Abstract
Carotid artery stenosis (CAS) often goes undetected until it reaches an advanced stage, which can result in serious complications. The present study evaluated the potential of long noncoding RNA (lncRNA) LINC01088 as a biomarker for CAS. 92 CAS patients and 92 healthy controls (Control group) were included. RT-qPCR was performed to assess the relative levels of LINC01088 and miR-195-5p. Receiver operating characteristic (ROC) curve was used to evaluate the diagnostic potential of LINC01088. The relationship between LINC01088 and miR-195-5p was identified by luciferase reporter assay. Proliferation, migration, and apoptosis in human aortic endothelial cells (HAECs) were assessed using CCK8, transwell, and flow cytometry assay. DAVID was employed for Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) analyses. CAS patients showed decreased LINC01088 expression and increased miR-195-5p expression compared to Control, with a negative correlation between their expression levels in CAS. LINC01088 demonstrated high sensitivity and specificity in distinguishing CAS patients from healthy individuals. LINC01088 directly targets miR-195-5p. Upregulation of LINC01088 reversed the effects of ox-LDL treatment, restoring proliferation and migration while reducing apoptosis in HAECs. However, miR-195-5p mimic reduced the protection of LINC01088 on HAECs proliferation, migration, and apoptosis. For miR-195-5p target genes, GO revealed protein metabolism pathways and KEGG highlighted the p53 and MAPK signaling pathways. The present study revealed the diagnosis value of LINC01088. LINC01088 reversed ox-LDL-induced proliferation, apoptosis, and migration by acting as sponges of miR-195-5p in HAECs. LINC01088 may serve as a protective biomarker in CAS progression.
Introduction
Carotid artery stenosis (CAS) refers to the narrowing or blockage of the common and internal carotid arteries, with atherosclerosis being the primary cause in approximately 90% of cases. 1 The carotid artery as key vessel supplying blood to the brain, when affected by CAS can cause symptoms such as blurred vision, limb numbness and weakness, and speech difficulties due to chronic ischemia and hypoxia from vascular narrowing. 2 In severe cases, cerebral infarction may result from the detachment of vascular plaques, causing blood vessel obstruction.2,3 Common surgical treatments include carotid endarterectomy and carotid artery stenting. 4 However, complications such as perioperative stroke, cardiac events, and cranial nerve damage can affect the success of carotid endarterectomy. The perioperative safety of carotid artery stenting and its long-term risk of adverse events remain areas of ongoing debate.4–6 Early detection of CAS and assessment of stenosis severity are critical for preventing and reducing cerebrovascular events. Digital subtraction angiography is considered the “gold standard” for diagnosing CAS, providing a clear view of the degree of stenosis. 7 However, its limitations—such as invasiveness, high cost, and time-consuming procedure—hinder its widespread application.7,8 Therefore, identifying reliable laboratory markers for the initial screening and evaluation of carotid artery stenosis is essential.
As a type of non-coding RNA, long non-coding RNAs (lncRNAs) can influence various biological functions and the progression of diseases through mechanisms such as DNA methylation, alternative splicing, mRNA stability modulation, and signaling pathway control.9,10 Increasing evidence indicates that lncRNA dysregulation is directly linked to cardiovascular diseases, playing a role in pathological processes including atherosclerosis, ischemia/reperfusion injury, and angiogenesis.11–13 For instance, the expression of H19 was associated with activated cell pyroptosis thereby aggravating atherosclerosis. 11 LncRNA OIP5-AS1 inhibits the ischemia-reperfusion injury by regulating miR-410-3p/Nrf2 pathway. 12 Understanding the regulatory mechanisms of lncRNAs is essential for developing new diagnostic and therapeutic strategies for carotid atherosclerosis. In the differential analysis using GEO public data, we found LINC01088 and further validated its relative expression in serum samples. It is reported that LINC01088 was upregulated in prostate tumor tissues and was related to the progression of the tumor by activating the PI3 K/AKT pathway. 14 LINC01088, as the ceRNA of miR-95/LATS2 regulated, inhibits the invasion and migration of gastric cancer cells. 15 Literature searches reveal that most studies on LINC01088 have focused on oncology,15,16 with no studies to date investigating its role in cardiovascular or cerebrovascular diseases.
