Abstract
Objective
Rare coagulation factor deficiencies (RCFDs) is a disease characterized by heterogeneous clinical manifestations and low prevalence. This study aimed to thoroughly analyze the clinical characteristics, routine laboratory results, and perioperative treatment plans of patients with RCFDs, enabling clinicians and laboratory professionals to manage these patients effectively.
Methods
Demographic data, laboratory diagnoses, clinical symptoms, bleeding symptoms, perioperative treatment regimens, and outcomes of patients diagnosed with RCFDs (including fibrinogen (FI), FV, FVII, FX, and FXI) were collected and analyzed from 2020 to 2024 at the first affiliated hospital of Xi’ an Jiaotong university.
Results
A total of 60 patients were diagnosed with RCFDs (male: female = 0.62: 1), aged from 1 year to 73 years. 9 patients were diagnosed with FXII deficiency. Fibrinogen disorders were the most common deficient factor (35, 58.3%), in which dysfibrinogenemia accounted for 20 cases, followed by FXI deficiency (10, 16.7%) and FVII deficiency (8, 13.7%). 63 patients (91.3%) had no prior bleeding tendencies, while 6 patients (8.7%) exhibited mucocutaneous bleeding symptom. Fresh frozen plasma (FFP), and fibrinogen concentrate are the main preoperative alternative therapy. Nearly 100% patients with dysfibrinogenemia and hypo-dysfibrinogenemia displayed prolonged TT and normal thromboelastometry parameters. Most of them were asymptomatic, but they may have postoperative bleeding manifestations.
Conclusion
The clinical manifestations of RCFDs are highly heterogeneous. It is difficult to judge the bleeding risk by coagulation factors activity in RCFDs. Fibrinogen disorders account for the highest proportion among all RCFDs. Patients with dysfibrinogenemia and hypo-dysfibrinogenemia may experience postoperative hemorrhage or thrombosis.
Keywords
Introduction
Rare coagulation factor deficiencies (RCFDs) include fibrinogen (FI), FII, FV, FVII, FX, FXI, FXIII, combined FV and FVIII and VK-dependent factor deficiency, but not hemophilia A, hemophilia B and von Willebrand factor. Since FXII does not cause bleeding, hemorrhagic disorders caused by deficiency of factors other than FXII are also referred to as rare bleeding disorders (RBDs).1,2
RBDs represent 3% ∼ 5% of congenital bleeding disorders and are primarily inherited in an autosomal recessive manner, FVII deficiency has the highest prevalence which can reach 1 in 500,000, and the lowest prevalence of FII and FXIII deficiency is about 1 in 2 million. 3 Congenital fibrinogen disorders (CFDs) is a RBD caused by mutations in FGA, FGB, and FGG. CFDs include afibrinogenemia, hypofibrinogenemia, dysfibrinogenemia and hypo-dysfibrinogenemia. 4 Dysfibrinogenemia is the most common type of CFDs characterized by fibrinogen dysfunction with an incidence of 15/100,000, which features large heterogeneity of clinical presentation.5,6 Because most patients do not have clinical manifestations, the prevalence rate is significantly underestimated.
With the rapid progress in laboratory diagnosis, molecular studies, the time to diagnosis of these diseases has moved forward significantly and the number of diagnoses has also increased dramatically. Clinically, they can be accompanied by asymptomatic to severe bleeding episodes, and there are a moderate or strong correlation between factor activity and bleeding severity in fibrinogen, FX, and FXIII deficiencies, but no correlation or weak correlation for FV, FVII, and FXI deficiencies.7,8 Currently, the main treatments for RCFDs include non-alternative treatments such as antifibrinolytic drugs and vitamin K and alternative treatments which include fresh frozen plasma (FFP), cryoprecipitate, prothrombin complexes (PCCs), coagulation factor concentrates, and recombinant coagulation proteins. 9
Currently, information on the clinical, genetic, and laboratory testing of RBDs is increasing, but there are no good laboratory indicators that can effectively predict the risk of bleeding for RBDs. And there is heterogeneous correlation between coagulation factor activity and bleeding severity in different RBDs, so a large amount of clinical data and bleeding prediction indicators need to be developed according to the classification of factor deficiency. Therefore, this study aimed to analyze the diagnosis and clinical profile of 60 patients with FI, FV, FVII, FX, FXI and 9 patients with FXII deficiencies of a single center from northwest China retrospectively. Because the lack of awareness of FXII deficiency among non-hematological clinicians can still lead to treatment delay and a large number of unnecessary alternative treatments, we included FXII deficiency in this study.
