Abstract
Background
Myocardial ischemia-reperfusion injury (MI/RI) poses a significant challenge during coronary revascularization. This research investigated alterations in oxidative stress and ferroptosis, alongside the impact of nicorandil on these aspects, among patients undergoing acute ST-elevation myocardial infarction (STEMI) and receiving primary percutaneous coronary intervention (pPCI).
Methods
121 patients with STEMI who were undergoing pPCI were included in the study, and we documented their thrombolysis in myocardial infarction (TIMI) blood flow grades before and after the procedure. The serum levels of creatine kinase-MB, creatinine, and N-terminal prohormone of brain natriuretic peptide (NTpro-BNP) were assessed. Additionally, we analyzed serum levels of SIRT1, HIF-1α, GPX-4, FTH1, FSP1, 4-HNE, and SI before pPCI and 6 h post-pPCI. Follow-ups were conducted 12 weeks after discharge, with major adverse cardiovascular events (MACEs) documented.
Results
The nicorandil group showed significantly improved TIMI blood flow grades and lower CK-MB and NTpro-BNP levels at 24, 48, and 72 h after PCI. Although preoperative SIRT1, HIF-1α, and ferroptosis-related indicators were similar, post-PCI, SIRT1 increased less and HIF-1α increased more in the nicorandil group. The nicorandil group exhibited higher GPX4, FTH1, and FSP1 levels, and lower levels of 4-HNE and SI. While no significant differences in left ventricular function were observed, the nicorandil group had lower LVEDD at 12 weeks. Importantly, the incidence of unstable angina and heart failure was significantly lower in the nicorandil group.
Conclusion
Administering nicorandil perioperatively during pPCI alleviates MI/RI, preserves cardiac and renal function, and reduces unstable anginarisk at 12 weeks post-PCI. These benefits of nicorandil may be attributed to its anti-oxidative stress and anti-ferroptosis effects.
Introduction
ST-segment elevation myocardial infarction (STEMI) is a severe cardiovascular condition triggered by the rupture of epicardial atherosclerotic plaque, instigating thrombosis that results in the necrosis of myocytes and collagen fiber denaturation.1,2 Over the past few decades, efforts in treating STEMI have primarily concentrated on structuring public health systems to ensure swift coronary revascularization of the affected artery and developing pharmaceutical interventions to sustain coronary blood flow.3–5 In clinical settings, primary percutaneous coronary intervention (pPCI) has emerged as the preferred method for reperfusion in STEMI patients. This approach reduces total ischemic time and diminishes mortality rates significantly.6,7 However, restoring blood flow to the ischemic heart muscle by reopening the infarct-related artery (IRA) during percutaneous coronary intervention (PCI) can unintentionally lead to additional myocardial cell damage. This occurrence, referred to as myocardial ischemia-reperfusion injury (MI/RI), can take place either alongside or following the procedure.1,8
Myocardial ischemia-reperfusion injury (MI/RI) is a condition where the restoration of anterior myocardial perfusion following the reopening of a blocked coronary artery can paradoxically worsen myocardial damage.9,10 Despite significant progress in understanding and treating MI/RI injuries, managing this condition remains a formidable challenge.11,12 Numerous cardioprotective strategies have been proposed to combat MI/RI, but as of now, none have demonstrated substantial improvements in the clinical outcomes of STEMI patients.13,14 Hence, there is a pressing urgent to explore novel treatments for tackling MI/RI damage. 15 Recent studies propose that ferroptosis, characterized by the buildup of iron-dependent lipid peroxides, plays a role in MI/RI.16,17 When reperfusion occurs, the abrupt increase in oxygen levels can stimulate the generation of reactive oxygen species (ROS) and consequent lipid peroxidation, which are pivotal components of ferroptosis. This cascade can exacerbate tissue damage within the heart.18,19 The connection between ferroptosis and the heightened oxidative stress response during MI/RI has garnered significant clinical interest and underscores the importance of investigating this relationship for potential therapeutic interventions.
Nicorandil is known for its ability to widen myocardial microcirculation, enhancing myocardial blood supply and potentially ameliorating MI/RI.20,21 However, its influence on ferroptosis remains unclear. Silent information regulatory factor 1, or SIRT1, a protein with deacetylation activity, plays a key role in regulating various biological processes such as energy metabolism, oxidative stress, aging, apoptosis, and autophagy.22–24 Recent research suggests that nicorandil may modulate SIRT1 expression, potentially reducing ferroptosis and thereby alleviating MI/RI linked to oxidative stress.25,26 Hypoxia-inducible factor 1 alpha (HIF-1α) is another crucial factor, highly expressed in ischemic and hypoxic conditions. By enhancing myocardial tolerance to hypoxia and diminishing infarct size, HIF-1α confers significant myocardial protection. 27 Consequently, a multitarget cardioprotective approach that combines different agents or interventions targeting various pathways at different stages (pre-, during, or post-pPCI) is vital. 28 Understanding the mechanisms of ferroptosis in the context of MI/RI could unveil potential therapeutic targets for mitigating the harm caused by this condition. In this study, we examined alterations in oxidative stress and ferroptosis-related markers, as well as the impact of nicorandil on these indicators in STEMI patients undergoing pPCI.
Materials and Methods
Subjects
This single-center, prospective, and randomized controlled intervention study recruited patients diagnosed with STEMI from January 2020 to December 2021 who received pPCI within 12 h of onset. We collected the following baseline patient characteristics: general clinical data, cardiac function grading, location of myocardial infarction, and time from symptom onset. Exclusion criteria included blood pressure below 90/60 mm Hg, cardiogenic shock, history of surgery, acute gastrointestinal bleeding, craniocerebral injury, unexplained stroke within the past month, and hematological or infectious diseases: severe trauma, severe liver and kidney dysfunction, autoimmune diseases, and mental illness. The research was officially registered in the Chinese Clinical Trial Registry under the registration number ChiCTR2100044120 on March 11, 2021. Prior to publication in this article, explicit written consent was acquired from the patients for the dissemination of their de-identified data.
