Abstract
Purpose
To systematically evaluate PEA's contribution to lower lobe hemoptysis, characterize associated systemic-to-pulmonary shunts (SPS), and assess the safety/efficacy of polyvinyl alcohol (PVA) particle embolization.
Materials and Methods
This retrospective cohort study analyzed 428 patients with PEA-related hemoptysis (19% of 2296 TAE cases) treated at the First Affiliated Hospital of Guangzhou Medical University (2014-2023). Digital subtraction angiography (DSA) mapped PEA anatomy and SPS, followed by tailored PVA embolization (90-1000 μm). Outcomes included technical success, recurrence rates, and complications. Sex-disaggregated and etiology-disaggregated analyses were performed.
Results
99.1% (424/428) of PEA-associated lesions localized to lower lobes, predominantly left-sided (43.5%). Immediate hemostasis achieved in 95%, 1-month recurrence rate 5.1% (22/428). Multivariate Cox regression identified predictors: massive hemoptysis (HR = 2.03), ectopic bronchial arteries (HR = 1.54), thrombocytosis (>215 × 109/L, HR = 1.27). Sex-disaggregated analysis revealed significant differences in weight (p < .001) and platelet counts (p = .023) between males and females. Etiology-based analysis showed higher recurrence rates in neoplastic (33.3%) and infectious-inflammatory (6.0%) etiologies compared to vascular diseases (0%). No spinal ischemia or bronchoesophageal fistulae.
Conclusion
PEA embolization using tailored PVA particles represents a safe and effective intervention for lower lobe hemoptysis, warranting systematic evaluation in all such cases. Sex-disaggregated analysis is essential for accurate risk stratification. Embolization outcomes are significantly influenced by underlying etiology, with infectious diseases requiring concurrent antimicrobial therapy and neoplastic lesions demanding anti-tumor treatment for durable outcomes.
Keywords
Summary Statement
The pulmonary esophageal artery (PEA) originating from the descending thoracic aorta and approximately 19% (428/2296) can be a source of hemoptysis in the disease involving the left lower lobe, and PEA embolization with polyvinyl alcohol (PVA) particles is safe and effective in treating hemoptysis.
Key Points
In this exploratory analysis, esophageal artery involvement was observed in approximately 19% (428/2296) of the hemoptysis cohort. Within this subgroup, lower lung abnormalities were detected in 99.1% (424/428) of cases, with left lower lobe manifestations being predominant (77%, 325/424).
PEA embolization with PVA particles is safe and effective in treating hemoptysis.
Upon conducting a follow-up with these patients and analyzing the data post-embolization of the proper esophageal artery, we discovered that identified weight, preoperative hemoptysis volume, preoperative duration, preoperative thrombocyte count, and the presence of ectopic bronchial arteries as significant predictors of hemoptysis recurrence.
Introduction
The 2020 Expert Consensus on Diagnosis and Treatment of Hemoptysis by the Hematology Group of the Beijing Medical Association Respiratory Committee categorizes hemoptysis as follows: massive (>500 mL/24 h or >100 mL in a single episode), moderate (100-500 mL), and minimal (<100 mL). Bronchial artery embolization (BAE) is established as the gold-standard minimally invasive intervention for massive or recurrent hemoptysis due to its efficacy in achieving hemostasis. 1 While bronchial arteries (BAs) remain the primary bleeding source, emerging evidence highlights the critical role of non-bronchial systemic arteries (NBSAs) in hemoptysis, particularly in chronic inflammatory or pleural-based pathologies. 2
NBSAs exhibit complex anatomic origins and hemodynamic behaviors that significantly impact both acute outcomes and long-term recurrence risks. 3 Among NBSAs, the pulmonary esophageal artery (PEA) warrants special attention. Although the PEA does not normally contribute to pulmonary perfusion, it undergoes compensatory hypertrophy in chronic pulmonary pathologies (eg, bronchiectasis, lung cancer, or pulmonary artery stenosis) when bronchial arterial supply becomes insufficient. 4 This adaptive angiogenesis establishes the PEA as a dominant contributor to hemorrhage in 19%–32% of lower lobe hemoptysis cases, yet its anatomic complexity and variable origins (eg, descending aorta, left gastric artery) lead to frequent oversight during BAE, 5 thus necessitating its systematic evaluation during embolization planning.
Despite its clinical significance, rigorous studies on PEA embolization remain sparse. This retrospective analysis of our institutional data aims to characterize PEA involvement in lower lobe hemoptysis and systemic-to-pulmonary shunts (SPS) formation and to assess the safety and efficacy of polyvinyl alcohol (PVA) particle embolization for PEA-associated bleeding, with particular attention to sex-specific and etiology-specific factors.
Materials and Methods
This single-center retrospective study received approval from the Institutional Review Board of the First Affiliated Hospital of Guangzhou Medical University. Given the retrospective nature of the study, the requirement for written informed consent from patients was waived.
Patients
Between April 2014 and August 2023, 2296 patients presenting with hemoptysis underwent transcatheter arterial embolization (TAE) to embolize both bronchial or non-bronchial systemic arteries following the failure of hemostatic treatment with medical drugs. Among 2296 patients, 428 (18.6%) demonstrated hypertrophied and tortuous esophageal arteries supplying pulmonary lesions, which promoted SPS formation. This pathologic phenomenon reflects the development of systemic-pulmonary anastomoses, wherein systemic arterial branches (eg, bronchial and esophageal arteries) abnormally connect to pulmonary arterial branches, establishing collateral pathways in chronic pulmonary pathologies. And then, these abnormal arteries were embolized. Patients who exhibited no significant abnormalities in the PEA during the procedure, as well as those whose PEAs were not embolized, were excluded from this study. Patient demographic and clinical data were extracted from the hospital's electronic health records. Imaging and procedural details were retrieved from the Picture Archiving and Communication System (PACS). Prior to the procedure, all patients underwent Computed Tomography Angiography (CTA), complete blood count, biochemical assays, and coagulation function tests. The patient selection flowchart is shown in Figure 1.

Flowchart of Patient Selection. the Flowchart Highlights a Systematic Exclusion Process (Unexplored PEA, Imaging/Technical Limitations, Incomplete Data) and Stratified Screening (Functional Confirmation of PEA), Culminating in 428 Patients Receiving PEA-Targeted Embolization. This Reflects Rigorous Clinical Decision-Making Regarding Indications, Technical Feasibility, and Data Completeness.