Patients and Methods
Study Design and Population
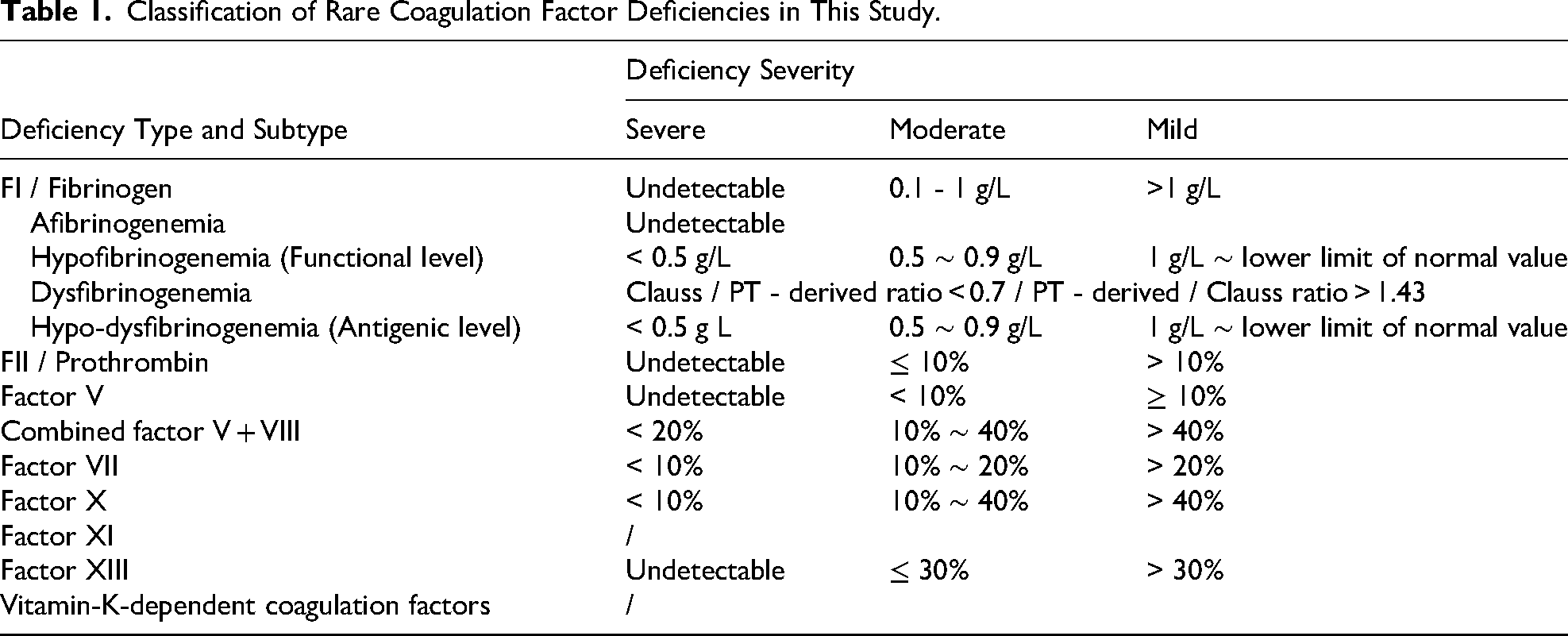
Data was collected from the First Affiliated Hospital of Xi’ an Jiaotong University, Shaanxi province in China from 2020 to 2024. Patients with RCFDs were included in the study. Hemophilia A and B and von Willebrand disease were excluded. All enrolled patients were diagnosed by one of the following methods: family investigation, genetic testing, or showing positive results on routine coagulation tests and coagulation factor assays in more than 2 repeated tests at different time periods using different detection systems after exclusion of other diseases (liver diseases, autoimmune diseases, infectious diseases and so on). Except for one patient with FX deficiency who might be acquired deficiency, the rest cases were all hereditary deficiency of coagulation factors. FXII deficiencies do not cause bleeding manifestations and are not included in the RCFDs. However, this subset of patients is included in this study because their deficiencies are rare and poorly understood by non-hematologists, leading to treatment delays and unnecessary alternative treatments for some patients. Demographic data, routine clotting indicators, factor levels, past history and alternative treatment for surgical history and others were collected. Finally, a total of 69 patients were enrolled in this study. According to the Chinese expert consensus on diagnosis and treatment of rare hereditary hemorrhagic diseases and the International Society of Thrombosis and Hemostasis (ISTH), the disease severity was classified according to the coagulation factor activity or plasma concentration (except for FXI), as shown in Table 1.4,10–13
Classification of Rare Coagulation Factor Deficiencies in This Study.
