Abstract
Percutaneous valve implantation or surgical replacement with mechanical or biological valves are standard therapies for severe valvular heart diseases. Prosthetic valve thrombosis, though rare, is a serious complication, particularly with mechanical prostheses. This study aimed to investigate the predictive value of platelet volume parameters, including mean platelet volume (MPV), platelet distribution width (PDW), and platelet-large cell ratio (P-LCR), for valvular thrombosis risk in patients undergoing valve replacement therapy. A retrospective cross-sectional study was conducted from May 2002 to May 2020, involving 108 patients with a history of mitral or aortic valve replacement and valvular thrombosis, and 216 controls with a history of valve surgery without valvular malfunction. PDW was significantly associated with an increased risk of thrombosis after adjusting for confounders, while MPV showed a clinical difference but did not reach statistical significance. P-LCR did not exhibit a significant association. These findings suggest PDW as a potential predictor of valvular thrombosis in such patients. The ease of measuring platelet volume parameters suggests their utility in routine hematological analysis for identifying patients at higher risk of valvular thrombosis post-replacement surgery. Further studies are warranted to validate these findings and explore additional laboratory markers, such inflammatory markers, for thrombotic risk assessment in this population.
Keywords
Introduction
Percutaneous valve implantation or surgical replacement with mechanical or biological valves are the gold standard and well-established therapies for most patients with severe valvular heart diseases. 1 Prosthetic valve thrombosis is a rare but serious cardiovascular complication following valve replacement, most often encountered with mechanical prostheses. The incidence of Obstructive prosthetic valve thrombosis (OPVT) for mechanical valves was estimated to be between 0.3% to 1.3% per patient year. 2 Thromboembolic events such as systemic emboli are more frequent, with incidence varying between 0.7% to 6%. 3 However, non-OPVT within the postoperative period is relatively frequent, with an incidence of 10%. 3 It has been shown that the overall incidence of clinical valve thrombosis in patients who underwent transcatheter aortic valve replacement was 2.8%. 4 Valvular thrombosis could be manifested by the development of dyspnea or worsening functional class, as well as systemic consequences such as stroke. Additionally, serum NT-proBNP levels could be elevated in these patients. 4 However, in cases with non-obstructive thrombosis, no specific symptoms may be observed. 1 Small non-obstructive thrombi highlight the importance of optimal anticoagulation therapy in the postoperative period.
The process of valvular thrombosis is not completely understood. Thrombosis begins with the aggregation of platelets, erythrocytes, and fibrin. Platelets secrete proinflammatory molecules with prothrombotic activity, contributing to the development of pathological valvular thrombosis. 5 The accumulation of platelets relies on the presence of platelet-bound von Willebrand factor (vWF) and fibrinogen. These components facilitate the cross-linking of adjacent platelets, ensuring stable platelet aggregation. This process is crucial for the formation of a primary hemostatic plug, known as a white thrombus, and is also a key factor in the development of pathological thrombi. Importantly, as the thrombus develops, platelets undergo further activation, transitioning from a pro-aggregatory to a pro-coagulant state. This shift allows for the assembly of coagulation complexes on the platelet surface, which are critical for the production of thrombin and fibrin. These processes promote secondary hemostasis, transforming the white thrombus into a red clot that is reinforced by a network of fibrin and trapped red blood cells (RBCs). 6
Mean platelet volume (MPV), platelet distribution width (PDW), and platelet-large cell ratio (P-LCR) are parameters of platelet volume that can be routinely determined in laboratories.7,8 It has been shown that the combined use of PDW and MPV could predict the activation of coagulation more efficiently. 9
It has been suggested that large platelets are metabolically more active than small platelets, and larger ones produce more β-thromboglobulin and thromboxane A2, associated with increased platelet activity, including upregulation of adhesion molecules and increased platelet aggregation.10,11 However, no study has evaluated the efficacy and value of platelet volume parameters, including MPV, PDW, and P-LCR, for the prediction of valvular thrombosis in patients undergoing valve replacement therapy. In this study, we investigated the value of MPV, PDW, and P-LCR in patients with a history of aortic and mitral valve replacement. Our study offers valuable insights into the interplay between a history of mitral or aortic valve replacement and the risk of valvular thrombosis, shedding light on potential factors influencing platelet-induced thrombosis. By elucidating these relationships, we pave the way for improved risk stratification and targeted interventions to mitigate thrombotic complications in patients with valve replacements.