MicroRNAs (miRNAs) can modulate mRNA levels by promoting transcript degradation and inhibiting translation. 15 lncRNAs can function as miRNA sponges or bind to target genes regulated by miRNAs. 17 These interactions ultimately influence protein expression and function, leading to changes in various cellular activities.18,19 miR-195-5p, located on chromosome 17p13.1, is part of the miR-15a/b/16/195/497 family. 20 It is reported that miR-195-5p levels were elevated in atherosclerosis patients’ serum compared to healthy individuals. 21 Furthermore, miR-195-5p has diagnostic value in CAS and may be a promising biomarker for the condition. 22 Based on previous predictions from the database, miR-195-5p is a potential target for LINC01088. However, the role of LINC01088 in CAS through the modulation of miR-195-5p remains to be substantiated with additional research.
This study seeks to elucidate the influence of LINC01088 and its associated regulatory target on CAS by examining the expression levels of molecular, alongside conducting cellular function assays. Our findings are expected to contribute valuable insights into the mechanisms underlying CAS development.
Materials and Methods
Study Subjects
184 subjects were recruited from 2020 to 2022 in The Second Affiliated Hospital of Zhejiang Chinese Medicine University Vascular Surgery Department. Among them, 92 cases were diagnosed with CAS. The informed consent was signed by each participant, and the study adhered to the Helsinki Declaration. Additionally, The Second Affiliated Hospital of Zhejiang Chinese Medicine University's Medical Ethics Committee approved this protocol.
Patients met the following criteria: (1) the degree of carotid stenosis is >50% as confirmed by CT angiography or magnetic resonance angiography examination; (2) no history of cerebrovascular disease; (3) no endocrine system diseases, major organ dysfunction, mental disorders
Sample Collection
Subjects who had enrolled were asked to provide fasting blood samples. After centrifuging the collected samples for 10 min at 4000 rpm to extract the serum, they were centrifuged again for 15 min at 12000 rpm to extract the cell debris. The extracted serum was then stored at −80 °C to preserve the integrity of the samples until further analysis.
Cell Culture and Transfection
Human aortic endothelial cells (HAECs, ATCC PCS-100-011) were incubated in Endothelial Cell Media supplemented with endothelial cell growth factors, 5% fetal bovine serum (FBS, Invitrogen, CA, Carlsbad, USA) at 37 °C with 5% CO2. The cells were treated with 100 µg/ml oxidized low-density lipoprotein (ox-LDL, Solarbio, Beijing, China). The overexpressed lentivirus vector of LINC01088 was obtained (System Biosciences, USA) and transfected using Lipofectamine 2000 (Invitrogen), using the RT-qPCR to assess transfection success.
Dual-Luciferase Reporter Assay
The relationship between LINC0108 and miR-195-5p was predicted from the lncRNASNP v3 (http://gong_lab.hzau.edu.cn/lncRNASNP3/#!/). The partial sequences of LINC0108 containing the putative binding sites of miR-195-5p cloned into the pmirGLO vectors (Promega, Madison, WI, USA). The transfection was performed using Lipofectamine 2000 (Invitrogen) and assessed by the dual-luciferase reporter assay system (Promega).
Real-Time qPCR
Total RNA was extracted from TRIzol reagent (Invitrogen) The synthesis of cDNA used PrimeScript RT reagent kit (RR036A, Takara, Dalian, China). The relative expressions of LINC01088 and miR-195-5p were conducted with SYBR Green qPCR SuperMix (Invitrogen). GAPDH and U6 were used as internal controls. The reactions were performed in an ABI7500 real-time PCR system (Applied Biosystems, Foster City, California, USA) and quantified by the 2−ΔΔCt method.
Cell Proliferation, Apoptosis, and Migration Assay
Cell Counting Kit 8 (CCK-8, Beyotime, Nanjing, China) was used to analyze the proliferation. 5 × 103 HAECs/well were seeded into a 96-well plat. 10 µl CCK-8 was added to per well after 48 h and incubated for 2 h. Using the microplate reader, the optical density at 450 nm was determined.