TAE Method
All procedures were performed by three interventional radiologists with over 10 years of experience, using a Siemens Artis Zee Floor/Artis Zee III digital subtraction angiography (DSA) system. Vascular access was achieved via the right femoral artery using the Seldinger technique with a 5F arterial sheath (Terumo Corporation, Tokyo, Japan). A 4F Cobra C2 catheter (Cordis Corporation, USA, assembled in Mexico) or 4F Yashiro catheter (Terumo) was advanced under fluoroscopic guidance to interrogate abnormal BAs and NBSAs, including the aortic arch, thoracic aorta, bilateral subclavian arteries (internal thoracic, thyrocervical, costocervical, and lateral thoracic branches), posterior intercostal arteries, inferior phrenic arteries, and PEAs (Figure 2). Ascending aortography with a 5F pigtail catheter (Cordis) was performed when necessary to identify occult pathologic vessels. Iopamidol (320 mg I/mL) served as the contrast agent.

A 32-Year-old Male Patient Presented with Intermittent Hemoptysis for Over Three Weeks. CT Imaging Revealed a Mass-Like Opacity in the Left Lower Lung, Suggestive of a Left Lower Lung Abscess. A (Esophageal Proper Artery) and B (Left Bronchial Artery): Angiography Showed no Apparent Abnormalities (Indicated by White Arrows). C (4F Cobra Catheter Angiography): the Left inferior Phrenic Artery (Black Arrow) Appears Dilated and Tortuous, with a Systemic-Pulmonary Shunt Observed at its Distal end (Black Arrowhead). D (Axial CT) and E (Coronal CT): Chest CT Demonstrates an Infectious Lesion in the Left Lower Lung with Incomplete Aeration (White Arrowheads), Consistent with Consolidative Changes and air-Fluid Levels.
Preoperative CTA was routinely conducted to localize bleeding sources, assess hemoptysis risk (eg, cavitary lesions indicating high risk), and map vascular anatomy, with particular attention to the diameter and course of the PEA in lower lobe pathologies. Intraprocedural DSA revealed hypertrophied and tortuous arteries supplying pathologic lung tissue, often forming SPS (Figures 3–5). SPS was classified as rapid (contrast reaching the pulmonary trunk within ≤4 cardiac cycles) or slow (>4 cycles), with rapid shunts prioritized for adjunctive coil embolization.
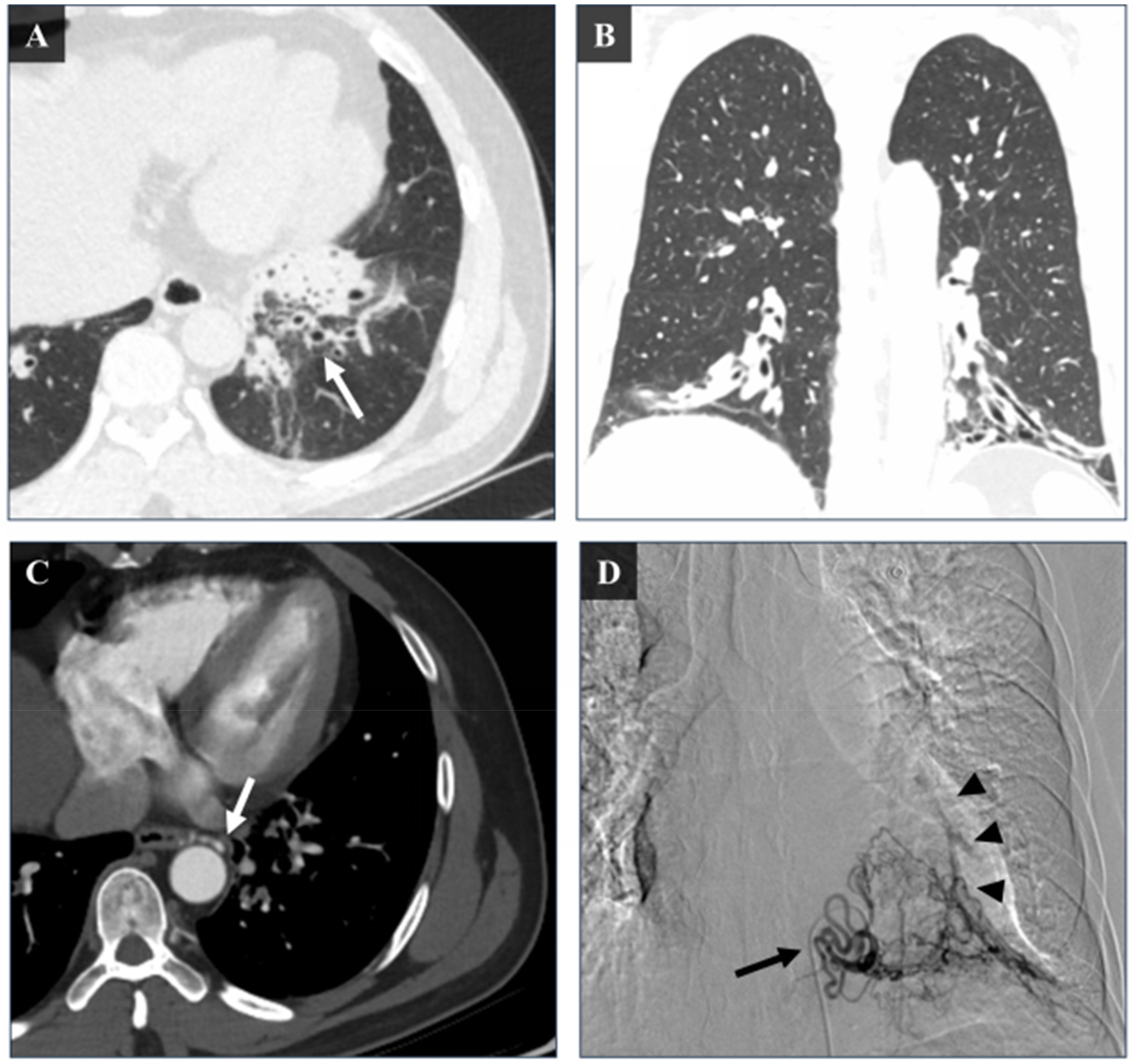
A -B: Multiple Bronchiectasis with Infection in the Basal Segments of the Left Lower Lobe. C: Enlargement of the PEA (White Arrow). D: Angiography of the PEA via a 4F Yashiro Catheter Demonstrates Hypertrophied, Tortuous, and Disorganized Branches (Black Arrow) Supplying the Left Lower Lobe Lesion. BPS is Observed, with Opacification of the Pulmonary Arterial Branches in the Basal Segments (Black Arrowhead).