Coagulation Assays
Citrated samples were immediately centrifuged to separate platelet poor plasma at 2000 g for 10 min. Laboratory diagnosis comprises both screening and diagnostic tests. Routine coagulation function screening tests based on clotting assay were initially conducted for 69 patients. The instrument is Sysmex CS-5100 fully automatic coagulation analyzer, along with its corresponding reagents. Activated partial thrombin time (APTT), Prothrombin time (PT), fibrinogen level, and Thrombin time (TT) served as screening tests, while a mixed test (1:1 mixture of normal plasma and patient plasma) functioned as an auxiliary test. Coagulation factor activity (F:C) acted as a diagnostic test. The activity and antigen levels of fibrinogen were determined by the Cluass and enzyme-linked immunosorbent assay (ELISA) methods, respectively.
Results
A total of 60 RCFDs and 9 FXII deficiency patients were enrolled in our study, mainly including 35 with fibrinogen disorders, 10 with FXI deficiency, 8 with FVII deficiency, 5 with FV deficiency and 2 with FX deficiency (Table 2). The average age at diagnosis for all RCFDs patients was 41 years (ranging from 1 year to 73 years), with 62% being female. There were 8 severe defects (26.7%), 7 moderate defects (23.3%), and 15 mild defects (50.0%). Of the 69 patients, 63(91.3%) were found and diagnosed by routine physical examination or preoperative screening, while only 6(8.7%) were identified and diagnosed due to bleeding manifestations or postoperative bleeding. This study did not identify any cases of FXIII deficiency, combined FV and FVIII or VK-dependent factor deficiency Fibrinogen disorder was the most prevalent. The comparison of routine coagulation tests and thromboelastogram parameters (which can indicate the function of fibrinogen) in patients with fibrinogen disorder revealed that all patients exhibited prolonged TT, markedly decreased fibrinogen-activity, and slightly decreased or normal fibrinogen antigen (Table 3). The PT and APTT of all 14 patients were within the reference range (PT: 12.72 ± 1.56, APTT: 29.63 ± 3.78, not shown in Table 3). 86% patients with dysfibrinogenemia and hypo-dysfibrinogenemia displayed normal thromboelastogram parameters, expect for 2 patients, which aligned with the absence of bleeding symptoms.
