Abstract
Background
To investigate the relationship between heart failure (HF) and the non-resolution of atrial thrombus detected in anticoagulated patients with non-valvular atrial fibrillation (NVAF).
Methods
This was a single-center, observational, retrospective, and prospective study. Anticoagulated patients with NVAF and atrial thrombus identified by transesophageal echocardiography or cardiac computed tomography angiography were consecutively enrolled. All patients received follow-up imaging within 6 months to assess the resolution of atrial thrombus. The primary endpoint was the resolution of atrial thrombus and the secondary endpoint was the occurrence of ischemic stroke, major bleeding, and all-cause death during the follow-up period.
Results
Among 8987 patients with NVAF scheduled for catheter ablation or cardioversion, 70 anticoagulated patients with atrial thrombus were final analyzed. The average age was 61.8±10.6 years, 62.9% of them were men, and 32 (45.7%) patients presented with HF. Within the 6-month follow-up period, atrial thrombus resolution was observed in 47 (67.1%) patients. The rate of atrial thrombus resolution was lower in patients with baseline HF (50.0% vs 81.6%). In the adjusted logistic regression analysis model, HF was independently associated with the non-resolution of atrial thrombus (adjusted OR: 5.38, 95% CI: 1.19-24.27). During the median follow-up period of 4.5 years, four ischemic stroke events occurred in four patients. None of the patients in this study experienced major bleeding events or death during follow-up.
Conclusions
HF was associated with the non-resolution of atrial thrombus detected in anticoagulated patients with NVAF. Further research is needed to identify optimal therapeutic approaches for this high-risk population.
Introduction
Atrial fibrillation (AF) increases the likelihood of atrial thrombus formation, which is the primary cause of thromboembolic complications and contributes to a poor prognosis.1,2 Anticoagulation therapy with vitamin K antagonists (VKA) or non-VKA oral anticoagulants (NOACs) is the standard therapy for stroke prevention in patients with AF. 3
For patients with atrial thrombus, current guidelines recommend continuous anticoagulation for a minimum of 3 weeks, followed by imaging to monitor the resolution of atrial thrombus. 3 However, even with sustained therapeutic anticoagulation, a residual risk of atrial thrombus formation persists, and it may continue despite intensified or extended anticoagulation therapy.4–6 Therefore, the non-resolution of atrial thrombus is a notable phenomenon that warrants further investigation.
Heart failure (HF) and AF often coexist, fostering the development of each other through shared pathophysiologic changes and risk factors.7,8 HF represents a hypercoagulable state, as explained by Virchow's triad, thereby promoting the formation of atrial thrombus. 9 The analysis of the ENSURE-AF study revealed an association between HF and the presence of left atrial (LA) thrombus in patients with non-valvular AF (NVAF). 10 Moreover, a sub-study analysis of the LATTEE registry showed that in patients with AF or atrial flutter (AFL), those with HF showed a prevalence of LA thrombus approximately three times higher than those without HF. 11 It was reported in a retrospective study that HF was associated with the non-resolution of LA thrombus in patients with NVAF. 12 However, in patients with NVAF who have atrial thrombus detected even after receiving sustained anticoagulation therapy, the impact of HF on the non-resolution of atrial thrombus remains unclear. The aim of the present study was to investigate the relationship between HF and the non-resolution of atrial thrombus in anticoagulated patients with NVAF.
Methods
Patients and Study Design
This was a single-center observational study. Patients with NVAF who had undergone at least 3 weeks of anticoagulation but still had atrial thrombus detected by transesophageal echocardiography (TEE) or cardiac computed tomography angiography (CTA) were consecutively included. Meanwhile, patients with prior percutaneous left atrial appendage (LAA) occlusion or surgical LAA excision were excluded. All the patients who underwent follow-up imaging within 6 months were finally analyzed. This study has a retrospective phase (from January 2012 to December 2018) and a prospective phase (from January 2019 to October 2023). The study protocol was approved by the institutional review board of the First Affiliated Hospital of Nanjing Medical University, Nanjing, China (Reference No. 2014-SR-113). Informed patient consent was obtained from all patients.
Atrial Thrombus Assessment
In the current study, all patients underwent either TEE or cardiac CTA examinations to assess the existence of atrial thrombus. The choice of the imaging modality was determined by patient preference and clinical discretion.
