Abstract
Disseminated intravascular coagulation (DIC) in patients with sepsis represents a critical condition. Thus, a simple and rapid diagnosis is required. The purpose of this study was to compare the performances of a recently developed Sepsis-Induced Coagulopathy (SIC) with the Japanese Association for Acute Medicine (JAAM) DIC. Four hundred nine patients with sepsis having coagulopathy and antithrombin activity of less than 70% and treated with antithrombin were retrospectively analyzed, and the SIC and JAAM-DIC criteria on days 1 (before treatment), 2, 4, and 7 were compared. The prevalence of JAAM-DIC on day 1 was significantly higher than that of SIC (91.4% vs 81.8%, P = .003), but there were no differences on days 2, 4, and 7. The mortality rates in the SIC and JAAM-DIC groups were both 23.3%. The specificity to 28-day mortality on day 1 was higher in the SIC group (15.8% vs 9.2%, P = .013). There were no differences in sensitivity on days 1, 2, 4, and 7. Mortality was significantly different between SIC-positive and SIC-negative groups on days 2, 4, and 7 (P < .01, respectively), while significant differences were seen between JAAM-DIC-positive and JAAM-DIC-negative groups only on days 4 and 7 (P < .05, .01, respectively). In summary, the SIC characteristics were similar to the JAAM-DIC group, and the classifications were comparable in terms of mortality prediction. The SIC scoring system is simple, easy to use, and adaptable to the new sepsis definitions and offers an important approach to evaluating patients in emergency and critical care settings.
Keywords
Introduction
Disseminated intravascular coagulation (DIC) and shock are the 2 major factors that lead to poor outcomes in sepsis. 1 A recent nationwide survey in Japan reported that in patients with sepsis in the intensive care unit, approximately one-third of patients exhibited shock and more than half exhibited DIC. 2 Anticoagulant therapy for DIC has been extensively studied in Japan, and recent studies have repeatedly shown that anticoagulant therapy is only effective in patients with severe coagulopathy. 3,4 Furthermore, subgroup analyses in the anticoagulant studies in sepsis including the KyberSept and PROWESS trials, a recombinant human activated protein C worldwide evaluation in severe sepsis study, reported a trend toward a greater risk reduction in mortality among patients with DIC than among patients without DIC. 5,6 Therefore, the selection of appropriate patients is important. Interestingly, the beneficial effects of anticoagulant therapy have been shown to correlate with the severity of the coagulopathy. 7 Since the release of the Japanese Society for Acute Medicine (JAAM) DIC diagnostic criteria, 8 studies examining the effect of anticoagulants in DIC have adopted the JAAM-DIC criteria, and many of them have reported beneficial effects. 1,9 Therefore, the JAAM-DIC criteria may recognize appropriate candidates for anticoagulant therapy. However, since the definition of sepsis was revised in 2016, 10 a need to update the DIC diagnostic criteria has been suggested. 11 One of the 4 components of the JAAM-DIC criteria is the Systemic Inflammatory Response Syndrome (SIRS) score, which is no longer included in the new sepsis definition and has been replaced by the Sequential Organ Failure Assessment (SOFA) score. 10
Sepsis-induced coagulopathy (SIC) was proposed in 2017 by the active members of International Society for Thrombosis and Haemostasis (ISTH) to categorize patients with “sepsis and coagulation disorders” and was designed to fit the new sepsis definition. 12 Sepsis-induced coagulopathy is not the first classification to use only 2 coagulation markers, that is, platelet count and the prothrombin time (PT)–international normalization ratio (INR). Kinasewitz et al 13 previously introduced a simple diagnostic system composed of these 2 classical hemostatic markers based on observations in a primate model of sepsis. 14 The SIC diagnostic criteria are simpler than JAAM-DIC diagnostic criteria and are composed of 3 items: platelet count, PT-INR, and the SOFA score. The SIC diagnostic criteria do not include SIRS item according to the new sepsis definition. 10 The SOFA score was included to confirm the presence of sepsis, not to reflect its severity; therefore, the score was regarded as 2 when the SOFA score was 2 or more. If the SIC criteria can identify a similar category of patients as the JAAM-DIC criteria, it would be more preferable because the SIC is composed of fewer items and that might be helpful to prevent the physicians to overlook the sepsis-associated DIC. Thus, the purpose of this study was to compare the prevalence and mortality of patients identified using the SIC or JAAM-DIC criteria. Also, the performances of both criteria were compared in terms of the prediction of mortality.
Patients and Methods
Data Collection
The data for this study were obtained from a multi-institutional postmarketing surveillance performed by Nihon Pharmaceutical Co Ltd (Tokyo, Japan). A total of 409 patients with sepsis-associated coagulopathy with an antithrombin activity of less than 70% and treated with antithrombin between April 2015 and May 2016 were enrolled. Each patient received 30 to 60 IU/kg/d of antithrombin concentrate (Nihon Pharmaceutical Co Ltd) for up to 3 consecutive days unless the patient died or treatment was stopped for any justifiable reason.
