Abstract
The activated partial thromboplastin time (APTT) assay is essential for evaluating coagulation. However, prolonged APTT may result from pathological conditions or contamination with unfractionated heparin (UFH), which can occur due to improper phlebotomy or use of heparinized lines—even in patients not on UFH therapy. Distinguishing between true pathology and heparin contamination is critical for patient care and appropriate laboratory reporting. This study assessed the effectiveness of Antihepca HRRS (AHCa, a Heparin Resistant Recalcifying Solution (HRRS)), in neutralizing heparin interference in APTT testing. APTT results using AHCa HRRS and standard 0.025 M CaCl2 were compared in 33 heparinized and 41 non-heparinized patient samples on Sysmex CN-6000 and Stago Start Max analyzers. Additionally, 30 random plasma samples with prolonged APTT were tested, and heparin-spiked plasmas were used to determine AHCa HRRS's neutralization limit. AHCa HRRS fully corrected prolonged APTT in samples containing up to 0.6 IU/mL UFH. In the 33 heparinized samples, AHCa HRRS reduced all APTT results to <40 s, eliminating the need for urgent reporting. Among the 30 prolonged APTT samples, 8 were corrected by AHCa HRRS (indicating heparin), 20 remained prolonged (suggesting other causes), and 1 was inconclusive. In non-heparinized samples, AHCa HRRS caused only minor APTT shortening (∼5%), confirming minimal impact on true results. Overall, AHCa HRRS effectively neutralizes UFH interference in APTT testing without compromising accuracy in non-heparinized samples. It offers a practical alternative to conventional calcium reagents and thrombin time for identifying heparin contamination in clinical laboratories.
Introduction
The activated partial thromboplastin time (APTT) assay is a crucial diagnostic tool for assessing the intrinsic pathway of the coagulation cascade. It is commonly employed to identify potential coagulation abnormalities, as prolonged APTT can indicate various conditions including liver disease, factor deficiencies, vitamin K deficiency, coagulation inhibitors, and disseminated intravascular coagulation (DIC).1,2 However, APTT prolongation is also frequently attributed to the presence of unfractionated heparin (UFH) in the patient's blood, which is used for anticoagulation therapy in clinical settings.
Current British guidelines recommend using an UFH calibrated anti-FXa assay over APTT for UFH monitoring at therapeutic ranges of 0.3–0.7 IU/ml, and state that APTT should not be used for patients with an abnormal baseline APTT. 3 However, the baseline APTT is often unknown. Contamination with UFH can arise from improper phlebotomy techniques and the use of heparinized lines. As a result, UFH contamination may occur even in patients not currently undergoing UFH therapy. 4 This highlights the importance of accurately distinguishing between pathological causes of prolonged APTT, such as bleeding disorders, and non-pathological causes, particularly heparin contamination, for effective patient management. 5
In a laboratory context, timely communication with clinical teams is essential when prolonged APTT results are found, as determining the underlying cause is beyond the laboratory staff's scope of practice and can require urgent action. As highlighted in recent reviews, in vitro heparin contamination from indwelling catheters remains a frequent pre-analytical challenge in clinical practice, often leading to diagnostic delays and unnecessary testing. 6 However, a prolonged APTT simply due to heparin contamination does not require the same urgent action by clinical staff. Therefore, a laboratory test capable of differentiating between pathological and heparin-induced APTT prolongation would significantly enhance communication flows and improve patient care, 7 especially when patient notes available to the laboratory are not up to date.
One promising solution to address this challenge is the use of Antihepca (AHCa), a heparin resistant recalcifying solution (HRRS, Haematex, Australia). 8 This reagent has been specifically designed to substitute for 0.025 M calcium chloride in the APTT assay while also possessing the capability to neutralize UFH in patient samples. If effective, AHCa could facilitate laboratories in determining whether prolonged APTT results are attributable to UFH or underlying bleeding or liver disorders.
Various alternative methods exist for assessing UFH presence, such as thrombin time and reptilase testing. In the presence of UFH, thrombin time is prolonged, while reptilase time remains unaffected. Combining these tests can help ascertain whether a patient is undergoing UFH therapy. 1 However, AHCa HRRS presents a more efficient approach due to its ability to neutralize UFH directly within the APTT assay, streamlining the process and providing a “baseline” APTT. This study evaluated the efficacy, limitations, and optimal use of AHCa HRRS in a laboratory setting. Specifically, the study assessed the effect of AHCa HRRS on APTT in non-heparinized samples, the percentage correction in heparinized samples, and the concentration limits of UFH neutralization.