Methods
Design and Setting
This retrospective cross-sectional study was conducted at a tertiary care hospital from May 2002 to May 2020 at a tertiary care hospital. The study received approval from the Ethics Committee of Tehran University of Medical Sciences (ethical code: IR.TUMS.THC.REC.1399.021). Before enrollment, written informed consent was obtained from all study participants.
Patient Characteristics
During the study period, patients with acute mitral and/or aortic valve malfunction were screened for inclusion and exclusion criteria. Inclusion criteria comprised (1) age ≥ 18 years old, (2) a history of mitral and aortic valve replacement or both, (3) mitral and/or valvular thrombosis, (4) normal complete blood count (CBC) three months after valve replacement, (5) normal echocardiography six months after valve replacement, and (6) ejection fraction > 45% in echocardiography. Exclusion criteria included (1) moderate or severe paravalvular failure of mitral and aortic valve after replacement, (2) simultaneous replacement of the tricuspid valve, (3) post-operative tricuspid valve stenosis (Mean gradient >3mm Hg), (4) patient prosthesis mismatch, (5) hemoglobin < 10 milligrams per deciliter (mg/dL), (6) thrombocytosis (platelet count > 450 000 per microliter), (7) thrombocytopenia (platelet count < 150 000 per microliter), (8) serum creatinine > 1.5 mg/dL, (9) increased levels of alkaline phosphatase, aspartate aminotransferase, and alanine transaminase (all laboratory findings assessed at admission), (10) hematologic disorders and malignancies, (11) and endocarditis. After identifying patients who met the inclusion and exclusion criteria, we selected the control group from our hospital database, comprising a population twice the size of the included group.
Data Collection at Recruitment
Patients’ characteristics, including demographics, comorbidities, clinical features, and laboratory findings, were retrieved from medical records. A trained team of researchers reviewed and extracted data for each patient.
Data and Outcome
MPV, PDW, and P-LCR were measured using blood samples and extracted from medical records. The laboratory parameters were measured in duplicate using the Sysmex XP-300 Hematology Analyzer. The outcome of interest was the presence of mitral or aortic valve thrombosis, assessed through esophageal echocardiography or the identification of thrombus on the valve during surgical intervention. Platelet volume parameters were compared between patients with valvular thrombosis and those with a history of mitral and aortic valve replacement but without valvular thrombosis.
Statistical Analysis
Descriptive statistics were utilized to summarize baseline characteristics, presenting mean ± standard deviation (SD) for normally distributed quantitative variables and median with interquartile range (IQR) for non-normally distributed quantitative variables. Independent t-tests were utilized to compare baseline characteristics between the two groups for continuous variables, while chi-squared tests were employed for qualitative variables to compare proportions between the two groups. Poisson and multivariable regression analyses were conducted to identify determinants associated with the occurrence of thrombosis. A threshold of statistical significance was set at p < 0.05.
Results
The data of patients were investigated from May 2002 to May 2020. A total of 898 patients were evaluated, and 108 patients were included in the study according to inclusion and exclusion criteria. The baseline and demographic characteristics of patients are summarized in Table 1. The case group comprised 108 patients (67 female and 41 male) with a history of mitral or aortic valve replacement and co-occurrence of valvular thrombosis. Conversely, the control group consisted of 216 patients (134 female and 82 male) with a history of valve surgery without valvular malfunction. There was no significant difference in gender distribution between the two groups (p = 0.99). The mean age of patients in the case group and control group was 53.72 ± 12.7 years and 50.32 ± 11.7 years, respectively (p = 0.018). Atrial fibrillation had a higher incidence among case group patients (33.3%) compared to control group patients (17.6%), and this difference was statistically significant (p < 0.01). Additionally, the history of coronary artery bypass graft (CABG) was significantly higher in the case group (28.7% vs 17.6%, p = 0.02). Furthermore, case group patients had almost the same frequency of pre-existing medical problems compared to the control group, including diabetes mellitus, hypertension, cerebral vascular accident (CVA), and a history of smoking (Table 1).