TEE is the gold standard for evaluating the presence of atrial thrombus in routine clinical practice. 13 On TEE, atrial thrombus was defined as a well-circumscribed echogenic mass attached to the atrial cavity or atrial appendage, visibly distinct from the underlying endocardium or pectinate muscles. The suspected atrial thrombus was examined from multiple planes (0°, 45°, 90°, and 135° views). All images were assessed by two experienced echocardiologists. Any discrepancies were resolved through consensus discussion or, if necessary, through consultation with another specialist.
Cardiac CTA is a reasonable alternative to TEE, as it has demonstrated comparable sensitivity and specificity for the detection of atrial thrombus. 14 In our study, we used a two-phase scan protocol, capturing a second set of images with a 60-s delay following the initial scan. The presence of atrial thrombus was identified if filling defects in the early scan persisted in the later scan. To minimize the influence of motion artifacts, we applied image reconstruction techniques. All images were interpreted independently by two radiologists. If there were discrepancies, a consensus discussion was held, and if deemed necessary, a senior radiologist performed an additional evaluation.
Definition of Heart Failure
The diagnosis of HF was made according to the 2021 European Society of Cardiology guidelines. 15 Patients with HF were identified based on the presence of typical symptoms and/or signs of HF, including but not limited to shortness of breath, fatigue, edema, and reduced exercise tolerance. Moreover, patients with HF exhibited objective evidence of structural and/or functional abnormalities of the heart and elevated natriuretic proteins, with B-type brain natriuretic peptide (BNP) levels exceeding 105 pg/mL or N-terminal prohormone of BNP (NT-proBNP) levels exceeding 365 pg/mL. Furthermore, HF were categorized based on the measurement of left ventricular ejection fraction (LVEF): LVEF ≤40% as HF with reduced ejection fraction (HFrEF), 41%–49% as HF with mildly reduced ejection fraction (HFmrEF), and ≥50% as HF with preserved ejection fraction (HFpEF).
As the HF status might change during follow-up, patients were divided into 3 groups: (1) the group without HF (Group A), comprising patients without HF at baseline and throughout the follow-up period; (2) the group with improved HF (Group B), including patients with HF at baseline who experienced improvement in HF symptoms and/or signs, increased LVEF, or decreased BNP levels; (3) the group with exacerbated HF (Group C), including patients without HF at baseline who developed HF during follow-up, and those with HF at baseline who showed no improvement in HF symptoms and/or signs, LVEF, or BNP levels.
Anticoagulation Adjustment
In this study, we did not have a standardized protocol across the entire study cohort. Anticoagulation management was individualized based on clinical judgment, with input from the treating physician. In practice, if atrial thrombus was observed, the decision to anticoagulation adjustment (for example, from NOAC to warfarin, using different NOAC, increasing NOAC dosage, or to a higher INR target if using warfarin) was made on a case-by-case basis. If thrombus resolution was not achieved during follow-up, we continued with the same individualized approach.
Follow-up and Endpoints
When atrial thrombus was identified, patients were required to receive appropriate anticoagulation therapy for at least 3 weeks. Additionally, follow-up TEE or cardiac CTA within 6 months was mandated. The primary endpoint was the resolution of atrial thrombus. The secondary endpoints included the occurrence of ischemic stroke, major bleeding, and all-cause death during the follow-up period. Major bleeding was defined according to the International Society on Thrombosis and Hemostasis criterion, 16 which included fatal bleeding and/or bleeding occurring in critical areas or organs, as well as instances resulting in a hemoglobin decrease of ≥20 g/L or necessitating a transfusion of at least 2 units of whole blood or red blood cells.
Statistical Analysis
In this study, continuous variables were presented as mean values with standard deviations or median values with quartiles and were compared using Student's t test or the Mann–Whitney U test, depending on their distribution. While categorical variables were displayed as frequencies and percentages, and comparisons were performed using the Chi-square test or Fisher's exact test, as appropriate.