Methods
Specimens
Residual plasmas from citrated vacutainers (Becton Dickinson, USA) were collected, considering factors such as icterus, lipaemia, and haemolysis, as these can interfere with photometric measurements. 9 Each sample was re-centrifuged at 4000 g for 10 min. Normal plasma samples were obtained from 41 patients not receiving anticoagulant therapy, with activated partial thromboplastin time (APTT) results falling within the reference range of 20.6 to 30.8 s. This reference range was derived from Aneurin Bevan Health Board values when using Actin FS on Sysmex CN-6000 analyzers. Heparinized plasma samples were obtained from patients undergoing UFH infusion therapy. Pooled normal plasma (PNP) for heparin-spiking studies was prepared from 30 normal individual donor samples. Therapeutic unfractionated heparin (UFH) diluted in saline to 10 IU/ml was used to prepare heparin-spiked plasmas. Plasmas were aliquoted and stored at −80 °C to preserve sample integrity for future analyses. Samples were fully thawed at 37 °C for 10 min and mixed prior to testing.
Testing
Automated analyses were performed using two Sysmex CN-6000 analyzers (Sysmex, Kobe, Japan), while semi-automated analysis employed the Stago Start Max (Diagnostica Stago, Asnieres sur Seine, France). Siemens Dade Actin FS APTT reagent (Siemens, Marburg, Germany) was used in both systems, with quality control passed using Siemens Ci-Trol 1 and 2 standards. Each methodology involved a similar series of incubation steps at 37 °C, followed by 0.025 M calcium chloride or AHCa HRRS additions. AHCa HRRS was donated by Haematex Research, Sydney, Australia. Thrombin time tests were carried out using Siemens Thromboclotin (Siemens, Marburg, Germany) to compare the ease and specificity of a currently used method for UFH detection. This had a reference range of 14–20 s (Aneurin Bevan health board) deemed normal and suggestive of a sample with no UFH, with samples above 20 s indicating likely presence of UFH. Ten control tests using AHCa HRRS and Ci-trol 1 were performed prior to experimental testing to ensure the accuracy and reliability of the analyzers’ performance.
A correction formula, which provides the percentage correction in APTT values when comparing results between samples treated with CaCl2 and AHCa HRRS, was utilized as follows:
There is a known 10% difference between the CN-6000 and Stago Start Max, as reflected in the 10% adjusted laboratory reference range, with the semi-automated technique being consistently 10% higher due to a difference in analyzer technique (Aneurin Bevan Health Board, 2024). This 10% was calculated upon validation of the Stago Start Max and subsequently verified by comparison with the Sysmex analyzer.
Ethics Statement
All testing was conducted at Grange University Hospital (GUH) in Wales, with ethics approval from the Aneurin Bevan Health Board. Additional ethics approval was obtained from Cardiff Metropolitan University (reference UG-9337).
Results
Discussion
A consistent difference in APTT times between AHCa HRRS and CaCl2 was observed throughout the study. After accounting for an inter-analyzer difference of 2.83%, AHCa HRRS produced shorter APTT times, with a mean difference of 0.6 s, exceeding the manufacturer's claim of 0.5 IU/ml. While statistically significant, this difference was not clinically relevant. The AHCa HRRS reduced 63.6% of results with APTT prolongation to within the normal range, which is crucial for laboratory reporting procedures.
