Abstract
Background
Hemogram is a novel biodegradable powdered hemostatic agent developed by binding calcium ions (Ca²+) to carboxymethyl starch (CM-starch) and applying granulation technology to enhance blood absorption and hemostatic efficacy. This study aimed to evaluate the clinical efficacy and safety of Hemogram compared with the commercially available agent Arista™ AH in patients undergoing gastrectomy.
Methods
This randomized controlled trial was conducted at Samsung Medical Center and included patients who experienced intraoperative oozing-type bleeding during gastrectomy. A total of 64 patients were enrolled and evenly randomized to receive either Hemogram (n = 32) or Arista™ AH (n = 32) for local hemostasis. The primary efficacy endpoint was the hemostatic success rate within 5 min. Secondary endpoints included hemostasis time, re-bleeding rate, number of hemostatic agent units used, transfusion volume, length of hospital stay, and incidence of adverse events.
Results
Hemostatic success within 5 min was achieved in 100% (32/32) of the Hemogram group and 96.9% (31/32) of the Arista™ AH group, with no statistically significant difference between the two (P > 0.05). The mean time to hemostasis was 1.25 ± 0.80 min in the Hemogram group and 1.52 ± 0.85 min in the Arista™ AH group. No re-bleeding events were observed in the Hemogram group, while one case of re-bleeding (3.3%) was reported in the Arista™ AH group. There were no significant differences between groups in terms of transfusion volume, length of hospitalization, or adverse event rates.
Conclusion
Hemogram demonstrated non-inferior hemostatic efficacy and safety compared to Arista™ AH in patients undergoing gastrectomy. These findings suggest that Hemogram is a promising and clinically applicable hemostatic agent for surgical use.
Introduction
Hemorrhage remains a common occurrence during surgical procedures and continues to be one of the leading causes of mortality in surgical patients. 1 While mild intraoperative bleeding can often be managed through direct compression and activation of the physiological coagulation cascade, more substantial hemorrhage may require the use of adjunctive hemostatic agents to minimize blood loss. 2 Traditionally, surgical hemostasis has been achieved using methods such as suturing, hemostatic clips, and energy-based devices like electrocautery. However, when these conventional techniques are insufficient, topical hemostatic agents are employed to achieve adequate bleeding control.
An ideal topical hemostatic agent should be biodegradable, biocompatible, easy to apply, and cost-effective. 3 Starch-based hemostatic agents have been proved to be effective due to their favorable biocompatibility and biodegradability profiles. 4 These microparticles function by absorbing plasma, concentrating clotting factors and platelets at the site of bleeding to accelerate coagulation. Arista™ AH (BD, USA), a commercially available starch-based hemostatic microparticle, has been widely used in various surgical settings for intraoperative bleeding control.
Despite its advantages, Arista™ AH has some limitations, particularly its relatively slow absorption rate, which can cause the particles to float on the surface of blood and hinder effective clot formation to block the bleeding spot.5,6 To overcome these drawbacks, several strategies have been explored, including physical modification to enhance porosity for faster plasma absorption, and the incorporation of metal ions to attract negatively charged erythrocytes and promote coagulation.7–9 Additionally, the introduction of carboxymethyl (CM) groups has been shown to improve hydrophilicity, thereby enhancing fluid absorption, while conjugation with calcium ions (a key factor in the coagulation cascade) has been reported to strengthen platelet aggregation and clot formation.10,11
The objective of this study is to compare the clinical hemostatic performance of the newly formulated granulated carboxymethyl starch and calcium ionic assembly (Hemogram) with that of Arista™ AH in patients undergoing gastrectomy. This investigation aims to evaluate the potential of Hemogram as a superior alternative in terms of intraoperative hemostatic efficiency and clinical applicability.
Methods
Study Population
The clinical trial was conducted at Samsung Medical Center and included patients who underwent gastric cancer surgery between September 2024 and May 2025. Patients were excluded if they were under 19 years of age, pregnant or breastfeeding, had major bleeding rather than exudative bleeding, were receiving immunosuppressive therapy, had chronic underlying diseases, or were taking anticoagulant medications. This study was conducted with the approval of the Institutional Review Board (IRB) of Samsung Medical Center and in accordance with ethical standards and patient confidentiality regulations (SMC 2024-03-112).