For apoptosis, the transfected was collected to prepare the 1 × 105 HAECs/mL cell suspension with buffer. Fluorescent dye was added to the cell suspension and incubated for 15 min in the dark. Using the flow cytometry (FACSCalibur, BD, USA) to count the cells.
Transwell (24-well transwell plates, Corning, USA) was used to assess the migratory potential. 2 × 104 HAECs/well were seeded into upper chambers (FBS-free medium). Medium with 10% FBS was added to the bottom chambers. After 48 h, fixed with 4% paraformaldehyde and stained with 0.1% crystal violet (Solarbio, China). The inverted light microscope (Olympus Corp.) was used to count the cells.
Bioinformatics Analysis
The GSE173719 was obtained in the GEO database for the search keyword “atherosclerosis lncRNA”, which was analyzed with GEO2R. Visualization of the differential expression results was done using the online website (https://bioinformatics.com.cn/). lncRNA was screened based on the GeneType and obtained the LINC01088. The target genes of miR-195-5p were forecasted based on miRBD, miRWALK, and starBase, and the intersection of the target genes was screened using a Venn plot. GO and KEGG analysis were performed on the DAVID 6.8 online tool.
Statistical Analysis
Each experiment was run at least in triplicate. The mean ± SD was used to express the results. The student's t-test was used to assess the differences between the two groups. The one-way ANOVA with Bonferroni's post hoc test was used to assess the differences of more than two groups. The Chi-square test evaluated the contribution of LINC01088 to the development of CAS. To evaluate the diagnostic value of LINC01088 in CAS, ROC analysis was performed. P < .05 denoted the statistically significant difference.
Results
Clinical Value of LINC01088
The general characteristics of the subjects are summarized in Table 1. No significant differences were observed between the Control and CAS patients regarding age, gender, smoking status, LDL-C, TC, or TG (P > .05, Table 1). However, CAS patients exhibited significantly higher SBP and DBP levels, as well as lower HDL-C levels compared to the Control (P < .001, Table 1).
Comparison of General Data Between the Disease Group and the Control Group.
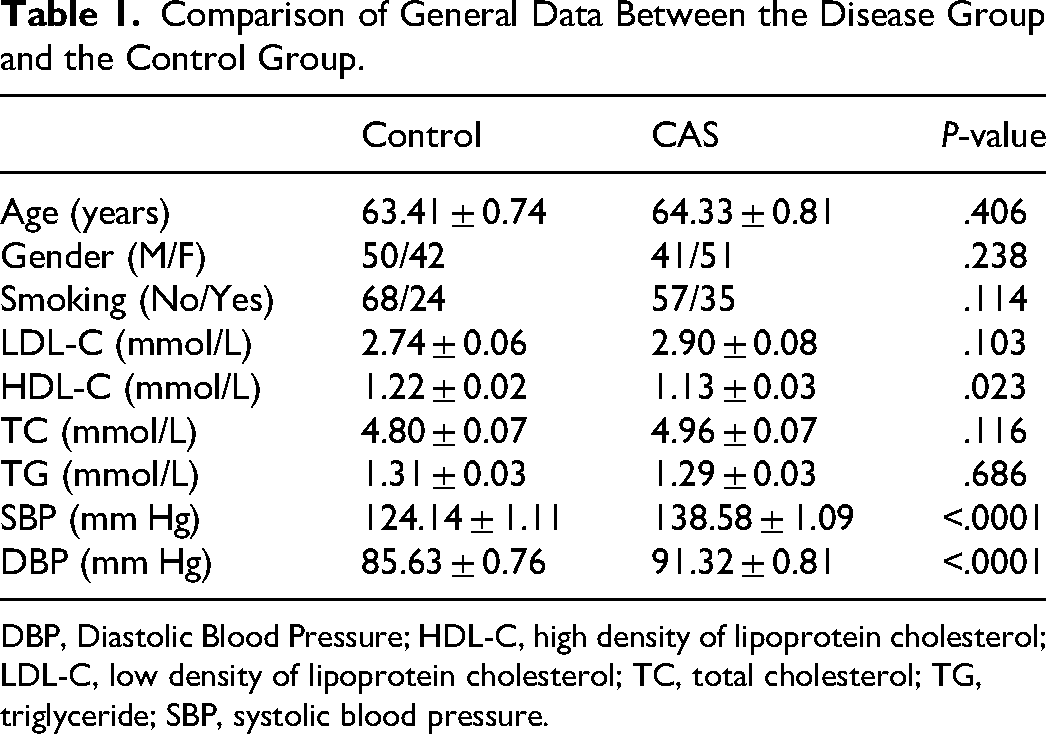
DBP, Diastolic Blood Pressure; HDL-C, high density of lipoprotein cholesterol; LDL-C, low density of lipoprotein cholesterol; TC, total cholesterol; TG, triglyceride; SBP, systolic blood pressure.