Laboratory Measurements and Diagnostic Criteria
The platelet count, fibrin/fibrinogen degradation products (FDPs, or
SIC and JAAM-DIC Criteria.a

Abbreviations: JAAM-DIC, Japanese Association for Acute Medicine Disseminated intravascular coagulation; SIC, Sepsis-Induced Coagulopathy; SIRS, systemic inflammatory response syndrome; SOFA, Sequential Organ Failure Assessment.
aSIC: Total SIC score is 4 or more with the sum of SOFA score and coagulation criteria exceeding 2. Total SOFA is the sum the 4 items (respiratory SOFA, cardiovascular SOFA, hepatic SOFA, and renal SOFA). JAAM-DIC: total score is 4 or more.
Ethics, Patient Consent, Study Permissions, and Consent to Publish
The postmarketing surveillance was performed under the supervision of the Japanese Ministry of Labour and Welfare and was conducted in accordance with the Declaration of Helsinki and Good Vigilance Practice and Good Post-Marketing Study Practice. Since the complete anonymization of personal data was performed upon data collection and the identification of individual patients was impossible, the ethical committee of Juntendo University waived the need to obtain informed consent and the patients’ agreement. For the same reason, the institutional committee judged that consent to publish was not required.
Statistical Analysis
The numerical values in the text and tables represent the median and interquartile range. Univariate associations were evaluated using the McNemar test, Fisher exact test, or unpaired Wilcoxon signed-rank test, as appropriate. The sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) for 28-day mortality were calculated at baseline (day 1) and on days 2, 4, and 7. A P value <.05 was considered to denote statistical significance. All the analyses were performed using SPSS 22.0 for Windows (IBM SPSS Inc, Chicago, Illinois).
Results
Study Population
Of the 409 patients included, 316 patients were alive at 28 days (28-day mortality, 22.7%). Table 2 shows the baseline characteristics of the patients. The median age of the survivors was 74 (67-82) years, compared with 80 (68-87) years for the nonsurvivors (P < .001). The ratio of male gender was significantly higher in the nonsurvivor group (P = .021). As for the coagulation profile at baseline, the platelet count (P = .708), FDP (P = .279),
Baseline Characteristics of the Patients.a
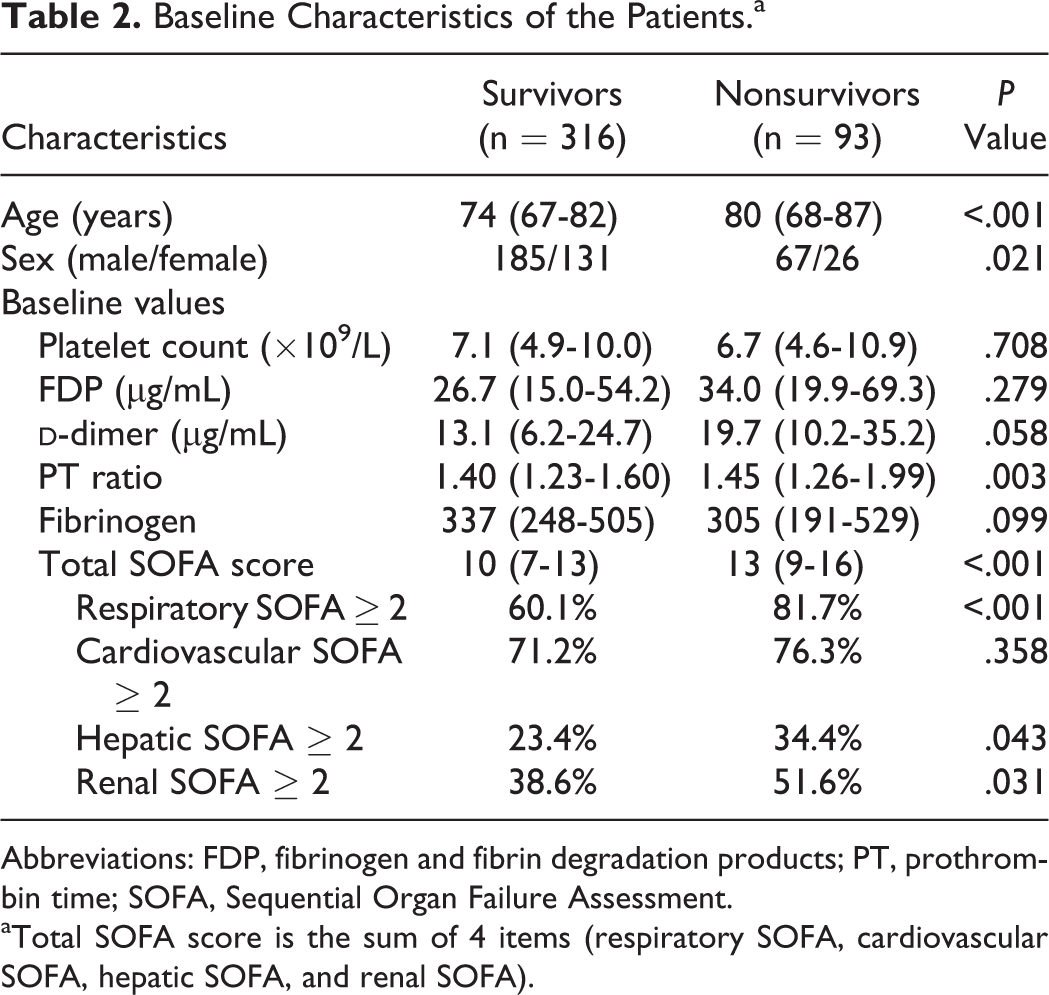
Abbreviations: FDP, fibrinogen and fibrin degradation products; PT, prothrombin time; SOFA, Sequential Organ Failure Assessment.