Grouping, Treatment, and Follow-up
All patients were randomly divided into the conventional treatment group (the control group) and the nicorandil group at a ratio of 1:1. Before undergoing PCI, some patients received thrombolytic therapy based on clinical indications. The use of thrombolysis was determined by the treating physician, considering factors such as time from symptom onset and contraindications. Thrombolysis can significantly affect outcomes by reducing the size of the myocardial infarction and improving coronary blood flow prior to PCI. This may lead to differences in the severity of myocardial injury and subsequent recovery, potentially influencing the observed clinical outcomes and biomarker levels between the nicorandil and control groups. Before receiving pPCI treatment, both groups of patients received oral administration of 40 mg of atorvastatin and a dual antiplatelet loading dose (300 mg of aspirin, 180 mg of ticagrelor, or 300 mg of clopidogrel). The choice between ticagrelor and clopidogrel was based on clinical guidelines and the treating physician's discretion, taking into account factors such as patient characteristics, contraindications, and potential drug interactions. Nicorandil was delivered via intravenous infusion at a rate of 2 mg/kg before the pPCI and continued for 24 h following the procedure. The control group received an intravenous infusion of normal saline at the same time. The administration was conducted under close monitoring, and adverse reactions were recorded. Common adverse reactions observed included hypotension, headache, and flushing. No serious adverse events were directly attributed to nicorandil treatment during the study period.
Each patient was provided with conventional medical treatment in accordance with clinical protocols, comprising β-blockers and ACE inhibitors as clinically warranted. The decision regarding the specific medications administered was made by the attending physician, with no notable differences in their application observed between the nicorandil cohort and the control group. We recognize that variations in medical therapy may influence clinical outcomes and have discussed this in relation to our findings. Two experienced interventional physicians conducted coronary angiography and interventional therapy of the criminal coronary artery. The right radial artery was generally selected for interventional therapy, and thrombus aspiration was performed during the operation as needed based on the coronary thrombus burden.
Stent thrombosis was delineated based on the criteria outlined by the Academic Research Consortium (ARC), categorizing it as definite, probable, or possible. In this study, we included only cases of definite stent thrombosis, which is characterized by the presence of ACS symptoms and angiographic evidence of thrombus at the site of the stent. This definition ensures a stringent assessment of stent thrombosis events in both the nicorandil and control groups. All patients underwent PCI using drug-eluting stents. A total of 150 stents were used across both groups, primarily consisting of sirolimus-eluting stents and paclitaxel-eluting stents. There were no significant differences in the type or number of stents used between the nicorandil group and the control group. The choice of stent type was made based on the interventional cardiologist's discretion, considering factors such as lesion characteristics and patient-specific factors. During the PCI procedure, pre-dilation and post-dilation techniques were employed as needed by the interventional cardiologists. Pre-dilation was used for significantly calcified or tight lesions to facilitate stent delivery, while post-dilation aimed to optimize stent expansion.
Following the operation, all patients received oral aspirin 100 mg q.d. combined with ticagrelor 90 mg b.i.d. (or clopidogrel 75 mg q.d.) for 12 months. Additionally, atorvastatin combined with ezetimibe was administered when necessary to achieve intensive lipid reduction. The β-blockers, nitrate esters, and angiotensin receptor neprilysin inhibitors (ARNI) or angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (ACEI/ARB) were administered based on the patient's condition. The levels of creatine kinase (CK-MB) isoenzyme, creatinine, and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) were measured before pPCI and at 24, 48, and 72 h afterward. A serum creatinine increase of greater than 44.2 µmol/L or 25% above the baseline level was classified as contrast-induced acute kidney injury (CI-AKI). 29 Serum levels of SIRT1, HIF-1α, GPX4, FSP1, FTH1, 4-HNE, and SI before and 6 h after pPCI were measured using the enzyme-linked immunosorbent assay (ELISA). All patients were examined by echocardiography before pPCI and 1 and 12 weeks after pPCI. The left ventricular end-diastolic diameter (LVEDD), left ventricular end-systolic diameter (LVESD), and left ventricular ejection fraction (LVEF) were measured and recorded. Additionally, all patients underwent outpatient follow-ups on 1, 4, and 12 weeks after discharge. Data from dynamic electrocardiogram monitoring, the level of NTpro-BNP, and major adverse cardiovascular events (MACEs, including cardiovascular death, unstable angina, stent thrombosis, NYHA class IV heart failure and malignant arrhythmia) were collected. In this study, MACEs were defined as a composite of cardiac death, myocardial infarction related to the target lesion, and target lesion revascularization. This definition ensures a comprehensive assessment of significant cardiovascular outcomes following the intervention. All patients were followed for a total duration of 12 weeks post-discharge, with evaluations at 1, 4, and 12 weeks. For patients lost to follow-up, we made multiple attempts to contact them via phone and scheduled appointments to gather necessary outcome data. We documented reasons for loss to follow-up and assessed any differences in baseline characteristics between those who completed the study and those who did not. Our analysis adheres to intention-to-treat principles to maintain the integrity of our findings.
Statistical Analysis
The data analysis was performed utilizing SPSS version 22.0. Intra-group comparisons were carried out using paired t-tests, while inter-group comparisons were conducted using independent sample t-tests. Categorical variables were represented as percentages (%) and assessed using either the Chi-square test or Fisher's exact test, depending on the data distribution. A p-value below 0.05 was deemed statistically significant. For time-to-event outcomes, survival probabilities were illustrated using the log-rank test and the Kaplan-Meier method. Moreover, Cox proportional-hazard models were employed to calculate Hazard Ratios (HR) along with 95% confidence intervals (CI) for comparing treatments.