Patients’ Demographics Characteristics.
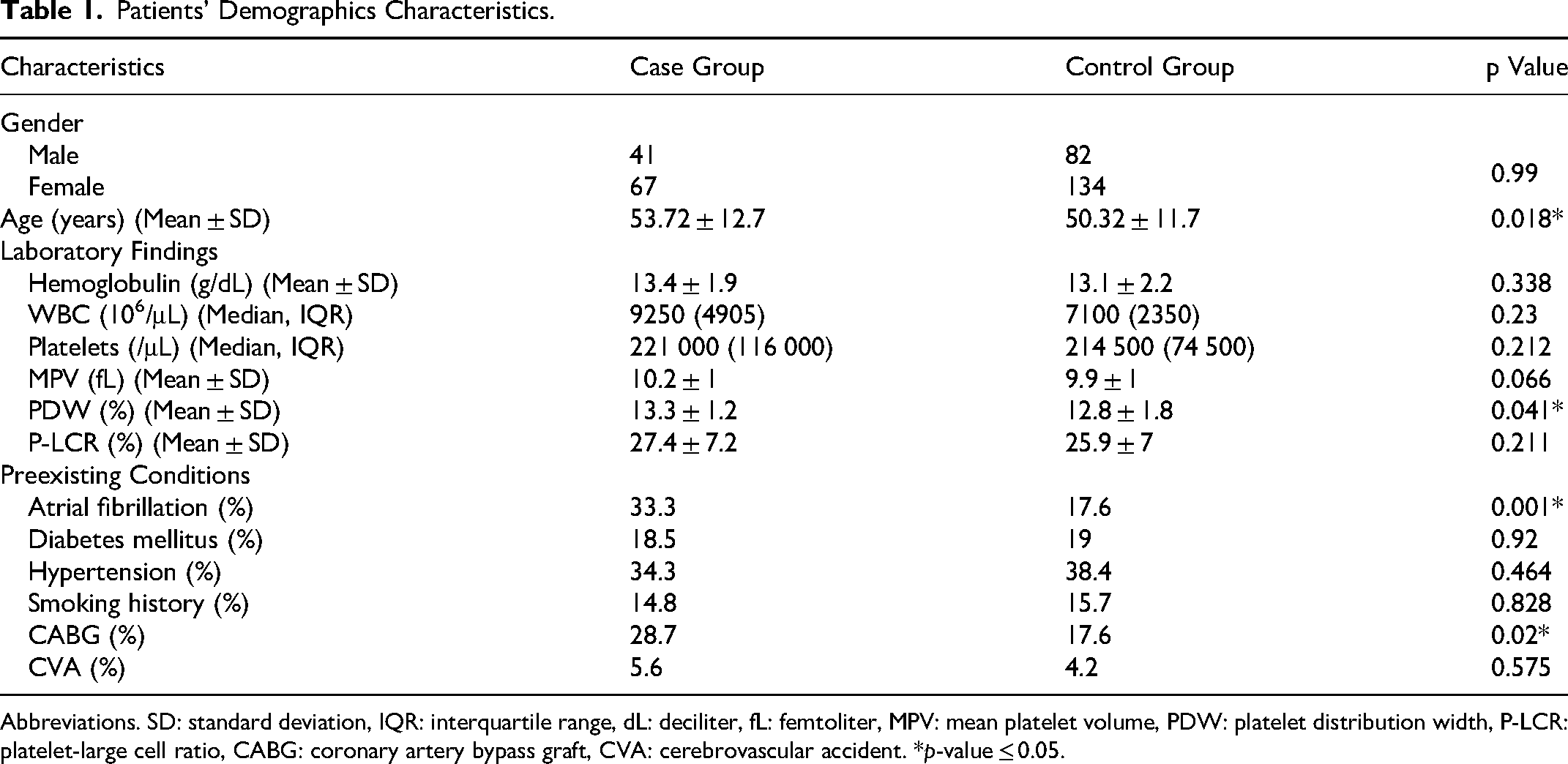
Abbreviations. SD: standard deviation, IQR: interquartile range, dL: deciliter, fL: femtoliter, MPV: mean platelet volume, PDW: platelet distribution width, P-LCR: platelet-large cell ratio, CABG: coronary artery bypass graft, CVA: cerebrovascular accident. *p-value ≤ 0.05.
