Abstract
Venous thromboembolism is a serious safety concern in women using combined oral contraceptives; ethinyl estradiol (EE) is widely used as an estrogen. Estetrol (E4) is a native estrogen with selective tissue activity and exclusively produced by the fetal liver. This study used a multicenter, randomized, open-label, active-controlled, parallel-group design to evaluate the effects of E4 combined with drospirenone (DRSP) on coagulation and fibrinolysis in Japanese patients with endometriosis. Participants were randomized to receive either E4 15 mg/DRSP 3 mg or EE 20 µg/DRSP 3 mg for 12 weeks. E4/DRSP and EE/DRSP were administered orally once a day in a cyclic regimen, ie, 24-day active use followed by a 4-day hormone-free period, and a flexible extended regimen, respectively, and blood coagulation and fibrinolysis markers were measured. The effect on coagulation and fibrinolysis was considerably less in the E4/DRSP group than in the EE/DRSP group. Major anticoagulant proteins, protein S (free, total) and tissue factor pathway inhibitor (free), were reduced following EE/DRSP treatment. Consequently, thrombin generation determined by the activated protein C sensitivity ratio was increased by approximately 4-fold in the EE/DRSP group than in the E4/DRSP group. Eventually, the fibrinolysis cascade was triggered to compensate for disturbed coagulation, and D-dimer levels were 4.7-fold higher in the EE/DRSP group than in the E4/DRSP group. This study demonstrated that the effect of E4/DRSP on the blood coagulation and fibrinolysis cascades was significantly less than that of EE/DRSP in participants with endometriosis, a disease of women of advanced and reproductive age (jRCT2080225090, https://jrct.niph.go.jp/en-latest-detail/jRCT2080225090).
Keywords
Introduction
Combined oral contraceptives (COCs) are used mainly for birth control, but are also prescribed to women of reproductive age with dysmenorrhea and endometriosis.1,2 The main safety concern with COCs is the increased risk of venous thromboembolism (VTE). In 1969, the British Committee on the Safety of Drugs reported a positive association between the use of synthetic oral estrogen use and increased VTE risk. 3 Therefore, the dosage of ethinyl estradiol (EE), a widely used estrogen in COCs, was intensively reduced, and the current preparations contain 10 to 30 µg of EE. Molecular modification of the progestin component also aimed to lower the side effects caused by the androgenic effects of the first- and second-generation progestins. This led to the commercialization of less androgenic COCs, including desogestrel (DSG) and gestodene (GSD) in the 80 s and drospirenone (DRSP) in the 2000s. Nevertheless, the use of these COCs was associated with an even higher incidence of VTE than COCs containing levonorgestrel (LNG).4–8 Interestingly, progestin-only pills (POPs) containing LNG or DSG do not increase the risk of VTE,9–12 suggesting that the problem is driven more by the estrogen in the combination than by the progestin itself.
Estetrol (E4) is a native estrogen produced exclusively by the human fetal liver and detectable in maternal blood and urine from the ninth week of gestation. 13 It has a 4- to 5-fold higher affinity to estrogen receptor (ER)α than ERβ. A unique characteristic of E4 is that it acts as an agonist of nuclear ERα and as an antagonist of ERα-dependent membrane-initiated steroid signaling (MISS).14,15 Consequently, it was termed the first native estrogen with selective tissue activity (NEST) and was classified differently from selective ER modulators (SERM). 16 Importantly, E4 15 mg in combination with 3 mg of DRSP was found to have little effect on sex hormone-binding globulin (SHBG) and all proteins produced in liver cells under the control of estrogens, such as angiotensinogen, cortisol-binding globulin, or thyroxin-binding globulin, suggesting that E4 could also have a minimal impact on protein levels in the coagulation and fibrinolysis cascades.17–19 An in-depth investigation of the coagulation profile of E4/DRSP showed that E4 had a lower impact on the global hemostasis process, which could translate into a lower VTE risk.18–24 Furthermore, Valéra et al reported that E4 protected against both arterial and venous thrombosis, and induced resistance to acute thromboembolism in mouse models. 25 These results are consistent with the clinical safety of E4/DRSP, as observed in Phase III studies outside Japan. Only a single VTE case was reported in 3417 women, resulting in a VTE incidence of 3.7/10 000 women years. 22 The US Food and Drug Administration (FDA) and the European Medicine Agency (EMA) have approved the inclusion of this for a contraceptive indication since 2021.26,27
In Japan, E4/DRSP combination has been clinically developed as treatment for dysmenorrhea and endometriosis-associated pelvic pain (EAPP). Endometriosis is a benign, estrogen-dependent, chronic inflammatory disease that is the most common cause of chronic pelvic pain in women aged >30 years. 28 In addition, it mainly causes secondary dysmenorrhea. Several international and Japanese guidelines recommend COCs as the first-line treatment to alleviate EAPP and menstrual pain that decreases patients’ quality of life (QOL).1,2,29,30 However, COCs that contain EE are known to increase the risk of VTE, and more cautious use needs to be considered, since aging is known to increase VTE risk by 3- to 5-fold.31–33 Therefore, a safe, well-tolerated COC is needed. E4 in association with DRSP has been found to have a lower impact on coagulation proteins than EE containing COCs,19,23 but data from women with endometriosis have not been obtained.
