Abstract
Background
Although hospital readmission after pulmonary embolism (PE) is common, there is limited evidence on the comparative risk of readmission between rivaroxaban and apixaban. This study compared the real-world risk of all-cause hospital readmission among patients with PE treated with rivaroxaban or apixaban.
Methods
This retrospective study used data from Mass General Brigham's Research Patient Data Registry (01/2013-05/2023) to identify adult patients newly initiated on rivaroxaban or apixaban during a PE-related hospitalization (discharge = index). Patients with venous thromboembolism in the 3 months prior to the index PE hospitalization were excluded. All-cause hospital readmissions at 30, 60, and 90 days post-index were assessed using Kaplan-Meier analysis and were compared between cohorts using hazard ratios (HRs), 95% confidence intervals (CIs), and p-values from Cox proportional hazards regression models. Inverse probability of treatment weighting was used to adjust for baseline confounding.
Results
In total, 686 rivaroxaban (mean age: 59.5; female: 50.1; Quan-Charlson comorbidity index: 1.51) and 2207 apixaban (mean age: 60.6; female: 50.8; Quan-CCI: 1.58) initiators were included. Rivaroxaban was associated with a 26% lower risk of all-cause hospital readmission at 30 days post-index (12.3% vs 16.5%; HR [95% CI]: 0.74 [0.58, 0.94]; P = .012). Risk of hospital readmission was also significantly lower at 60 days (17.0% vs 22.3%; HR [95% CI]: 0.74 [0.61, 0.91]; P = .004) and 90 days post-index (21.6% vs 25.6%; HR [95% CI]: 0.81 [0.68, 0.98]; P = .029).
Conclusions
Rivaroxaban was associated with significantly lower risk of all-cause hospital readmission within 90 days post-discharge from PE-related hospitalization than apixaban.
Keywords
Introduction
Venous thromboembolism (VTE), which comprises deep vein thrombosis (DVT) and pulmonary embolism (PE), 1 is estimated to affect up to 900 000 individuals in the United States (US) annually, among which 40% are diagnosed with PE with or without DVT.2,3 VTE recurrence is common, 2 affecting 17.3% of patients with PE and 9.5% of those with DVT only. 4 Moreover, 10% of patients with PE die within 30 days of initial diagnosis, 5 and those surviving discharge have over 14% risk of hospital readmission within 30 days of diagnosis.6,7 Unplanned all-cause 30-day readmission, defined as hospitalization for any cause within 30 days of discharge after initial hospitalization, may be a critical outcome among patients with VTE. 6 A US study reported frequent unplanned all-cause 30-day readmissions among patients following an initial VTE event, which were associated with an 8.1% mortality rate. 6
The rate of 30-day all-cause hospital readmissions has recently become an important measure in hospital systems, as increased rates of hospitalization are associated with considerable costs and may be reflective of quality of care.8–10 Given the frequent hospital readmissions among patients with PE, treatments that may help reduce this burden represent an important mitigation strategy. Rivaroxaban and apixaban, two direct oral anticoagulants (DOACs), have been increasingly used to treat PE.11,12 However, comparative evidence regarding the rates of hospital readmissions among patients treated with rivaroxaban versus apixaban remains limited, and prior studies have primarily assessed the rate of hospital readmissions due to VTE events and major bleeding among patients initiated on VTE treatments.13–15 Therefore, the aim of this study was to evaluate and compare the 30-day risk of all-cause hospital readmission between patients with PE treated with rivaroxaban versus apixaban using real-world evidence. Risk of all-cause hospital readmission at 60 and 90 days after the PE hospitalization was also assessed.
