Abstract
Objectives
Multiple myeloma (MM) is a hematologic malignancy comprising approximately 10% of all blood cancers. Patients with MM are at risk for disseminated intravascular coagulation (DIC), a serious complication characterized by systemic coagulation activation, leading to microthrombi, organ dysfunction, and severe bleeding. This study aims to investigate the incidence of DIC among MM patients and identify risk factors associated with DIC development. We also sought to develop a predictive formula for assessing DIC risk.
Methods
A retrospective analysis was conducted on MM patients. Logistic regression analysis was used to identify factors significantly associated with DIC. The predictive power of the logistic regression model was evaluated using receiver operating characteristic (ROC) curve analysis.
Results
The incidence of DIC among hospitalized MM patients was 16.8%. Significant factors identified by logistic regression analysis included prothrombin time (PT), fibrin degradation products (FDP), and D-dimer levels. ROC curve analysis indicated that the predictive model had strong discriminatory power, with an area under the curve (AUC) of 0.927. A predictive formula for the probability of DIC occurrence was developed based on the logistic regression model.
Conclusions
The predictive formula developed in this study offers a tool for early identification of MM patients at high risk of DIC. While the model demonstrates strong predictive capability, further validation and refinement are required to improve its accuracy and clinical application.
Keywords
Introduction
Multiple myeloma (MM) is a significant hematologic malignancy characterized by the clonal proliferation of plasma cells within the bone marrow.1–3 Globally, MM accounts for approximately 1% of all cancers and about 10% of hematologic malignancies. In China, the incidence of MM has been rising, with recent studies indicating an annual incidence rate of approximately 1.15 per 100,000 person-years. 3 The prevalence of MM standardized by the China 2000 population census data was 5.81 per 100,000 population, with higher rates observed in males than in females1. The burden of MM in China has been increasing, reflecting both improved diagnostic capabilities and an aging population.4,5
The disease burden of MM in China is substantial, as the disease impacts both patient quality of life and healthcare resources. Patients with MM typically present with symptoms such as bone pain, anemia, renal insufficiency, and hypercalcemia, necessitating comprehensive management strategies. 6 The economic burden associated with MM treatment, including chemotherapy, stem cell transplantation, and supportive care, poses considerable challenges for the healthcare system. 7
Disseminated intravascular coagulation (DIC) is a severe complication that can arise in various clinical settings, including infection, trauma, and malignancies such as hematologic disorders. 8 DIC is characterized by systemic activation of the coagulation cascade, leading to microthrombi formation throughout the vasculature and resulting in multiple organ dysfunction due to ischemia and infarction. Concurrently, the consumption of platelets and coagulation factors can lead to severe bleeding complications.9,10 In hematologic malignancies, coagulation abnormalities are common, particularly in diseases such as acute promyelocytic leukemia (APL), where DIC is frequently observed. While MM is not associated with the same high incidence of DIC as APL, patients with MM are nonetheless at risk of developing coagulopathies, including hypofibrinolysis and DIC, due to disease progression, treatment-related factors, and comorbid conditions. 11 The underlying mechanisms involve the release of procoagulant factors by malignant cells and the effects of treatments that can exacerbate coagulopathy. 12
Coagulation abnormalities are common in patients with MM. The disease itself disrupts normal hemostasis due to the proliferation of malignant plasma cells. 12 Additionally, treatments for MM, including chemotherapy and novel agents like immunomodulatory drugs and proteasome inhibitors, can further contribute to coagulation dysfunction. DIC in MM patients is a critical concern, with studies reporting varying incidence rates. The presence of DIC at diagnosis or during the course of the disease is associated with a poor prognosis, significantly impacting patient morbidity and mortality. 13 The pathogenesis of coagulopathies, including DIC, in MM is multifactorial. Procoagulant activity is driven by systemic inflammation, endothelial damage, and the expression of tissue factor by malignant plasma cells, which can lead to both thrombotic and hemorrhagic complications.6,8,14
Predicting the occurrence of DIC and its outcomes in MM patients remains a clinical challenge. The diagnosis of DIC relies on a combination of clinical and laboratory criteria, including elevated levels of D-dimer and fibrin degradation products (FDP), prolonged prothrombin time (PT), and thrombocytopenia. Despite advancements in diagnostic methodologies, the management of DIC in MM patients is complicated by the dual risks of thrombosis and hemorrhage.14,15
Recent studies have focused on developing predictive models for MM patients.16–19 Logistic regression analysis and receiver operating characteristic (ROC) curve analysis are commonly used statistical methods to evaluate the association between various clinical parameters and the risk of developing DIC. Predictive models aim to facilitate early intervention and improve clinical outcomes by enabling targeted management strategies.10,20,21 Diagnosing DIC in MM is particularly challenging due to the overlapping features with MM-related bone marrow dysfunction and the multifactorial nature of coagulopathies. Existing DIC scoring systems, though valuable, do not fully account for the unique pathophysiological mechanisms in MM, including elevated monoclonal immunoglobulins, hyperviscosity, and endothelial damage. These limitations underscore the need for improved, MM-specific diagnostic tools for DIC. Current knowledge gaps include understanding how MM-specific factors contribute to DIC onset and progression, making the development of tailored diagnostic strategies critical for early identification and management.