In this Phase II, clinical pharmacology study, the effects on coagulation and fibrinolysis of E4/DRSP in a cyclic regimen and of EE/DRSP in a flexible extended regimen were investigated in Japanese patients with endometriosis.
Material and Methods
Study Design
This study used a multicenter, randomized, open-label, active-controlled, parallel-group design in Japanese participants diagnosed with endometriosis. The aim was to compare the effects of E4/DRSP and EE/DRSP on coagulation and fibrinolytic parameters. The treatment period was 12 weeks in both groups.
This study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice (GCP) protocols and met all local legal and regulatory requirements. The protocol was reviewed and approved by the institutional review board of each study site, and written, informed consent was obtained from all participants.
Participants
Participants aged between 20 and 50 years who were diagnosed with endometriosis were enrolled in this study. The clinical diagnoses were based on laparotomy/laparoscopy, transvaginal ultrasonography, or magnetic resonance imaging (ie, presence of ovarian endometriomas). Another eligibility criterion was EAPP Visual Analogue Scale (VAS) scores ≥40 mm during two consecutive menstrual cycles in the baseline observation period. Other eligibility criteria are described in Supplemental Materials.
Treatment
Eligible participants were randomly allocated to either the E4 15 mg/DRSP 3 mg group or the EE 20 µg/DRSP 3 mg group in a 1:1 ratio balanced for baseline VAS score (< 60 mm or ≥ 60 mm) and comorbidities (none, uterine fibroids, or adenomyosis) to eliminate potential biases between groups. Immediately after randomization, the participants took one tablet per day at a comparable time of the day from the first day of menstruation. In the E4/DRSP group, participants were treated with a cyclic regimen, ie, a 24-day oral administration of E4/DRSP tablets, followed by a 4-day hormone-free interval with placebo tablets per cycle, for three consecutive cycles. In the EE/DRSP group, the participants received a daily oral tablet for 12 weeks (84 days) according to the dosage on the labeling. In both groups, a 56-day follow-up was scheduled after treatment. Stratified randomization codes were developed by the Interactive Web Response System using a permuted-block design managed by an office independent of the clinical investigators and other stakeholders.
Evaluating Endpoints
The participants were requested to visit the clinical sites during the baseline observation and treatment periods every 4 weeks. Blood samples were collected before randomization and at the first and third visits to measure blood coagulation and fibrinolysis markers. These included antithrombin (AT), D-dimer, prothrombin time (PT), fibrinogen, soluble fibrin monomer complex (SFMC), activated partial thromboplastin time (APTT), total protein S (PS) activity and antigen, PS specific activity (PS activity/PS antigen), free PS antigen, free tissue factor pathway inhibitor (TFPI) antigen, activated protein C sensitivity ratio (APCsr), activated coagulation factor V (FVa), FVIIa, FXa, protein C (PC) activity, plasminogen activity, tissue plasminogen activator (tPA)-plasminogen activator inhibitor-1 (PAI-1) complex (total PAI-1), prothrombin fragment 1 + 2 (F 1 + 2), and SHBG. Total PS activity and antigen, PS specific activity, and free PS antigen levels were measured at Sinotest Corp., Tokyo, Japan. APTT-based APCsr (APCsr) 34 and endogenous thrombin potential (ETP)-based normalized APCsr (nAPCsr) 35 were measured at QUALIblood, Namur, Belgium. Other markers were measured at LSI Medience Corp., Tokyo, Japan. Assay procedures and reference ranges are summarized in Supplemental Table 1.