Results
Patient Enrollment and Baseline Characteristics
Out of the initial 121 patients, 61 were in the nicorandil group, and 60 were in the control group, all of who completed the follow-up. Three patients from the nicorandil group and four from the control group were unable to be tracked during follow-up. Notably, there were no notable differences in the selection of antiplatelet therapy between the two groups, ensuring treatment comparability. It is acknowledged that the choice of stent utilized could influence clinical results, encompassing restenosis rates and adverse events. Furthermore, there were no significant differences in the application of dilation techniques across the groups. Throughout the follow-up period, the occurrence of MACEs did not exhibit significant differences between the nicorandil and control groups. The consistent follow-up enabled a comprehensive evaluation of clinical outcomes in both treatment cohorts. No significant statistical differences in baseline characteristics were observed between the two groups (Table 1).
Baseline Characteristics of the Two Groups.a
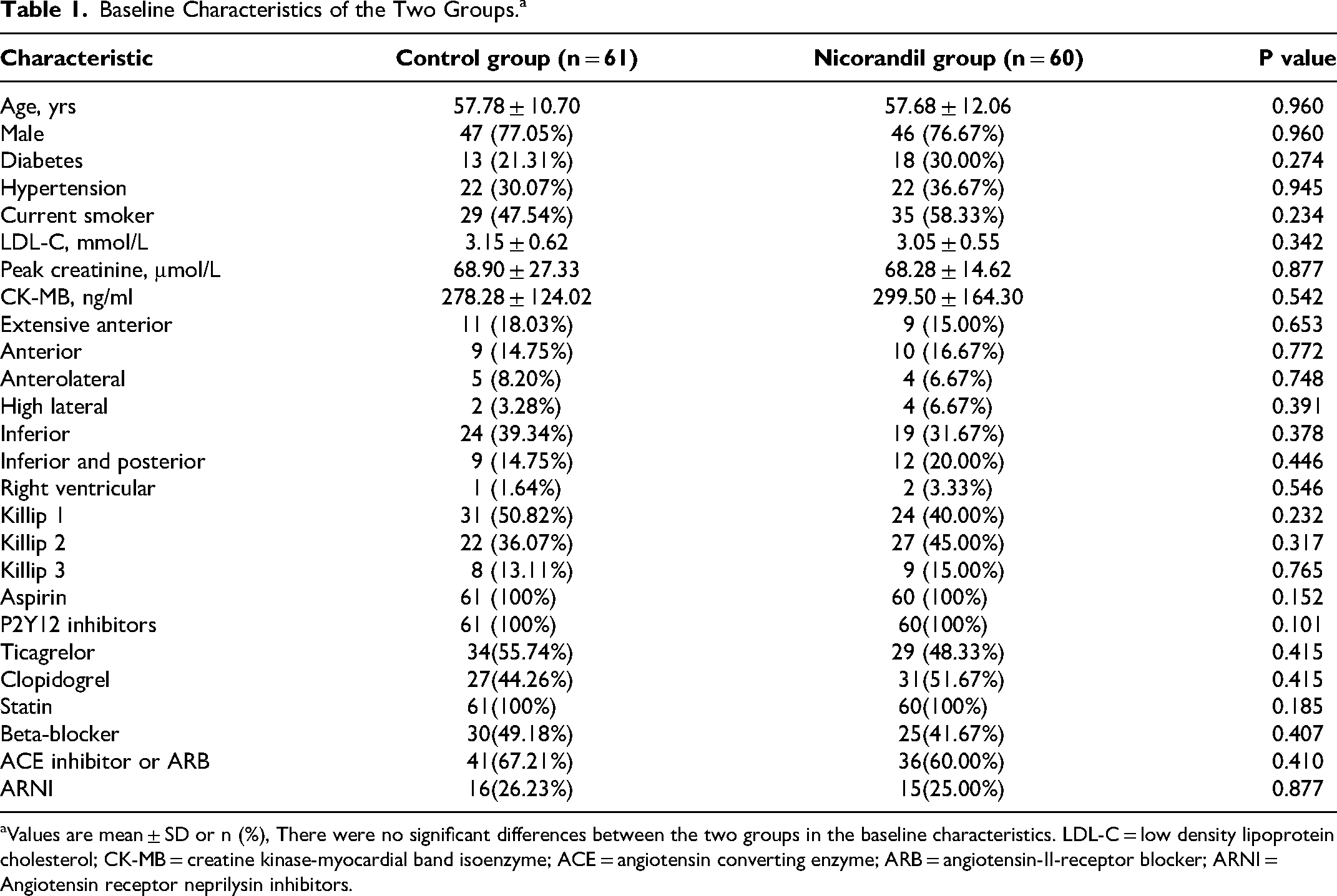
Values are mean ± SD or n (%), There were no significant differences between the two groups in the baseline characteristics. LDL-C = low density lipoprotein cholesterol; CK-MB = creatine kinase-myocardial band isoenzyme; ACE = angiotensin converting enzyme; ARB = angiotensin-II-receptor blocker; ARNI = Angiotensin receptor neprilysin inhibitors.
Coronary Intervention Results
Preoperative TIMI blood flow grades for the target vessel, assessed after coronary angiography (CAG) and before the start of pPCI, were similar between the two groups. However, the nicorandil group showed significantly improved postoperative TIMI blood flow grades compared to the control group. There were no significant differences between the two groups regarding the number of diseased vessels, target vessel distribution, thrombus aspiration, symptom-to-balloon time, door-to-balloon (D2B) time, or the volume of contrast agent used during pPCI (Table 2).