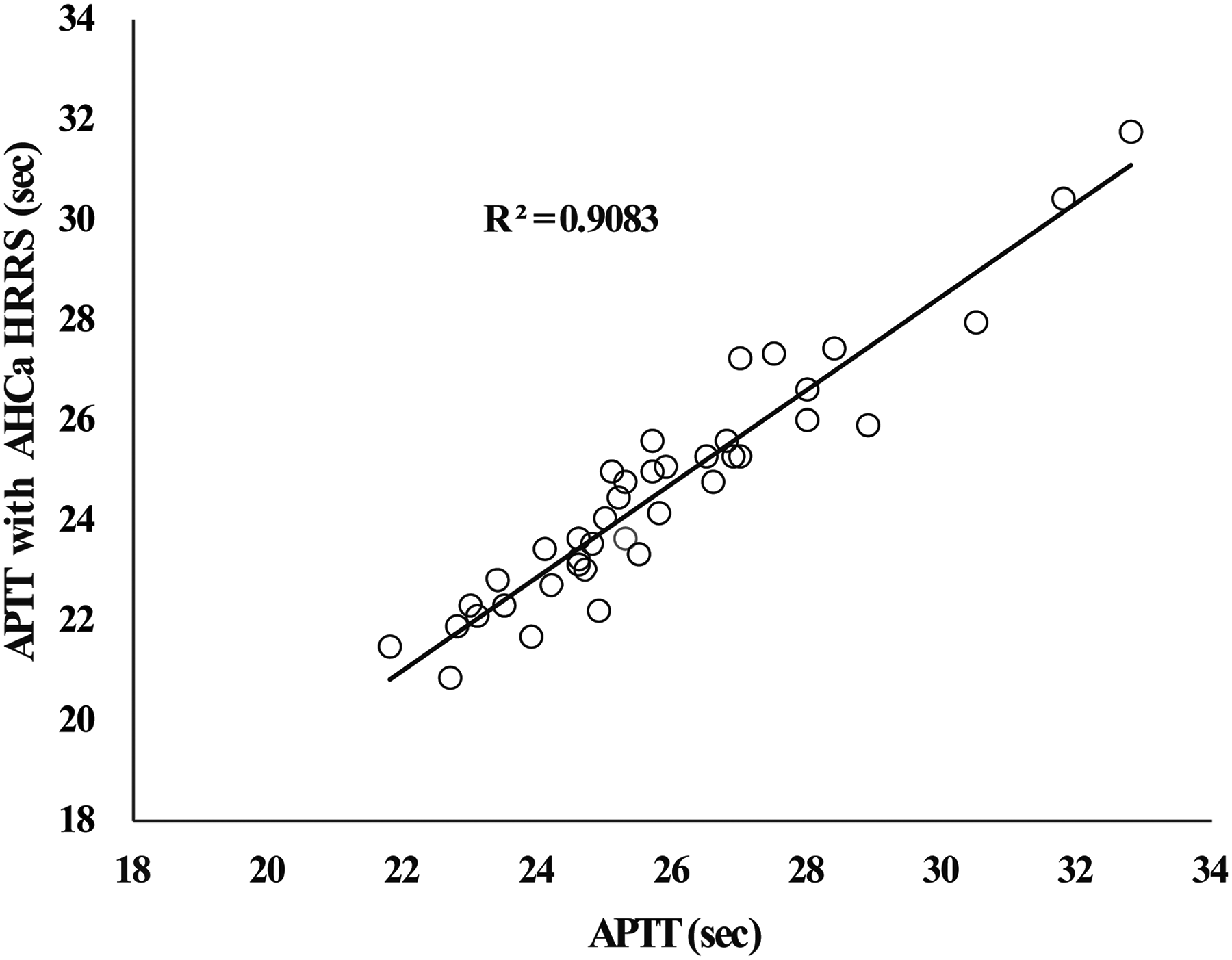
Correlation Between Regular APTT Results on 41 Normal and Near-Normal non-Heparinized Patient Plasmas and APTTs Using AHCa HRRS.

Effect of Heparin Spiked into a Pooled Normal Plasma on Regular APTT (Open Circles) and Also on APTT Using AHCa HRRS (Closed Circles).