The present study aimed to provide a comprehensive analysis of the incidence of DIC among MM patients as well as the risk factors, and predictive models for DIC in these patients. By examining a large cohort and employing robust statistical methodologies, we sought to enhance our understanding of the clinical characteristics and outcomes associated with DIC in this patient population, with the goal of ultimately improving the management and prognosis of MM patients at risk for this severe complication.
Materials and Methods
Study Design and Setting
This study was conducted as a retrospective cohort analysis at First Hospital of Shanxi Medical University aimed at investigating the incidence of, risk factors for, and a predictive model for DIC in patients with MM. The study included inpatients newly diagnosed with MM between August 2017 to December 2023.
A priori power analysis was conducted using G*Power software (version 3.1.9.7) to determine the required sample size for logistic regression analysis. Based on anticipated effect sizes from preliminary data, an alpha level of 0.05, and a desired power of 0.80, a minimum of 100 patients (including approximately 20 cases of DIC) were required to detect significant associations between predictor variables and the occurrence of DIC.
Participants
Patients were included in the study if they had a confirmed diagnosis of MM based on International Society of Thrombosis and Hemostasis (ISTH) overt DIC diagnostic criteria 22 and had complete medical records including baseline characteristics and laboratory parameters. The ISTH criteria were applied exclusively to patients presenting with clinical suspicion of DIC, including indicators such as excessive bleeding or relevant complications. Patients were categorized based on their diagnosis or complication type at admission, with distinctions made between initial diagnoses and treatment-related admissions. Exclusion criteria were incomplete medical records and diagnoses of other hematologic malignancies or conditions that could confound the diagnosis of DIC.
Data Collection
Data were collected on baseline characteristics and laboratory parameters for each patient. Baseline characteristics included age, sex, and medical history. Laboratory parameters comprised white blood cell (WBC) count, lymphocyte count, neutrophil count, platelet count, PT, activated partial thromboplastin time (APTT), and levels of hemoglobin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total bilirubin, total protein, albumin, calcium, phosphorus, lactate dehydrogenase (LDH), blood urea nitrogen (BUN), creatinine, FDP, D-dimer, and fibrinogen (FIB). Laboratory values were recorded at the time of hospital admission unless otherwise noted.
Statistical Analysis
Baseline characteristics and laboratory parameters were compared between the DIC group (n = 20) and the control group (n = 99) in the MM patient population. Continuous variables were expressed as either mean ± standard deviation (SD) for normally distributed data, or median with interquartile range (IQR) for non-normally distributed data. Comparison of continuous variables between groups was performed using the Welch two-sample t-test or the Wilcoxon rank sum test, as appropriate based on the distribution of the data. Categorical data were compared using Pearson's chi-squared test.
The sample size was calculated to ensure adequate power to detect significant predictors of DIC occurrence. Given the expected incidence rate and the number of predictor variables in the logistic regression model, the calculated sample size provides sufficient power to validate the predictive model.
Logistic regression analysis was employed to explore the association of various factors with the occurrence of DIC. The regression model adjusted for age, sex, and specific laboratory measures, and odds ratios (ORs) with 95% confidence intervals (CIs) were calculated to quantify the strength of the associations. The model's predictive power was evaluated using ROC curve analysis, with the area under the curve (AUC) value serving as a summary measure of the model's ability to discriminate between DIC occurrence and non-occurrence. An AUC value closer to 1 indicates excellent model performance, whereas a value closer to 0.5 suggests no discriminative ability.
The statistical analysis was conducted using Python 3.8, and all tests were two-tailed with a significance level set at p < 0.05.
Results
Baseline Characteristics
The baseline characteristics of patients with MM were compared between those who developed DIC (n = 20) and the control group (n = 99). The results are summarized in Table 1. Patients in the DIC group had a mean age of 67 ± 10 years, compared to 65 ± 10 years in the control group, with no significant difference observed (p = 0.410). The incidence rate of DIC among hospitalized MM patients was 16.8%, as depicted in Figure 1. This highlights that a significant proportion of MM patients are at risk of developing DIC.

Incidence rate of Disseminated Intravascular Coagulation (DIC) among Multiple Myeloma (MM) patients.
Baseline Characteristics.