Demographic and Clinical Data of 60 RCFDs and 9 FXII Deficiency Patients.
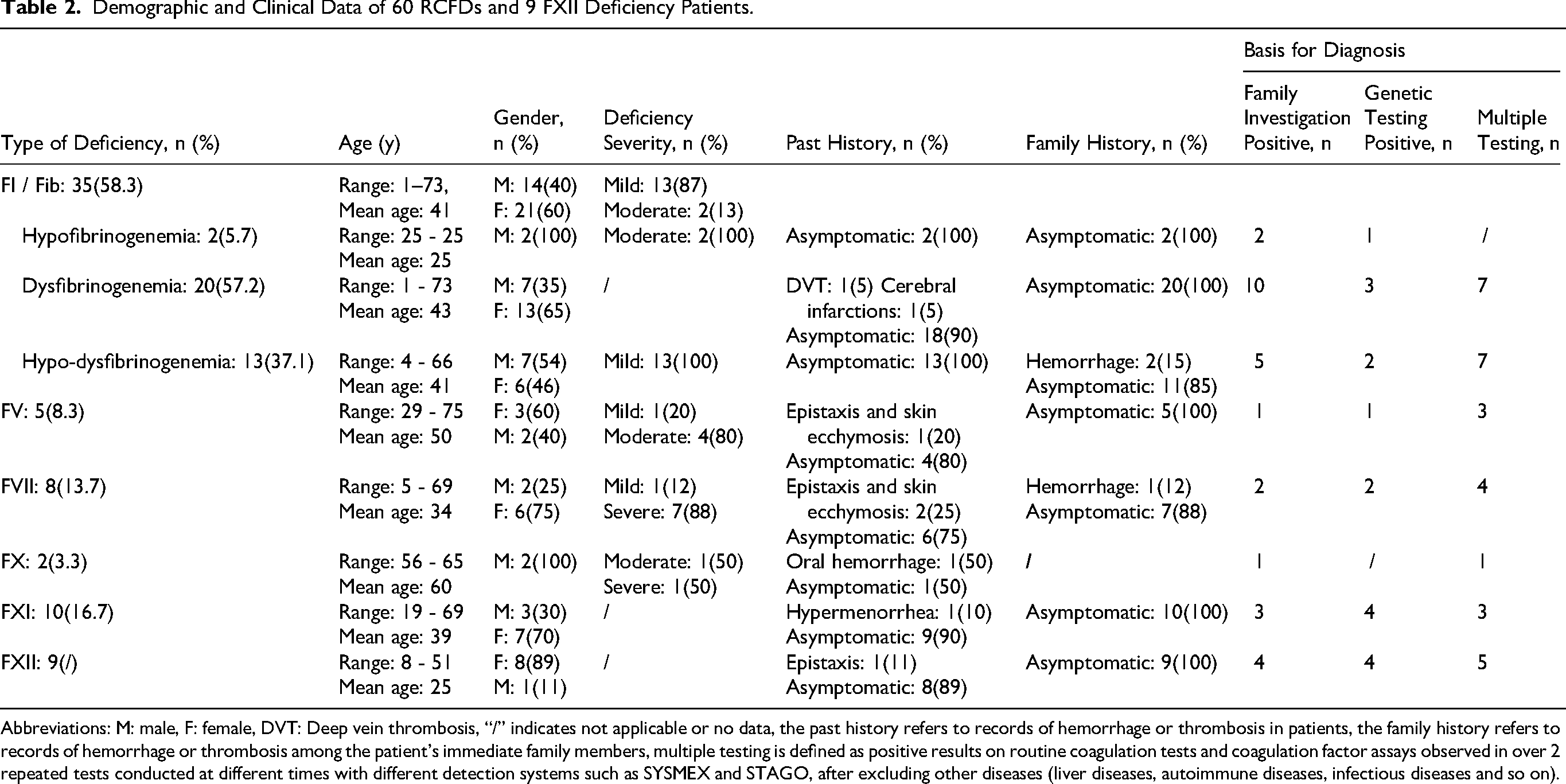
Abbreviations: M: male, F: female, DVT: Deep vein thrombosis, “/” indicates not applicable or no data, the past history refers to records of hemorrhage or thrombosis in patients, the family history refers to records of hemorrhage or thrombosis among the patient's immediate family members, multiple testing is defined as positive results on routine coagulation tests and coagulation factor assays observed in over 2 repeated tests conducted at different times with different detection systems such as SYSMEX and STAGO, after excluding other diseases (liver diseases, autoimmune diseases, infectious diseases and so on).
Preoperative Routine Coagulation Results and Thromboelastogram Results in 11 Dysfibrinogenemia and 3 Hypo-Dysfibrinogenemia Patients.

Abbreviations: M: male, F: female, Fib: fibrinogen, TT: Thrombin time, TEG: thromboelastogram, “/” indicates not applicable.
Next, Table 4 presents the routine laboratory coagulation indicators and clinical outcomes before and after replacement therapy in 11 patients with dysfibrinogenemia, 3 patients with hypo-dysfibrinogenemia and 1 with hypofibrinogenemia. The research indicates that, in most cases, clinicians will elevate the fibrinogen level to a safe range of at least 1.5 g/L before the operation. In cases with minimal surgical trauma, preventive supplementation is not provided. There are significant differences in intraoperative and postoperative bleeding among individuals receiving preoperative alternative therapy. More than half of the patients experienced minimal or no bleeding, and with no thrombosis occurring after surgery. 2 patients had a total bleeding volume exceeding 700 mL. However, 2 patients developed deep vein thrombosis (DVT) or cephalic vein thrombosis after the operation.
Routine Coagulation Indicators and Clinical Outcomes of 15 Patients with Fibrinogen Disorder.
