stroke in a total of 204 patients, with 117 in the early group and 87 in the late group. The analysis showed no significant favoring of the late introduction of anticoagulants (OR = 1.13, 95% CI [0.83 to 1.53], P = 0.44). (Figure 3) The included studies were homogeneous (P = 0.34, I2 = 11%). Publication bias was observed, as indicated by the funnel plot. (Supplementary Figure 3)

Comparison Between Early Anticoagulation and Late Anticoagulation in AF Patients Regarding the Risk of Intracerebral Hemorrhage.
Bleeding
In our analysis, eight studies reported bleeding after the index stroke as a complication in a total of 258 patients, 120 of whom were in the early group and 138 were in the late group. The analysis presented results that show no significant favoring of the early use of anticoagulants (OR = 0.87, 95% CI [0.67 to 1.12], P = .27), and there was no heterogeneity among the included studies (P = .53, I2 = 0%). (Figure 4)

Comparison Between Early Anticoagulation and Late Anticoagulation in AF Patients Regarding the Risk of Bleeding.
Mortality
Seven studies reported the incidence of death during the follow-up period following the index stroke in a total of 231 patients, out of which 102 were in the early group, and 129 were in the late group. The data revealed insignificant differences in the risk of mortality between the two groups (OR = 0.94, 95% CI [0.72 to 1.24], P = .68), and the included studies were homogeneous (P = .36, I2 = 9%). (Figure 5)

Comparison Between Early Anticoagulation and Late Anticoagulation in AF Patients Regarding the Risk of Mortality.
TIA or minor Stroke
Eight studies reported the incidence of TIA or minor stroke during the follow-up period after the index stroke in a total of 7078 patients, 3151 of whom were in the early group and 3927 were in the late group. There was no statistically significant difference in the incidence of TIA or minor stroke between the two groups (OR = 0.99, 95% CI [0.57 to 1.74], P = .98), and the included studies were minimally heterogeneous (P = .20, I2 = 28%). (Figure 6)

Comparison Between Early Anticoagulation and Late Anticoagulation in AF Patients Regarding the Risk of TIA or minor Stroke.
Thrombosis
Only 4 of the included studies reported the incidence of thrombosis after the index stroke in a total of 5687 patients, of which 2739 were in the early group and 2948 were in the late group. The data revealed results that were insignificantly in favor of the early introduction of anticoagulants (OR = 0.87, 95% CI [0.59 to 1.27], P = .47), and there was no heterogeneity among the included studies (P = .42, I2=0%). (Figure 7)