In the GSE173719, 132 up-regulated and 11 down-regulated differentially expressed genes were identified. LINC01088 had the highest fold change among lncRNAs and was found to be downregulated (Figure 1A, Supplementary table). Compared to the Control group, LINC01088 expression was markedly decreased in the CAS (P < .001, Figure 1B). The ROC curve analysis revealed an AUC value of 0.850 (95% CI, 0.792-0.906), indicating a strong ability to distinguish CAS from the Control (Figure 1C).

LINC01088 significantly decreased in CAS. (A) Volcano plot showed that LINC01088 was down-regulated in the differentially expressed analysis of GSE173719. (B) Bar plot showed the expression of LINC01088 was down-regulated in the serum of CAS patients (n = 92) compared to the Control group (n = 92). (C) ROC curve analysis showed the diagnostic performance of LINC01088. Data are presented as the mean ± SD. ***P < .001 versus Control. CAS, Carotid artery stenosis; ROC, receiver operating characteristic.
To explore the clinical utility of LINC01088, the CAS patients were categorized into two groups based on the mean relative level of LINC01088. The analysis revealed significant correlations between LINC01088 expression and several clinical parameters including HDL-C (P = .038, Table 2), SBP (P = .040, Table 2), and stenosis degree (P = .005, Table 2). No significant relationships between serum LINC01088 levels and age, gender, smoking, LDL, TC, TG, or DBP (P > .05, Table 2).
Association of Features. with Patients’ Clinicopathological Features.

DBP, Diastolic Blood Pressure; HDL-C, high density of lipoprotein cholesterol; LDL-C, low density of lipoprotein cholesterol; TC, total cholesterol; TG, triglyceride; SBP, systolic blood pressure.
Impact of the LINC01088/miR-195-5p Axis on HAECs Proliferation, Apoptosis, and Migration
Compared to healthy volunteers, CAS patients exhibited a significant increase in miR-195-5p expression (P < .001, Figure 2A). The relationship between LINC01088 and miR-195-5p levels showed a significantly negative correlation (r = -0.788, Figure 2B). The potential binding sites of LINC01088 and miR-195-5p were shown in Figure 2C based on the lncRNASNP v3. Luciferase reporter assays demonstrated that overexpression of miR-195-5p inhibited the relative luciferase activity of LINC01088-WT while having no influence on LINC01088-MUT (P < .001, Figure 2D). The results indicated that LINC01088 directly targeted miR-195-5p.