Procedural Characteristics.a
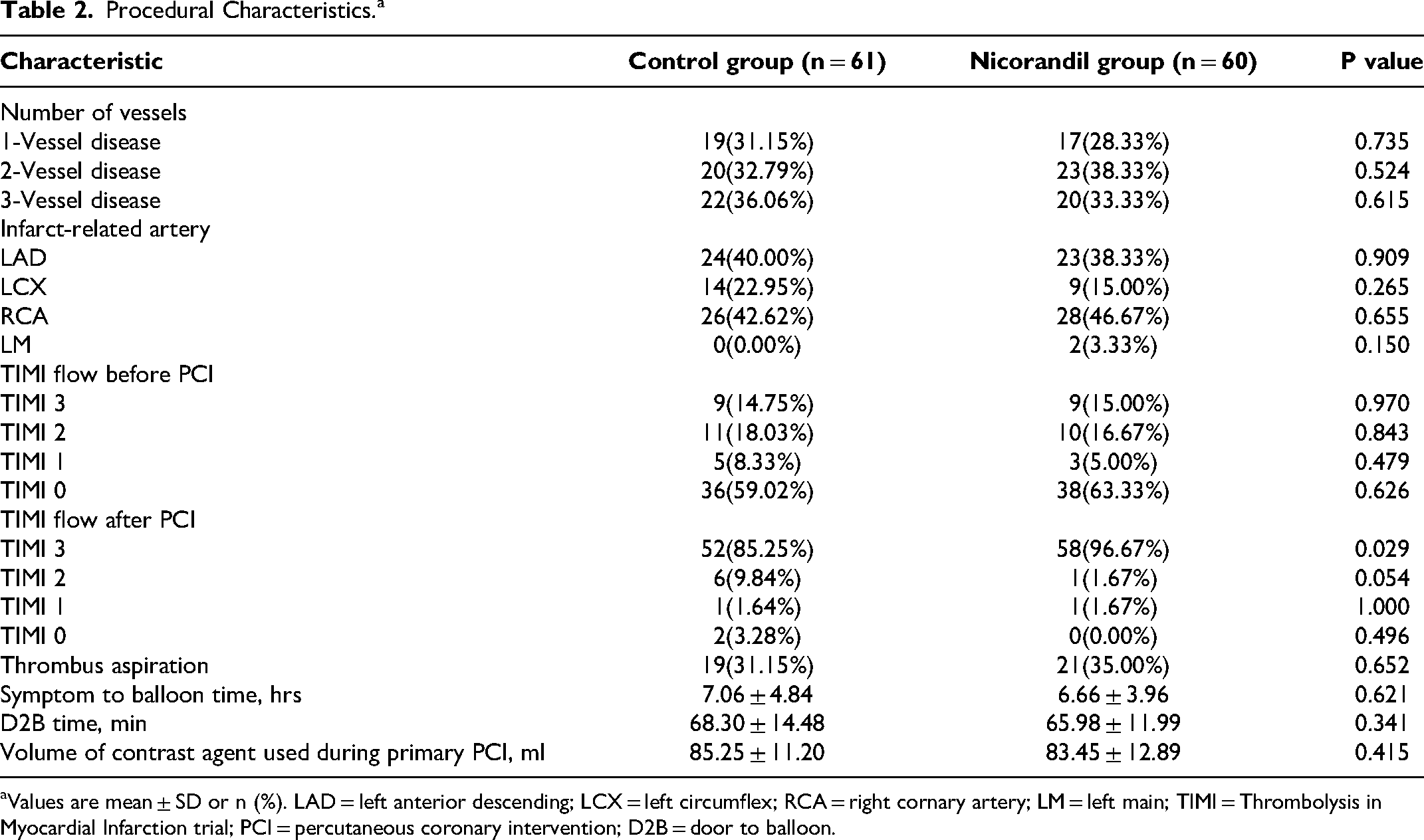
Values are mean ± SD or n (%). LAD = left anterior descending; LCX = left circumflex; RCA = right cornary artery; LM = left main; TIMI = Thrombolysis in Myocardial Infarction trial; PCI = percutaneous coronary intervention; D2B = door to balloon.
Laboratory Test Results
The CK-MB levels in the nicorandil group exhibited a notable decrease at 24-, 48-, and 72-h post-PCI. Similarly, NT-proBNP levels were significantly diminished in the nicorandil group in contrast to the control group across all three-time intervals. While serum creatinine levels showed a gradual rise in both groups, reaching a peak at 24–48 h, the creatinine levels in the nicorandil group were markedly lower at each time point (P < 0.05) (Figure 1).
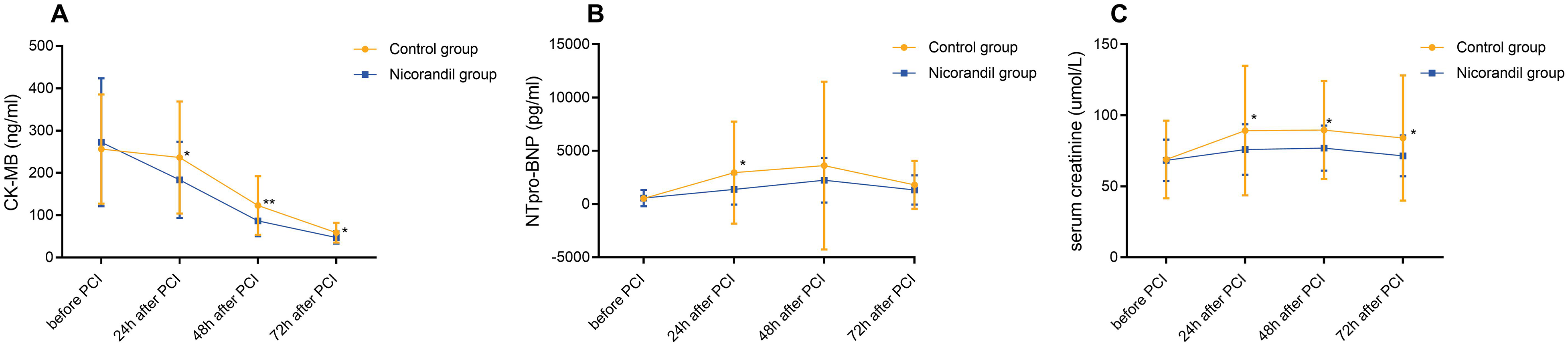
Changes in serum levels of CK-MB, NTpro-BNP, and serum creatinine in the two groups. A: Changes in CK-MB before and after PCI; B: Changes in NTpro-BNP before and after PCI; C: Changes in serum creatinine before and after PCI. The lines represent the mean values of each index for the respective groups over time. CK-MB = creatine kinase-myocardial band isoenzyme; NTpro-BNP = N-terminal prohormone of brain natriuretic peptide; PCI = percutaneous coronary intervention.