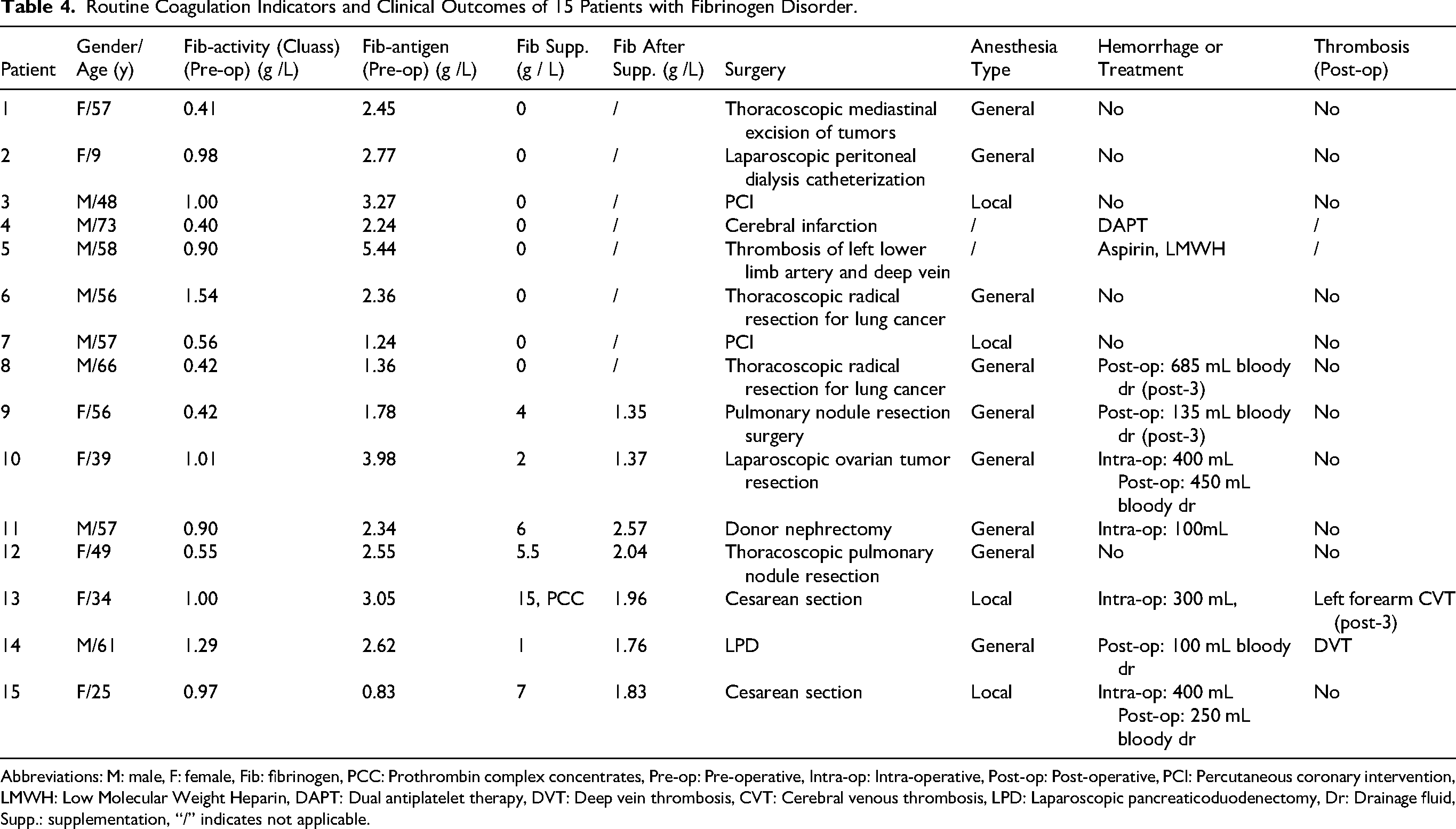
Abbreviations: M: male, F: female, Fib: fibrinogen, PCC: Prothrombin complex concentrates, Pre-op: Pre-operative, Intra-op: Intra-operative, Post-op: Post-operative, PCI: Percutaneous coronary intervention, LMWH: Low Molecular Weight Heparin, DAPT: Dual antiplatelet therapy, DVT: Deep vein thrombosis, CVT: Cerebral venous thrombosis, LPD: Laparoscopic pancreaticoduodenectomy, Dr: Drainage fluid, Supp.: supplementation, “/” indicates not applicable.
In Table 5, we provide information on the coagulation factor activities, preoperative alternative therapy, clinical bleeding manifestations, and clinical outcomes of 7 RCFDs (other than fibrinogen) and 2 FXII deficiency patients. More than 60% of patients requiring surgery or female delivery received corresponding alternative therapy and experienced minimal bleeding afterward. One FV deficiency patient experienced severe bleeding although received alternative therapy. One FXI deficiency patient who did not receive alternative therapy before surgery developed severe postoperative bleeding manifestations. Another one with FXI deficiency relied on FFP to control hypermenorrhea.
Hemorrhage Outcomes After Alternative Therapy of 9 Inpatients with RCFDs.