Domestic NOACs were approved for clinical use since 2022 in China. The proportions of baseline HF and the utilization of NOACs before and after the introduction of domestic NOACs were compared in this study. In addition, we compared the proportion of heart failure among patients at different age groups. The patients were divided into two subgroups, aged <55 years and ≥55 years, based on the study protocol of the Swedish Heart Failure Register. 17
Univariable odds ratio (OR) with 95% confidence intervals (CIs) for the risk factors included in the CHA2DS2-VASc scheme were calculated for the prediction of atrial thrombus non-resolution. Moreover, univariate and multivariable logistic regression analyses were employed to investigate the relationship between HF and the non-resolution of atrial thrombus in patients with NVAF. The demographic characteristics, clinical characteristics, LA diameter (LAD), and anticoagulation adjustment were subsequently adjusted. All statistical analyses were performed using SPSS, version 26.0. P-values were calculated as two-sided, and those less than 0.05 were considered statistically significant.
Results
Overall Population Characteristics
In the retrospective phase of this study, 5334 patients with NVAF scheduled for catheter ablation or cardioversion were admitted to our center. Among these patients, 70 (1.3%) anticoagulated patients with NVAF and atrial thrombus were assessed for eligibility. After excluding those with a prior history of left atrial appendage (LAA) intervention (n = 2), without follow-up imaging (n = 30), and those who underwent follow-up imaging over 6 months (n = 6), 32 patients were included in the final analysis. In the prospective phase of our study, 3653 patients with NVAF scheduled for catheter ablation or cardioversion were admitted to our center. A total of 52 (1.4%) anticoagulated patients with NVAF were detected to have atrial thrombus. After excluding one patient with a history of LAA intervention, five patients without informed consent, and two patients with incomplete data, 44 patients were enrolled. During follow-up, 3 patients refused to receive any follow-up imaging and another 3 underwent follow-up imaging over 6 months. Finally, 38 patients from the prospective phase were analyzed (Figure 1).

Patient flow chart.
The mean age of the total 70 patients was 61.8 ± 10.6 years, with 62.9% being men. Among the study population, 54 (77.1%) patients had non-paroxysmal AF, 32 (45.7%) had HF, 15 (21.4%) had a history of stroke/transient ischemic attack (TIA), 33 (47.1%) had hypertension, and 57 (81.4%) patients exhibited elevated levels of BNP. The mean CHA2DS2-VASc score was 2.7 ± 1.8, the mean creatinine clearance rate (CCR) was 83.7 ± 28.3 mL/min, and the mean LAD was 44.9 ± 6.2 mm. Atrial thrombus was predominantly located in the LAA (92.9%, 65/70), with 4 in the left atrial cavity and 1 in the right atrial cavity. At baseline, 25 (35.7%) patients were on warfarin, 15 (21.4%) on dabigatran, and 30 (42.9%) on rivaroxaban (Table 1). The usage of NOAC gradually increased among the study population from 2012 to 2023 (Figure S1). Figure 2 shows a higher prevalence of HF (75.0% vs 37.0%, P = 0.007) and a greater proportion of patients using NOACs (100.0% vs 53.7%, P < 0.001) at baseline in the domestic NOAC era (year 2022 and after), compared to before 2022.

The proportion of patients with heart failure and Non-vitamin K antagonist oral anticoagulant utilization among the study population before and after domestic NOAC era.
Baseline Characteristics of Patients with Atrial Thrombus and Non-Valvular Atrial Fibrillation.