Oxidative Stress and Ferroptosis-Related Parameters
There were no differences noted in preoperative SIRT1, HIF-1α, and ferroptosis-related markers between the two groups. Following PCI, both groups displayed a significant rise in SIRT1 and HIF-1α levels at the 6-h mark (P < 0.01). However, the increase in SIRT1 was less pronounced in the nicorandil group in contrast to the control group, while HIF-1α levels were notably higher in the nicorandil group. GPX4 levels experienced a significant increase at 6 h post-PCI within the nicorandil group but remained unaltered in the control group (P < 0.01). Subsequent to the procedure, FTH1 and FSP1 levels exhibited no change in the control group but demonstrated a substantial increase in the nicorandil group (P < 0.01). Despite both groups manifesting a marked reduction in 4-HNE and SI levels at 6 h post-PCI, the nicorandil group showcased notably lower levels compared to the control group (Figure 2).
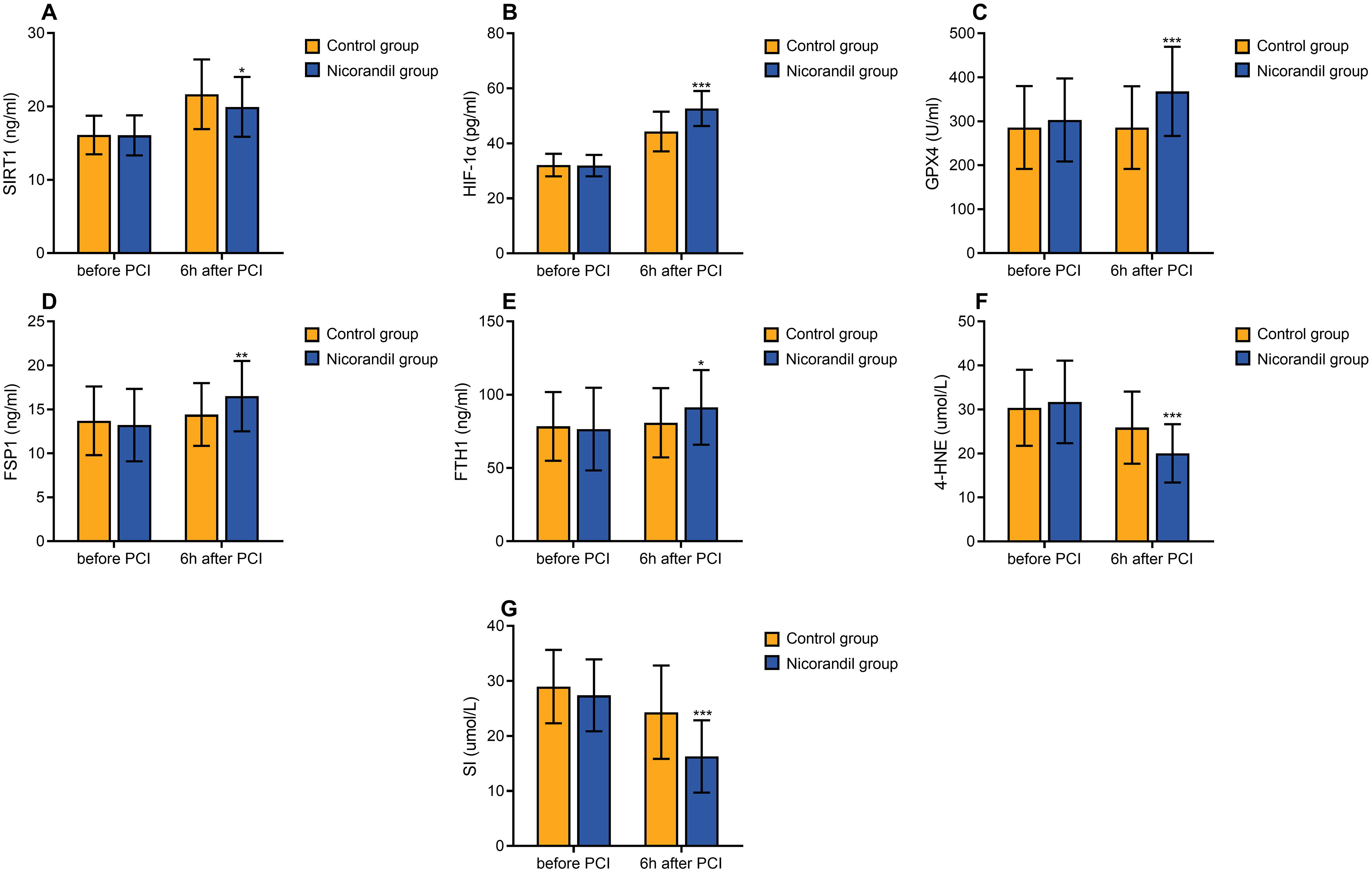
Changes of serum level of SIRT1, HIF-1α, GPX4, FTH1, FSP1, 4-HNE and SI of the two groups. A: Changes of SIRT1 before and 6 h after PCI; B: Changes of HIF-1α before and 6 h after PCI; C: Changes of GPX4 before and 6 h after PCI; D: Changes of FTH1 before and 6 h after PCI; E: Changes of FSP1 before and 6 h after PCI; F: Changes of 4-HNE before and 6 h after PCI; G: Changes of SI before and 6 h after PCI. SIRT1 = silent information regulatory factor 1 or sirtuin 1; HIF-1α= hypoxia-inducible factor 1 alpha; GPX4 = glutathione peroxidase 4; FTH1 = ferritin heavy chain 1; FSP1 = erroptosis suppressor protein 1; 4-HNE = 4-hydroxynonenal; SI = serum iron; PCI = percutaneous coronary intervention.