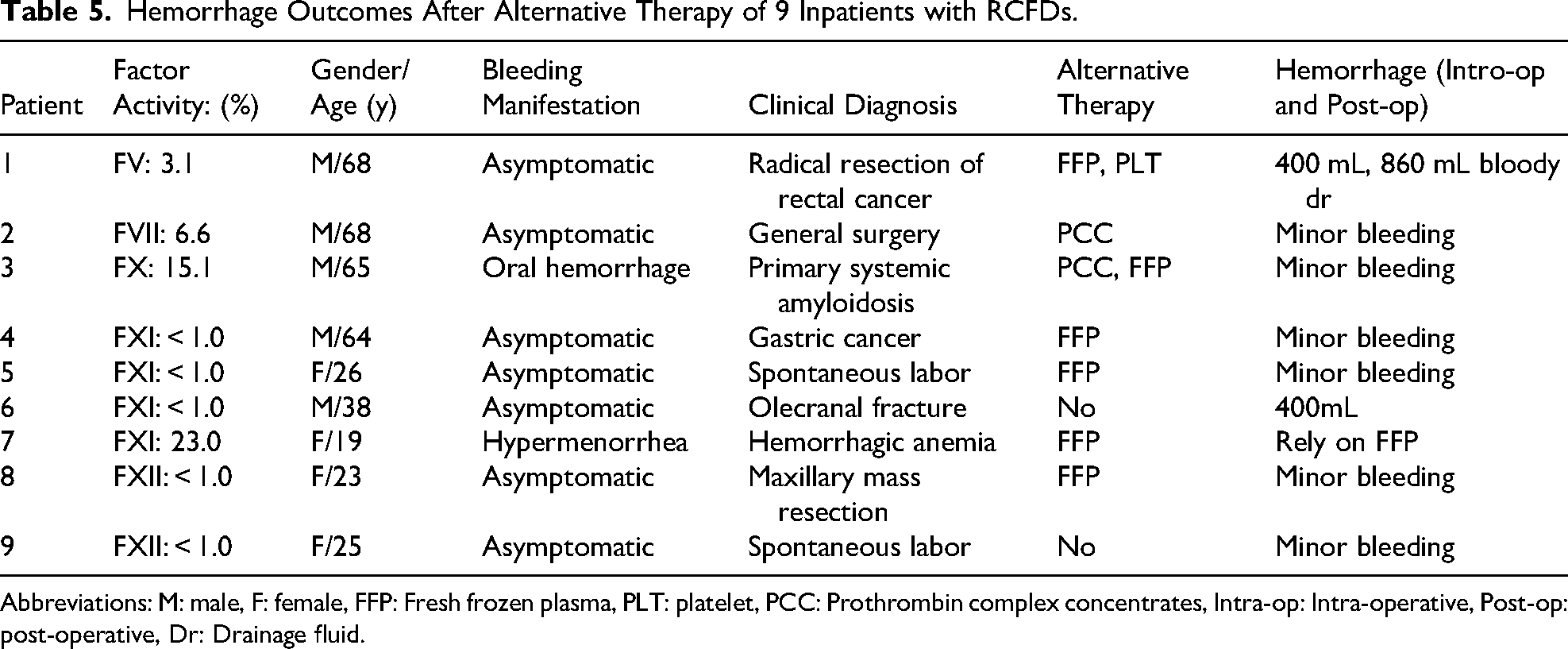
Abbreviations: M: male, F: female, FFP: Fresh frozen plasma, PLT: platelet, PCC: Prothrombin complex concentrates, Intra-op: Intra-operative, Post-op: post-operative, Dr: Drainage fluid.
Discussion
RCFDs pose a challenge in hematology clinicians and laboratory professionals due its rarity, poor correlation between factor activity and bleeding severity, and lack of guidelines. To our knowledge, this is the first report containing the largest number of patients with rare hereditary hemorrhagic disease in northwest China.
91.3% RCFDs was detected by physical examination or preoperative screening, and only 8.7% was diagnosed by bleeding manifestations, conforming to the characteristics of bleeding heterogeneity in RCFDs. The ratio of male/female is close to 1:1 in previous study,3,14 while in this study, women have a higher incidence of the disease possibly because they face a greater risk of bleeding from menstruation and childbirth, which increases the likelihood of diagnosis.