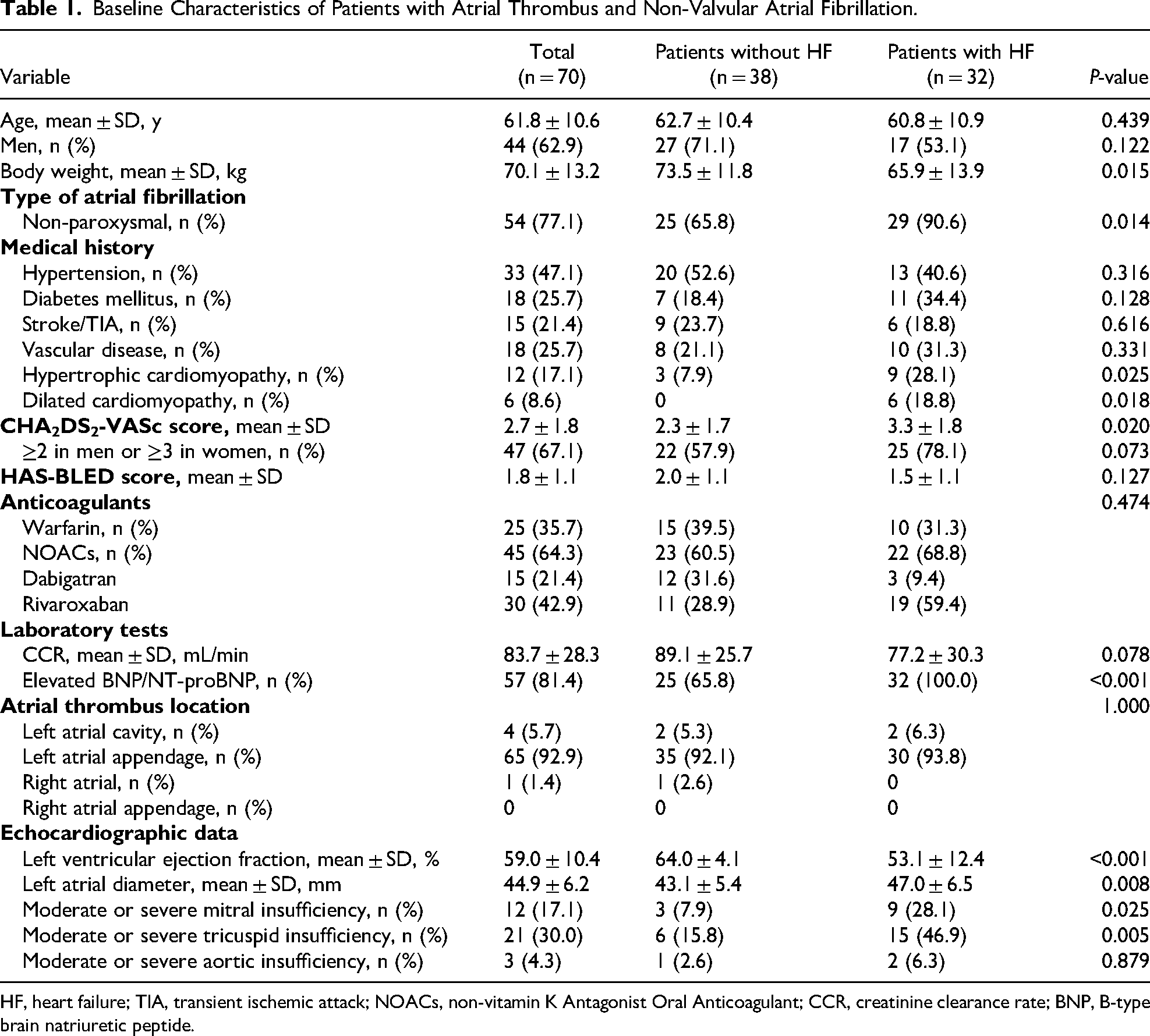
HF, heart failure; TIA, transient ischemic attack; NOACs, non-vitamin K Antagonist Oral Anticoagulant; CCR, creatinine clearance rate; BNP, B-type brain natriuretic peptide.
Heart Failure Population Characteristics
Among the 32 patients diagnosed with HF at baseline, 5 (15.6%) had HFrEF, 7 (21.9%) had HFmrEF, and the remaining 20 (62.5%) patients exhibited HFpEF. Patients with HF were more likely to have non-paroxysmal AF (90.6% vs 65.8%, P = 0.014), elevated levels of BNP (100.0% vs 65.8%, P < 0.001), moderate-to-severe mitral insufficiency (28.1% vs 7.9%, P = 0.025), and moderate-to-severe tricuspid insufficiency (46.9% vs 15.8%, P = 0.005). Additionally, the HF group had lower body weight (65.9 ± 13.9 kg vs 73.5 ± 11.8 kg, P = 0.015), higher CHA2DS2-VASc scores (3.3 ± 1.8 vs 2.3 ± 1.7, P = 0.020), lower LVEF (53.1 ± 12.4% vs 64.0 ± 4.1%, P < 0.001), and larger LAD (47.0 ± 6.5 mm vs 43.1 ± 5.4 mm, P = 0.008). However, no difference in the frequency of NOAC usage or the distribution of atrial thrombus location between the two groups was observed (Table 1).