Statistical Analyses
Statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). No formal sample size estimation was conducted because of the exploratory nature of this study. However, assuming a medium effect size of 0.5 and a dropout rate of 15%, 40 participants per group were required to have statistical power of 80% or greater. The individual relative changes from baseline to the end of treatment (%) of each marker were primarily used to assess the effects of E4/DRSP and EE/DRSP on the blood coagulation and fibrinolysis parameters. An analysis of variance (ANOVA) model was applied with treatment group as a fixed effect, and least squares (LS) means were inferred with two-sided 95% confidence intervals (CIs) for each treatment. Furthermore, within the framework of ANOVA, LS mean and a two-sided 95% CI were implied for the between-group difference of each marker, ie, E4/DRSP - EE/DRSP. No imputation method was used for missing data.
For SFMC, the proportion of participants whose test results were over the lower limit of the reportable value (3 µg/mL) was also evaluated, and Fisher's exact test was used because no quantitative test results were reported when the measured values were less than the limit. Other measures are expressed as mean ± standard deviation values. All analyses were conducted in the full analysis set consisting of participants who took the study drugs at least once and in whom data on blood coagulation and fibrinolysis markers were obtained.
Results
Participants
The first visit of the first participant was on March 5, 2020. From the 18 clinical sites, 88 eligible participants were randomized. The full analysis set population consisted of 86 participants: 45 in the E4/DRSP group and 41 in the EE/DRSP group. Early termination occurred in four participants due to withdrawal by participants in the E4/DRSP group and adverse events and others in the EE/DRSP group (Figure 1).
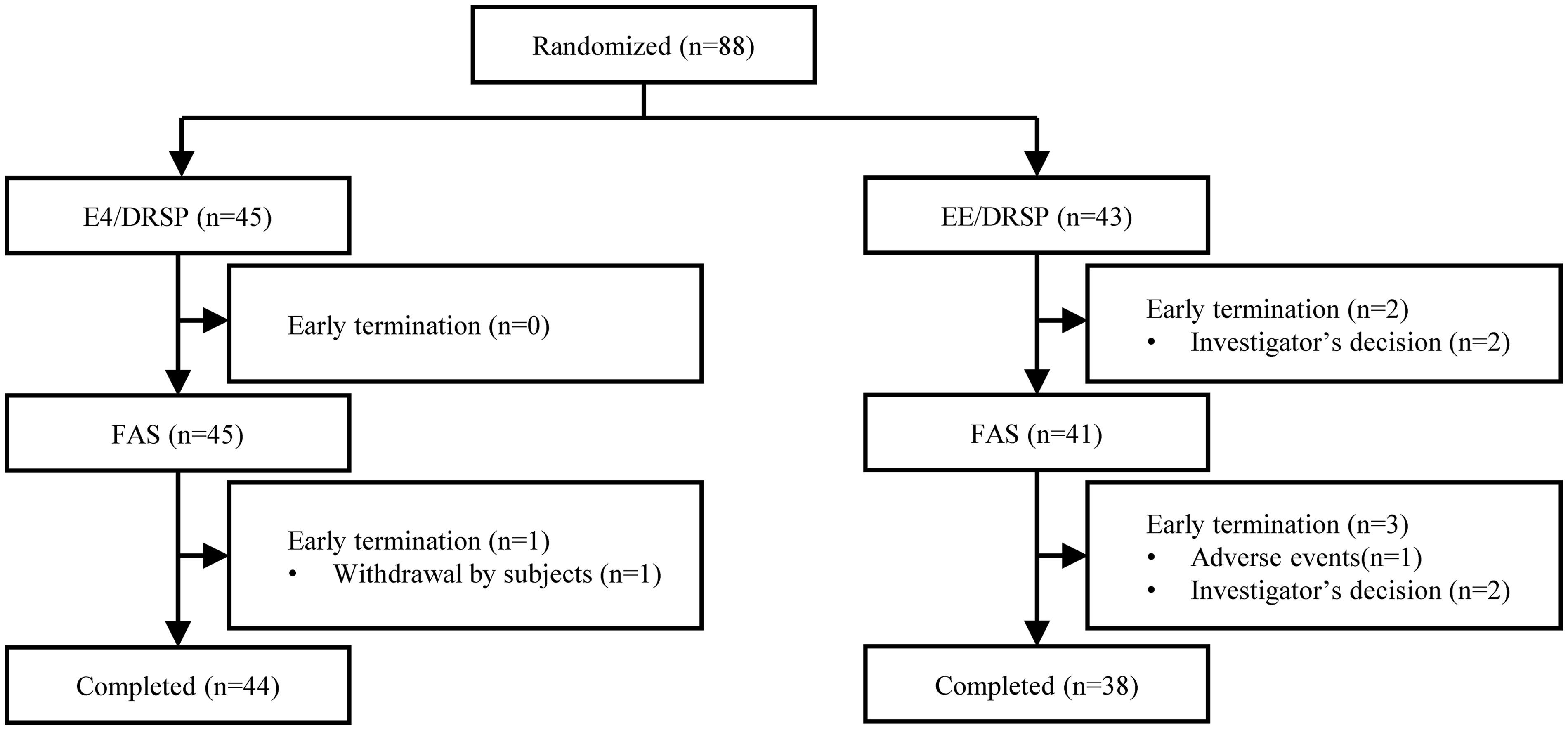
Patient flowchart.
