Abstract
Surgery of gynecologic malignancies often increases the incidence of Venous thromboembolism (VTE). TAT, TM, PIC, t-PAIC are considered to be potential monitoring significance for the change of coagulation and fibrinolytic balance with gynecological malignant tumors. We aimed to explore TAT, PIC, TM, t-PAIC as diagnostic and predictive new marker of postoperative VTE for patients undergoing surgery of gynecologic malignancies and evaluate its related high-risk factors. 103 cases of gynecological surgery were selected. The malignant tumor patients were divided into VTE and non-VTE group. All patients were detected by chemiluminescence immunoassay for TAT, TM, PIC and t-PAIC before and d1, d3 after operation. One month after surgery, the incidence rate of deep vein thrombosis(DVT) in malignant tumor group was 10.20%. Before operation, PIC, t-PAIC levels in malignant tumor group were significantly higher than those in benign tumor group (P = .025, P = .030). D3 after operation, TAT, TM, PIC and t-PAIC levels in malignant tumor group were significantly higher than those in benign tumor group (P < .0001, P = .036, P = .037, P < .0001). PIC level of the VTE group was significantly higher than that of the non-VTE group in malignant patients (P < .0001). Logistics regression analysis showed that pre-PIC and post-PIC were independent factors of VTE. The AUC of pre-PIC and post-PIC were 0.95, 0.941, with a sensitivity of 100%, 100% and a specificity of 86.4%, 88.6%. As a new predictive biomarker for VTE after the gynecologic malignant surgery, pre-PIC and post-PIC levels are the independent risk factors of DVT and has accurate diagnostic value.
Introduction
Malignant tumors are often accompanied by hypercoagulable state, which puts the affected patients at substantial risk for thrombosis. Cancer-related venous thromboembolism (VTE) remains the second leading cause of death in patients with malignant tumors.1,2 Surgery due to gynecologic malignancy is frequently associated with higher incidence of VTE, with the main causes being tumor location deep in the pelvic cavity, extensive surgery-related trauma, and postoperative hemodynamic changes.
Currently, due to imitations of various methods of detecting blood-related disorders, D-dimer serves as the most common marker for monitoring coagulation. However, it is an exclusive diagnostic index for symptoms suggestive of thrombotic diseases after thrombosis, and there is an urgent need for economic and efficient VTE markers for early diagnosis of hypercoagulable state during or even before the state of thrombosis.
Thrombomodulin (TM), thrombin antithrombin complex (TAT), tissue plasminogen activator/inhibitor-1 complex (t-PAIC), and plasmin-antiplasmin complex (PIC) play a vital role in the regulation of the process of venous thrombosis. 3 These four markers combined can comprehensively reflect the degree of vascular endothelial injury, detect the activation of the coagulation and fibrinolytic system, 4 and demonstrate the abnormal changes of the peripheral blood system before thrombosis, which is of great clinical significance for VTE prevention.5,6 Therefore, in this study, we investigated TAT, PIC, TM, t-PAIC as diagnostic and predictive markers of VTE in patients undergoing surgery due to gynecologic malignancies as well as the related high-risk factors for developing postoperative VTE.
Material and Methods
Study Population
Conforming the Enhancing the QUAlity and Transparency Of health Research (EQUATOR) network guidelines, we retrospectively studied our case series. And the clinical information of all cases can be consulted in the hospital's medical history archive. Inpatients with benign or malignant tumor, diagnosed and admitted for the initial surgery (laparotomy or laparoscopic surgery) at the Gynecology Department of Renji Hospital, School of Medicine, Shanghai Jiao Tong University, between March and June 2021, were included in the study. The exclusion criteria were as follows: (1) chronic liver disease; (2) prolonged or >1 month-long use of coagulation modifying drugs; (3) familial thrombophilia; (4) concomitant infectious diseases; (5) history of chemotherapy within the past 3 months, or radiotherapy or surgery within the last 2 weeks; (6) other serious medical or surgical diseases or related tumors.
Among the included patients, 49 presented with malignancy (19 - ovarian cancer, 11 - endometrial cancer, 19 – cervical cancer) and comprised the malignant group, whereas 54 presented with benign disease and served as benign control group. All patients received anticoagulant therapy after surgery, according to their Caprini score 7 and were followed up to 1 month after the operation.