A 48-Year-old Woman with a 40-Year History of Recurrent Cough and Sputum, and Over a Decade of Hemoptysis (up to 50 ml/day), has Been Diagnosed with Bronchiectasis and Infection. Figure A-B Illustrates the Esophageal Intrinsic Artery Emerging from the Left Anterior Wall of the Thoracic Aorta (White Arrow). Figure C Shows the Esophageal Intrinsic Artery Originating Near the Lower Edge of the Sixth Thoracic Vertebra (Yellow Line and White Arrowhead). Figure D, on March 27, 2020, Selective Angiography of the PEA was Performed During Transarterial Embolization (TAE). Imaging Showed an Enlarged Main Artery (Black Thin Arrow) Running Inward and Downward, with Increased and Disorganized Distal Branches Supplying Blood to a Lesion in the Left Lower Lung. This Vascular Setup Creates a SPS with a Branch of the Left Lower Pulmonary Artery (Black Arrowhead). Figure E Shows Peripheral Embolization with 300μm PVA Particles, Resulting in Angiographic Images That Confirm no SPS and a Significant Decrease in Primary Blood Flow. Dilated and Tortuous Left Bronchial Artery (F) and Left inferior Phrenic Artery (G), Accompanied by Localized SPS. Figure H Details the Patient's Chronic Gastritis History; a Follow-up Gastroscopy on April 1, 2023, Five Days After TAE, Confirmed Chronic non-Atrophic Gastritis, with Smooth Esophageal mucosa and Good Motility.

A 71-Year-old man with a Three-Year History of Left Lower Lobe Small Cell Lung Cancer, Treated with Radiotherapy and Chemotherapy, Presented with two Days of Hemoptysis, Producing up to 10 mL of Blood in 24 h. Chronic Inflammation was Also Present in Both Lungs. Figure A Shows Selective PEA Angiography from November 27, 2020. Imaging Shows an Enlarged Main Trunk of the Esophageal Artery (Thin Black Arrow) Moving Inward and Downward, and Increased, Disorganized Distal Branches Supplying the Left Lower Lung. This Vascular Anomaly Forms a SPS with a Branch of the Left Lower Pulmonary Artery (Black Arrowhead). Figure B Shows the Occlusion of Distal Branches with 200 μm and 300 μm PVA Particles, Causing Main Trunk Blood Flow Stagnation Under Fluoroscopy. Angiography Later Confirms no Visible SPS and Significantly Reduced Main Trunk Blood Flow. Figure C Highlights the Esophageal Artery Originating from the Left Anterior Wall of the Thoracic Aorta (White Arrow). Figure D Shows the PEA Branching Point Near the Middle of the T6 Vertebra (Yellow Line and White Arrowhead). in Figure E, Dated November 19, 2021, one Year After TAE, the Patient Reported Left Upper Abdominal Discomfort. A Gastroscopy Revealed Chronic non-Atrophic Gastritis, with the Esophageal mucosa Appearing Normal.
Superselective catheterization of target vessels was achieved using microcatheters (1.9 Fr FineDrive [PIOLAX], 2.5 Fr Cantata [COOK], 2.7 Fr Progreat [Terumo], or 2.8 Fr Maestro [Merit]), followed by embolization with polyvinyl alcohol (PVA) particles (90-1000 μm) tailored to lesion characteristics: smaller particles (90-500 μm) occluded distal vessels while preserving proximal trunks, whereas larger particles (500-1000 μm), coils, or gelatin sponge addressed aneurysms, high-flow SPS, or anatomic constraints (eg, intercostobronchial trunks). PEA supply patterns were categorized as left lower lobe, right lower lobe, bilateral lower lobes, or non-lower lobe contributions. Arteries supplying the spinal cord (eg, Adamkiewicz artery) were strictly avoided.
All protocols prioritized real-time DSA-guided superselective catheterization to minimize non-target embolization, with technical refinements progressively optimized through 428 consecutive procedures to maximize safety and clinical efficacy.
Definition of Research Endpoints
Clinical outcomes including clinical success (defined as complete hemoptysis cessation for ≥1 month post-embolization), 6 recurrence rates (hemoptysis requiring urgent reintervention such as repeat TAE), need for additional therapies, and major complications were evaluated. Preoperative time was defined as the interval between hospital admission and intervention, while surgical delay was classified as >24 h from admission to procedure. Electronic medical records were systematically reviewed to collect demographics, procedural details (angiographic findings, embolized arteries, embolic agents), time metrics (preoperative time, surgical delay), prognostic factors (bleeding etiology, embolization completeness), and complications (eg, fever, stroke, systemic embolization). Angiographic success required superselective catheterization with post-embolization contrast extravasation absence.
Statistical Methods
Data analysis was conducted using R (v4.4.1; www.R-project.org) and SPSS 25.0. Normality of continuous variables was assessed via the Shapiro-Wilk test, with normally distributed data reported as mean ± standard deviation (SD). Cox proportional hazards regression identified predictors of post-embolization hemoptysis recurrence, with significance thresholds set at p < .05. Optimal cutoffs for continuous variables (eg, weight, BMI) were determined using X-tile software (v3.6.1; Yale University), 7 stratifying patients into low- and high-risk groups. Recurrence-free survival was estimated via Kaplan-Meier curves and compared using the log-rank test (G-rho rank test, ρ = 1).
To address multicollinearity, variance inflation factors (VIF) 8 were calculated for all variables after univariate Cox regression. Variables with VIF >10 were excluded to eliminate redundant predictors. Remaining non-collinear variables were incorporated into multivariate Cox regression to derive adjusted hazard ratios (HRs).
Sex-disaggregated analysis was performed for all continuous variables using Student's t-test or Mann-Whitney U test as appropriate. Etiology-based subgroup analysis was conducted to evaluate recurrence rates and long-term outcomes across different disease categories.
Results
Patient Characteristics
Among 2296 patients with hemoptysis, 19% (428/2296) exhibited PEA involvement. The cohort undergoing PEA embolization comprised 242 males and 186 females (mean age: 54.3 years; mean BMI: 23.5 kg/m2). Diagnoses were categorized as follows: non-neoplastic infectious/inflammatory disease (74.1%, 317/428), primary/metastatic tumors (2.1%, 9/428), non-malignant vascular disease (4.2%, 18/428), tumor with infection (4.2%, 18/428), infection with vascular involvement (14.3%, 61/428), tumor with vascular invasion (0.2%, 1/428), concurrent tumor-infection-vascular disease (0.2%, 1/428), and occult hemoptysis (0.7%, 3/428). Comorbidities included hypertension (11.7%, 50/428) and diabetes (6.3%, 27/428). Preoperatively, 28.0% (120/428) presented with fever, and hemoptysis severity was classified as massive (12.4%, 53/428), moderate (39.3%, 168/428), or mild (48.4%, 207/428). Interventional therapy was performed urgently (<24 h) in 18.0% (77/428) and electively (>24 h) in 82.0% (351/428) following medical stabilization (Table 1).