Participants’ background characteristics were comparable between the E4/DRSP and EE/DRSP groups (Table 1). The participants’ average age and body mass index were 34.9 ± 7.6 years and 20.8 ± 2.4 kg/m2, respectively. Of the 86 patients, 24 (27.9%) and 16 (18.6%) participants also had adenomyosis and uterine fibroids, respectively. Both adenomyosis and uterine fibroids were observed in six participants (7.0%).
Demographics and Baseline Characteristics (FAS).
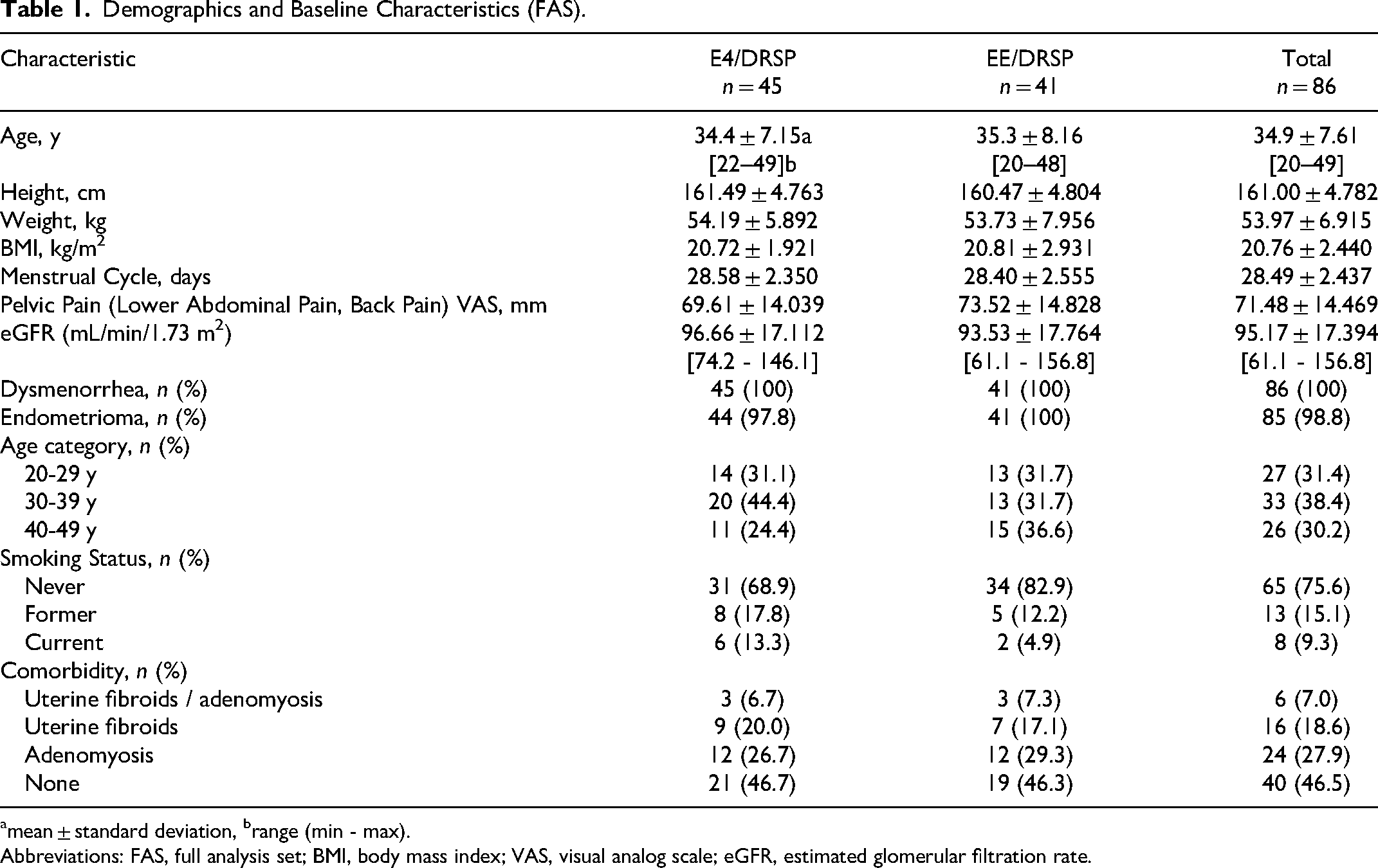
mean ± standard deviation, brange (min - max).