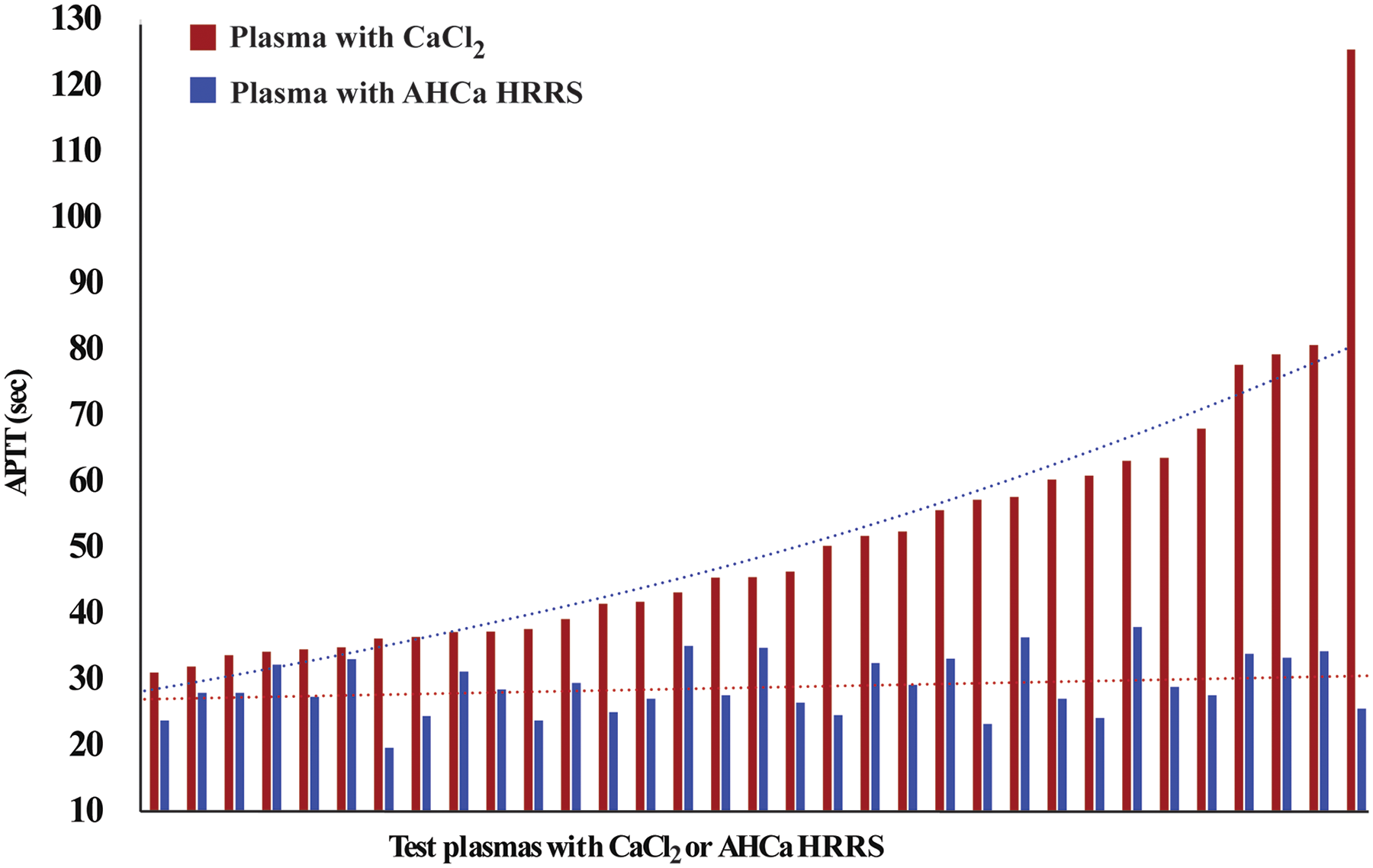
APTT Results on 33 Plasmas from Patients on U/F Heparin Infusion (Orange Bars) in Increasing Order. Corresponding APTT Results with AHCa HRRS Shown Similarly (Blue Bars). Trend Lines Shown for Both Series.

APTT Results on 30 Random Plasmas with Abnormal/Prolonged APTT Plotted Against Corresponding APTTs with AHCa HRRS. Plasmas Giving Thrombin Time (TT) Results Above 50 s are Shown as Orange Markers. the Single Triangle Denotes an Unexpected Result with Long TT but no Significant APTT Change with AHCa HRRS. the “x” Denotes Results with Normal Plasma.
The semi-automated methods using the Stago STartMax produced longer APTT times than the automated Sysmex CN-6000 when the known 10% difference was not accounted for. However, both techniques effectively reduced APTT times with AHCa HRRS, with no significant differences after accounting for the 10% variance. Additionally, while thrombin time confirmed heparin contamination, some results indicated potential underlying disorders affecting APTT, highlighting AHCa HRRS's utility in distinguishing non-heparin-related abnormalities.