Surgical procedures were categorized by invasiveness: minimally invasive (laparoscopic) versus open surgery. Procedure types and durations were recorded to assess potential confounding effects. Deep vein thrombosis (DVT) was diagnosed using color Doppler ultrasound performed by certified radiologists. Diagnostic criteria included: (1) non-compressibility of the vein, (2) absence of flow on Doppler imaging, and (3) direct visualization of thrombus within the vessel lumen. All suspected cases were independently reviewed by two radiologists to ensure diagnostic accuracy. Symptomatic patients underwent bilateral lower extremity ultrasound, while asymptomatic patients were screened based on clinical suspicion or elevated biomarker levels. Local Ethics Committee approved of the study (No.KY2020-183).
Methods
On one day before operation, first and 3th day after the operation, 2.7 ml of fasting venous blood were collected and mixed with 0.3 ml of 3.2% sodium citrate 10 times. Whole blood was centrifuged at 2000xg for 10 min. The upper-phase plasma was separated and stored at −80°C, then tested regularly at one-week intervals. TAT, PIC, TM and t-PAIC were detected using Sysmex HISCL-5000 automatic chemiluminescence instrument. Upon enrollment, all patients received VTE-preventive or treatment measures, as per their Caprini RAM score. All clinical records were obtained from the hospital information center. Caprini score was calculated based on established risk factors including age, BMI, surgery type, and comorbidities. VTE prophylaxis included intermittent pneumatic compression and low-molecular-weight heparin (enoxaparin 40 mg/day) for patients with Caprini score ≥3.
Statistical Analysis
Statistical analyses were conducted using SPSS 20.0 software and Graphpad Prism 6. For continuous variables, data were summarized as mean ± standard deviation (SD), facilitating the understanding of central tendency and dispersion. Categorical data, on the other hand, were presented as proportions to provide a clear representation of the distribution of cases within each category.
To compare the differences between the two groups, Student's t-test was employed for continuous variables, allowing for the assessment of statistical significance in mean differences. For categorical variables, the chi-square test was utilized to examine associations and differences in proportions.
Furthermore, logistic regression analysis was performed to identify potential risk factors associated with the outcome variable. Both univariate and multivariate models were constructed. The univariate analysis served as an initial screen to determine which variables were significantly associated with the outcome, while the multivariate analysis provided a more comprehensive understanding by considering the effects of multiple variables simultaneously.
The diagnostic accuracy of the index was evaluated using the receiver operating characteristic (ROC) curve. This analysis enabled the assessment of sensitivity and specificity across various cut-off points, ultimately aiding in the determination of the optimal threshold for clinical decision-making.
Statistical significance was established at a P-value of <.05, ensuring rigorous interpretation of the results.
Results
Out of the 103 patients, 49 (mean age: 55.65 ± 4.02 years) presented with malignancy (study group) and 54 (mean age: 49.98 ± 5.17 years) with benign tumor (control group). Within one month of follow-up after surgery, deep vein thrombosis (DVT) developed in only 5 cases in the study group and no cases of pulmonary embolus (PE) were observed. On average, DVT developed on postoperative day 13.2. Next, the study group was further subdivided into VTE group (5 cases of DVT diagnosed using Color Doppler) and no-VTE group (44 cases). The clinical and pathological characteristics of the study group, with and without VTE, are summarized in Table 1.
Clinical and Pathological Characteristics in the Study Group, With and Without VTE.

Baseline Levels of TAT, TM, PIC, t-PAIC and DD in Patients with Benign and Malignant Tumors
At baseline, the initial TAT, TM, PIC, t-PAIC levels varied between the study group (malignant tumor) and the control group (benign tumor). PIC (0.709 ± 0.116 c.f. 1.075 ± 0.111, P = .025), t-PAIC (5.472 ± 0.314 c.f. 7.131 ± 0.711, P = .030) and DD (0.18 ± 0.21 c.f.0.58 ± 0.98, P = .004) levels were significantly higher in the study group as compared to controls (Table 2).
Baseline (Before Surgery) TAT, TM, PIC, t-PAIC, DD Levels.