The Alteration of Anticoagulation Regimen After the Identification of Atrial Thrombus
After the identification of atrial thrombus, the anticoagulation regimen was altered in 21 patients and remained unchanged in the other 49 patients (Figure S2). Among the 49 patients who did not alter their anticoagulation regimen, 18 increased their NOAC dosage, 14 maintained the NOAC dosage, and the remaining 17 continued with warfarin as the anticoagulant. In total, 56 patients received anticoagulation adjustment.
Heart Failure and Atrial Thrombus Resolution
During the 6-month follow up, a total of 70 patients underwent TEE or cardiac CTA. In the first examination, 43 patients had their atrial thrombi resolved. Among the left 27 patients whose thrombi did not resolve, 7 underwent a second examination, during which 4 additional patients had their thrombi resolved. Therefore, the overall thrombus resolution rate was 67.1% (47/70). The median follow-up imaging period was 3 months (IQR: 3-5 months). Among patients who underwent examination at 1, 2, 3, 4, 5, and 6 months, the thrombus resolution rates were 66.7% (4/6), 80.0% (8/10), 61.1% (22/36), 40.0% (2/5), 50.0% (2/4), and 56.3% (9/16), respectively (Table S1). Patients who did not receive follow-up imaging (n = 33) or had follow-up imaging over 6 months (n = 9) were less likely to have vascular diseases (9.5% vs 25.7%, p = 0.037) compared to those who underwent follow-up image within 6 months. No significant differences were observed in other baseline characteristics between the two groups (Table S2).
Patients with HF exhibited a significantly lower rate of atrial thrombus resolution (50.0% vs 81.6%, P = 0.005). Notably, the resolution rate was lowest among patients aged <55 years with HF (33.3%) and highest among patients aged <55 years without HF (85.7%), as shown in Figure S3. Additionally, among the risk factors included in the CHA2DS2-VASc scheme, only HF was associated with the non-resolution of atrial thrombus, with an OR of 4.43 (95% CI: 1.51–12.96, P = 0.007, Figure 3). After adjusting for demographic characteristics, clinical characteristics, LAD, and anticoagulation adjustment, HF remained significantly associated with the non-resolution of atrial thrombus (adjusted OR: 5.38, 95% CI: 1.19–24.27, P = 0.029, Table 2).

Forest plot of odds ratio of risk factors included in the CHA2DS2-VASc scheme in relation to the non-resolution of atrial thrombus.
Univariate and Multivariable Logistic Regression Analysis to Show the Association Between Heart Failure and the Non-Resolution of Atrial Thrombus.

CI, confidence interval.
Model 0: Unadjusted.
Model 1: Adjusted for demographic characteristics, including age, sex.
Model 2: Model 1 + adjustment for clinical characteristics, including hypertension, diabetes mellitus, prior stroke/ transient ischemic attack, vascular disease, cardiomyopathy, and non-paroxysmal atrial fibrillation.
Model 3: Model 2 + left atrial diameter.
Model 4: Model 3 + anticoagulation adjustment.
In this study, 35 patients did not have HF at baseline and during follow-up (Group A), 25 patients had improved HF (Group B), and 10 patients had exacerbated HF (Group C). The resolution rate of atrial thrombus was lower among patients in either Group B or Group C, compared with those in Group A (56.0% vs 82.9%, P = 0.023, and 40.0% vs 82.9%, P = 0.022). There was no significant difference in the resolution rate of atrial thrombus between the Group B and Group C (56.0% vs 40.0%, P = 0.630) (Figure 4).