Abbreviations: FAS, full analysis set; BMI, body mass index; VAS, visual analog scale; eGFR, estimated glomerular filtration rate.
Blood Coagulation and Fibrinolysis Markers
In Figure 2, the relative changes from baseline in the blood coagulation and fibrinolysis parameters (LS means and two-sided 95% CIs) following 12 weeks of treatment with E4/DRSP or EE/DRSP are depicted. The between-group differences (LS means and two-sided 95% CI), ie, E4/DRSP - EE/DRSP, are shown in Figure 3. All results are shown with two-sided 95% CIs in Table 2. Summary statistics for each marker are shown in Supplemental Table 2.
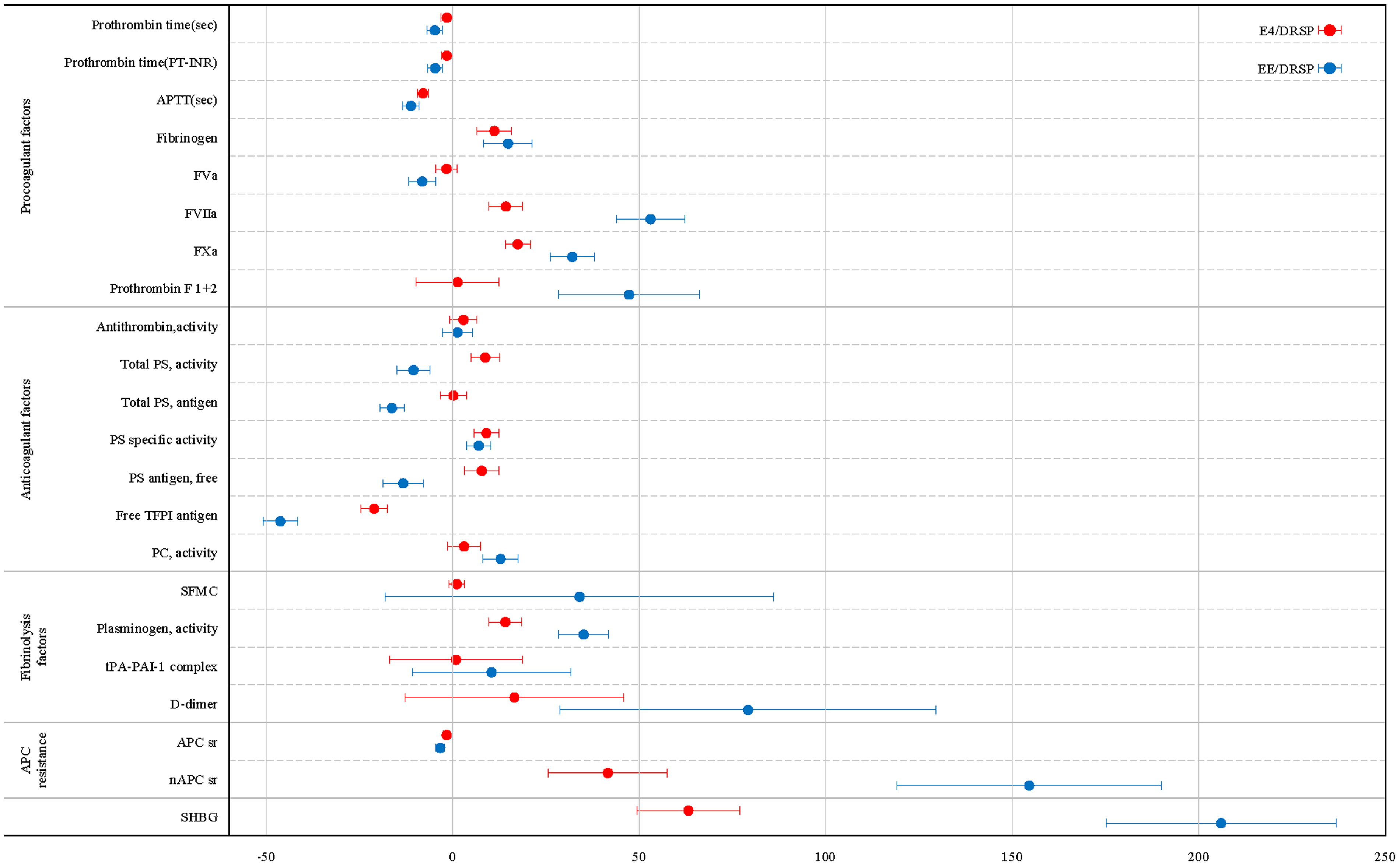
Relative changes from baseline in blood coagulation and fibrinolysis makers following 12-week administration of E4/DRSP and EE/DRSP in Japanese participants with endometriosis (LSmeans with two-sided 95% CIs).