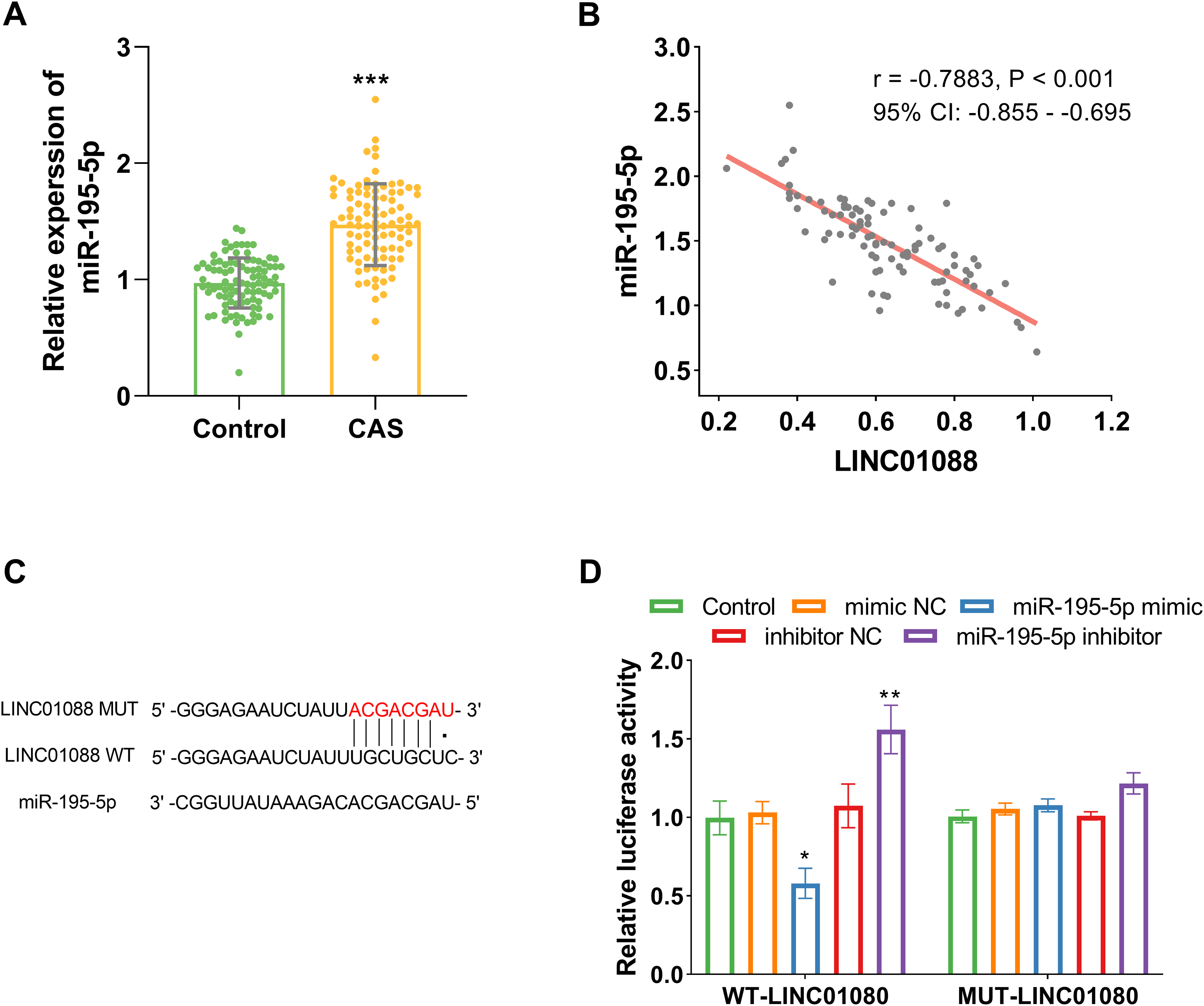
LINC01088 directly targets mir-195-5p. (A) The abundance of miR-195-5p was increased in the serum of CAS patients (n = 92). (B) The negative correlation between the expression of LINC01088 and miR-195-5p in CAS. (C) The potential binding sites for LINC01088 and miR-195-5p by lncRNASNP database. (D) The relationship between LINC01088 and miR-195-5p was verified by dual-luciferase. Data are presented as the mean ± SD (n = 3). *P < .05, **P < .01, ***P < .001 versus Control. CAS, Carotid artery stenosis; HAECs, Human aortic endothelial cells.
Next, the expression of molecular was verified in the cell experiments. HAECs were treated with ox-LDL to establish a CAS-related cell model. Ox-LDL treatment resulted in LINC01088 decreased and miR-195-5p increased, mirroring the expression patterns observed in serum samples (P < .001, Figure 3A). Ox-LDL treatment inhibited HAECs proliferation and migration while promoting apoptosis (P < .05, Figure 3B to D). Conversely, LINC01088 overexpression significantly enhanced cell proliferation and migration while reducing apoptosis (P < .05, Figure 3B to D). Co-regulation experiments showed that miR-195-5p mimics reversed the impact of LINC01088 on HAECs proliferation and migration, as well as its inhibition of apoptosis (P < .05, Figure 3B to D), suggested that miR-195-5p mediated the impact of LINC01088 on HAECs activities.