aTotal SOFA score is the sum of 4 items (respiratory SOFA, cardiovascular SOFA, hepatic SOFA, and renal SOFA).
A comparison of the performances such as sensitivity, specificity, PPV, and NPV for 28-day mortality between SIC and JAAM-DIC is shown in Table 3. Although the specificity on day 1 was higher for SIC (15.8% vs 9.2%, P = .013), no other statistical differences were seen for any of the items between the SIC and JAAM-DIC criteria.
Comparison of the Performances for Predicting 28-Day Mortality of SIC and JAAM-DIC.
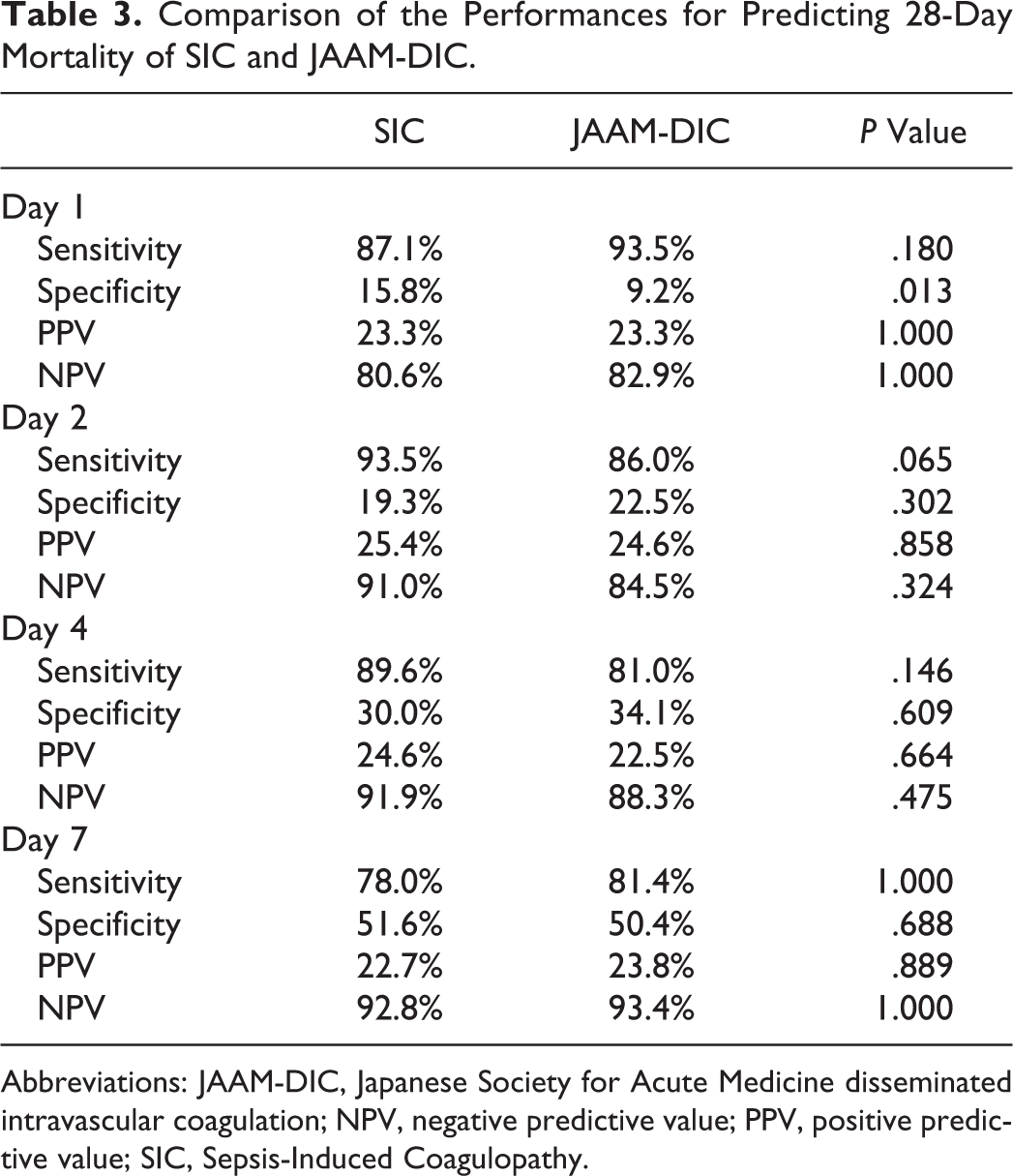
Abbreviations: JAAM-DIC, Japanese Society for Acute Medicine disseminated intravascular coagulation; NPV, negative predictive value; PPV, positive predictive value; SIC, Sepsis-Induced Coagulopathy.