Changing Trends of TAT, TM, PIC, t-PAIC Levels After Surgery
After surgery, the changing trends of these new markers were continuously monitored on postoperative days 1 and 3 in both groups (Figure 1, Table 3). On postoperative day 1, only the t-PAIC levels increased significantly in the study group, which was statistically different between the two groups (P < .0001). On postoperative day 3, statistically significant differences were noted TAT, TM, PIC, t-PAIC levels between the two groups (P < .0001, P = .036, P = .037, P < .0001, respectively). However, the change in DD levels on postoperative days 1 and 3 was not statistically significantly different between the study group and controls (P = .267, P = .277).
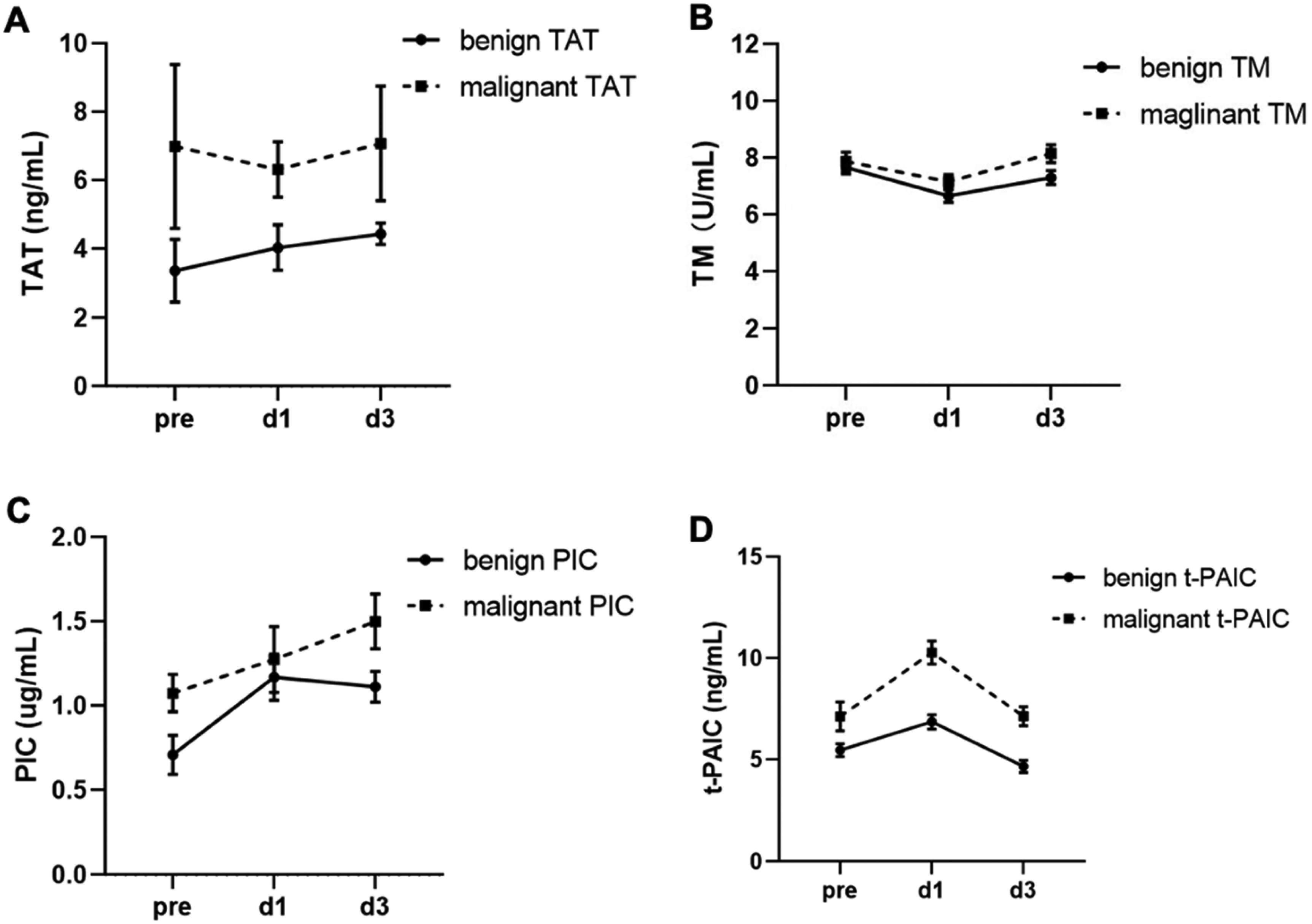
Changing trends in TAT, TM, PIC, and t-PAIC levels on postoperative days 1 and 3. A: The TAT levels in the malignant group were significantly higher compared to the benign controls, with a statistically significant difference between the two groups (P < .0001). B: The TM levels were notably elevated in the malignant group compared to the benign group, showing a statistically significant difference (P = .036). C: PIC levels in the malignant group were higher than in the benign controls, demonstrating a statistically significant variance (P = .037). D: The t-PAIC levels in the malignant group surpassed those in the benign group, with a highly significant statistical difference (P < .0001). (Note: pre refers to baseline measurements; d1 represents the first postoperative day; d3 signifies the third postoperative day.).