Patient Enrolment and Randomization
Based on previous reports indicating a hemostatic success rate of 94.4% for Arista™ AH in general surgical settings, the expected hemostatic success rate of the agent was set at 95% in this study. According to prior clinical data on Hemogram applied in gastric cancer surgery, the rate of bleeding-related complications (eg, intra-abdominal bleeding) was reported to be approximately 1.2%, allowing the estimated hemostatic success rate for Hemogram to be set at 98%. Assuming a 10% difference in success rates between the two groups, with a one-sided significance level of 2.5% and a statistical power of 80%, a total sample size of 64 patients (32 per group) was calculated to be necessary. A total of 67 patients scheduled to undergo gastric cancer surgery were initially screened for enrollment. Among them, 3 patients were excluded due to changes in the surgical plan or withdrawal of consent. Ultimately, 64 patients were randomized in a 1:1 ratio into the test group (Hemogram) and the control group (Arista™ AH), with 32 patients allocated to each group (Figure 1). Fixed randomization was employed, with stratification by sex (male/female), age (<60 / ≥60 years), and surgical approach (open/laparoscopic surgery), resulting in 8 strata. Within each stratum, a block size of 6 was used, and treatment assignments were determined using a random number table based on the Moses-Oakford algorithm. Blinding was not applied in this study.

Flow-Chart of Study Subject Selection.
Evaluation of Hemostasis
Throughout the study, surgical techniques and postoperative management were standardized. All patients received a topical hemostatic agent during surgery, with Hemogram used in the test group and Arista™ AH in the control group. The bleeding site was adequately covered with the assigned agent, and compression was applied for one minute using gauze moistened with saline and wrung out. Hemostasis was considered successful when no additional bleeding occurred, no blood spread onto the applied powder, and no further treatment such as reapplication or repeated compression was required. Hemostasis time was measured in one-minute intervals. Bleeding time was monitored for up to five minutes, and hemostasis was considered unsuccessful if bleeding was not controlled within this period.
Data Collection
Data collection for this clinical trial was conducted in accordance with a pre-approved protocol. All participants provided written informed consent, and eligibility was confirmed during a screening visit based on predefined inclusion and exclusion criteria. Eligible patients were enrolled and evaluated across four time points: preoperative visit, day of surgery, postoperative week 1, and postoperative week 4.
Collected data included demographic characteristics (age, sex, height, weight, medical history), surgical details (type and approach method of surgery, bleeding characteristics, application of Hemogram or Arista™ AH), hemostatic outcomes (hemostatic success rate, time to hemostasis, rebleeding), product usage (amount of hemostatic agent used, transfusion requirement and volume), recovery-related outcomes (ICU and hospital stay), and adverse events (surgical complications, device-related reactions such as allergic responses or infections).
All data were obtained from the electronic medical records (EMR) and clinical report forms (CRF) and were entered either manually or electronically by the investigators. Collected data were anonymized and transferred to a dedicated research database. Data accuracy was ensured through double-checking and independent monitoring, and all adverse or serious adverse events were reported.
Statistical Analysis
Clinical findings of the enrolled patients were compared using appropriate statistical methods. Categorical variables are presented as frequencies and percentages. Chi-square test or Fisher's exact test were conducted for categorical data, while Student's t-tests were used to determine statistical significance for continuous variables. Statistical significance was determined with a threshold of P < .05. Statistical analyses were performed using SPSS version 29.0 for windows ((IBM Corporation, Armonk, NY, USA).
Conflict of Interest Statement
This work was supported by ‘Supporting Project to Clinical evaluation Domestic Medical Devices in Hospitals’ funded by ‘Ministry of Health and Welfare (MOHW, Republic of Korea)’ and ‘Korea Health Industry Development Institute (KHIDI, Republic of Korea)’. The sponsor had no role in the study design, data collection, analysis, or manuscript preparation. The authors declare no other conflicts of interest related to this work.