The 28-day mortality rate for patients with SIC at baseline was 23.3%, which was identical to that of JAAM-DIC on day 1 (Table 4). The mortality rate among the SIC-positive patients was significantly higher than that among the patients who were negative for SIC on day 2 (25.4% vs 9.0%, P = .002). For JAAM-DIC, the difference in mortality was not statistically significant on day 2 (24.6% vs 15.5%, P = .081). The mortality differences were significant between patients with SIC and those without SIC and between patients with JAAM-DIC and those without JAAM-DIC on days 4 and 7.
Comparison of Mortality in SIC and Mortality in JAAM-DIC.
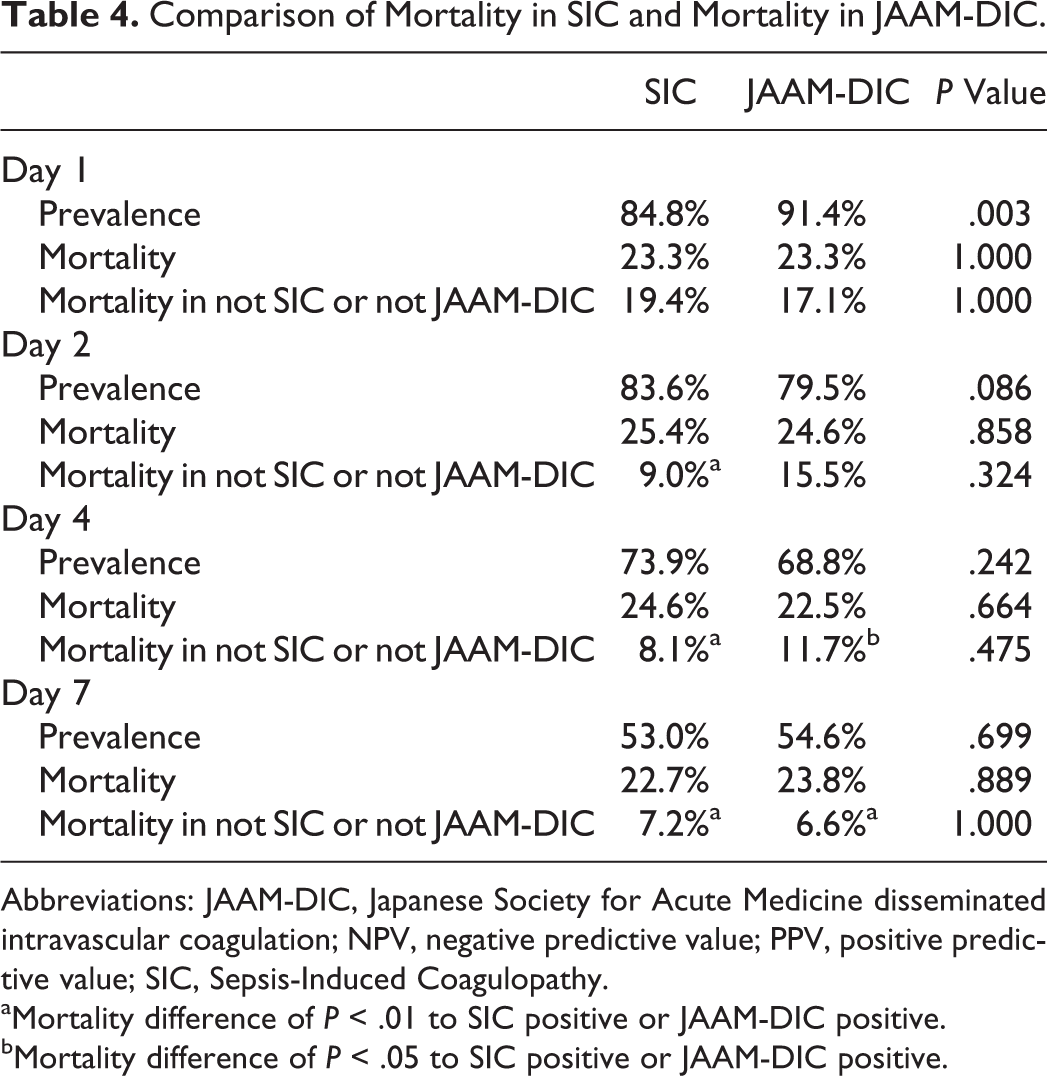
Abbreviations: JAAM-DIC, Japanese Society for Acute Medicine disseminated intravascular coagulation; NPV, negative predictive value; PPV, positive predictive value; SIC, Sepsis-Induced Coagulopathy.
aMortality difference of P < .01 to SIC positive or JAAM-DIC positive.
bMortality difference of P < .05 to SIC positive or JAAM-DIC positive.