Changes in TAT, TM, PIC, t-PAIC, DD Levels After Surgery.

(d1: postoperative day 1; d3: postoperative day 3)
Pre- and Postoperative TAT, TM, PIC, t-PAIC, DD Levels in VTE or no-VTE Patients with Malignant Tumor
Five patients from the study group developed DVT within the first postoperative month. Therefore, we compared the levels of the four new markers of VTE group with no-VTE group in malignant tumor patients at baseline and on postoperative day 3. The results showed that only the PIC, DD levels (including pre- and post- d3) were statistically different between VTE and no-VTE groups in patients with malignancy (P < .0001, P < .0001; P = .002, P = .002) (Table 4).
Comparison of Pre- and Post-Operative TAT, TM, PIC, t-PAIC, DD Levels in VTE and no-VTE Patients with Malignant Tumor.
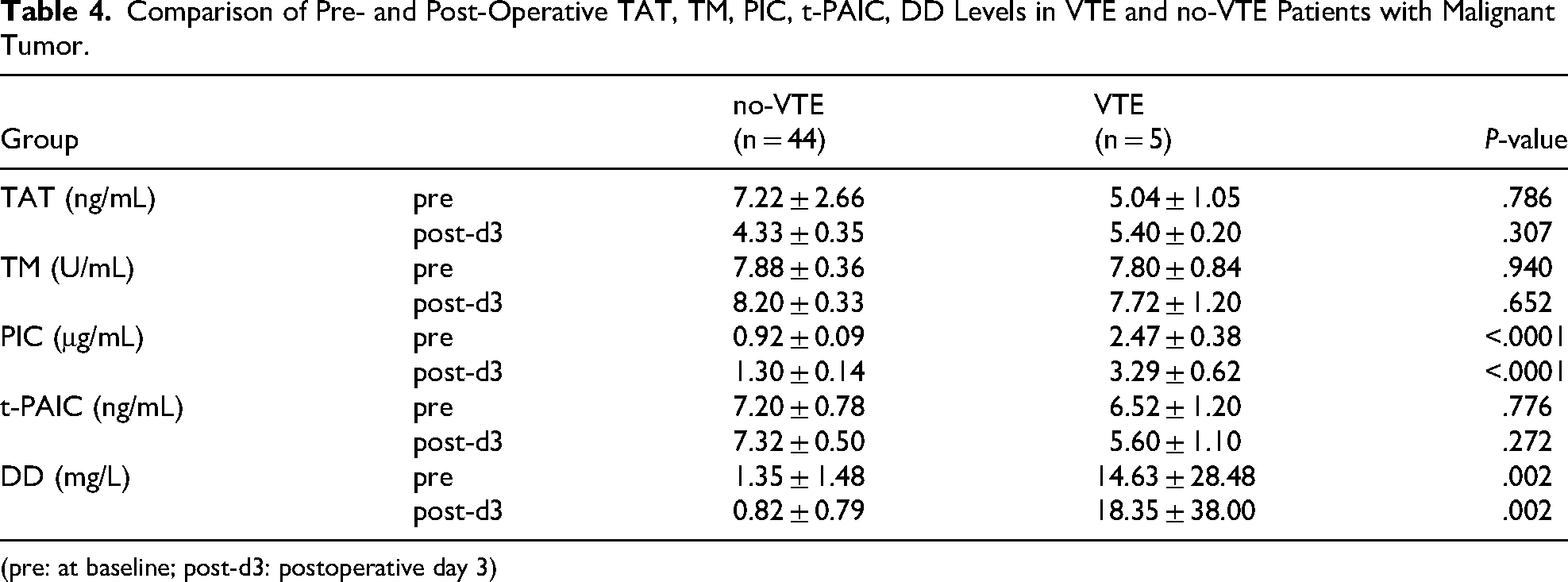
(pre: at baseline; post-d3: postoperative day 3)
Relationship Between Clinical Indices and VTE in Patients with Malignant Tumors and its Diagnostic Efficiency
Univariate logistic regression revealed that the Caprini score as well as pre-PIC and post-PIC levels were indicative of the risk for developing VTE after surgery for gynecological malignancy (Table 5). Furthermore, multivariate logistic regression analysis showed that pre-PIC and post-PIC levels were independent prognostic markers for developing VTE after malignant tumor surgery (Table 6).