Results
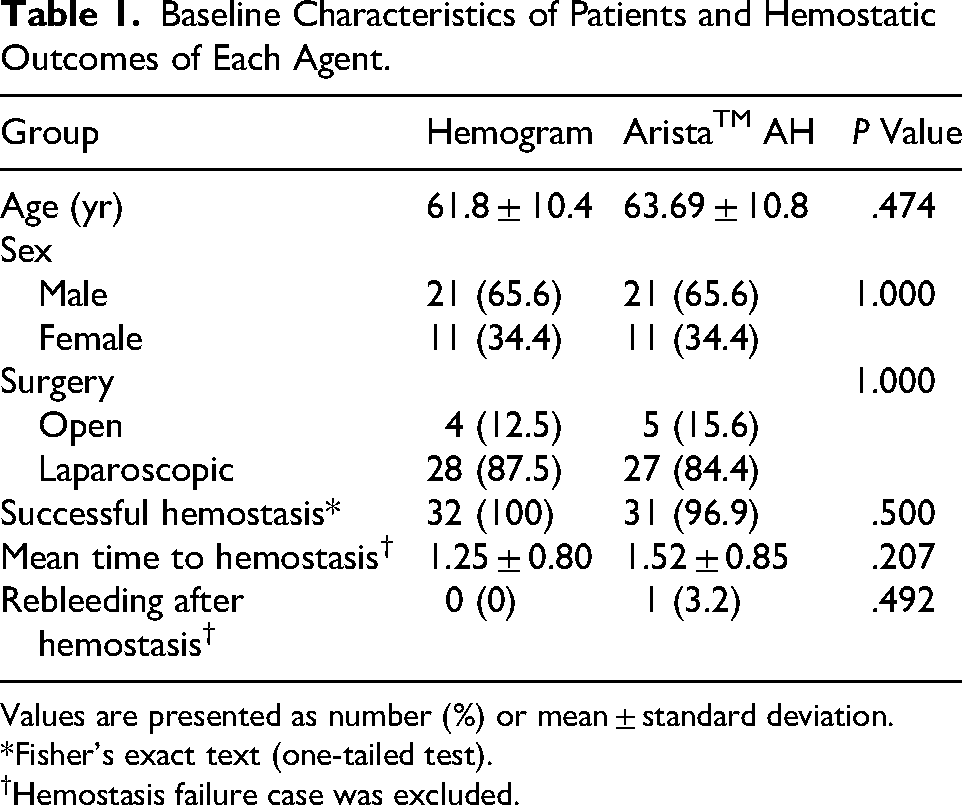
Sixty-seven patients were randomized, and 2 patients withdrew their consent. As a result, 33 patients were allocated into the Hemogram group and 32 into the Arista group. One patient in the Hemogram group was excluded after allocation, leaving 32 patients in the Hemogram group and 32 patients in the Arista group for final analysis (Figure 1). Comparison of demographic features revealed no significant difference in the mean age, gender ratio, surgical approaches between the groups (Table 1).
Baseline Characteristics of Patients and Hemostatic Outcomes of Each Agent.
Values are presented as number (%) or mean ± standard deviation.
*Fisher's exact text (one-tailed test).
Hemostasis failure case was excluded.
In the analysis of hemostatic outcomes, the rate of successful hemostasis was not significantly different between the Hemogram group (100%) and the Arista group (96.9%; Table 1). The mean time to hemostasis were not significantly different between the Hemogram group (1.25 ± 0.80 min) and the Arista group (1.52 ± 0.85 min; Table 1). As for the time to hemostasis, 87.5% of the Hemogram group and 67.7% of the Arista group achieved a complete hemostasis within 1 min (Figure 2). 0ne case of the Arista group developed a re-bleeding after initial assessment of complete hemostasis while the Hemogram group showed no case of re-bleeding.

Distribution of Time to Haemostasis in Each Group.