Results of Selective PEA Exploration
A total of 428 abnormal PEAs were identified, with 50 (11.7%) demonstrating collateral connections to bronchial artery branches (Figure 6), 7 (1.6%) to left gastric artery branches, 10 (2.3%) to subdiaphragmatic artery branches, and 3 (0.7%) exhibiting tripartite anastomoses among the PEA, bronchial arteries, and subdiaphragmatic arteries. Contrast injection into the PEA revealed tortuous vessels originating from the descending thoracic aorta, coursing along the aortic anterior wall and penetrating thickened pleura to supply lower lobe lesions. SPS were observed in 84.8% (363/428) of patients at distal PEA branches, with pseudoaneurysms in 12 cases (2.8%) manifesting contrast extravasation and associated paroxysmal cough during angiography.
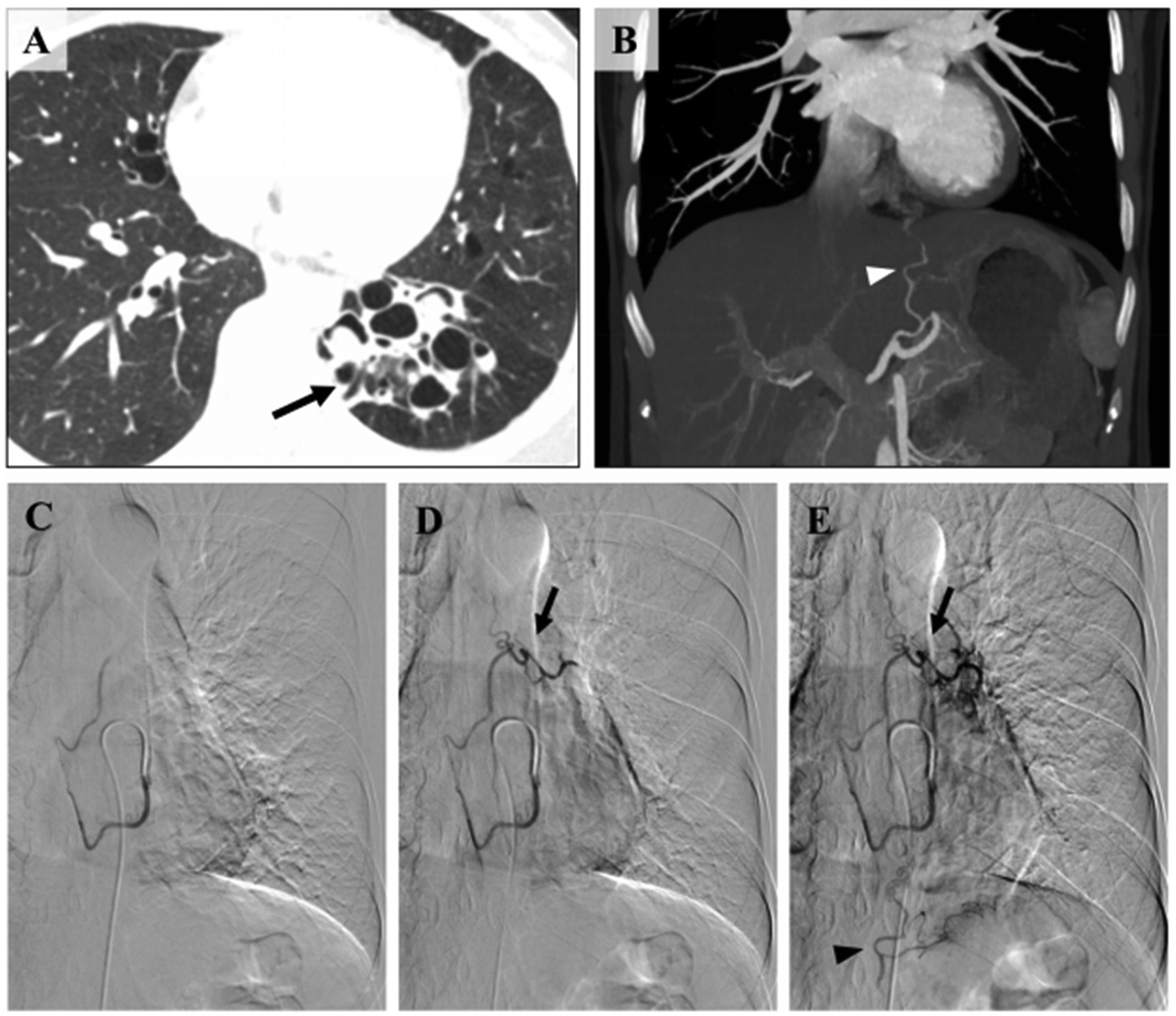
A: Chest CT Demonstrates Bronchiectasis with Infection in the Left Lower Lobe (Black Arrow). B: CT Angiography Reconstruction Reveals Hypertrophy of the Esophageal Branch of the Left Gastric Artery (White Arrowhead). C-E: Three Sequential Phases of the Same Angiographic Procedure Show Anastomoses Between the PEA, Left Bronchial Artery (Black Arrow), and Esophageal Branch of the Left Gastric Artery (Black Arrowhead).
PEA supply distribution was categorized as follows: left lower lobe (186/428, 43.5%), right lower lobe (99/428, 23.1%), bilateral lower lobes (139/428, 32.5%), and non-lower lobe regions (4/428, 0.9%) including left upper lobe (1), right upper lobe (1), and right middle lobe (2). All PEAs contributed to multi-arterial perfusion of pulmonary lesions.
Embolization was performed on all culprit vessels—bronchial, intercostal, subdiaphragmatic, intrathoracic, and esophageal arteries—using PVA particles. Particle size selection was tailored to lesion characteristics, with smaller particles (90-300 μm) used for distal occlusion and larger particles (300-1000 μm) or hybrid approaches for high-flow targets (Table 2). Persistent SPS after small-particle embolization necessitated adjunctive large-particle administration until complete flow stasis was achieved.
Baseline Characteristics of Patients.

Includes bronchiectasis, tuberculous pneumonia, and other infections confirmed by microbiologic/histologic criteria.
Histologically confirmed squamous cell carcinoma, adenocarcinoma, small cell carcinoma, or metastases.
Encompasses pulmonary artery embolism, stenosis, and vascular malformations diagnosed by CTA/DSA, which can lead to hemoptysis, and this category of disease is characterized by the absence of overt inflammatory processes or neoplastic formations within the pulmonary system.
Lung neoplasms (as above) with superimposed pneumonia confirmed by BAL culture/biopsy.
Non-neoplastic infections (eg, tuberculosis) complicated by vascular erosion or thrombosis.
Tumors invading or compressing pulmonary vessels (eg, lung adenocarcinoma with pulmonary vein invasion).
Cases involving concurrent tumor, infection, and vascular disease were confirmed through multidisciplinary review by a team of interventional radiologists, pathologists, and infectious disease specialists, integrating histologic, imaging, and microbiologic evidence.
Hemoptysis without structural or microbiologic etiology after exhaustive workup.
Intraoperative Information for TAE.