Univariate Logistic Regression Analysis of the Correlation Between Clinical Indices and VTE.

Multivariate Logistic Regression Analysis of the Prognostic Markers in Patients with VTE.

The areas under the curve (AUC) for pre-PIC and post-PIC were 0.95 and 0.941, respectively. The cut-off values were 1.409 mg/L and 1.788 mg/L, respectively. Pre-PIC and post-PIC demonstrated higher diagnostic value for VTE, with 100% sensitivity for both markers and the specificity of 86.4% and 88.6%, respectively (Table 7). In contrast, the AUC for pre-DD and post-DD were only 0.675 and 0.711, respectively, with a sensitivity of 60% and 80% and a specificity of 88.6% and 61.4%, respectively.
Comparison of the Diagnostic Efficacy of D-Dimer and PIC for VTE in Patients with Malignant Tumors.

Discussion
Cancer-associated thrombosis remains to be the major cause of mortality among cancer patients, 8 with venous thromboembolism (VTE) as the most common complication. A relationship among malignant tumor, hypercoagulable state and thrombosis was first detected as early as the nineteenth century. Thrombosis is a multifactor, continuous complication affecting the coagulation, fibrinolytic, and endothelial systems.9,10 At present, D-dimer is the most common clinical marker used to monitor the coagulation status in the patients. However, the value of the D-dimer marker lies in its ability to exclude thrombosis, and its detection rates are affected by numerous factors and they lack specificity. Therefore, the search for early-detection, reliable methods of diagnosing VTE continues to be the focus of attention of various authors. The absence of PE cases may reflect rigorous perioperative prophylaxis or limited follow-up duration. PE screening (eg, CT pulmonary angiography) was only performed on symptomatic patients, potentially underestimating subclinical events.
TAT has been recognized as a sensitive marker of thrombin production and an indicator of the activation of the coagulation system.11,12,13,14 PIC is an indicator of activation of the fibrinolytic system, TM can monitor the function of the endothelial cells, 15 while t-PAIC not only reflects abnormalities in the fibrinolytic system, but also the endothelial damage. 16 All of them are important markers in the process of venous thrombosis, as they can be significantly elevated before thrombus and, consequently, can be easily detected. 17
The specific mechanism of coagulation and fibrinolytic system imbalance caused by malignant tumors remains complex.10,18 Patients with malignancy often present with hypercoagulable state, which puts them into very high-risk for thrombosis group. Our results revealed elevated levels of PIC and t-PAIC before surgery in the study group as compared to benign controls, and the same observations were made for DD levels. Our findings also provide further evidence that patients with gynecologic malignancy do indeed have a persistent state of chronic hypercoagulable state before surgery, due to the existence of compensatory hyperfibrinolysis and the aggravation of the vascular endothelial cell damage.
It is common knowledge that operative procedures can increase the risk of developing VTE19,20 due to preoperative mFI (modified Frailty Index) of patients, intraoperative bleeding, transfusion, postoperative vascular endothelial injury and stress activation of the coagulation and fibrinolytic systems, especially in case of surgery for gynecological malignancy. In addition, prolonged bedrest after surgery will further exacerbate the hypercoagulable state in the affected patients.