The laboratory results indicated that the white blood cell (WBC) count and hemoglobin levels between the two groups did not differ significantly (see Table 1). The median (IQR) platelet count per μL in the case group and control group was 221 000 (116 000) and 214 500 (74 500), respectively (p = 0.212). Unadjusted results revealed that a higher MPV (RR: 0.31, 95% CI: 0.07, 0.55, p = 0.012) and PDW (RR: 0.51, 95% CI: 0.07, 0.94, p = 0.024) were associated with an increased risk of thrombosis. Conversely, P-LCR (RR: 1.55, 95% CI: −0.04, 3.12, p = 0.056) did not exhibit a statistically significant association with thrombosis occurrence. However, the association of PDW with an increased risk of thrombosis remained statistically significant after adjustment for age, atrial fibrillation, and CABG (RR: 0.47, 95% CI: 0.02, 0.93, p = 0.041). Additionally, while there was a clinical difference in MPV between the two groups, it did not reach statistical significance (RR: 0.2, 95% CI: −0.02, 0.47, p = 0.06) (Table 2).
Evaluating the Determinants Associated with Increased Risk of Thrombosis After Mitral and/or Aortic Valve Replacement. Adjusted Risk Ratios for Thrombosis Occurrence, Accounting for age, CABG, and AF.

Abbreviations: CABG: coronary artery bypass graft, AF: arterial fibrillation, RR: risk ratio, CI: confidence interval, MPV: mean platelet volume, PDW: platelet distribution width, P-LCR: platelet-large cell ratio. *p-value ≤ 0.05.
Discussion
We observed that higher PDW is associated with an increased risk of valvular thrombosis in patients with a history of mitral or aortic valve replacement, even after adjusting for potential confounders such as age, atrial fibrillation, and history of CABG. However, MPV and P-LCR did not demonstrate an association with an increased risk of valvular thrombosis after adjusting for confounders. These findings suggest PDW as a promising factor for predicting valvular thrombosis in patients with a history of mitral or aortic valve replacement.