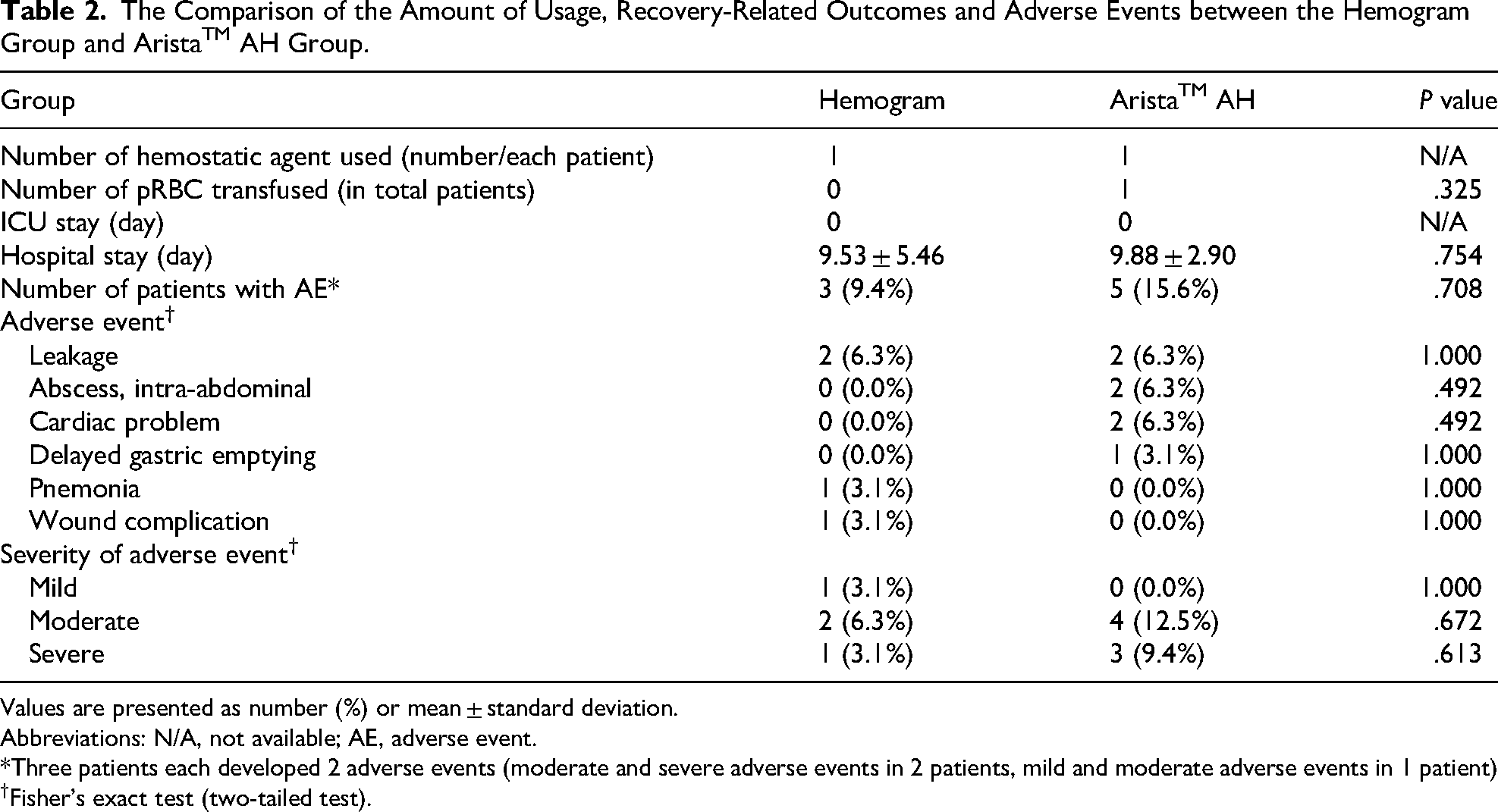
No significant difference was found in the number of hemostatic agents used, number of packed RBC transfused, and the length of hospital or ICU stay between the groups (Table 2). Adverse events were developed in 3 patients (9.4%) of the Hemogram group and 5 patients (15.6%) of the Arista group. Leakages of anastomosis or duodenal stump sites were developed in 2 cases (6.3%) in each group and intra-abdominal abscess was observed in 2 cases (6.3%) of the Arista group. However, we did not find any statistically significant difference in the incidence of each adverse event and the severity of adverse events between the groups (Table 2). All the adverse events were not seem to be caused by or directly related to the usage of both hemostatic agent and we could not find any kind of allergic reaction of infection in this study.
The Comparison of the Amount of Usage, Recovery-Related Outcomes and Adverse Events between the Hemogram Group and AristaTM AH Group.
Values are presented as number (%) or mean ± standard deviation.
Abbreviations: N/A, not available; AE, adverse event.
*Three patients each developed 2 adverse events (moderate and severe adverse events in 2 patients, mild and moderate adverse events in 1 patient)
Fisher's exact test (two-tailed test).