This retrospective study found that fibrinogen disorders (58.3%) were the most reported RCFDs, with a prevalence of dysfibrinogenemia reaching 33.3%, higher than the proportion from Sudan (23.4%), 14 and significantly higher than the data from World Hemophilia Federation (WHF) and the European Network of the Rare Bleeding Disorders (EN-RBD) (8%).15–17 This might be attributed to the combined use of Clauss and PT-derived methods which offers high sensitivity and specificity in diagnosis of congenital dysfibrinogenemia.11,13
There were 15 cases of hypofibrinogenemia and hypo-dysfibrinogenemia in 35 cases of fibrinogen disorders, and the other 20 cases were asymptomatic dysfibrinogenemia (other than 2 with thrombosis events). Congenital dysfibrinogenemia is the most common type clinically in fibrinogen disorders. The global average incidence rate is 1.1% and 1% in Asia, with currently no data available for China. 18 Our study showed that dysfibrinogenemia had up to 57.1% of all fibrinogen disorders, lower than 79.6% in another Chinese report, while higher than 44.7% from 16 countries.19,20 It is reported that 50%∼65% of dysfibrinogenemia is asymptomatic, with 20%∼25% of them suffering from bleeding tendencies, 10%∼15% patients suffering from thrombogenic phenotype with compound genotypes, and occasionally about 2% with both bleeding and thromboembolism.13,21,22 In this study, the incidence of asymptomatic dysfibrinogenemia was much higher than other RCFDs. An exon/genomic data analysis involving 140,000 individuals revealed that the global prevalence of recessively-inherited fibrinogen deficiencies may be 10-fold previously reported, thus the true prevalence of it may be significantly higher than previous inferences. 18
All the patients with dysfibrinogenemia and hypo-dysfibrinogenemia exhibited prolonged TT, normal APTT and PT, significantly decreased fibrinogen activity and normal fibrinogen antigen, similarly to other studies.20,23 86% patients with dysfibrinogenemia and hypo-dysfibrinogenemia displayed normal thromboelastogram parameters, which aligned with the absence of bleeding symptoms. Wei et al found the MA of TEG was not significantly different compared with normal people, 24 consistent with our research showing that the MA of the 14 patients were all within the normal range. Zhou et al found parameters of TEG assays under non-pregnant status might predict the occurrence of obstetric complications, which could help to decide whether replacement therapy is required. 25 The abnormal K and angle value of the only cesarean section patient indicated low fibrinogen function. Based on the results of the routine coagulation tests, although the patient had no positive hemorrhage or thrombosis history, the clinician still considered that the postoperative cesarean section involved a large incision and a high risk of hemorrhage. Therefore, replacement therapy was provided before, during, and after the operation, with the postoperative fibrinogen antigen concentration peaked at 7.77 g/L. In addition, considering the patient's possible history of infusion on the left side and the high incidence of thrombosis during the puerperium period, the patient suffered left forearm thrombosis 48 h after delivery. For such patients, the past history and post-supplementation fibrinogen antigen concentration measured by the PT-derived method may be potential risk factors for postoperative thrombosis. Another patient supplemented with only 1-unit fibrinogen before the operation, ended up with mild bleeding, but DVT after the surgery. For patients with dysfibrinogenemia, a more comprehensive assessment is needed during the perioperative period, including past history, family history, specific gene mutation sites, indicators predicting bleeding risk, the characteristics of the surgery and so on.12,26
FFP, cryoprecipitate, and fibrinogen concentrate are the main preoperative alternative therapy, consistent with our study.27,28 5 out of 8 dysfibrinogenemia and hypo-dysfibrinogenemia patients did not receive alternative therapy and no bleeding or thrombosis occurred. Among the 7 patients who received alternative therapy, 2 with normal TEG parameters still suffered from massive bleeding during and after the operation although fibrinogen had been replenished to more than 1 g / L. So, even if the fibrinogen activity levels are more than 1 g/L in patients with dysfibrinogenemia, and nearly 85% of their TEG results are also normal, clinicians cannot have complete confidence. A study including 166 CFD cases from 16 countries based on the Prospective Rare Bleeding Disorders Database (PRO-RBDD) also revealed that there is no association between fibrinogen activity and bleeding severity in qualitative deficiencies (dysfibrinogenemia) (P = 0.69). 19 Conventional coagulation tests and TEG are not sufficiently effective in predicting the risk of postoperative bleeding in dysfibrinogenemia patients without a prior history, posing a significant challenge for laboratories and clinical practice.
The second most factor deficiency was FXI (16.7%), close to the proportion from North India (18.2%) and WHF (26.5%).3,17 Various studies have documented variable clinical presentation of RCFDs, poor correlation with factor levels or genotype.7,29 Similarly, not all FXI deficiency cases at similar level presented with same bleeding episodes in our study. The manifestation of 4 FXI deficiency patients observed are consistent with earlier reports, 10 which noted post-operative bleeding and increased menstruation. Compared with 2 patients received FFP, the patient who did not receive therapy experienced severe post-operative bleeding. FFP is a nonspecific alternative therapy used in many patients with FXI deficiency undergoing major surgery, because specific FXI concentrates are not readily available worldwide. Only a female developed moderate anemia due to hypermenorrhea and relied FFP, align with the study reporting bleeding manifestation could be mild to moderate. 29 And abnormal uterine bleeding is common in women with FXI deficiency. 30 Wiewel-Verschueren S et al found that most FXI deficiency patients had a mild deficiency and hypermenorrhea accounted for 7%∼67%. 31 For females during the reproductive period, personal history of bleeding was reported the strongest predictor of perioperative or obstetric bleeding, rather than with FXI levels. 32 Thus, the bleeding score systems (BSSs) or bleeding assessment tools (BATs) might be useful. 33
FVII deficiency (13.7%) take the third spot. The bleeding symptoms of FVII deficiency patients observed in this study are the most commonly reported symptoms: epistaxis and skin ecchymosis. 34 FVII deficiency is the most common recessively inherited RCFDs, with thousands of FVII deficiency patients throughout the world.10,35–38 A study from China revealed that 89.6% had FVII:C levels below 10%, and major bleeding events occurred solely in these patients. 34 In contrast, those with minor or no symptoms and FVII:C levels greater than 10% often delay seeking medical attention, leading to underestimated number of patients with FVII deficiency in China.