Methods
Data Source
Eligible patients were identified from the Mass General Brigham Research Patient Data Registry (MGB RPDR) PE Data Mart (January 2013–May 2023). The database contains combined inpatient and outpatient clinical data for over 7 million patients and 3 billion records sourced from electronic health record (EHR) data of 8 major healthcare facilities associated with the Harvard Medical School in Massachusetts, specifically Massachusetts General Hospital, Brigham and Women's Hospital, Brigham and Women's Faulkner Hospital, Massachusetts Eye and Ear Hospital, McLean Hospital, Newton-Wellesley Hospital, Salem Hospital (formerly North Shore Medical Center), and Spaulding Rehabilitation Hospital. Data include demographics, providers, visits, diagnoses, medications, procedures, laboratories, as well as prescriptions ordered by providers within the MGB hospital system (including inpatient and outpatient facilities). This study was approved by the MGB Institutional Review Board.
Study Design and Population
A retrospective observational design was used to evaluate and compare outcomes among patients with PE treated with rivaroxaban or apixaban (Figure 1). The date of the first prescription of rivaroxaban or apixaban was defined as the initiation date, and the baseline period was defined as the 6 months prior to the initiation date (including the initiation date). The initiation date was required to occur during a hospitalization with a diagnosis of PE, defined as the index PE hospitalization. The index date was defined as the day of the discharge from the index PE hospitalization. Study outcomes were evaluated during the observation (follow-up) period, which spanned from the index date to the earliest between switch to (or addition of another) oral anticoagulant (OAC), treatment discontinuation, end of clinical activity or data availability, active cancer, or death, whichever occurred first. Due to limited information on days of supply of prescriptions, the discontinuation date was defined as the date of the last prescription of the index agent plus the median time between consecutive prescriptions of the index agent.
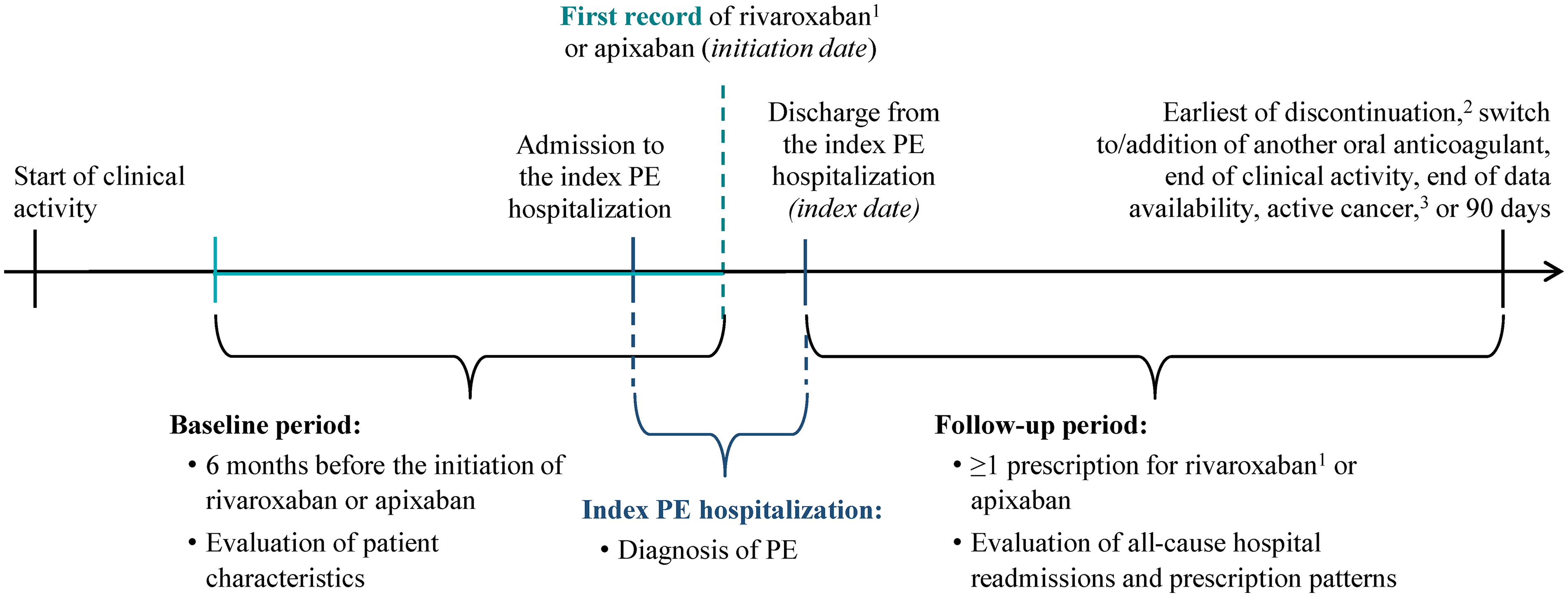
Study design. Abbreviation: PE: pulmonary embolism. Notes: 1. Only 15 mg and 20 mg prescriptions were considered for the identification of rivaroxaban initiation. 2. Discontinuation was defined as the last prescription of the index agent plus the median gap between prescriptions, evaluated per agent per patient. 3. Observation was censored at active cancer, which was identified by the presence of both cancer diagnosis and treatment (at the later of the two dates).