This comprehensive approach ensured complete angiographic occlusion in 92.5% (396/428) of cases, with no instances of spinal artery embolization due to strict avoidance of vessels supplying the Adamkiewicz artery.
Embolization Treatment of PEA and Complications
Embolization was performed using PVA particles (90-300 μm or 300-1000 μm) based on distal PEA vessel diameter to achieve occlusion and eliminate SPS, preserving proximal trunks. Postprocedural complications included transient chest pain (24/428, 5.6%) managed with NSAIDs, fever (137/428, 32.0%) treated with supportive care, and contrast-related urticaria (25/428, 5.8%). Three patients (0.7%) developed pulmonary thromboembolism that was treated successfully with low-molecular-weight heparin. No severe complications (contrast nephropathy, gastrointestinal bleeding, bronchial-esophageal fistula, stroke, or paraplegia) occurred (Table 3).
Postoperative Complications.

Attributable to post-embolization syndrome (inflammatory response to ischemic tissue) or concurrent pulmonary infection.
Caused by vascular ischemia or diaphragmatic irritation from adjacent embolized vessels.
Occurred post-compression bandage removal, likely due to dislodged lower-limb deep vein thrombi (DVT) from prolonged immobilization.
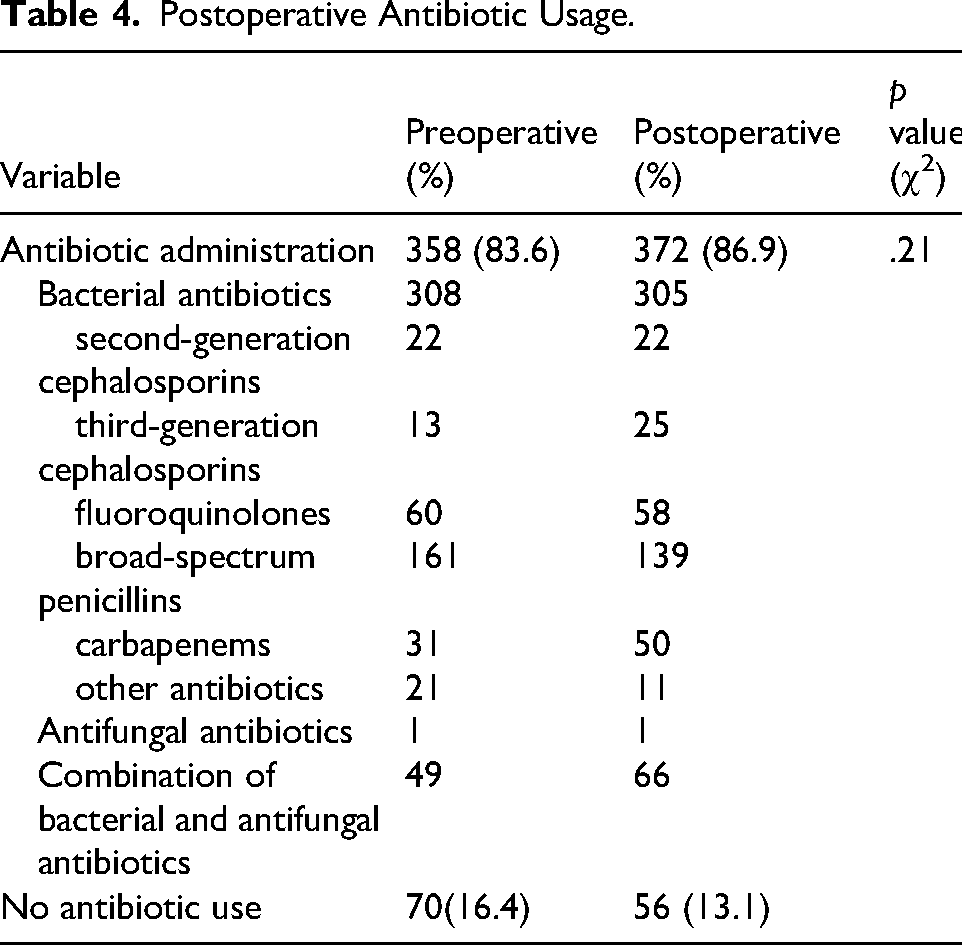
Antibiotics were administered to 83.6% (358/428) preoperatively and 86.9% (372/428) postoperatively (p = .21), with 56 patients (13.1%) receiving no postoperative antibiotics (Table 4).
Postoperative Antibiotic Usage.
Results of Arterial Embolization Treatment and Follow-up Outcomes
No patients experienced significant hemoptysis post-embolization, including those with active bleeding at intervention. During a median follow-up of 34 months (range: 5-95), 22 patients (5.1%) exhibited recurrence within 1 month: 9 (2.1%) required repeat embolization due to technical failure (persistent bleeding), while 13 (3.0%) had self-limited minor hemoptysis managed conservatively.
Univariate Cox regression identified continuous predictors of recurrence, with X-tile-derived optimal cutoffs: weight ≤54.6 kg, BMI ≤18.07 kg/m2, hospitalization >15 days, preoperative delay >5 days, and platelet count >215 × 109/L. Based on these cutoff points, patients could be classified into low-risk and high-risk groups for recurrent hemoptysis, as shown in Figure 7. After excluding collinear variables (VIF >10, Table 5), multivariate analysis retained five predictors: low weight (HR = 0.61, 95%CI:0.46-0.81), massive preoperative hemoptysis (HR = 2.03, 1.27-3.23), preoperative delay (HR = 0.71, 0.51-1.00), elevated platelets (HR = 1.27, 0.93-1.74), and ectopic bronchial arteries (HR = 1.54, 1.13-2.11) (Figure 8).