5 (8.3%) FV deficiency were identified, approximately equal to the proportion from WHF (9%). 17 In a study from North Pakistan, FV (28%) deficiency emerged as the most prevalent condition, followed by FX (26%) deficiency, possibly due to elevated consanguinity region. 39 This study reported a patient with FV deficiency who suffered from epistaxis, skin ecchymosis, which are two of the most common manifestation of FV deficiency.10,40 Another surgical patient with FV deficiency still suffered from severe bleeding despite receiving FFP and PLT. Postoperative bleeding might be attributed to the failure to continue supplementing FFP strictly in accordance with the guidelines.
Only 2(3.3%) FX deficiency were identified. Spontaneous CNS bleeds were the most commonly reported type of severe hemorrhage for patients with hereditary FX deficiency, described in 40 studies. 41 However, in our study, one moderate FX deficiency patient presented with oral hemorrhage, the other severe FX deficiency patient diagnosed with multiple myeloma was asymptomatic. The former one had systemic amyloidosis (AL) as primary disease, where amyloid deposits in peripheral blood vessels and organs and adsorbed circulating FX, leading to an acquired FX deficiency with an incidence of 7% to 14%. Therefore, for patients with FX deficiency, genetic testing may be needed for further clarification.
Our study identified no cases of FII deficiency or FV + VIII deficiency, which may be attributed to the rarest inherited coagulation disorder, with a prevalence of 1 in 2 million. 16 This study also did not identify any cases of and FXIII deficiency, which could be due to the rarity of FXIII deficiency cases, the lack of specific diagnostic reagents, and the limited number of laboratories performing these tests.42,43
FXII deficiency (n = 9), an autosomal recessive inheritance, does not cause bleeding. On the contrary, it may lead to thrombotic diseases by reducing fibrinolytic activity in the body, whose incidence rate in the population remains unclear. 44 In our study, except for one with epistaxis, the rest were asymptomatic. Most reports also have indicated that FXII deficiencies individuals do not display abnormalities in bleeding, and often go undiagnosed for decades without apparent clinical phenotype, 45 align with ours. However, Demidova Ekaterina et al reported that mild bleeding accounted for more than 50% patients with FXII deficiency. 46 Although in vitro prolonged APTT due to FXII deficiencies is well known to the hematologists but surgeons still tend to transfuse plasma for correcting APTT, such as FFP replacement therapy both in our and previous studies.46,47
It should be particularly noted that our results reflect RCFDs characteristics in this northwest China center. Caution is advised when applying these findings to regions with different healthcare resources or genetic backgrounds, as disease presentation and management may vary. Genetic tests were not performed on all fibrinogen disorders cases in our study. Despite the poor relationship between the genotype and phenotype of dysfibrinogenemia patients, the genetic tests are useful for diagnosis of various fibrinogen deficiencies. Future work will also incorporate genetic sequencing to clarify genotype–phenotype relationships. While this single - center study is limited by sample size, future work will explore multi - center collaborations in Northwest China to validate findings.
In summary, this article provides a comprehensive analysis of the clinical characteristics, laboratory results, and perioperative treatment plans for patients with RCFDs in northwest China, empowering clinicians and laboratory professionals to manage these individuals effectively. It is important to assess coagulation abnormalities prior to surgical procedures, which may necessitate a consultation with hematologists. Clinicians and laboratory professionals should raise awareness for RCFDs to improve the accuracy of diagnosis and explore more effective perioperative management due to the heterogeneous bleeding manifestations of RCFDs.
Footnotes
Acknowledgements
None.
Ethical Considerations
Our study was approved by the Ethics Committee in Clinical Research (ECCR) of the First Affiliated Hospital of Xi’an Jiaotong University (XJTUAF2025LSYY-564).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Li Yuan designed the study, provided instruction and revised the manuscript. Junling Li conducted the statistical analysis and wrote the manuscript. Yanxia Liu collected the data. All authors critically reviewed and approved the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data analyzed during the current study are available from the corresponding author upon reasonable request.