Frailty was assessed using the 11-item modified Frailty Index (mFI) based on preoperative comorbidities. In particular, the frailty assessment of gynecological oncological patients is fundamental for the efficient prediction of the risk of post-operative complications, even their outcome in terms of Overall Survival(OS) and Disease-Free Survival(DFS). 21 Our exclusion criteria inherently minimized frailty-related confounders. Additionally, the mean age of the malignant group (55.65 ± 4.02 years) and benign controls (49.98 ± 5.17 years) indicated a non-elderly population with lower baseline frailty risk. Therefore, the impact of frailty on perioperative VTE was considered negligible.
On the other hand, we detected an inconsistency in the activation of the coagulation and fibrinolytic systems in benign and malignant gynecological surgery. On postoperative day 3, TM, TAT, PIC and t-PAIC levels were significantly elevated in the study group patients as compared to benign controls. It proves that surgery on the malignant tumor itself resulted in a new activation of the coagulation and fibrinolytic system in the body, which may easily induce VTE. D-dimer, the most commonly used clinical indicator for monitoring the coagulation status in patients, does not have a similar advantage to TM, TAT, PIC and t-PAIC levels. This also confirms that D-dimer is a negative predictor of thrombotic disease in clinical practice.
PIC is an activation marker of fibrinolysis22,23 and is regarded as a satisfactory indicator of enhanced fibrinolytic state, with a half-life of approximately 6 h. 24 Fibrinolytic activity refers to the so-called secondary fibrinolysis, which occurs after a fibrin clot forms in vivo, so elevated PIC levels provide evidence for thrombosis. 25 In this study, we found significant differences in pre-PIC (at baseline, before surgery) and post-PIC, D-dimer (on postoperative day 3) levels between VTE and no-VTE groups. Furthermore, a multivariate analysis showed that only pre- and post-PIC levels were two independent prognostic factors for VTE in patients with gynecological malignancy. Our findings are consistent with the research of Ikeda et al, who found that PIC levels and tumor diameter were two important independent determinants of VTE risk in patients with malignant tumors. 26
Unlike D-dimer (a late marker of fibrin degradation), PIC reflects real-time fibrinolysis activation. This explains its superior sensitivity (100% vs 60%-80%) in detecting pre-thrombotic states, suggesting PIC could guide early prophylaxis in high-risk patients before D-dimer elevation. The results of ROC analysis in our study showed that the areas under the curve (AUC) of pre-PIC and post-PIC values were 0.95, and 0.941, respectively, which is consistent to previous studies. Pre-PIC and post-PIC showed a much better diagnostic value for VTE than DD, with a sensitivity of 100%, 100% and a specificity of 86.4%, 88.6%, respectively. Thus, we believe that they are the optimal diagnostic and predictive markers of VTE.
The primary limitation of this study is the relatively small sample size (n = 103), particularly the low incidence of VTE events (5 cases) in the malignancy group. This limits the statistical power of our analysis and the generalizability of our findings. Future studies with larger cohorts are needed to validate the proposed cut-off values for pre- and post-PIC and to confirm the predictive value of PIC in a broader population. Secondly, these four new markers can be further applied to the perioperative monitoring of other malignant tumors in human body, deepening their clinical application value. Third, surgical parameters (eg, operative approach, duration) were not included in the analysis. As extensive surgeries for advanced malignancies may independently increase VTE risk, future studies should integrate surgical trauma quantification (eg, using the Surgical Complexity Score). Last but not least, we observed 5 cases of DVT but no cases of pulmonary embolism (PE). The absence of PE may be attributed to the relatively small sample size and the low incidence of VTE in our cohort. Additionally, the use of standardized VTE prophylaxis measures in all patients may have further reduced the risk of PE. Future studies should aim to include a larger population to better assess the predictive value of PIC for both DVT and PE.
In conclusion, it was found that TAT, TM, PIC, t-PAIC levels have played an important role in monitoring the occurrence of perioperative thrombosis in gynecological malignant tumors. In particular, PIC can be used as an optimal diagnostic and predictive marker of venous thrombosis and help reduce the incidence and mortality of venous thrombosis, as well as prolong the survival time of the affected patients.