Values are presented as Median with Interquartile Range (25th to 75th quantiles) unless specifically marked as mean ± SD. Statistical analyses were conducted accordingly.
Welch two-sample t-test.
Wilcoxon rank sum test.
Pearson's chi-squared test.
Laboratory Parameters
WBC count, lymphocyte count, and neutrophil count showed no significant differences between the DIC and control groups (p > 0.05 for all). However, significant differences were noted in the hemoglobin levels (90 ± 18 g/L vs 101 ± 25 g/L, p = 0.033), ALT levels (26 U/L [15, 33] vs 17 U/L [11, 23], p = 0.037), and AST levels (26 U/L [19, 44] vs 22 U/L [15, 27], p = 0.016). The platelet count was significantly lower in the DIC group (39 × 10^9/L [32, 43] vs 77 × 10^9/L [41, 99], p < 0.001). PT and APTT were significantly prolonged in the DIC group compared with the control group (PT: 18.25 s [15.00, 20.63] vs 12.70 s [11.95, 14.20], p < 0.001; APTT: 33 s [28, 39] vs 28 s [25, 32], p = 0.017). The FDP level was higher in the DIC group than in the control group (13 µg/mL [6, 35] vs 6 µg/mL [3, 11], p = 0.004), although no significant difference was observed in the D-dimer levels of the two groups (2.2 µg/mL [1.0, 4.7] vs 1.6 µg/mL [0.5, 3.3], p = 0.209).
Logistic Regression Analysis and Predictive Model Performance
Logistic regression analysis identified several factors significantly associated with the occurrence of DIC in MM patients (Table 2). PT, FDP level, and D-dimer level were significantly associated with DIC occurrence. PT had an OR of 1.957 (95% CI [1.460, 2.623], p = 6.99e-06); FDP had an OR of 1.187 (95% CI [1.056, 1.335], p = 0.004); and D-dimer had an OR of 0.710 (95% CI [0.542, 0.929], p = 0.012).
Multivariate Logistic Regression Analysis.
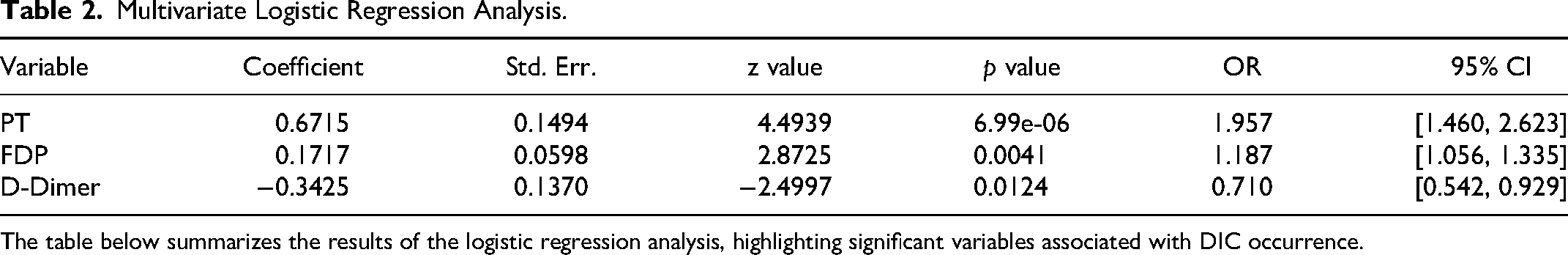
The table below summarizes the results of the logistic regression analysis, highlighting significant variables associated with DIC occurrence.
The predictive power of the logistic regression model was evaluated by ROC curve analysis (Figure 2). The AUC for the model was 0.927, indicating a high discriminative ability for predicting the occurrence of DIC among MM patients.

Receiver Operating Characteristic (ROC) Curve. The ROC curve below demonstrates the model's ability to distinguish between DIC occurrence and non-occurrence. The area under the curve (AUC) provides a measure of the model's overall performance.
Predictive Formula
The coefficients obtained from the logistic regression model were used to construct a predictive formula for the probability of DIC occurrence. The logit transformation was applied to express the probability in a logistic function format. Based on the logistic regression analysis, the predictive formula for the occurrence of DIC in MM patients is: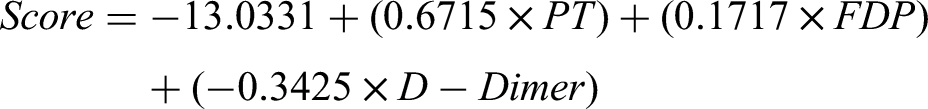
To facilitate clinical decision-making, we determined an optimal cutoff probability value using Youden's index derived from the ROC curve analysis. A cutoff score of
Discussion
This study has provided a comprehensive analysis of the incidence of risk factors for DIC in MM patients as well as developed a predictive model for DIC in these patients. We found that DIC is a significant complication in hospitalized MM, with an incidence rate of 16.8%. Our predictive model demonstrated good discriminative ability, indicating that while useful, further refinements are necessary to improve its accuracy.