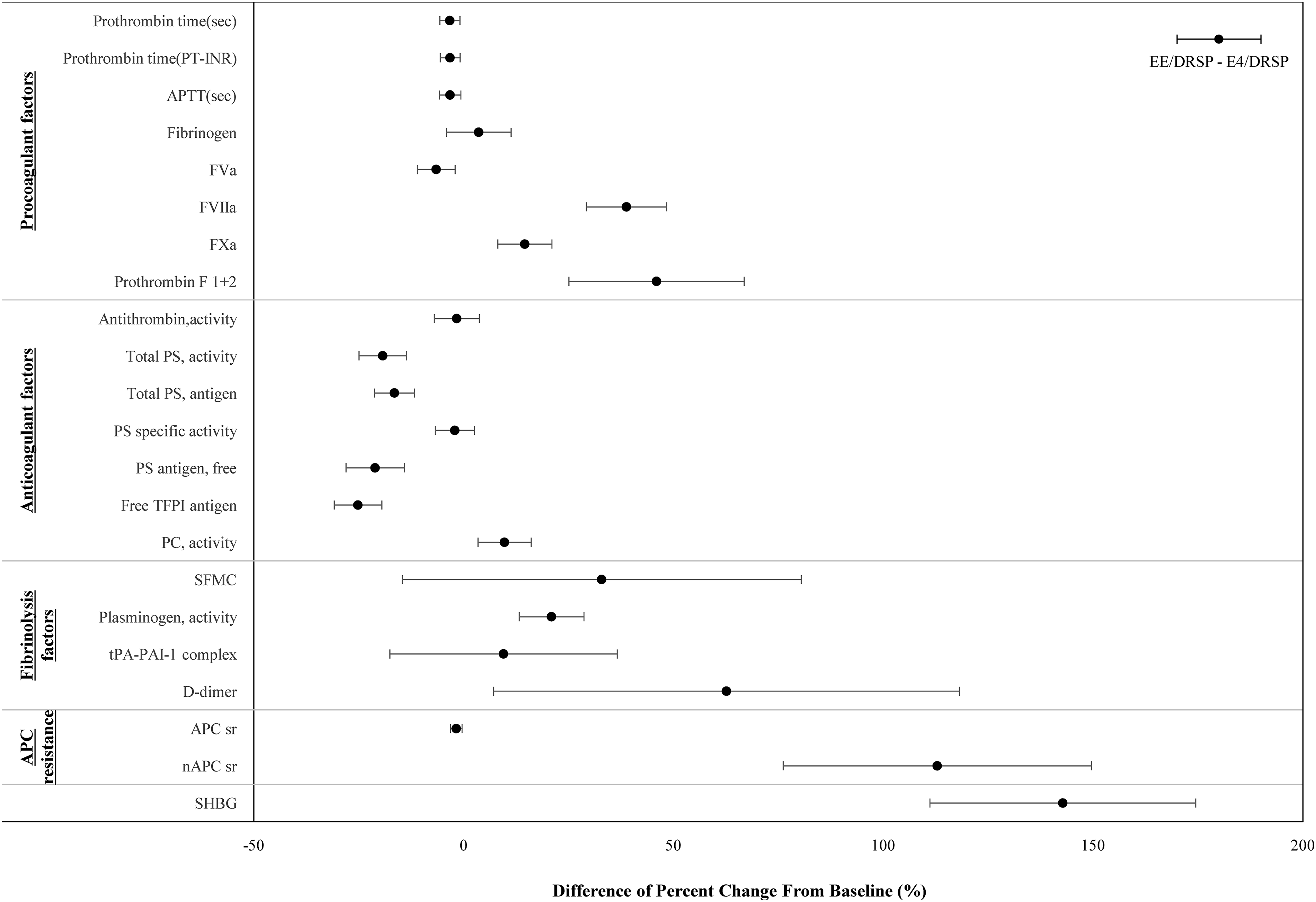
Group differences in change from baseline in blood coagulation and fibrinolysis markers between E4/DRSP and EE/DRSP following 12-week administration in Japanese participants with endometriosis (LSmeans with two-sided 95% CIs).