Left Ventricular Function
We found that before and one week after PCI, no significant differences in LVEDD and LVEF were discerned between the two groups. Likewise, at the 12-week juncture post-PCI, there was no significant difference in LVEF between the groups. However, during the 12-week evaluation post-PCI, the nicorandil group demonstrated a markedly diminished LVEDD in contrast to the control group (Figure 3).
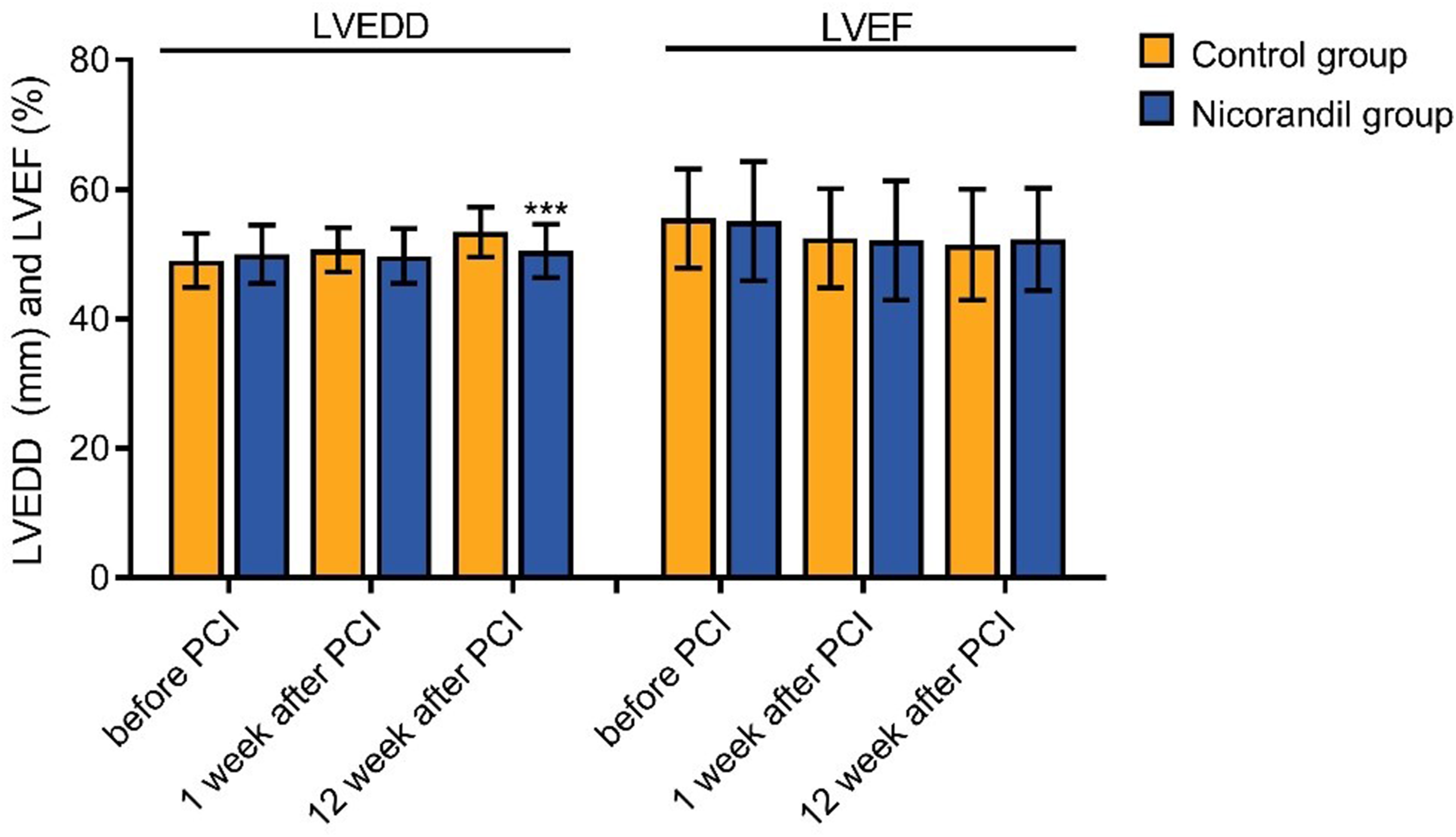
Changes of left ventricular function of the two groups. Panel shows the changes of LVEDD and LVEF before, 1 week after PCI and 12 weeks after PCI of the two groups. LVEDD = left ventricular end-diastolic diameter; LVEF = left ventricular ejection fraction; PCI = percutaneous coronary intervention.
The Endpoints
Although by the 12-week mark post-PCI, the nicorandil group did not display a significantly reduced occurrence of MACEs in comparison to the control group(P>0.05),but the nicorandil group has a better clinical trend.This better clinical trend in the nicorandil group was stemmed from a lower prevalence of unstable angina (P < 0.05) and NYHA class IV heart failure (P = 0.05) within the nicorandil cohort (Table 3). Conversely, no noticeable difference was observed between the two groups regarding the frequency of cardiovascular death, stent thrombosis, and malignant arrhythmia (Figure 4).