Discussion
Powder-type hemostatic agents can be broadly classified into four categories: cellulose-based, gelatin-based, starch-based and those containing plasma-derived proteins or thrombin.12,13 These agents are characterized by their ease of use, ability to be applied over a wide area, rapid onset of action, and excellent absorbability. In both open and laparoscopic surgeries, cellulose-based and plasma-derived protein or thrombin-combined powder hemostats have recently gained widespread clinical use. While they are not effective in controlling massive or arterial bleeding, their efficacy has been well demonstrated in achieving hemostasis of capillary and small vessel bleeding.
Arista™ AH (BD, USA), a commercially available starch-based hemostatic microparticle, has been widely used in various surgical settings for intraoperative bleeding control. Arista™ AH has been reported to achieve successful hemostasis in nephrectomy and splenic injury, showing significantly superior hemostatic efficacy compared to simple compression.14,15 Additionally, it is characterized by excellent absorbability and minimal residual infection or inflammation at the site of powder application compared to other hemostatic agents. 16 Also, in liver surgery, Arista® AH showed a reduction in estimated blood loss (EBL), a shorter postoperative hospital stay, and earlier removal of the JP drain, suggesting a faster recovery. 17 This is likely a characteristic of powder-type agents, where the fine particles effectively penetrate diffuse bleeding sites within the tissue to promote hemostasis. As a result, estimated blood loss (EBL) is reduced, and because the applied powder is well absorbed without remnants, it minimizes additional tissue inflammation, leading to earlier removal of the intra-abdominal drain. However, some studies have reported that its hemostatic effect may be inferior to that of other agents, such as Surgicel® Powder.18,19 This may be due to certain limitations of Arista™ AH, particularly its relatively slow absorption rate, which can cause the particles to float on the surface of blood and hinder effective clot formation at the bleeding site.
In the present study, we evaluated the efficacy and safety of a novel starch-based hemostatic agent. Hemogram (ATC, Theracion biomedical, Seongnam, Republic of Korea) was developed by combining CM-starch with calcium ions (Ca²+) and further granulating the microparticles using a binder to enhance hemostatic efficacy. Granulation was employed to overcome limitations of existing microparticles, such as poor penetration into the bleeding site and surface clot formation, by increasing particle size and facilitating deeper infiltration into the wound. This approach is expected to improve the effectiveness of topical hemostasis and reduce the risk of re-bleeding.
Although there were no statistically significant differences between the Hemogram and Arista™ AH across the three parameters (successful hemostasis, mean time to hemostatsis, re-bleeding after hemostasis), the Hemogram group demonstrated a higher rate of successful hemostasis, a shorter time to achieve hemostasis, and a lower incidence of re-bleeding. This agent may inherit the advantages of the conventional Arista® AH, such as good absorbability, minimal tissue swelling, reduced inflammation, and decreased serous fluid accumulation at the surgical site, while also demonstrating the potential for improved hemostatic efficacy.
Limitation
A limitation of this study is the relatively small sample size included in the trial. This results in limited statistical power and restricts the generalizability of the findings, indicating that further studies with larger cohorts are necessary to draw more robust conclusions. Additionally, despite randomization, the small number of cases raises the possibility of uneven distribution of certain cases into specific hemostatic agent groups. Furthermore, maintaining consistency in the selection of bleeding sites has inherent difficulties, which constitutes another limitation of this study. Therefore, to definitively establish the non-inferiority or superiority of Hemogram, larger-scale studies with adequate sample sizes are warranted. Nevertheless, this study holds significance by conducting a randomized controlled trial to compare the hemostatic efficacy of a novel powder agent that addresses some limitations of Arista® AH—an agent known for effective control of intraperitoneal micro-bleeding with minimal adverse effects—thereby suggesting the potential for improved powder-type hemostatic materials.
Conclusions
This randomized controlled trial evaluated the clinical efficacy and safety of Hemogram, a newly developed biodegradable hemostatic agent, in comparison with the Arista™ AH. Among patients undergoing gastrectomy with oozing-type intraoperative bleeding, Hemogram demonstrated equivalent performance with no statistically significant differences compared to Arista™ AH. Hemogram achieved a 100% hemostatic success rate within 5 min, with no product-related adverse events reported, supporting both its safety and clinical utility.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Korea Health Industry Development Institute (KHIDI, Republic of Korea), Ministry of Health and Welfare (MOHW, Republic of Korea), (grant number Not applicable).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