Several studies have examined the risk factors of valvular thrombosis in patients with a history of valve replacement. It has been proposed that various factors may contribute to transcatheter and surgical bioprosthetic valve thrombosis, including inadequate antithrombotic therapy, diminished antiplatelet effect, prothrombotic states, and elevated platelet reactivity. 12 OPVT represents a severe complication in patients with mechanical valves despite achieving therapeutic international normalized ratio (INR) levels. Studies have indicated that patients with OPVT exhibit heightened platelet reactivity compared to their counterparts without thrombotic complications, potentially predisposing them to increased thrombotic risk. 13 Studies demonstrated that patients with cerebral venous sinus thrombosis and brain parenchymal lesions identified via MRI displayed elevated MPV and PDW levels.14,15 Furthermore, increased MPV, PDW, and P-LCR have been observed in patients with acute myocardial injury and unstable angina compared to individuals with stable coronary artery disease and healthy controls. 16 Additionally, MPV, PDW, and P-LCR has been shown to be predictors for thrombosis in autogenous arteriovenous fistula. 17 Similarly, higher MPV, not platelet count, was associated with increased risk of ischemic stroke. 18
Bluestein et al demonstrated that severe and mild valvular malfunction can increase platelet activity by up to 8- and 3-fold, respectively. However, their assessment of platelet activity utilized real-time assays and prothrombin time measurements. 19 Icli et al reported increased MPV among 40 patients with endocarditis and demonstrated an elevated risk of thromboembolic events in patients with higher MPV, a risk that decreased following endocarditis treatment and MPV reduction. 20
Mangapally et al indicated that large platelets exhibit greater activity than small platelets, a phenomenon that persists even after in vitro exposure to aspirin. They found that alpha-granule protein release, surface protein activation, dense granule content, and thromboxane synthesis were significantly higher in large platelets compared to small ones. 21 Large platelets produce thromboxane A2 and β-thromboglobulin, which have prothrombotic properties and are related to increased platelet activation. 14 Moreover, these two molecules increase the expression of adhesion molecules and aggregation of platelets. 11 MPV and PDW are indicators of platelet volume. Additionally, platelet activation is associated with morphological changes in platelets, such as pseudopod formation and spherical shape, and PDW may also indicate these changes. 22
Similar to aforementioned studies, our study yielded similar results; however, the difference in MPV was not statistically significant after adjusting for confounding variables. Additionally, Tekbas et al investigated MPV in patients with prosthetic mitral valve thrombosis (37 patients) compared to patients with normal prosthetic valves (62 subjects) and healthy individuals (69 subjects). They found that MPV was increased in patients with prosthetic valves (with or without thrombosis) compared to healthy individuals. However, there was no significant statistical difference in MPV between patients with prosthetic mitral valve thrombosis and those with normal prosthetic mitral valves, consistent with our findings. 23
This study represents the first investigation into the association of platelet volume indices with the risk of valvular thrombosis in patients with a history of mitral or aortic valve replacement. It is important to note that this was a single-center, retrospective cross-sectional study, and residual confounding due to unobserved factors such as INR cannot be entirely excluded. We adjusted the results for CABG, age, and AF. However, the platelet activation status in coronary artery disease patients after CABG may be restored compared to before surgery. 24 According to significant difference of CABG between two groups, we decided to adjust it as a confounder. Furthermore, we did not assess other markers of platelet activation and aggregation, fibrinolytic status, or thrombotic status. Future research should focus on establishing the relationship between platelet volume parameters, specifically MPV and PDW, and valvular thrombosis through larger prospective studies. Investigating the mechanisms behind thrombus formation and considering inflammatory markers will provide deeper insights. Multicenter studies could improve the applicability of findings across various populations. Incorporating routine monitoring of platelet volume parameters in clinical practice may enhance early detection and intervention, ultimately improving outcomes for patients undergoing valve surgeries.
Platelet factors, alongside other thrombosis risk factors, play a crucial role in assessing thrombotic risk. Individuals exhibiting higher PDW and MPV, indicative of increased platelet activity, face elevated risks of thromboembolic events. Therefore, it appears advantageous to administer aspirin to individuals at higher risk and to focus on platelet factors, particularly PDW and MPV, for risk identification. These parameters can be conveniently measured through routine hematological analysis, offering a straightforward, simple, and cost-effective means of predicting valvular thrombosis risk in patients with a history of mitral or aortic valve replacement. Based on the findings of this study, individuals with elevated PDW and MPV values can be readily identified during routine hematological analysis and may potentially benefit from preventive treatment strategies.
Footnotes
Acknowledgements
None.
Data Availability
The data supporting the findings of this study are available on request from the corresponding author and with permission from the Tehran University of Medical Sciences, Tehran, Iran.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Consent to Participate
This study was approved by the Ethics Committee of Tehran University of Medical Sciences (Tehran, Iran). Written informed consent was obtained from the patients to publish this report in accordance with the journal's patient consent policy.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