Adult patients were selected in the study population based on the following inclusion criteria: initiated rivaroxaban (15 mg or 20 mg) or apixaban during a hospitalization with ≥1 diagnosis of PE (see Table S1 for diagnosis codes), had a prescription of the index agent as the first OAC prescribed after discharge from the index PE hospitalization, and had ≥6 months of clinical activity before the admission to the index PE hospitalization. Patients meeting any of the following criteria were excluded: had a dispensing for an OAC in the 6 months prior to the index PE hospitalization; had a diagnosis of VTE (see Table S1 diagnosis codes) in the 3 months prior to the index PE hospitalization; had active cancer (defined as having both a cancer diagnosis and treatment), a mechanical heart valve, or a diagnosis of antiphospholipid syndrome in the 6 months prior to or during the index PE hospitalization; had a knee or hip replacement surgery in the 35 days prior to or on the initiation date; or had pregnancy or severe loss of kidney function (estimated glomerular filtration rate [eGFR] < 30) in the 6 months prior to or on the initiation date.
Patients included in the study population were assigned to mutually exclusive rivaroxaban and apixaban cohorts based on the medication prescribed on the initiation date. If both medications were prescribed on the initiation date, the first OAC prescription after discharge from the index PE hospitalization was used to define treatment cohort.
Statistical Analysis
Patients in the apixaban cohort were weighted using inverse probability of treatment weighting (IPTW) to match their baseline characteristics with those of the rivaroxaban cohort and estimate the average treatment effect on the treated (ATT). Weights were calculated using propensity scores derived from a logistic regression model predicting the probability of being treated with rivaroxaban based on the following patient characteristics: age; sex; race; year of initiation date; Quan-Charlson comorbidity index (Quan-CCI; 0-24) 16 ; modified Registro Informatizado de Enfermedad TromboEmbólica (RIETE) bleeding score (0-8) 17 ; major bleeding identified using the Cuningham algorithm 18 ; VTE and bleeding risk factors; comorbidities, medications, and procedures with prevalence ≥5% in either cohort; eGFR; hemoglobin level; and red blood cell, white blood cell, and platelet counts. Differences in baseline characteristics before and after IPTW-ATT were assessed between cohorts using standardized differences (std. diff.).
The risk of all-cause hospital readmission was assessed up to 90 days after discharge from the index PE hospitalization using Kaplan-Meier analysis to account for varying length of follow-up between patients. Risk of all-cause hospital readmission was reported at 30, 60, and 90 days post-discharge from the index PE hospitalization and compared between cohorts using hazard ratios, 95% confidence intervals (CI), and p-values from Cox proportional hazards regression models. In addition, the most frequent admission or primary diagnosis codes observed during readmissions were described among all patients.
All analyses were conducted using Software SAS Enterprise Guide Version 7.15.
Results
Study Population
In total, 686 rivaroxaban and 2207 apixaban users were hospitalized with PE and met the study eligibility criteria (Figure S1). After weighting, the mean age was approximately 60 years, about half of the patients were female, the mean Quan-CCI score was 1.51 and 1.58 among rivaroxaban and apixaban users, respectively, and the mean RIETE bleeding score was 2.31 and 2.39 (Table 1; see Table S2 for additional characteristics). During baseline, the most prevalent comorbidities among rivaroxaban and apixaban cohorts were hypertension (50% and 52%) and hyperlipidemia (35% and 36%), a majority of patients had a prescription for non-oral anticoagulants (88% and 90%) or cardiovascular medication (73% and 76%), and the mean eGFR was 91 in both cohorts.