Comparison Between Early Anticoagulation and Late Anticoagulation in AF Patients Regarding the Risk of Thrombosis.
Discussion
There is a debate regarding the optimal time for administration of anticoagulants, either VKAs or non-VKAs, after acute ischemic stroke in patients with AF. In this review of 17 included studies, we aimed to compare the benefits and harms associated with the early and late use of anticoagulants in those patients. We found that the early introduction of anticoagulants was associated with a lower incidence of recurrent ischemic stroke events. However, there was no significant difference between the two administration time points regarding Bleeding, ICH, mortality, TIA, and thrombosis.
Real-world data emphasize the importance of guideline-adherent anticoagulation practices to improve stroke prevention in AF. Our findings further reinforce the clinical need for timely initiation consistent with current guideline frameworks. 39 Prevention of ischemic stroke recurrence is crucial, making early anticoagulant administration preferred over late administration; according to our findings, the lower odds of recurrence were observed in the early intervention group. These findings are consistent with the two previously published meta-analyses by Babu 2024 et al 40 (OR 0.72, 95% CI 0.52 to 0.98) and Jiang 2023 et al 41 (OR: 0.68; 95% CI: 0.55,0.84; P = .0003). These results are consistent with those obtained from the ELAN trial, 24 in which the early DOAC led to recurrence in only 1.4% of the early group compared to 2.5% in the late one (OR 0.57, 95% CI 0.29 to 1.07). These findings suggest the superiority of early anticoagulant use over late administration; however, there was significant heterogeneity in the pooled analysis among studies. This heterogeneity may arise from differences in study design between the included studies, variations in the administered drugs, and the differing definitions of both early and late. Additionally, the AF cause was specified as non-valvular, whereas others did not mention a specific etiology. Additionally, the patients were not stratified according to whether they were of high-risk or low-risk profiles for recurrent ischemic stroke.
Mizoguchi 2020 et al 23 found that early anticoagulant use was associated with higher stroke events by 4.5% compared to late use by 2.5% (OR 1.8, 95% CI 0.68 to 4.82). In the study by De Marchis 2021 et al, 30 1.1% of the early group experienced recurrence compared to 0.5% in the late group. Those findings do not align with the meta-analyses and the other studies included.
Regarding ICH, the most serious complication associated with the use of DOAC after ischemic stroke leads to increased mortality and morbidity, 42 resulting in a tendency to delay the use of DOAC after ischemic stroke. In the ROCKET-AF trial, 43 rivaroxaban was postponed for at least 14 days after a minor AIS and 90 days after a major ischemic stroke, while the ARISTOTLE study 44 started apixaban use at least 7 days after any ischemic stroke. Most studies that delayed the use of DOACs were conducted in the pre-DOAC era, when heparin, low-molecular-weight heparin (LMWH), and aspirin were in use.
Our meta-analysis suggests that the use of early treatment is inferior to late treatment in ICH; however, there was no statistically significant difference between the two groups, and no heterogeneity was observed in the pooled analysis. These findings were similarly observed in the previous two meta-analyses. The TIMING trial 32 applied DOAC and found that the early treatment group (within 4 days) was non-inferior to the late group (within 5-10 days) regarding the incidence of ICH. It is worth noting that the incidence of ICH is not only related to the timing of drug administration but also has a multifactorial etiology, including the size and location of the infarction and patient characteristics such as the presence of a bleeding tendency.
The data obtained from the observational studies only provide uncertain directions, and the low quality of evidence makes it challenging for stroke physicians every day to determine if they should initiate the anticoagulant in AF-related ischemic stroke or not. Field experts still advocate for patient-personalized decisions, making them highly dependent on the physician's judgment. 45
The 2021 AHA/ASA guidelines recommend delaying the initiation of DOAC beyond 14 days in high-risk patients to reduce the risk of ICH. In contrast, patients at low risk of ICH may initiate DOAC after 2-14 days to reduce the risk of RIS. 46 Differences in guideline recommendations stem from studies conducted in the pre-DOAC era and do not differentiate between DOAC and VKA. 26
Despite the proven benefits of VKAs, their narrow therapeutic window and bleeding complications such as diffuse alveolar hemorrhage have prompted a shift toward DOACs for stroke prevention in AF. 47 DOACs have demonstrated superior safety and practicality compared to VKAs in post-stroke anticoagulation. Their predictable pharmacokinetics, rapid onset of action, and lack of need for INR monitoring enable earlier and more consistent initiation. Studies such as TIMING 32 and ELAN 24 have shown that DOACs initiated within 4 days are non-inferior to delayed initiation (5-10 days) in terms of ICH risk and superior in reducing ischemic recurrence. Conversely, VKA-based regimens, as reflected in older trials like ROCKET-AF 43 and ARISTOTLE, 44 were typically delayed due to concerns about labile INR and higher hemorrhagic risk. Hence, early DOAC initiation—particularly within ≤7 days—may be preferable in clinically stable patients with mild-to-moderate strokes, while VKA use warrants a more conservative, delayed approach. The findings of this review highlight the evolving role of early and optimized anticoagulation