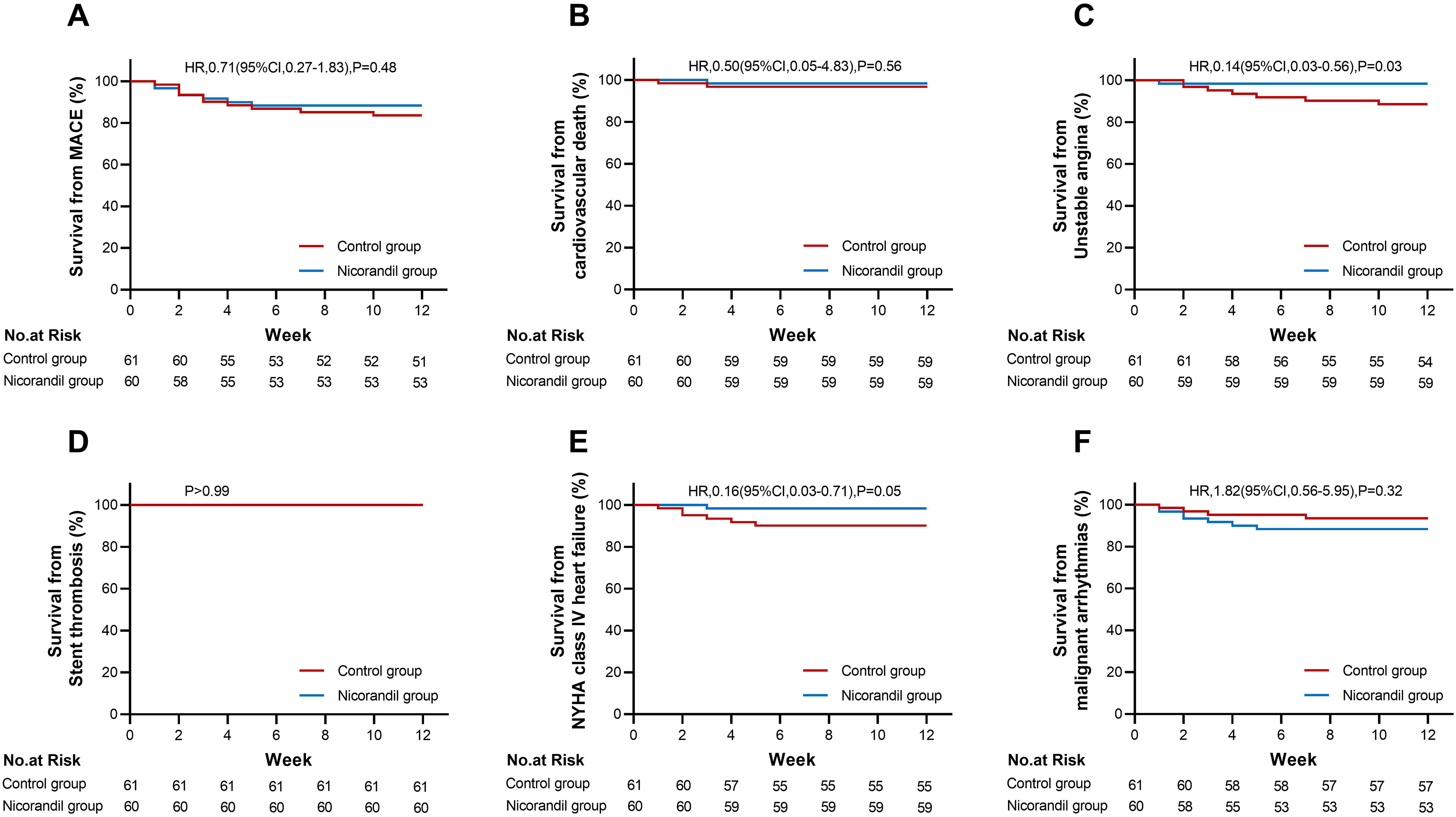
Kaplan-Meier curves for the endpoints. Panel A shows Kaplan-Meier estimate of the survival rate of the MACE. Panels B, C, D, E and F show the components of MACE (cardiovascular death, unstable angina, stent thrombosis, NYHA class IV heart failure and malignant arrhythmia), respectively. MACE = major adverse cardiovascular events; NYHA = New York Heart Association; PCI = percutaneous coronary intervention; HR = hazard ratio; CI = confidence interval.
Clinical Endpoints at 12 Weeks.a

Values are n (%) for occurrences of both first events and total events; bMACE denotes the composite of cardiovascular death, unstable angina, Stent thrombosis, NYHA class IV heart failure and malignant arrhythmia. MACE = major adverse cardiovascular events; CI = confidence interval; NYHA = New York Heart Association.
Myocardial Infarction and Target Lesion Revascularization
The study outcomes included data on MI and Target Lesion Revascularization (TLR). However, there were no significant differences in the incidences of MI or TLR between the nicorandil group and the control group. While specific incidence rates were not provided for these outcomes, it was evident that both groups demonstrated similar outcomes regarding MI and TLR.
Discussion
The present study found that in STEMI patients after pPCI, levels of SIRT1 and HIF-1α increased, while levels of 4-HNE and SI decreased. However, levels of GPX4, FSP1, and FTH1 remained unchanged. The perioperative application of nicorandil improved coronary TIMI blood flow, reduced myocardial infarct size, and protected both cardiac and renal function. Additionally, it reduced the incidence of postoperative angina pectoris and acute heart failure 12 weeks after PCI. The beneficial effects of perioperative nicorandil application include the inhibition of SIRT1, an increase in HIF-1α, GPX4, FTH1, and FSP1, and a reduction in 4-HNE and SI, ultimately inhibiting myocyte ferroptosis.