The number of patients diagnosed as having SIC on day 1 was 347, which was similar to the number of patients diagnosed as having JAAM-DIC (n = 374). Overall, 92.5% of the SIC patients overlapped with the JAAM-DIC patients, while 85.8% of the JAAM-DIC patients overlapped with the SIC patients, on day 1. The numbers of patients were similar for both sets of criteria, and the mortalities of the patients with SIC but without JAAM-DIC tended to be higher than those with JAAM-DIC but without SIC on days 2, 4, and 7 (Figure 1).
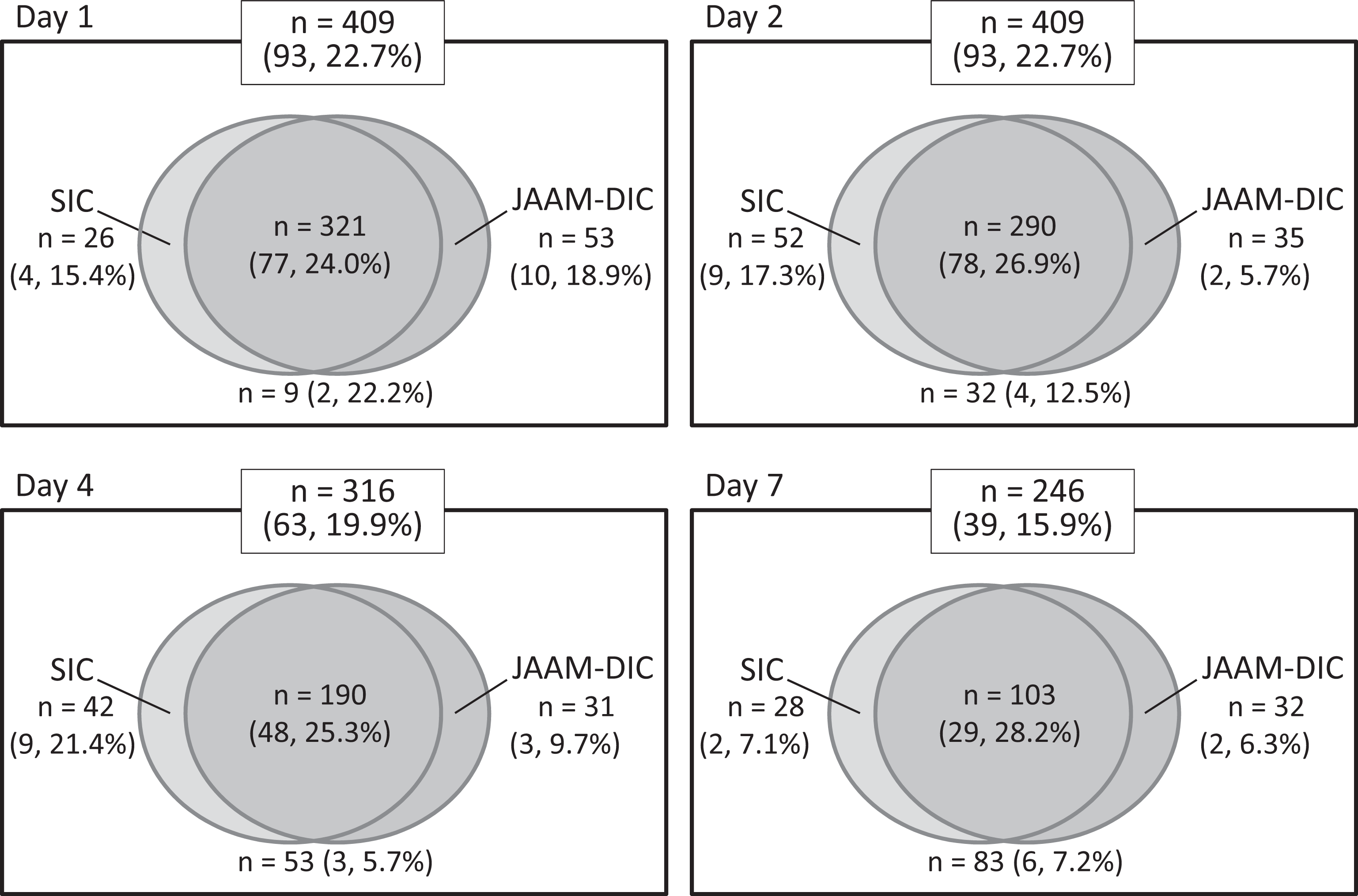
Distribution and mortality of patients according to the SIC and JAAM-DIC diagnostic criteria. Each figure shows a comparison of the SIC and JAAM-DIC criteria. The numbers represent the number of cases in each category, and the numbers in parentheses are for nonsurvivors and the mortality rate. DIC indicates disseminated intravascular coagulation; JAAM, Japanese Association for Acute Medicine; SIC, sepsis-induced coagulopathy.