Selected Baseline Demographic and Clinical Characteristics.

Abbreviations: BMI: body mass index; eGFR: estimated glomerular filtration rate; IPTW: inverse probability treatment weighting; PE: pulmonary embolism; Quan-CCI: Quan-Charlson comorbidity index; RIETE: Registro Informatizado de Enfermedad TromboEmbólica; SD: standard deviation; Std. diff.: standardized difference.
Notes:
The IPTW approach based on the propensity score was used to estimate the average treatment effect for the treated population (ATT). Weights were trimmed over 99% of the distribution, whereby weights larger than the 99th percentile were replaced by that threshold value.
For continuous variables, the standardized difference is calculated by dividing the absolute difference in means of the control and the case by the pooled standard deviation of both groups. The pooled standard deviation is the square root of the average of the squared standard deviations. For dichotomous variables, the standardized difference is calculated using the following equation where P is the respective proportion of participants in each group: |(Pcase-Pcontrol)| / √[(Pcase(1-Pcase)+Pcontrol(1-Pcontrol))/2].
The on-treatment period spanned from the day after discharge from the index PE hospitalization to the earliest between discontinuation, switch to or addition of another oral anticoagulant, end of clinical activity or data availability, active cancer, and 90 days. The discontinuation date was defined as the last prescription of the index agent plus the median gap between prescriptions (ie, 54 and 63 days for rivaroxaban and apixaban, respectively). Active cancer was defined as the presence of both cancer diagnosis and treatment; the later of the two dates was used.
The index PE was defined as “unprovoked” in the absence of malignancy, neurosurgery, orthopedic surgery, trauma, acute spinal cord injury, fracture, estrogen therapy, pregnancy/postpartum state, or oral contraceptive use in the 6 months prior to the admission date to the index PE hospitalization.
Reference: Quan, H. et al Coding Algorithms and for Defining Comorbidities Data in ICD-9-CM Administrative. Medical Care. 2011;43(11):1130–9.
Reference: Ruiz-Gimenez N, et al Predictive variables for major bleeding events in patients presenting with documented acute venous thromboembolism. Findings from the RIETE Registry. Thromb Haemost. 2008;100(1):26–31.
Major bleeding was identified using the Cunningham algorithm. Reference: Cunningham, A et al An automated database case definition for serious bleeding related to oral anticoagulant use. Pharmacoepidemiol Drug Saf. 2011;20(6):560–6.
Obesity (BMI ≥30 or diagnosis) was based on having ≥1 diagnosis of obesity or ≥1 BMI measurement ≥30 kg/m2 anytime during the baseline period.
The last observed baseline measurement was used for the evaluation of lab/vital values.
Chronic kidney disease stage was evaluated with the eGFR using the CKD-EPI 2021 equation.
All-Cause Hospital Readmission
Patients in the rivaroxaban cohort had significantly lower risk of 30-day all-cause hospital readmission after discharge from their index PE hospitalization than those treated with apixaban (Figure 2). Specifically, the risk of all-cause hospital readmission was 26% lower at 30 days (12.3% vs 16.5%; HR [95% CI]: 0.74 [0.58, 0.94]; P = .012) post-discharge from the index PE hospitalization. In addition, the risk of all-cause hospital readmission among patients with PE treated with rivaroxaban versus apixaban was 26% lower at 60 days (17.0% vs 22.3%; HR [95% CI]: 0.74 [0.61, 0.91]) and 19% lower at 90 days (21.6% vs 25.6%; HR [95% CI]: 0.81 [0.68, 0.98]) post-discharge from the index PE hospitalization (both P < .05). The most frequent diagnoses during readmission within 30 days of discharge from the index PE hospitalization were VTE, cardiac arrhythmia, breathing abnormalities, abnormal findings on diagnostic imaging of lung, throat or chest pain, and pleural effusion (Table S3). Major bleeding was not a primary driver of rehospitalization and the proportion of patients who experienced major bleeding during their first hospital readmission within 30 days did not differ substantially between rivaroxaban and apixaban cohorts.