strategies in patients with AF. Evidence from large-scale patient-level meta-analyses, particularly the COMBINE AF collaboration reported by Carnicelli et al 48 , provides robust data supporting the superiority of DOACs over warfarin for stroke prevention in nonvalvular AF. In that analysis of 71 683 patients, standard-dose DOACs significantly reduced the risks of stroke or systemic embolism (HR 0.81, 95% CI 0.74-0.89), all-cause mortality (HR 0.92, 95% CI 0.87-0.97), and intracranial bleeding (HR 0.45, 95% CI 0.37-0.56) compared with warfarin, without increasing major bleeding. These results underscore the clinical value of initiating DOAC therapy early, especially in patients without prior VKA exposure and those with lower creatinine clearance, who derived the greatest net benefit. Furthermore, younger patients and those with lower body weight appeared to have a more favorable bleeding profile, reinforcing the safety of DOACs across demographic subgroups.
Across the included studies, early anticoagulation was typically initiated within 3-7 days after the index event, aligning with the EHRA-ESC “1-3-6-12-day rule”, 8 which recommends starting OAC 1 day after TIA, 3 days after minor stroke, 6 days after moderate stroke, and 12 days after severe stroke. Our findings indicate that initiating OAC within this early window significantly reduced recurrent ischemic stroke risk without a parallel increase in ICH. Conversely, evidence for severe strokes remains less conclusive, supporting guideline recommendations for delayed initiation up to 12-14 days in large infarcts. The AHA/ASA (2018, 2021) 9 similarly advises initiating OAC between 4 and 14 days, depending on the risk of hemorrhagic transformation.
According to our meta-analysis, the early treatment group experienced a lower incidence of bleeding events compared to the late treatment group. However, there was no statistically significant difference between the two groups. The last meta-analysis, 40 which included only studies that applied DOAC, found no significant difference between the two groups in major or non-major hemorrhagic events (P = .26) and (P = .21). Jiang 2023 et al 41 found no significant differences in the incidence of hemorrhagic events (P = .26). Our findings are consistent with those of previous meta-analyses, despite the differences in the OAC strategies applied. The recent literature suggests that DOACs are superior to VKAs in reducing the risk of bleeding, with similar therapeutic effects. 49
There was no statistically significant difference in mortality between the two groups; however, Wilson et al 25 found higher mortality rates in the late group compared to the early group, and they attributed this to the delay in administering OAC in patients with more severe strokes and comorbidities. However, the most common causes of death were non-vascular causes, highlighting the non-importance of OAC timing effect on mortality.
TIA events were comparable between the two groups, with a tendency to increase in the early group. The pooled analysis was heterogeneous, and this heterogeneity was resolved by removing Koge 2022 et al, 31 who applied rivaroxaban after reperfusion therapy.
Strengths and Limitations
Our study has the most comprehensive, up-to-date evidence synthesized from a meta-analysis with larger sample size and more included studies than previous meta-analyses; however, our study has several limitations: (1) the heterogeneity of the included study designs; (2) only three randomized controlled trials were included; (3) many OAC strategies were applied in the included studies such as VKA, LMWH, and DOAC; (4) different definitions of early and late across the included studies; (5) stroke severity was not addressed in the analysis, as there was no data categorized according to the stroke severity in the included studies; additionally the late group was associated with higher morbidity patients.
Conclusion
Our study highlights the importance of early anticoagulant administration in preventing ischemic stroke recurrence. Despite the non-significant difference regarding ICH, early anticoagulants may be preferred as they are associated with lower odds of developing ICH. Additionally, there was no significant difference between the two groups in terms of bleeding, mortality, TIA, or thrombosis, with a comparable number of events in both groups. The current evidence is limited due to heterogeneity among the studies included in the analysis, and it cannot definitively conclude the timing of anticoagulant administration. Further, RCTs should address the limitations in recent literature to make a more robust decision.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251410869 - Supplemental material for Anticoagulation Timing After Stroke in Atrial Fibrillation: Evidence from a Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296251410869 for Anticoagulation Timing After Stroke in Atrial Fibrillation: Evidence from a Systematic Review and Meta-Analysis by Mostafa Hossam El Din Moawad, Suhel Batarseh, Abdelaziz A. Awad, Raghad Amro, Ahmed Farid Gadelmawla, Omar A. Kaoud, Dina Essam Abo-elnour, Muhammad Daoud Tariq, Osama Omar Ballut, Judy Ahmed Abdullah, Ammar Mektebi, Ibrahim Serag, Ahmed Alattar, Ibraheem M. Alkhawaldeh, Hamza A. Abdul-Hafez, Mohamed Abouzid, Mohamed O Mohamed and Osama Bisht in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Contributions
Mostafa Hossam El Din Moawad, Suhel Batarseh, Raghad Amro, Ahmed Farid Gadelmawla, Omar A. Kaoud, Dina Essam Abo-elnour, Muhammad Daoud Tariq, Osama Omar Ballut, Judy Ahmed Abdullah, Ammar Mektebi, Ibrahim Serag, Ahmed Alattar, Ibraheem M Alkhawaldeh, Hamza A. Abdul-Hafez, Mohamed Abouzid, Mohamed O Mohamed, Osama Bisht: contributed equally
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