Our findings align with the general trend observed in previous studies, which show an incidence of nearly 20% for DIC in MM patients. 23 The Global Burden of Disease Study reported a continuous increase in the MM incidence in China from 2006 to 2016, reflecting the rising disease burden observed in our study.3,24 This is consistent with global data indicating an increasing MM incidence, especially in developed countries due to better diagnostic capabilities and an aging population.
While PT, FDP, and D-dimer are well-established predictors of DIC and are included in standard scoring systems, our predictive model offers added value by tailoring these factors specifically to multiple myeloma patients. MM-specific pathophysiological processes, such as endothelial dysfunction and monoclonal protein dysregulation, necessitate a more targeted approach to DIC prediction. Although FDP assays are not universally available, their inclusion in high-risk settings could improve the sensitivity of DIC detection in MM patients. Ongoing research is focused on identifying alternative biomarkers that may enhance the model's applicability in various clinical environments.
Our study revealed a significant 16.8% incidence of DIC in hospitalized MM patients and identified key risk factors such as PT, FDP level, and D-dimer level. These findings are consistent with previous research showing that coagulation abnormalities exacerbate the risk of DIC in MM patients. 25 The logistic regression model, demonstrated good predictive ability with an AUC of 0.927, and while it can offer a useful tool, there is a need for further refinement to enhance its accuracy.26,27
Previous research has also highlighted the significant impact of DIC in hematologic malignancies. Similar to our results, other studies have shown that DIC exacerbates both thrombotic and hemorrhagic events, complicating clinical management. The pathogenesis involving the release of procoagulant factors by malignant cells and systemic inflammation has been well-documented.11,23 Systemic inflammation can lead to bleeding by causing endothelial damage and disrupting the balance of coagulation and fibrinolysis, which exacerbates hemorrhagic complications in conditions like sepsis-induced DIC. 28 Although the incidence of DIC in MM is not as high as in other hematologic malignancies such as APL, MM patients remain at risk of developing coagulopathies, including DIC, particularly in the advanced stages of the disease or when undergoing treatments that impact endothelial function and coagulation pathways.
The predictive formula developed in this study can be integrated into clinical workflows to identify MM patients at elevated risk for DIC. Clinicians can input relevant laboratory parameters (PT, FDP, D-Dimer) into the formula to calculate a risk score. Utilizing the established cutoff probability value of
This study has several limitations. Firstly, the retrospective design might introduce selection bias, as only patients with complete medical records were included. This limitation could mean some cases with incomplete data were missed, affecting the generalizability of the findings. Furthermore, the study was conducted at a single center, which may limit the applicability of the results to patient populations with different demographic characteristics and to regions with different healthcare practices. The predictive model, although it showed good accuracy, requires further validation in larger, multicentric cohorts to enhance its predictive power. Additionally, relying solely on standard laboratory parameters might not fully capture the complexity of DIC pathogenesis, suggesting a need for incorporating novel biomarkers in future models.
Future research should focus on multicenter prospective studies to validate the predictive model and ensure its applicability across diverse clinical settings. Incorporation of advanced biomarkers and molecular profiling could provide a more nuanced understanding of DIC in MM, potentially improving predictive accuracy and patient outcomes. Future models may benefit from incorporating MM-specific biomarkers or predictors beyond the ISTH criteria to improve diagnostic specificity.
Exploring the impact of newer therapeutic agents on the incidence and management of DIC in MM patients is also essential. With the advent of novel immunotherapies and targeted treatments, understanding their influence on coagulation pathways will be crucial for optimizing treatment protocols and mitigating DIC risks.
Conclusion
In conclusion, this study underscores the significant incidence and impact of DIC in MM patients, highlighting critical risk factors and providing a predictive model for early identification of MM patients at increased risk. While our findings contribute valuable insights into the clinical management of MM, ongoing research and refinement of predictive tools are essential for improving patient outcomes. The predictive formula facilitates early identification and stratification of MM patients at high risk for DIC, thereby enabling timely interventions and personalized management strategies to improve patient outcomes. By addressing the identified limitations and advancing our understanding of DIC pathogenesis, we can better tailor interventions and enhance the prognosis for MM patients at risk of this severe complication.
Footnotes
Author Contributions
All authors contributed to the concept and design of the study. R.B., M.F., and M.H. wrote the study protocol and coordinated the research. R.B. performed all statistical analyses and prepared the initial draft of the manuscript. R.B., M.F., and M.H. wrote the first subsequent iterations. L.L. and H. provided critical feedback on ensuing drafts to refine the final manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