Figure 2 shows the relationships between SIC and JAAM-DIC and the mortality rates. The mortality rate was 17.4% for an SIC score of 4 at baseline, and it gradually increased as the score increased, reaching a maximum of 27.3% for a score of 6. For the JAAM-DIC, the mortality rate was 24.7% for a score of 4 but it decreased for scores of 5 and 6 points and then increased again for scores of 7 points. In the case of SIC, the mortality rate seemed to increase along with an increase in the score on days 2, 4, and 7. In contrast, the relationships were not linear for JAAM-DIC (Figure 2 and 3).
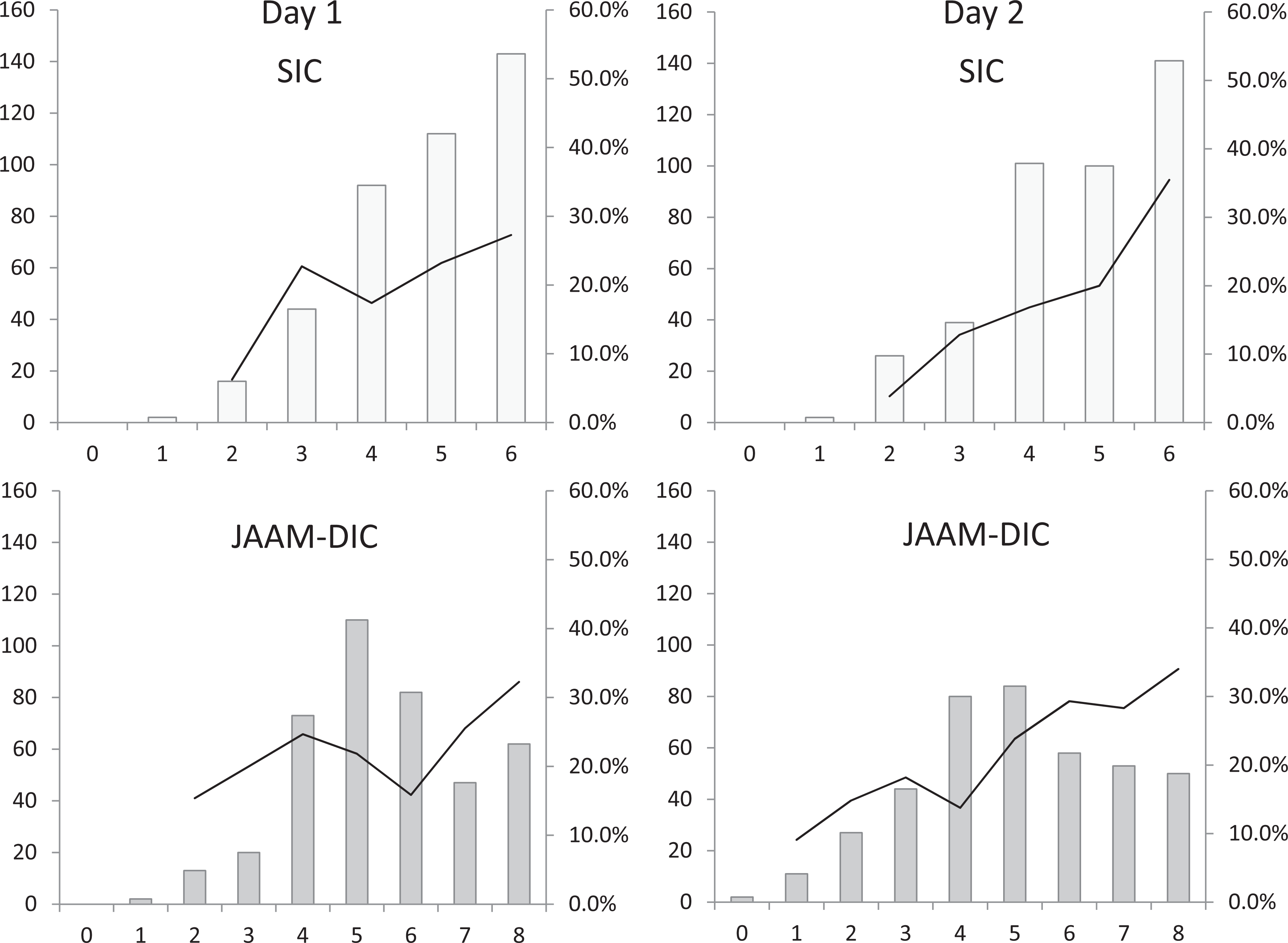
Patient numbers and mortality rates according to the SIC and JAAM-DIC score classifications. The patient distributions (bars) and the mortality rates (lines) are plotted according to the SIC scores (above) and the JAAM-DIC scores (below). The x-axis represents the score. The mortality rate increased as the SIC score increased except for score 3 on day 1. For the JAAM-DIC criteria, the mortality rate was higher for score 4 than for scores 5 or 6 on day 1. DIC indicates disseminated intravascular coagulation; JAAM, Japanese Association for Acute Medicine; SIC, sepsis-induced coagulopathy.