Ferroptosis, a novel type of programed cell death, has become a research focus in MI/RI.30,31 Cardiomyocyte ferroptosis plays a crucial role in cardiovascular diseases. GPX4, a key molecule in ferroptosis, serves as an essential marker and an effective indicator of antioxidant capacity.32,33 FSP1, a glutathione-independent ferroptosis inhibitor, can compensate for glutathione deficiency. Recently discovered, FSP1 works synergistically with GPX4 to inhibit ferroptosis. 34 Additionally, HIF-1α is the most sensitive marker for acute myocardial hypoxia and has a protective effect on acute coronary ischemia.35,36 Our study found that timely and effective reperfusion therapy increased serum levels of GPX4 and FSP1, enhancing antioxidant capacity. The perioperative application of nicorandil further elevated levels of GPX4, FSP1, and HIF-1α, thereby inhibiting ferroptosis and protecting cardiomyocytes. This effect may be related to nicorandil's unique dual-channel action in improving myocardial microcirculation.
There are limited reports on changes in serum SIRT1 levels following direct PCI combined with nicorandil treatment. miRNA-34a is highly upregulated in cardiomyocytes of rats with acute myocardial infarction and induces cardiomyocyte apoptosis. 37 SIRT1 is one of the target genes of miRNA-34a.38,39 Myocardial ischemia and hypoxia upregulate SIRT1 expression through the AMP-activated protein kinase (AMPK) pathway, thereby protecting cardiomyocytes from ischemia-reperfusion injury. 40 Nicorandil could downregulate miRNA expression, upregulate SIRT1 expression, reduce cardiomyocyte apoptosis and fibrosis, and inhibit myocardial remodeling. 41 Our data showed that SIRT1 levels continued to rise after re-canalization of STEMI infarct-related vessels, which might be related to the progression of STEMI and the pathophysiological changes caused by direct PCI. Furthermore, SIRT1 levels significantly increased after nicorandil treatment and were notably higher than in the control group, indicating that nicorandil combined with direct PCI could further enhance SIRT1 levels and provide myocardial protection. However, the precise mechanisms underlying the protective effects of nicorandil warrant further exploration. During ferroptosis, ferritin levels increase in parallel with SI levels.
We observed that FTH1 in STEMI patients after emergency intervention exhibited synchronous changes, indicating reliable remission of ferroptosis. Following re-canalization of infarcted vessels in acute myocardial infarction patients, cardiomyocytes experience reperfusion injury, which nicorandil can alleviate. Additionally, re-canalizing occluded coronary arteries significantly reduces 4-HNE levels and alleviates oxidative stress. The combined use of nicorandil further reduces 4-HNE levels, suggesting that nicorandil provides myocardial protection by reducing oxidative stress in cardiomyocytes. The significant reduction in MACEs in the nicorandil group compared to the control group highlights the drug's potential to improve short-term outcomes following PCI. Specifically, the decrease in unstable angina and NYHA class IV heart failure suggests that nicorandil enhances cardiac stability post-PCI. Although there were no significant differences in cardiovascular death, stent thrombosis, or malignant arrhythmia, these findings indicate that nicorandil may effectively reduce certain adverse events, contributing to better clinical outcomes in STEMI patients.
Limitations
There are several limitations in the current study that should be acknowledged. Firstly, the research was conducted at a single center, which may limit the generalizability of the results. Findings from a single institution may not fully represent the broader population due to potential variations in patient demographics, clinical practices, and treatment methodologies across different centers. Secondly, the small sample size of 121 patients could hinder the statistical power of the study. A small sample may not adequately represent the population and could lead to Type I or Type II errors, impacting the robustness of the conclusions drawn regarding the efficacy of nicorandil. The follow-up duration of only 12 weeks post-PCI restricts our understanding of the long-term clinical outcomes associated with nicorandil treatment. While short-term outcomes are informative, it is crucial to evaluate the potential for late adverse events or sustained benefits beyond this period. Although the study employed randomization, there may still be inherent biases in patient selection and treatment adherence. Factors such as patient preferences, physician recommendations, or clinical judgment could influence which patients were enrolled or how they were treated, potentially skewing the results. Additionally, the study design did not include blinding of participants or investigators, which may introduce bias in outcome assessment. Participants’ expectations regarding treatment could influence their reported outcomes, and investigators’ knowledge of treatment assignments may affect data collection and analysis. Reliance on laboratory tests and clinical assessments may also introduce variability in measurements. For instance, serum biomarker levels can be affected by various factors, including timing of sample collection, patient hydration status, and assay variability, which may impact the consistency of the results. The study focused on specific clinical outcomes and biomarkers, which may not capture the full spectrum of patient health and recovery. Other important factors, such as quality of life, functional status, and patient-reported outcomes, were not assessed and could provide valuable insight into the treatment's overall impact. Finally, the findings may not be applicable to all populations, particularly given potential differences in demographics, comorbidities, and treatment responses in diverse patient groups. Future studies should aim to include a more varied population to enhance external validity. In light of these limitations, we emphasize the need for further research to validate our findings in larger, multi-center trials with extended follow-up periods and comprehensive assessment of clinical outcomes.
Conclusion
In summary, despite the enhanced anti-oxidative stress and anti-ischemia activity, ferroptosis persists in STEMI patients after pPCI. Therefore, administering nicorandil during the perioperative period of pPCI can alleviate MI/RI, protect cardiac and renal function, and reduce the risk of unstable angina12 weeks post-PCI. These benefits of nicorandil may be attributed to its anti-oxidative stress and anti-ferroptosis effects.
Footnotes
Acknowledgements
The authors would like to thank the professors in the central laboratory of the Second Hospital of Hebei Medical University for their contributions to the data analysis of this experiment.
Authors’ Contributions
SJ wrote the manuscript. SJ and YL conceived and designed the experiments. DT, XJ and WZ analyzed the data. HJ and JZ collected and provided the sample for this study. All authors have read and approved the final submitted manuscript.
Availability of Data and Materials
The data-set generated and analyzed during the current study will be available one years later and is also available now from the corresponding author on a reasonable request.
Consent for Publication
Not Applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Subjects (or their guardians) have given their written informed consent. The current research was approved by the Ethics Committee of The Second Hospital of Hebei Medical University (HDFY-LL-2021-045).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