The APTT of pooled plasma with AHCa HRRS was 25.5 s, representing the upper threshold for complete neutralization. A heparin concentration of 0.6 IU/ml produced an APTT of 69.4 s with CaCl2, corresponding to a percentage correction of 101.5%. However, results showed that AHCa HRRS remained effective up to heparin concentrations of 1.575 IU/ml, reducing prolonged APTTs to below the critical telephoning threshold of 45 s. AHCa HRRS also lowered APTTs over 180 s to interpretable ranges of 120–160 s for heparin concentrations up to 2.3 IU/ml. At 2.40 IU/ml, AHCa HRRS failed to yield interpretable results. Linear interpolations were used to determine the concentration of heparin when APTT is at 25.5 s, giving a result of 0.611 IU/ml, which can be defined as the AHCa HRRS limit of neutralization.
As heparin concentrations increased, the final APTTs obtained with AHCa HRRS also rose, indicating that partial neutralization could still yield prolonged APTTs. Higher heparin concentrations resulted in lower percentage corrections, but values over 180 s led to inaccuracies. The maximum recorded APTT for patients on UFH therapy was 125 s, with a mean of 51.3 s (APTT ratio 2.05), which falls within AHCa HRRS's 0.611 IU/ml neutralization range. This suggests that for most clinical samples, AHCa HRRS will effectively neutralize heparin-induced prolongation totally.
This study did not explore whether increasing the volume of AHCa HRRS for severely prolonged APTTs would improve neutralization. Future investigations could assess this. AHCa HRRS is compatible with both automated and semi-automated analyzers. However, users should account for slight differences in APTT times between techniques. Semi-automated methods, while slower, may be used without reprogramming fully automated analyzers. 10 AHCa HRRS can be added as needed in labs with occasional use cases, minimizing contamination risk when proper protocols are followed. In vitro heparin contamination—for instance, from blood drawn via a heparin-flushed indwelling line without adequate sample discard, or from contamination with intravenous fluids—can cause falsely prolonged APTT results.11–14 In these scenarios, or when the clinical history is unclear, the AHCa HRRS test is critical for distinguishing pre-analytical errors from true coagulation abnormalities. 15 By establishing clear acceptance criteria, AHCa HRRS's ability to differentiate between pathological causes and heparin contamination can reduce the need for repeat testing and urgent clinician reporting, while improving patient care.
It should also be noted that the spiking of plasma with UFH is less robust than using samples from patients who have been on UFH infusion, as it does not account for in vivo interactions. In addition, no anti-Xa assays were used in this investigation.
Conclusion
AHCa HRRS was evaluated for its application within laboratory settings that routinely analyze patient APTTs. The reagent effectively reduced APTT values in patients with prolonged APTTs due to UFH from therapy or contamination. Importantly, AHCa HRRS did not significantly alter results for samples devoid of heparin, proving its utility in replacing CaCl2. Nevertheless, it was noted that the greater the APTT prolongation from heparin, the higher the final APTT result after using AHCa HRRS, which may limit the reportability of AHCa HRRS results. Overall, AHCa HRRS shows promise in identifying the causes of APTT prolongation and detecting heparin contamination. However, results should not be reported to clinical staff as they may not accurately reflect the in vivo clotting ability of the patients. Each laboratory contemplating the use of AHCa HRRS should carefully weigh its advantages and drawbacks to develop appropriate standard operating procedures. Recently AHCa HRRS has been CE marked under In vitro Diagnostic Regulation (IVDR) and is accessible for general use, therefore it could now be easily implemented within clinical laboratories.
Footnotes
Acknowledgements
Elizabeth Crosby is grateful for the support of Javier Bethencourt, voluntary blood donors, and the donation of AHCa HRRS from Haematex Research. Haematex Research is grateful to Suzanne Kelly for the field support.
Author Contributions
Elizabeth Crosby: Performed and designed research, analyzed and interpreted data, and wrote the manuscript. Thomas Exner conceptualized the study and proof read the article. Manita Dangol assisted with manuscript submission, proofreading and formatting the article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Open Practices Statement
The data that support the findings in this study are available from the corresponding author upon reasonable request.
Relationship Disclosures
Elizabeth Crosby declares no conflict of interest. Thomas Exner is the managing director and Manita Dangol is an employee at Haematex Research Pty. Ltd. Manita Dangol is an employee at Haematex Research Pty. Ltd.