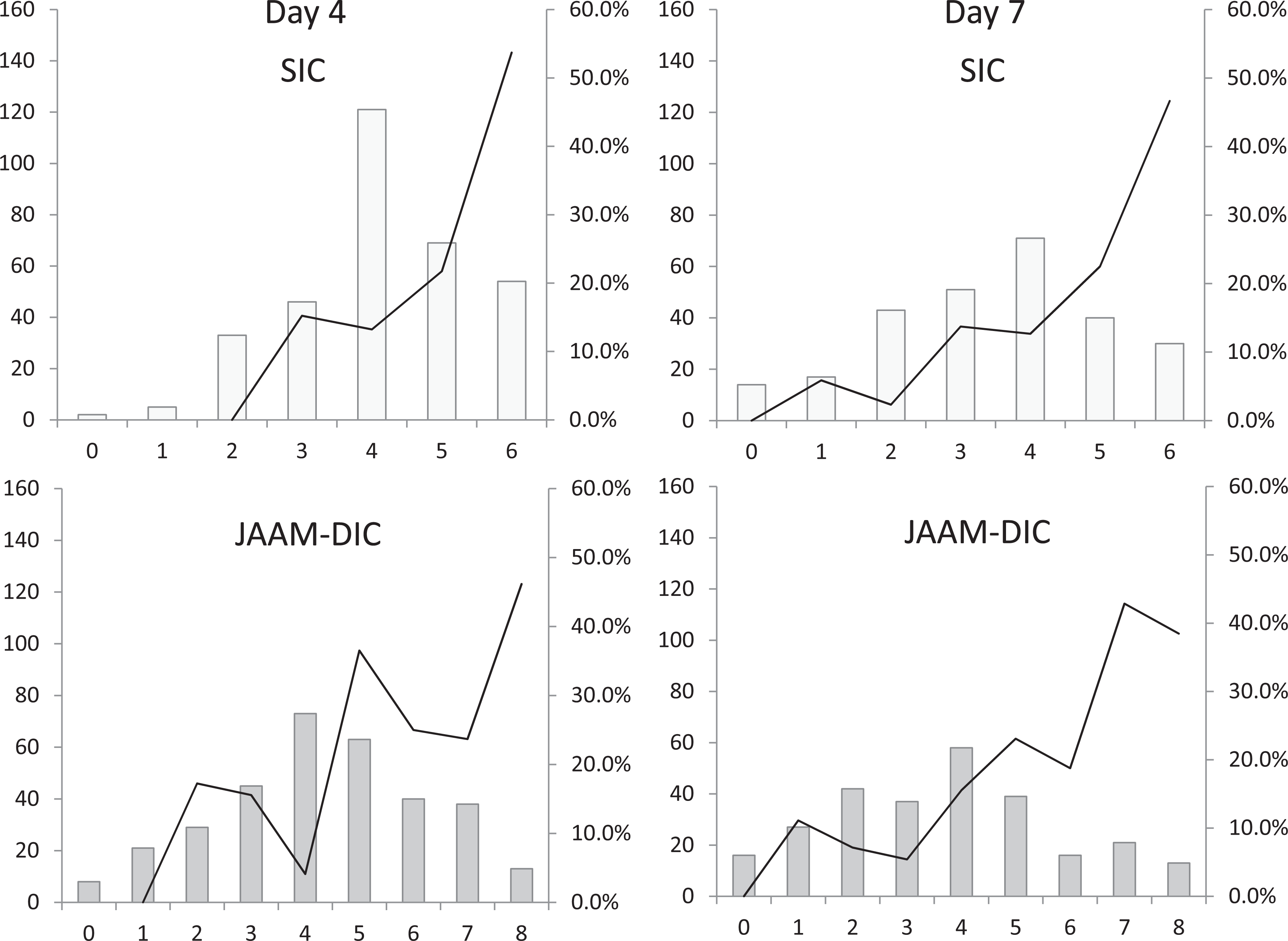
Patient numbers and mortality rates according to the SIC and JAAM-DIC score classifications.
Discussion
Disseminated intravascular coagulation is one of the most common but severe complications of sepsis, 15,16 and the ensuing coagulopathy plays a pivotal role in the development of organ dysfunction. Therapy of DIC with anticoagulants targets both the coagulation disorder and also the organ dysfunction that ensues. 17,18 However, patients who are likely to benefit from anticoagulant therapy must be identified. The JAAM-DIC has been shown to be an appropriate method to evaluate for anticoagulant therapies, 4,9 and antithrombin and recombinant thrombomodulin are recommended by the Japanese guidelines for the management of DIC. 19,20 However, the SIRS score is one of the 4 components of the JAAM-DIC criteria, and with the revised sepsis criteria, new DIC criteria are needed. 11 Sepsis-induced coagulopathy is one such possible candidate; therefore, the purpose of the present study was to examine the characteristics and the performance of the SIC criteria by comparing them with those of the JAAM-DIC criteria.