Kaplan-Meier rehospitalization rates. Abbreviation: CI: confidence interval. Note: 1. Number of patients still observed at the specific point in time and with no evidence of re-hospitalization.
Discussion
This real-world retrospective study of EHR data evaluated and compared the short-term risk of all-cause hospital readmission outcomes among patients with PE treated with rivaroxaban or apixaban. The results showed that, compared to apixaban, rivaroxaban was associated with a significantly lower risk of all-cause hospitalization in the 30 days following discharge from patients’ initial PE hospitalization. Risk of rehospitalization was also significantly lower among rivaroxaban users at 60 days and 90 days post-discharge.
DOACs are the mainstay for the treatment of PE.19–21 Previous literature comparing the effectiveness of DOACs, including rivaroxaban and apixaban, has largely focused on the rate of recurrent VTE. For instance, a prospective clinical study directly comparing rivaroxaban and apixaban found that there was no significant difference in the rate of VTE recurrence among treatments (apixaban: 2.3%; rivaroxaban: 2.0%), 22 with similar results reported in two meta-analyses and a cohort study.23–25 To our knowledge, this study is among the first to compare 30-day all-cause hospital readmissions, an increasingly important metric in hospital systems,8,9 among patients with PE treated with rivaroxaban and apixaban. As such, these results provide insights into a scarcely researched outcome in patients with PE.
Hospital readmissions constitute a major issue for the US healthcare system. Around 20% of all Medicare beneficiaries are readmitted within 30 days of hospital discharge, incurring estimated healthcare costs of $15 billion per year. 8 In 2010, the Centers for Medicare and Medicaid Services introduced the Hospital Readmissions Reduction Program with the aim of reducing avoidable readmissions and improving patient care by financially penalizing hospitals when they exceeded expected 30-day readmission rates.26,27 Thus, the rate of 30-day hospital readmissions may be used to assess hospital quality, 10 as higher readmission rates may reflect poorer quality of patient care and additional strain on the healthcare system.6,28–31 Among patients with PE, unplanned all-cause 30-day readmission may be an important outcome, 6 especially considering that the risk for recurrent VTE is highest in the first month after initial thrombosis. 32 Indeed, previous studies have reported that approximately 10%–16% of patients are readmitted to the hospital within 30 days of discharge following PE.6,7,33,34 VTE readmissions are often associated with other conditions (eg, sepsis, cancer, pneumonia, or procedural complications); however, a considerable proportion of readmissions may be due to VTE or a directly-related disease process.6,7 In the present study, VTE was the most common diagnosis during readmission within 30 days of the index date, most of which were attributable to PE. Other common diagnoses during readmission, such as cardiac arrhythmia, breathing issues, chest pain, and pleural effusion, may occur following PE,35–37 or result from procedural complications or sepsis associated with PE treatments, such as surgical embolectomy or inferior vena cava filter placement.6,38,39 Although bleeding is known to be a common complication following treatment of PE with anticoagulation, major bleeding identified using the Cunningham algorithm was not observed to be a driver of 30-day readmission in this study.7,18 Taken together, these findings suggest that there may be a need for improved management of PE.7,33
Literature investigating the impact of DOAC use on 30-day readmission among patients with PE is lacking. A retrospective US study evaluated whether oral anticoagulant use was a risk factor for all-cause 30-day hospital readmission among all patients discharged from a single academic center, regardless of the cause of the initial hospitalization. 8 In that study, warfarin was associated with significantly increased risk of hospital readmission (odds ratio: 1.36) within 30 days of discharge, while treatment with DOACs rivaroxaban, apixaban, and dabigatran were not. 8 However, the study did not compare the risk of 30-day hospital readmission between DOACs, nor was it limited to patients with PE. In the present study, rivaroxaban was associated with a 26% lower risk of readmission than apixaban within 30 days of discharge from the index PE hospitalization. Differences in the length of lead-in dosage and daily dosing schedule may be a factor in the observed readmission rates. Specifically, the length of lead-in dosage for the treatment of DVT and/or PE is 3 weeks for rivaroxaban (15 mg twice daily) and 1 week for apixaban (10 mg twice daily). Following lead-in dosing, rivaroxaban is taken once daily while apixaban continues to be taken twice daily,40,41 which could potentially be associated with lower adherence.42,43 Studies comparing rates of hospital readmission between rivaroxaban and apixaban are scarce, and further research is warranted to investigate potential reasons for differences in risk of all-cause hospital readmission. Nonetheless, these results may help inform treatment strategies to help improve management of patients with PE, as well as reduce costs associated with early readmissions.