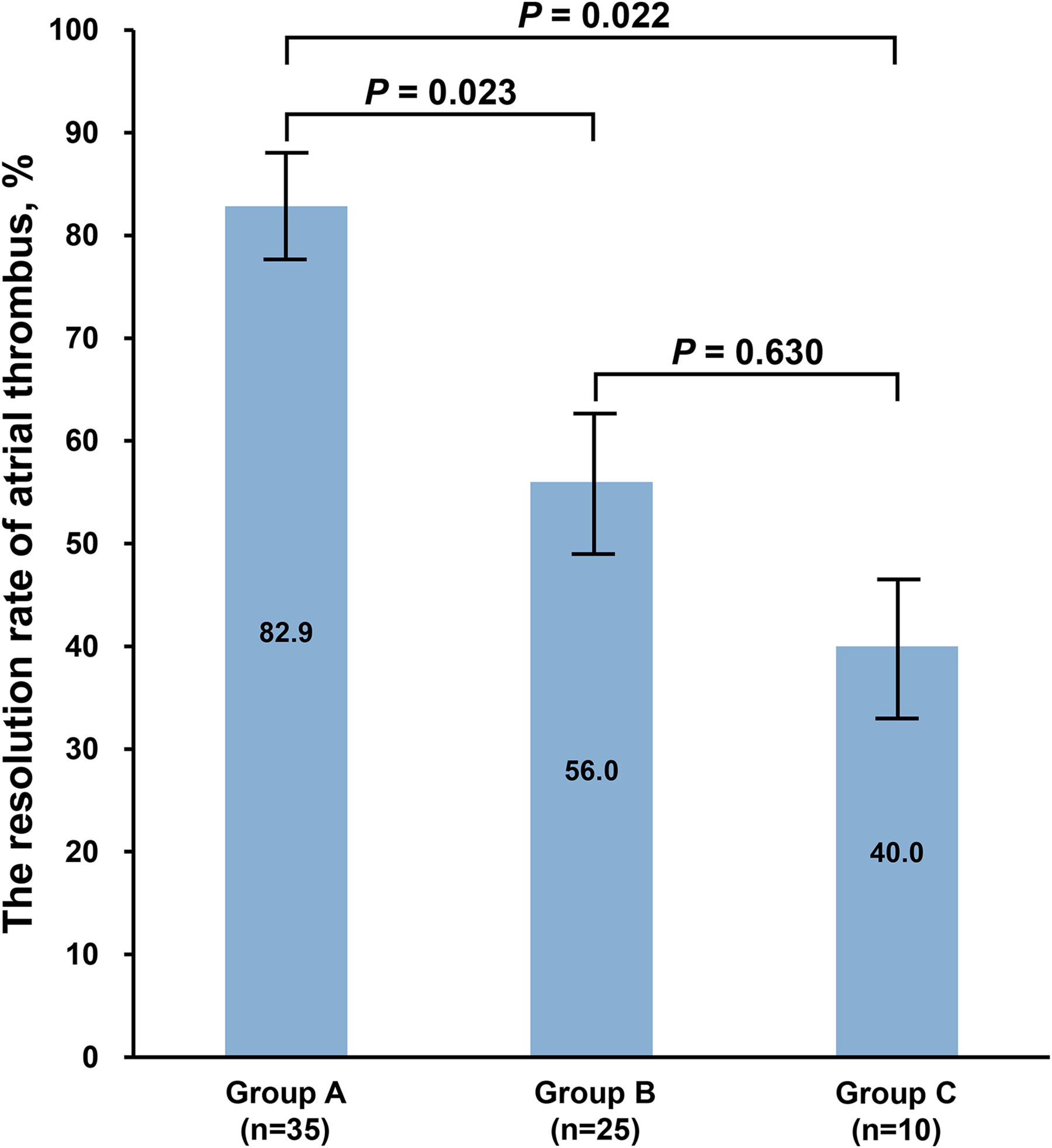
The resolution rate of atrial thrombus according to the changes in heart failure status.
Follow-up Outcomes
During the median follow-up period of 4.5 years (IQR: 2.1–7.4 years), four ischemic stroke events occurred in four patients, resulting in an event rate of 1.1 per 100 person-years. Among the four ischemic stroke events, two occurred after the interruption of OACs, one occurred on the same day after undergoing AF ablation, and another one occurred while under chronic anticoagulation. Among the two patients who interrupted OAC therapy, one was a 70-year-old woman with non-paroxysmal AF, hypertension, and coronary artery disease. She was mildly frail, with a CHA2DS2-VASc score of 4, a HAS-BLED score of 3, and a CCR of 92.44 ml/min. Echocardiography revealed an LVEF of 66.3% and an LAD of 39 mm. She lived alone and had been using warfarin as her anticoagulant. She used to regularly visit the hospital to have her medication dispensed. However, severe weather prevented timely access, resulting in an interruption of her anticoagulation therapy. The other patient who interrupted OAC therapy was a 67-year-old man with non-paroxysmal AF and prior ischemic stroke. His CHA2DS2-VASc score was 3, HAS-BLED score was 3, and CCR was 86.52 ml/min. Echocardiography showed an LVEF of 59.0% and an LAD of 55 mm. He was treatment with dabigatran. His interruption was due to poor adherence to the twice-daily dosing regimen. In both cases, the interruptions in OAC therapy were made as patient decisions. None of the patients who experienced ischemic stroke events had concomitant HF (Table S3). None of the patients in this study experienced major bleeding events or death during follow-up.