The incidence of DIC changes depending on the diagnostic criteria. It increases with the less specific criteria and decreases with the strict criteria. For example, the JAAM-DIC criteria were designed for diagnosing DIC at a relatively early stage of coagulopathy, and the incidence was more than twice that reported for the ISTH overt-DIC criteria. 21 In the present study, the mortality and the incidence of SIC were similar to those of JAAM-DIC, and this result was consistent with a previous study. 12 To establish the SIC criteria, clinical data were obtained from a nationwide survey for recombinant thrombomodulin, and similar results were obtained in the present study despite the difference in anticoagulants and the newer data set. Thus, we believe that the SIC criteria identify a patient population similar to that identified by the JAAM-DIC criteria.
The role and potential efficacy of anticoagulants for the treatment of sepsis remains a subject of significant debate. 22,23 This may be due to analyses reporting the effects are dependent on disease severity. 24 Tagami et al examined the effect of antithrombin administration in peritonitis-associated DIC using data from the Japanese Diagnosis Procedure Combination and reported a significant reduction in mortality (27.6% vs 19.9%). 25 Based on this report, we hypothesized that DIC would be an appropriate target, with an estimated mortality rate of between 20% and 30%. Our current study found that both the SIC and the JAAM-DIC could identify patients with this degree of severity.
In the present study, we also found that the performance of SIC was comparable with that of JAAM-DIC in terms of predicting 28-day mortality; however, the differences in mortality between the positive and negative groups were more evident for the SIC criteria. Both the SIC and the JAAM-DIC criteria utilize a scoring system, and the SIC score appears to reflect disease severity more linearly than the JAAM-DIC. This is likely due to the JAAM-DIC criteria that include fibrin-related markers, which did not differ significantly between survivors and nonsurvivors. This modification was possible because SIC targeted only the coagulation disorder in patients with sepsis. The coagulopathy secondary to infection is characterized by the excessive suppression of fibrinolysis arising from the overproduction of plasminogen activator inhibitor-1, and therefore, fibrin-related marker could be eliminated. 26 However, DIC was defined as “an acquired syndrome characterized by the intravascular activation of coagulation with loss of localization arising from different causes” and the ISTH overt-DIC diagnostic criteria (Supplemental 1) were released according to this definition. 21 Since SIC is the specific category for sepsis, we have to keep that difference in our mind. In addition, the SIRS score did not differ significantly between survivors and nonsurvivors in the present study. The SIC criteria did not include this parameter and showed a better linearity consisting of only 2 readily available laboratory parameters.
In summary, DIC may be underdiagnosed in emergency and critical care settings. The SIC diagnostic criteria were specifically established to identify SIC using a more simplified approach. Our present study suggests that SIC could help further improve diagnostic criteria for sepsis-associated DIC. In particular, since the SIC criteria can identify patients with a similar severity, it may be useful for improving the identification of appropriate candidates for anticoagulant therapy.
Limitations
The present study has several limitations. First, since the data set was obtained from a postmarketing survey for antithrombin, a selection bias likely exists. The patients diagnosed as having sepsis with DIC were enrolled, but SIC without DIC might not be enrolled. Thus, the predictive performance should also be examined in those patients. Second, all the patients were treated with antithrombin, which might have affected the outcome. We have to remind that all patients examined in this study had decreased antithrombin activity, and therefore, SIC score was SIC and antithrombin activity of less than 70% and that JAAM DIC was JAAM DIC score and antithrombin activity of less than 70%. The diagnostic performance of SIC should also be examined in patients not receiving anticoagulants. However, anticoagulant therapy is commonly applied in Japan and the present study has also been done in the patients treated with antithrombin. Third, since the present study was a retrospective analysis, the usefulness of SIC remains to be determined in a future prospective study.
Conclusion
The severity of SIC was comparable to that of JAAM-DIC. The SIC scoring system has a similar performance for the prediction of 28-day mortality, compared to the JAAM-DIC scoring system. The SIC is simple, includes revised sepsis definitions, and can be readily used in emergency and critical care settings. Additional prospective studies of this scoring system are warranted to further define its potential utility.
Supplemental Material
Supplemental Material, Suppl_1 - Sepsis-Induced Coagulopathy and Japanese Association for Acute Medicine DIC in Coagulopathic Patients with Decreased Antithrombin and Treated by Antithrombin
Supplemental Material, Suppl_1 for Sepsis-Induced Coagulopathy and Japanese Association for Acute Medicine DIC in Coagulopathic Patients with Decreased Antithrombin and Treated by Antithrombin by Toshiaki Iba, Makoto Arakawa, Jerrold H. Levy, Kazuma Yamakawa, Hiroyuki Koami, Toru Hifumi, and Koichi Sato in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
This work was performed using data from a postmarketing surveillance conducted by Nihon Pharmaceutical Co Ltd. Our institution does not require ethical approval for reporting individual cases or case series. Informed consent for patient information to be published in this article was not obtained because the complete anonymization of personal data was performed upon data collection and the identification of individual patients was impossible.
Acknowledgments
The authors thank K. Kinoshita for the operation of the infrastructure that was used to collect the data. The authors also thank all the institutes that cooperated with this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.A. is an employee of Nihon Pharmaceutical Co Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Ministry of Education, Culture, Sports, Science and Technology-Supported Program for the Strategic Research Foundation at Private Universities 2017.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