Effect of E4/DRSP and EE/DRSP on Blood Coagulation and Fibrinolysis systems a .
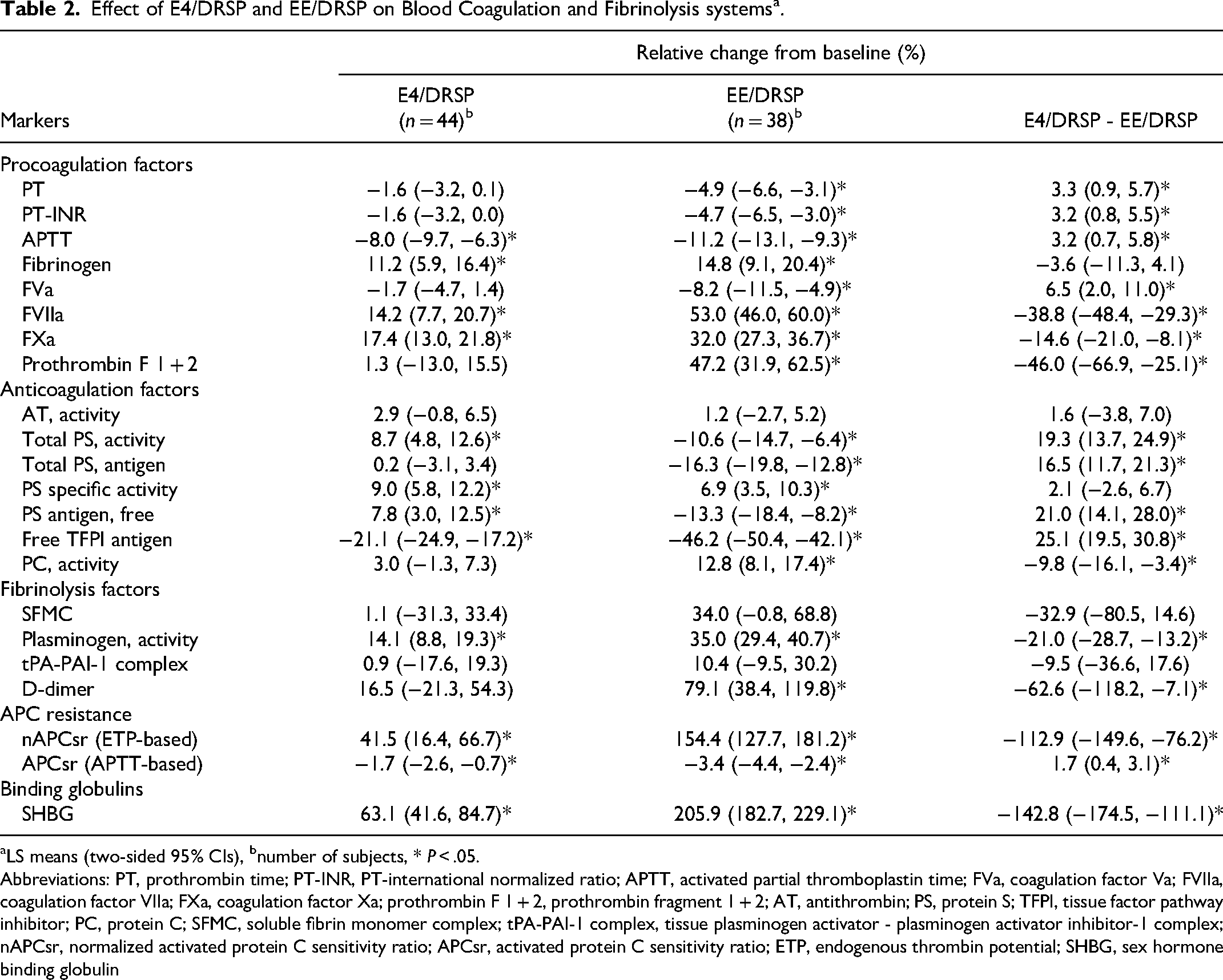
LS means (two-sided 95% CIs), bnumber of subjects, * P < .05.