Limitations
The results of this study should be considered in the context of certain limitations. First, the possibility of coding inaccuracies in EHR data, such as incorrect entry of diagnosis, procedure, and drug codes, cannot be excluded and may have led to misidentification. However, it is anticipated that the impact on rivaroxaban- and apixaban-treated patients would be similar. Second, all patients included in this study received treatment in a well-resourced health system in Massachusetts, which may impact the generalizability of the present findings to other regions and environments. Third, although IPTW-ATT was used to balance patient demographics and clinical characteristics in both cohorts, there remains a possibility for residual confounding resulting from unobserved heterogeneity. Fourth, as there was limited information in the prescription data on days of medication supply, the last recorded prescription for the index DOAC plus the median gap between prescriptions was used as a proxy for discontinuation, which may imperfectly reflect medication use. Moreover, although all patients had a prescription for the index DOAC at the time of index PE hospitalization and post-discharge, patients may not have filled their prescription or taken their medication as prescribed. Fifth, the most common diagnosis codes during first hospital readmission are purely descriptive and are not meant for direct comparison between the two cohorts.
Conclusions
This real-world, retrospective study found that treatment with rivaroxaban was associated with a significantly lower risk of all-cause readmission compared to apixaban within 30 days after discharge from PE hospitalization. Risk of all-cause hospital readmission was also lower among rivaroxaban users up to 90 days after discharge. Further investigation is needed to determine possible reasons for differences in rates of readmission between rivaroxaban and apixaban.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251327592 - Supplemental material for Real-World Assessment of All-Cause Hospital Readmissions among Pulmonary Embolism Patients Treated With Rivaroxaban Versus Apixaban
Supplemental material, sj-docx-1-cat-10.1177_10760296251327592 for Real-World Assessment of All-Cause Hospital Readmissions among Pulmonary Embolism Patients Treated With Rivaroxaban Versus Apixaban by Veronica Ashton, Guillaume Germain, Julien Boudreau, Manasvi Sundar, Sean D MacKnight, Shawn Murphy, Yichuan G. Hsieh and François Laliberté in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Medical writing assistance was provided by professional medical writer, Roxanne Wosu, MASc, an employee of Groupe d’Analyse, Ltée, a consulting company that has provided paid consulting services to Janssen Scientific Affairs, LLC, which funded the development and conduct of this study and manuscript.
Ethical Considerations
This study was approved by the MGB Institutional Review Board.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article. This study was funded by Janssen Scientific Affairs, LLC. The study sponsor was involved in several aspects of the research, including the study design, interpretation of data, and approval of the final manuscript.
Conflicting Interests
Veronica Ashton is an employee of Janssen Scientific Affairs, LLC. Guillaume Germain, Julien Boudreau, Manasvi Sundar, Sean D. MacKnight, and François Laliberté are employees of Groupe d’Analyse, Ltée/Analysis Group, Inc., a consulting company that provided paid consulting services to Janssen Scientific Affairs, LLC., the study sponsor. Shawn Murphy and Yichuan G. Hsieh are employees of Mass General Brigham, which received funds for this study.
Data Availability
The data supporting the findings of this study are available from MGB RPDR. Restrictions apply to the availability of these data, which were used under license for this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