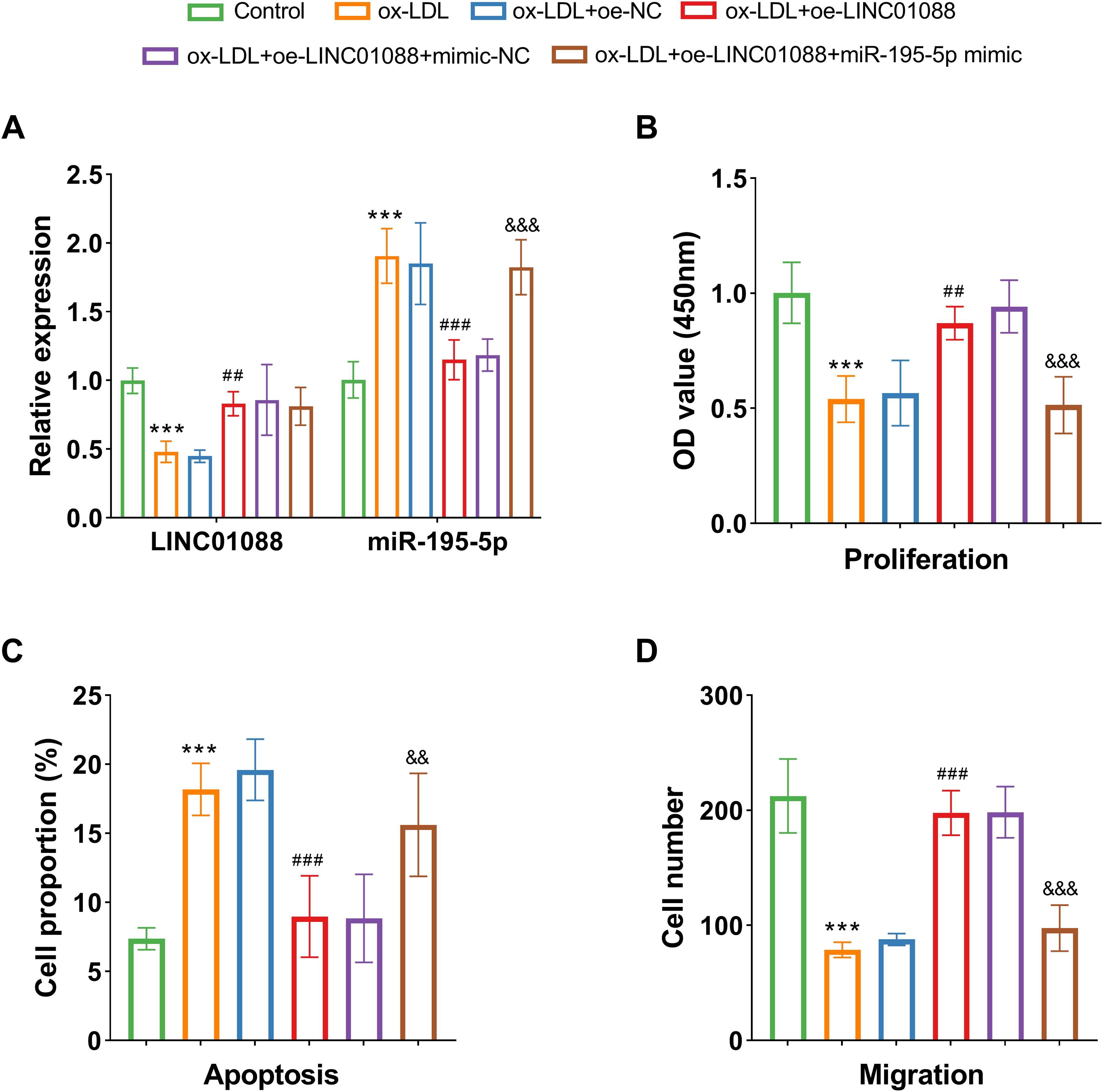
Up-regulated LINC01088 reversed the suppression of proliferation and migration and the promotion of apoptosis due to ox-LDL treatment in HAECs. (A) The levels of LINC01088 and miR-195-5p after co-transfection. (B-D) The levels of proliferation (B), apoptosis (C), and migration (D) after co-transfection under ox-LDL treatment in HAECs. Data are presented as the mean ± SD (n = 5). ***P < .001 versus Control,## P < .01, ### P < .001 versus ox-LDL + oe-LINC01088, ## P < .01, ### P < .001 versus ox-LDL, && P < .01, &&& P < .001 versus ox-LDL + oe-LINC01088. CAS, Carotid artery stenosis; HAECs, Human aortic endothelial cells; ox-LDL, oxidized low-density lipoprotein.