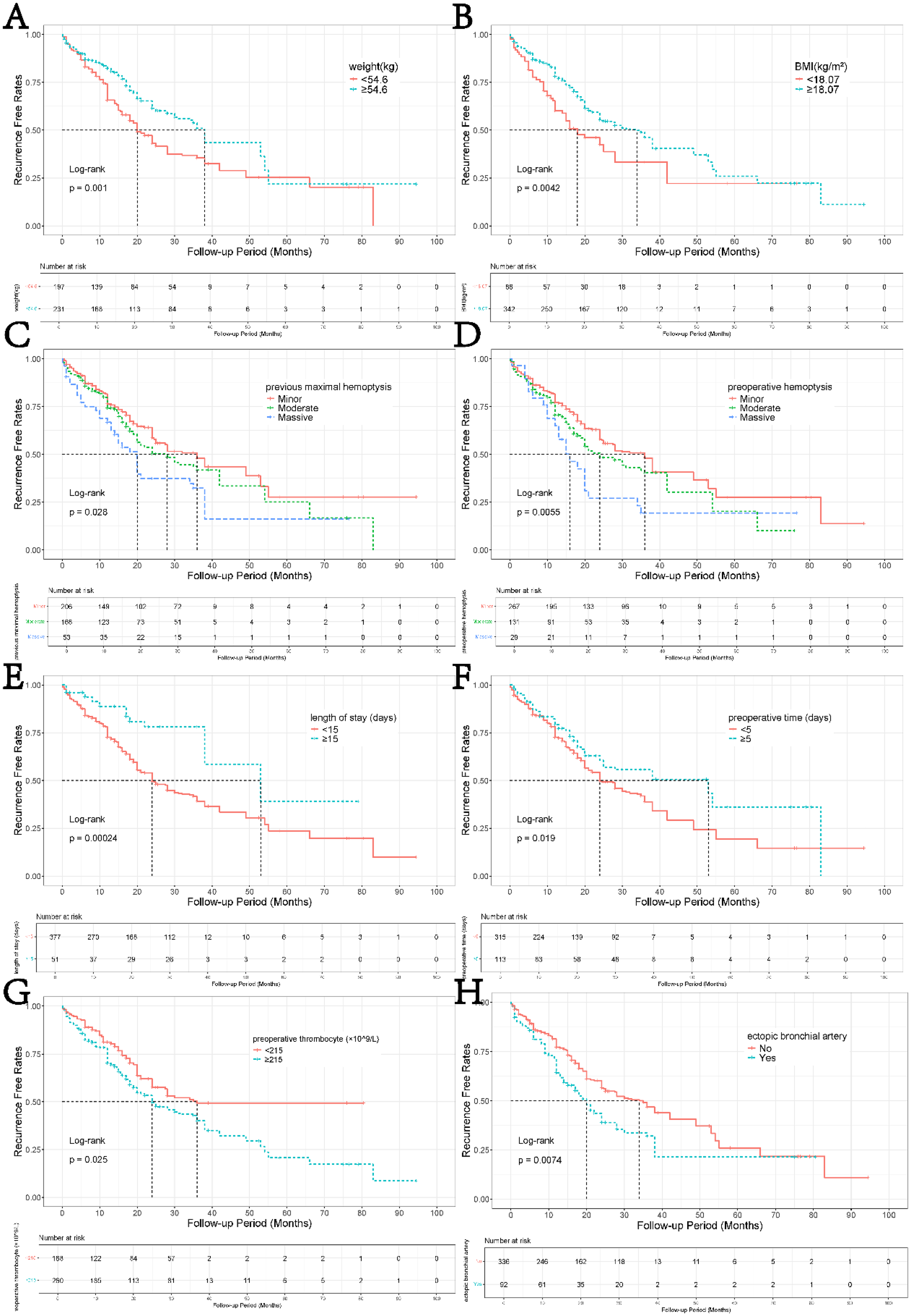
Survival Curves Evaluating Predictors of Hemoptysis Recurrence-Free Survival. Key Findings Include: Lower Recurrence-Free Survival was Associated with Body Weight >54.6 kg (p = .001), BMI >18.07 (p = .004), Hospital Stay >15 Days (p < .001), Delay >5 Days to Embolization (p = .02), and Preoperative Platelet Count <215 × 109/L (p = .03). Higher Recurrence-Free Survival Correlated with History of Significant Hemoptysis (p = .028), Preoperative Significant Hemoptysis (p = .006), and Intraoperative Detection of Ectopic Bronchial Arteries (p = .007). the Results Suggest That Clinical Factors (eg, Prolonged Hospitalization, Delayed Intervention) and Hematologic Markers (eg, low Platelets) may Reflect Disease Severity or Delayed Treatment, While Prior Hemoptysis Episodes and Ectopic Vessel Identification Could Indicate Targeted Therapeutic Efficacy. Statistical Significance (p < .05) Underscores the Clinical Relevance of These Variables in Predicting Recurrence Risk.

Forest Plot of Risk Factors Associated with Hemoptysis Recurrence, with the Following Key Interpretations: 1. Patients with ≥54.6 kg Exhibited a Significantly Lower Recurrence Risk (HR = 0.61, 95% CI: 0.46-0.81; p < .001), Suggesting Higher Body Weight is a Protective Factor. 2. Platelet Levels ≥215 × 109/L Showed no Significant Association with Recurrence (HR = 1.27, 95% CI: 0.94-1.71), as the Confidence Interval overlaps 1. 3. A delay of ≥5 Days Trended Toward Reduced Recurrence Risk (HR = 0.71, 95% CI: 0.51-1.00; p≈.05), Though Statistical Significance was Marginal. 4. Massive Hemoptysis was Strongly Associated with Higher Recurrence Risk (HR = 2.03, 95% CI: 1.27-3.23; p < .001), while Moderate Hemoptysis Showed No Significant Effect (HR = 1.27, 95% CI: 0.93-1.74). 5. Presence of Ectopic Arteries Significantly Increased Recurrence Risk (HR = 1.54, 95% CI: 1.13-2.11; p = .007), Highlighting its Role as a Prognostic Marker.
Univariate Cox Regression Analysis and Calculation of VIF.