Discussion
In this study, we found that nearly half of the patients with NVAF who still had atrial thrombus despite anticoagulation also presented with HF. We observed that 67.1% of atrial thrombus were resolved within the 6-month follow-up period. Notably, patients with HF exhibited a lower rate of atrial thrombus resolution in comparison to those without HF. This association between HF and non-resolution of atrial thrombus persisted even after adjusting for other important covariates. Furthermore, the risk of ischemic stroke was relatively low during the long-term follow-up in this group of patients with HF.
Anticoagulation therapy is of vital importance for stroke prevention in patients with NVAF but cannot completely eliminate the risk of atrial thrombus formation.4,18,19 Based on our finding, approximately 1.3–1.4% of patients who received at least 3 weeks of anticoagulation still had atrial thrombus detected by TEE or cardiac CTA. These results highlighted the importance of pre-procedural imaging test before cardioversion or AF ablation procedures to rule out any existing atrial thrombus. This approach can potentially reduce the peri-procedural risk of thromboembolic events, particularly in patients with HF who are at higher risk of non-resolution of atrial thrombus, as shown in our study.
Previous studies have demonstrated the superiority of NOACs over VKA in stroke prevention in patients with NVAF, especially in Asian patients.20–22 Moreover, NOACs, such as rivaroxaban, have also been demonstrated to be safe and effective in resolving atrial thrombus. 23 In China, although NOACs were approved for clinical use in 2009, their widespread usage was limited primarily due to affordability constraints in the last decade. Therefore, warfarin continued to be the primary OAC in use until the expanded use of generic NOACs. In our study, we found an increased usage of NOACs and a higher proportion of HF cases among the patients with atrial thrombus detected after the year of 2022, when generic NOACs were added to the list of essential medicines in China. In the warfarin era, the formation of atrial thrombus might be mainly attributed to poor adherence to warfarin and failure to maintain therapeutic international normalized ratio.24,25 Given the ease of administration and pharmacokinetic stability advantages associated with NOACs, 26 the increased prevalence of HF further underscored its role in promoting the formation of atrial thrombus in the NOAC era.
In a study by Wu et al, 27 after a median follow-up period of 4.2 months, LA thrombus resolution was observed in 25 out of 44 (56.8%) patients. Another pooled analysis of 619 subjects revealed that the resolution rate of LA thrombus in patients treated with warfarin for at least 3 weeks was 63.7%. 28 Moreover, a recent retrospective real-world study found that among patients who underwent repeat TEE, 66.7% (122/183) showed the resolution of LAA thrombus. 29 The resolution rate of atrial thrombus reported in our study was 67.1%, aligned with previous studies. For the treatment of anticoagulation-resistant atrial thrombus, switching to another NOAC was considered the preferred initial strategy, followed by transitioning to a VKA, low-molecular-weight heparin, or an adjustment in NOAC dosage, with LAA closure reserved as the last resort, as outlined in the European Heart Rhythm Association physician survey. 30 However, in our study, anticoagulation management was individualized based on clinical judgment, with no standardized protocol for adjusting the anticoagulation therapy across the entire cohort.