GO and KEGG Analysis
Using bioinformatics analysis, the downstream pathways were explored. A total of 1,419, 7,530, and 2764 target genes for miR-195-5p were identified from miRBD, miRWALK, and starBase, respectively, resulting in 593 overlapping target genes through the Venn diagram (Figure 4A). The overlapping genes were analyzed by KEGG and GO enrichment analysis. KEGG analysis enriched the pathways including the p53 signaling pathway, MAPK signaling pathway, focal adhesion, autophagy, and spinocerebellar ataxia (Figure 4B). GO enrichment analysis reveals that the focus is primarily on protein metabolic pathways. In the Biological Process category, enrichment is predominantly observed in diverse catabolic processes. Within the Cellular Component category, enrichment is mainly found in protease complexes. In the Molecular Function category, enrichment is prominently associated with protease activity (Figure 4C).

KEGG and go functional enrichment analysis of mir-195-5p target genes. (A) Screen the targeted genes of miR-195-5p from miRBD, miRWALK, and starBase. (B) KEGG enrichment pathways of targeted genes. (C) GO enrichment analysis of targeted genes. GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes.
Discussion
CAS primarily arises from plaque accumulation during carotid atherosclerosis, and the progression of these plaques can lead to cerebrovascular events, posing serious threats to patient health and life. 4 lncRNAs play significant roles in carotid atherosclerosis. 23 In the sequencing data analysis, LINC01088 emerged as the most downregulated lncRNA, indicating its potential as a candidate in CAS research, and its downregulation in serum was further verified. Correlation analysis with clinical indicators showed significant associations between LINC01088 and both HDL-C and SBP. HDL-C exerts protective effects against cardiovascular disease by slowing atherosclerotic plaque formation through antioxidant, anti-adhesion, and anti-thrombotic mechanisms. 24 Low HDL-C levels were reported to be associated with an increased risk of CAS. 25 Additionally, research indicates that stroke risk increases with rising SBP in symptomatic carotid atherosclerosis patients. 26 SBP was identified as a risk factor for cerebrovascular events in carotid atherosclerosis and stenosis patients through LASSO-Logistic regression analysis. 27 Medical imaging assessments of stenosis provide direct insights into vascular patency, 28 and LINC01088 was found to be closely associated with stenosis severity. Using ROC analysis, a method frequently applied to evaluate diagnostic performance, LINC01088 demonstrated strong accuracy in distinguishing CAS patients from healthy controls. Based on these findings, it was speculated that LINC01088 plays a critical role in CAS pathogenesis.