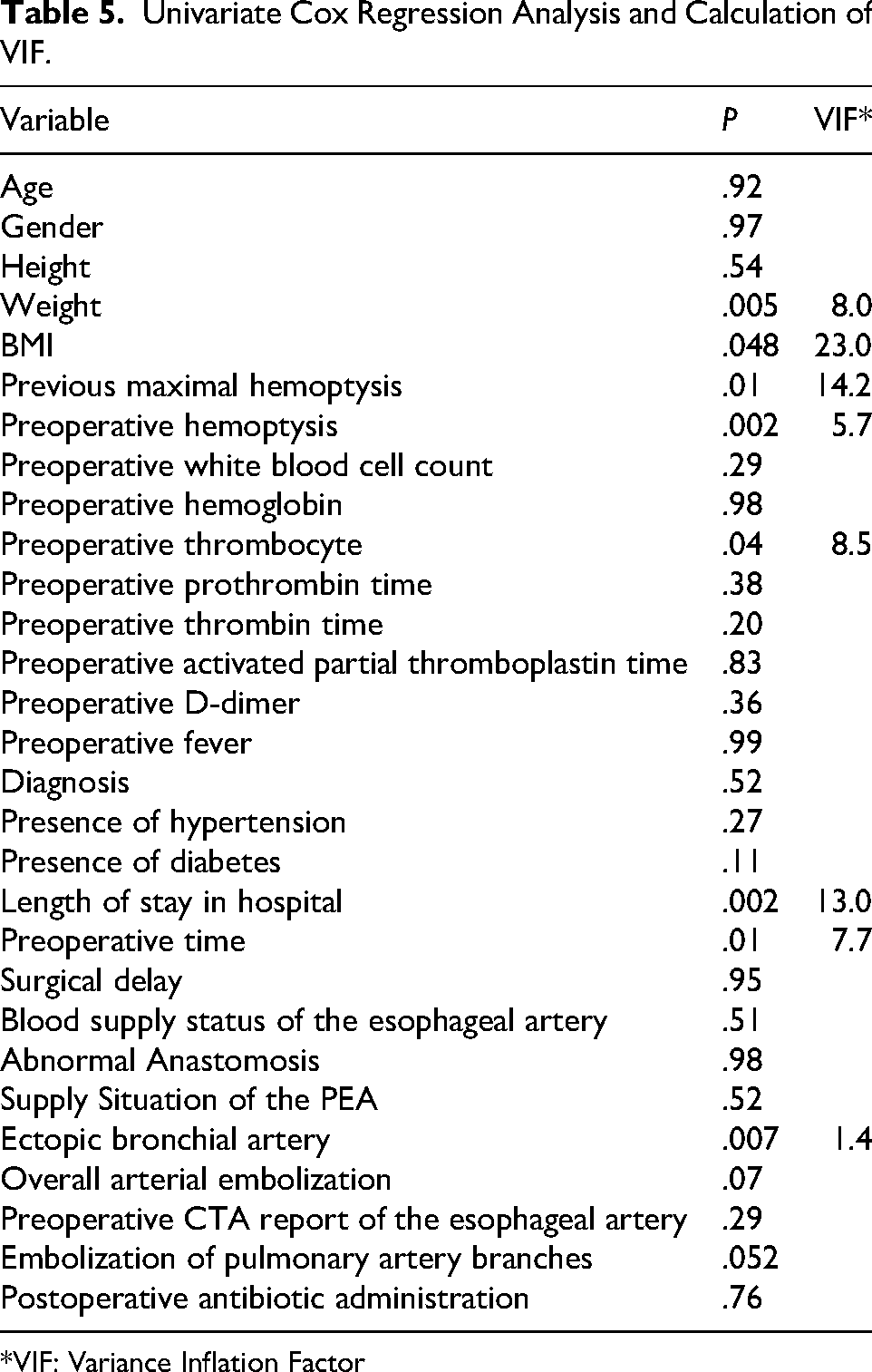
*VIF: Variance Inflation Factor
Sex-Disaggregated Analysis
Significant differences were observed between male and female patients in weight (males: 67.2 ± 10.1 kg vs females: 54.8 ± 8.3 kg, p < .001) and platelet counts (males: 258.3 ± 112.4 × 109/L vs females: 281.6 ± 125.7 × 109/L, p = .023). When stratified by sex, the previously identified weight cutoff of 54.6 kg was found to be more applicable to female patients, while male patients showed different weight distribution patterns related to recurrence risk.
Etiology-Based Analysis
Recurrence rates varied significantly by underlying etiology. Neoplastic lesions showed the highest recurrence rate (33.3%, 3/9), followed by infectious-inflammatory diseases (6.0%, 19/317), while vascular diseases had no recurrences (0%, 0/18). Patients with infectious etiologies who received appropriate antimicrobial therapy had significantly lower recurrence rates compared to those without targeted treatment (4.2% vs 12.8%, p = .037).
Postoperative upper endoscopy in 20 patients (4.7% of cohort) revealed: 30% had upper abdominal pain, 50% had reflux symptoms, and 20% underwent routine surveillance; normal mucosa (17/20, 85%), Barrett's esophagus (1/20, 5%), reflux esophagitis (2/20, 10%) (Table 6). No procedure-related esophageal fistulae or ischemic complications were observed.
Examination Results of Gastrointestinal Endoscopy in 20 Postoperative Patients.

Discussion
Exploration of the Significance of PEA
PEA embolization demonstrates clinically significant improvements over conventional BAE, achieving 95% immediate hemostasis (vs 90% benchmark 9 ) with substantially lower recurrence (5.1% vs 10%-58%3,5), highlighting its specific utility in lower lobe hemoptysis characterized by dominant non-bronchial collaterals. This improvement likely reflects refined procedural expertise, multidisciplinary collaboration (eg, rapid anesthesia support for unstable patients), and systematic evaluation of both BAs and NBSAs, including the PEA.10,11 The left lower lobe predominance (43.5%) in our cohort aligns with known anatomic vulnerabilities. The dependent positioning of the left lower pulmonary lobe predisposes it to chronic mucus stasis and recurrent infections, driving compensatory angiogenesis through non-bronchial pathways.
A critical limitation of isolated BA embolization lies in the interconnected terminal vasculature between BAs and NBSAs (eg, PEA, intercostal arteries). While BA embolization may achieve immediate hemostasis, recurrence rates escalate due to compensatory hypertrophy of NBSAs, which rapidly reconstitute blood supply to pathologic lung tissue. In our cohort, 29% of patients required hybrid embolization (small + large particles) to address residual SPS arising from NBSA collateralization. Proactive embolization of all culprit arteries—guided by real-time DSA—reduces recurrence risks by eliminating “escape routes” for collateral recruitment.
Sex-specific considerations emerged as a key finding, with significant differences in weight (p < .001) and platelet counts (p = .023) between males and females. These findings underscore the importance of sex-stratified risk assessment in hemoptysis management. 12
Etiology-driven outcomes revealed distinct recurrence patterns: neoplastic lesions showed the highest recurrence (33.3%), followed by infectious-inflammatory diseases (6.0%), while vascular diseases had no recurrences. This emphasizes that embolization technique alone is insufficient—concurrent management of underlying etiologies is essential for durable outcomes.
The pathophysiological rationale for PEA embolization extends beyond immediate hemostasis. By disrupting the angiogenesis-hemorrhage cycle perpetuated by chronic inflammation, embolization provides a therapeutic window for definitive management of underlying pathologies.
Our study addresses a significant gap in interventional radiology by establishing the clinical importance of routine PEA assessment—an aspect frequently overlooked in conventional embolization protocols. The systematic approach described herein represents a paradigm shift in managing complex lower lobe hemoptysis.
PEA Anatomy and Pathophysiologic Role in Hemoptysis
The PEA, anatomically divided into cervical, thoracic (upper/lower), and abdominal segments, 13 primarily supplies the lower esophagus but assumes a compensatory role in chronic pulmonary pathologies. Originating from the T6-T9 thoracic aorta, the PEA descends along the esophageal wall, forming anastomoses with bronchial, left gastric, and diaphragmatic arteries. In our cohort, 60 PEA branches demonstrated direct connections to bronchial arteries, while 5 linked to phrenic arteries and 1 to the left gastric artery, corroborating its integration into systemic-pulmonary collateral networks. These anastomoses, though critical for ischemic compensation, complicate embolization due to overlapping vascular territories and DSA imaging challenges in delineating fine collaterals.