CHA2DS2-VASc score is the most commonly used stroke risk stratification scheme in patients with NVAF. Di et al 31 discovered that patients with LA thrombus resolution exhibited a lower CHA2DS2-VASc score. Nevertheless, our results showed that within the CHA2DS2-VASc scheme, only HF had predictive capabilities regarding atrial thrombus resolution. HF is often associated with atrial enlargement, atrial fibrosis, and poor atrial function, leading to slower blood flow and increased stasis within the atria.32,33 These changes can create a more favorable environment for thrombus formation and make it harder for existing thrombi to be resolved by anticoagulation therapy. In addition, HF is often accompanied by systemic inflammation and endothelial dysfunction.34–36 These conditions can enhance the pro-thrombotic state and promote thrombus stability, making anticoagulation less effective. Patients with atrial thrombus also exhibited greater atrial fibrosis and impaired atrial function, which can in turn lead to exacerbated heart failure.37–39 Furthermore, our findings indicated that with the improvement of HF status, the likelihood of atrial thrombus resolution increased. This suggests that patients with atrial thrombus and concurrent HF may benefit from adherence to guideline-directed medical therapies for HF.
Interestingly, we found that the resolution rate of atrial thrombus was lowest among younger patients with HF. This could suggest underlying primary atrial cardiomyopathy or global myocardial disease, or genetic predispositions in these younger patients, leading to a more pro-thrombotic state. Further studies are required to understand the unique pathophysiological mechanisms in this subgroup.
In patients with LA/LAA thrombus, the one-year all-cause mortality and the incidence of ischemic stroke were both recorded at 4.9%. 40 However, the evidence regarding the relationship between the presence of LA thrombus and the risk of all-cause mortality and ischemic stroke remained controversial. In our study, four patients experienced ischemic stroke events during follow-up. Notably, two of these strokes occurred after anticoagulation therapy was interrupted. Consistent with Van der Horst et al, 41 who identified male sex, younger age, reduced DOAC dose, twice-daily dosing, and treatment with apixaban or dabigatran as risk factors for nonadherence, one of our patients experienced treatment interruption due to poor adherence to a twice-daily dabigatran regimen. This highlights the challenges of complex dosing schedules in anticoagulation adherence. These findings underscored the importance of maintaining continuous anticoagulation therapy in patients with NVAF, a recommendation supported by previous studies.42–45
Our findings suggest that AF patients with concurrent HF may require more aggressive or tailored anticoagulation strategies to achieve effective thrombus resolution. Further research is needed to identify optimal therapeutic approaches for this high-risk population.
This study had several limitations. Firstly, this study was conducted in a single center and involved a relatively small sample size, which may limit the statistical power and generalizability. Secondly, our study included both retrospective and prospective phases. The retrospective nature of part of the study could introduce biases related to historical data collection and variability in patient management over time. Thirdly, in the multivariable logistic regression analyses, some confounding factors, such as BMI, were not adjusted due to the lack of available data, which might have a slight impact on the interpretation of the results. Lastly, while the study notes the increasing use of NOACs, direct comparisons between NOAC and warfarin outcomes in thrombus resolution were not performed. Further subgroup analyses comparing the efficacy of different anticoagulation strategies would be informative.
Conclusions
HF was associated with the non-resolution of atrial thrombus detected in anticoagulated patients with NVAF. Further research is needed to identify optimal therapeutic approaches for this high-risk population.
Supplemental Material
sj-docx-1-cat-10.1177_10760296251324922 - Supplemental material for Heart Failure and the Non-Resolution of Atrial Thrombus Detected in Anticoagulated Patients with Non-Valvular Atrial Fibrillation
Supplemental material, sj-docx-1-cat-10.1177_10760296251324922 for Heart Failure and the Non-Resolution of Atrial Thrombus Detected in Anticoagulated Patients with Non-Valvular Atrial Fibrillation by Shu Yang, Jie Yang, Ning Chen, Chang Cui, Jincheng Jiao, Li Zhu, Mingfang Li, and Minglong Chen in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Contributions
All authors contributed to the design and data interpretation, and all provided input in each draft of the manuscript.
Data Availability
All data and material supporting this article will be available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent Statements
Ethical approval to report this case series was obtained from the institutional review board of the First Affiliated Hospital of Nanjing Medical University, Nanjing, China (Reference No. 2014-SR-113). Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the National Natural Science Foundation of China (grant number 82270329).
Prior Presentations
Poster, Heart Failure as a Risk Factor for Non-resolution of Atrial Thrombus in Patients with Non-valvular Atrial Fibrillation. 16th Asia Pacific Heart Rhythm Society Scientific Session in Conjunction with CardioRhythm 2023, 31 August - 3 September 2023, Hong Kong.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