Subsequently, the potential role of LINC01088 in CAS was investigated. Ox-LDL, generated through the oxidation of LDL, is known for its contribution to atherosclerotic plaque formation and its tendency to increase plaque instability. 29 Ox-LDL is commonly used to simulate atherosclerosis in vitro studies.11,30 Vascular endothelial cells, located in the vascular intima and integral to the vascular wall, are in direct contact with blood. 31 Vascular endothelial cells are the primary protective barrier of the vascular intima. 7 Endothelial cell dysfunction is closely related to inflammation response, foam cell formation, and functional changes in smooth muscle cells, all of which promote plaque development on the vascular wall.31–33 In HAECs, exposed to ox-LDL, abnormal cellular activities were observed, including reduced cell proliferation and migration and increased apoptosis. However, upregulation of LINC01088 mitigated these effects, restoring cell proliferation, migration, and apoptosis to levels closer to those under control conditions. This indicates that LINC01088 exerts a protective effect on cells subjected to ox-LDL stimulation, highlighting its potential as a biomarker for CAS. The molecular mechanism of LINC01088 was further examined. miRNAs, which negatively regulate gene expression, significantly impact cerebrovascular diseases, with lncRNA-miRNA interactions serving as a common regulatory way in disease progression.17,33,34 For instance, LINC01088 regulates gastric cancer cell invasion and migration by sponging miR-95 15 and inhibits ovarian epithelial cell tumorigenesis by targeting miR-24-1-5p. 16 Database analysis identified miR-195-5p as a candidate target, and its expression was confirmed in serum further verifying that LINC01088 directly targets miR-195-5p. In cellular experiment, upregulation of miR-195-5p inhibited the protective impact of LINC01088 on HAEC activities, including proliferation, migration, and apoptosis. These findings suggest that LINC01088 may influence CAS progression by serving as the sponge of miR-195-5p.
The existence of miRNA binding sites across various RNA transcripts supports the increasingly recognized hypothesis of competing endogenous RNAs (ceRNAs).17,35 According to this concept, all RNA transcripts with miRNA binding sites can interact and regulate one another by competing for shared miRNAs. By functioning as ceRNAs, lncRNAs can mitigate miRNA-mediated repression of downstream target mRNAs.36–38 In this study, we utilized online prediction tools to identify mRNAs potentially targeted by miR-195-5p and conducted functional enrichment analysis on these targets. GO enrichment analysis revealed associations with protease activity, ubiquitin processes, and other functions related to protein degradation metabolism. KEGG enrichment analysis highlighted the p53 and MAPK signaling pathways, which are prominent research areas in various diseases. However, a limitation of this study is that we did not further validate the predicted mRNAs and signaling pathways. Furthermore, our investigation primarily focused on cell models, lacking in vivo verification using animal models. The findings from this study are based on in vitro experiments using HAECs, which are representative of endothelial cell behavior under pathological conditions. However, further studies involving animal models and clinical samples are necessary to confirm the generalizability of these results to human populations. Further research should focus on validating the molecular mechanism and potential therapeutic strategies in larger cohorts and exploring their long-term efficacy and safety. Additionally, investigating the molecular mechanisms underlying the interaction between LINC01088 and miR-195-5p in more detail could provide deeper insights into the pathogenesis of CAS.
Conclusion
In summary, this study demonstrated the diagnosis value of lncRNA LINC01088 and revealed that LINC01088 could affect the CAS progression by targeting the miR-195-5p, which could be a therapeutic target. LINC01088 reversed ox-LDL-induced proliferation, apoptosis, and migration in HAECs, suggesting LINC01088 may serve as a protective biomarker in CAS. This study provides novel insights into the progression of CAS and highlights the potential for developing new treatments for CAS
Supplemental Material
sj-xlsx-1-cat-10.1177_10760296251319281 - Supplemental material for LINC01088 Targets miR-195-5p to Promote Proliferation and Migration and Reduce Apoptosis in the Inhibition of Carotid Artery Stenosis
Supplemental material, sj-xlsx-1-cat-10.1177_10760296251319281 for LINC01088 Targets miR-195-5p to Promote Proliferation and Migration and Reduce Apoptosis in the Inhibition of Carotid Artery Stenosis by Huoquan Tang, Shuo Sun, Yali Zhang, Ying Jin, Caijiao Wang, Chunchun Xu, Yanfeng Zhang, Li Chen and Defeng Wu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Not Applicable.
Consent to Participate
The informed consent was signed by each participant.
Consent for Publication
Not applicable.
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
The Second Affiliated Hospital of Zhejiang Chinese Medicine University's Medical Ethics Committee approved this protocol and the study adhered to the Helsinki Declaration.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