Abnormal PEA Radiographic Manifestations
Typically, the esophageal artery doesn't supply blood to the lungs. But in cases of chronic inflammation in the lower lobe or lingula involving the visceral pleura, the bronchial arteries may fall short. This can cause the esophageal artery to step in and supply blood to the affected lung tissue via the pulmonary ligament or thickened mediastinal pleura. 14 The PEA can contribute to massive hemoptysis by supplying blood to the bronchial arteries via its branches in the esophagus. During embolization, if a lesion in the left lower lobe or lingula is near the mediastinal pleura and the affected pulmonary segment has poor blood supply, the PEA's involvement should be considered, requiring selective angiography. The esophageal artery typically curves rightward and runs down the esophagus. If angiography shows the artery is enlarged, twisted, and branching to supply blood to pulmonary lesions through thickened pleura, or if there's a shunt to the pulmonary artery, embolization is needed. Due to connections between esophageal and bronchial arteries or the left gastric artery, it is crucial to control low-pressure injections during embolization to prevent embolic particles from refluxing into the aorta and causing ectopic embolization.
Selection of Embolization Materials
The selection of embolic agents in TAE requires careful consideration of anatomic complexity and long-term outcomes. While PVA particles remain the cornerstone for durable occlusion, prior studies have highlighted risks associated with non-selective embolization. Munk et al 15 reported bronchoesophageal fistulas following simultaneous embolization of bronchial and upper esophageal arteries using PVA, attributed to ischemic necrosis of adjacent tissues. Similarly, Hsu and Su 16 documented bilateral fistulas after gelatin sponge embolization, underscoring the need for meticulous vessel selection. In our cohort, graded PVA particles (90-1000 μm) were tailored to vessel diameter and flow dynamics: smaller particles (90-300 μm) targeted distal shunts to prevent collateral recruitment, while larger particles (300-1000 μm) or coils addressed high-flow segments.
Coils, though permanent, induce transient thrombosis and may recanalize over weeks due to thrombus resolution. 17 Thus, they are reserved for proximal protective embolization in shared trunks (eg, intercostobronchial arteries) but avoided in bleeding vessels requiring durable occlusion. Instead, gelatin sponge or coil—absorbable yet effective for temporary flow diversion—was deployed to occlude distal intercostal segments when superselective bronchial artery catheterization was unfeasible.
Complications and Management After Embolization
PEA embolization using PVA particles demonstrated a favorable safety profile in this cohort. Post-procedural complications included transient chest pain (24/428, 5.6%), self-limiting fever (137/428, 32.0%), and contrast-related urticaria (25/428, 5.8%), all managed conservatively. Three patients (0.7%) developed pulmonary thromboembolism, successfully treated with low-molecular-weight heparin. Upper endoscopy in 20 patients revealed preserved esophageal mucosa in 85% (17/20), Barrett's esophagus in 5% (1/20), and mild reflux esophagitis in 10% (2/20). Notably, no cases of gastrointestinal bleeding, perforation, or bronchoesophageal fistula—historically reported complications of esophageal artery embolization—were observed, likely attributable to our protocol's emphasis on superselective microcatheterization and small-particle embolics (90-500 μm PVA), which minimize transmural ischemia. This contrasts with studies reporting dysphagia (0.7-18.2%) 4 or fistulae 18 when using liquid embolics (eg, ethanol) or gelatin sponge, which risk simultaneous occlusion of bronchial and esophageal microvasculature. 19
The absence of reperfusion or delayed complications underscores the efficacy of distal PVA embolization in achieving durable occlusion while preserving proximal arterial integrity. Our findings align with emerging consensus that particulate agents, particularly PVA, balance efficacy and mucosal preservation in mediastinal-pleural pathologies.
In patients with hemoptysis involving the pulmonary esophageal artery (PEA), recurrence correlates strongly with identifiable risk factors, including ectopic bronchial arteries, massive hemoptysis (>200 mL/24 h), elevated platelet counts (>215 × 109/L), and abbreviated hospitalization (<15 days). These factors collectively reflect the interplay of chronic inflammation, hypoxia-driven angiogenesis, and incomplete embolization. Notably, bronchial arteries account for 90% of massive hemoptysis cases, 19 yet 58% of patients experience recurrence within 30 days post-TAE due to reperfusion, residual collaterals, or neoangiogenesis. 21
The presence of ectopic bronchial arteries, as observed by Kenkichi et al, 22 amplifies vascular complexity in chronic inflammatory diseases (eg, bronchiectasis, tuberculosis), where hypoxia-inducible factor-1α (HIF-1α) and vascular endothelial growth factor (VEGF) drive compensatory arterial proliferation. This pathologic angiogenesis establishes redundant collateral networks, predisposing to early recurrence if not comprehensively embolized. In our cohort, patients with ectopic arteries exhibited 1.54-fold higher recurrence risk (p = .02), underscoring the need for pan-arterial embolization protocols.
Reactive thrombocytosis, prevalent in 32% of our patients, aligns with infection-induced megakaryocyte stimulation23,24 and tumor-related hypoxia.25,26 The time frame of thrombocytosis in our cohort (typically within 48-72 h of hemoptysis) suggests a combination of post-hemorrhagic re-equilibration and true reactive thrombocytosis. Chronic bronchial infections in bronchiectasis likely perpetuate platelet overproduction, exacerbating microthrombosis and endothelial injury—a cycle further aggravated by immobilization and malnutrition in advanced disease. Weight loss, observed in 28% of patients, stems from the catabolic effects of chronic inflammation, including anorexia, metabolic dysregulation, and muscle wasting.27,28
Limitations
This study has several limitations. First, selection bias may arise from categorizing PEA embolization as a supplemental procedure, potentially underrepresenting cases where PEA was the dominant bleeding source. Second, as a single-center retrospective analysis, external validity is constrained by heterogeneous pre- and postoperative management protocols across departments (eg, variable antibiotic regimens, inconsistent follow-up schedules), which may confound long-term efficacy assessments. Third, while CTA is invaluable for initial vascular mapping, its limited spatial resolution and dynamic contrast timing can obscure small or tortuous NBSAs, particularly PEAs with atypical origins or collateral networks. Future multicenter prospective studies employing standardized protocols and advanced imaging modalities (eg, dual-energy CTA, 4D flow MRI 29 ) are warranted to validate these findings.
Conclusion
PEA embolization using tailored PVA particles represents a safe and effective intervention for lower lobe hemoptysis, warranting systematic evaluation in all such cases. Our data demonstrate that tailored PVA particle embolization achieves durable hemostasis (95% immediate success rate) with minimal complications, reinforcing its role as a first-line intervention. Crucially, long-term outcomes depend on concurrent management of underlying etiologies: infectious diseases require targeted antimicrobial therapy, while neoplastic lesions demand anti-tumor treatment. Sex-disaggregated analysis is essential for accurate risk prediction, as male and female patients demonstrate different patterns of weight distribution and platelet counts related to recurrence risk.
Footnotes
Abbreviations
Ethics considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