Abbreviations: PT, prothrombin time; PT-INR, PT-international normalized ratio; APTT, activated partial thromboplastin time; FVa, coagulation factor Va; FVIIa, coagulation factor VIIa; FXa, coagulation factor Xa; prothrombin F 1 + 2, prothrombin fragment 1 + 2; AT, antithrombin; PS, protein S; TFPI, tissue factor pathway inhibitor; PC, protein C; SFMC, soluble fibrin monomer complex; tPA-PAI-1 complex, tissue plasminogen activator - plasminogen activator inhibitor-1 complex; nAPCsr, normalized activated protein C sensitivity ratio; APCsr, activated protein C sensitivity ratio; ETP, endogenous thrombin potential; SHBG, sex hormone binding globulin
Procoagulant Factors
After 12 weeks of treatment, fibrinogen, FVIIa, and FXa levels were higher in the EE/DRSP group than in the E4/DRSP group. Relative changes from baseline were 14.8% and 11.2% for fibrinogen, 53.0% and 14.2% for FVIIa activity, and 32.0% and 17.4% for FXa activity in the EE/DRSP and E4/DRSP groups, respectively. The LS mean differences between groups (E4/DRSP - EE/DRSP) were significant for FVIIa and FXa, at −38.8% and −14.6%, respectively (both P < .05). Prothrombin F 1 + 2 increased by 47.2% in the EE/DRSP group compared with baseline, whereas no significant change was observed in the E4/DRSP group, leading to a significant LS mean difference of −46.0% (P < .05). In addition, the effect on FVa levels was slightly greater in the EE/DRSP group than in the E4/DRSP group.
Anticoagulation Factors
EE/DRSP and E4/DRSP reduced free TFPI antigen levels by −46.2% and −21.1% compared with baseline, respectively, and the LS mean difference (E4/DRSP - EE/DRSP) was significant (25.1%, P < .05). Similarly, free and total PS antigen levels were significantly decreased by −13.3% and −16.3%, respectively, from baseline in the EE/DRSP group (P < .05). E4/DRSP did not change or even marginally increased PS antigen levels (7.8% and 0.2% for free and total PS, respectively), leading to significant between-group differences (both P < .05). No change in AT activity was observed in the E4/DRSP and EE/DRSP groups. A slight increase in PC activity was observed after 12-week treatment in the EE/DRSP group, but not in the E4/DRSP group, with a significant between-group difference (−9.8%, P < .05).
Fibrinolysis Factors
EE/DRSP increased D-dimer levels up to 79.1% from baseline after 12-week treatment. In contrast, D-dimer elevation was limited to 16.5% in the E4/DRSP group, leading to a significant between-group difference of −62.6% (P < .05). SFMC levels were higher in the EE/DRSP group than in the E4/DRSP group, and the proportion of participants with SFMC ≥3 µg/mL was 15.8% in the EE/DRSP group and only 2.3% in the E4/DRSP group (P = .045). Some changes followed a slight increase in plasminogen levels in both groups; however, the elevation was less in the E4/DRSP group than in the EE/DRSP group, including total PAI-1 (tPA-PAI-1 complex).
Activated Protein C Resistance
The ETP-based assay (expressed as the normalized APC sensitivity ratio, nAPCsr) was increased by 154.4% from baseline in the EE/DRSP group, but only 41.5% from baseline in the E4/DRSP group at week 12. The between-group difference of −112.9% was significant (P < .05). The values for nAPCsr were 2.22 ± 0.78 for E4/DRSP and 4.03 ± 1.12 for EE/DRSP, and limited changes were observed in the APTT-based assay (APCsr) in both groups, as expected.
Sex Hormone Binding Globulin
SHBG was increased much more in the EE/DRSP group (205.9% from baseline) than in the E4/DRSP group (63.1% from baseline) following 12 week treatment, resulting in a significant between-group difference (P < .05).
Discussion
This Phase II study aimed to compare the effects of E4/DRSP on coagulation and fibrinolysis in Japanese participants with endometriosis with those of EE/DRSP which has already been approved for patients with EAPP in Japan. 36 The results showed that the E4/DRSP group exhibited fewer changes in coagulation and fibrinolysis parameters than the EE/DRSP group.
Endometriosis is an estrogen-dependent disease and a major cause of pelvic pain and infertility, with a prevalence ranging from 2% to 11% in asymptomatic women of reproductive age and up to 50% in women with infertility and chronic pelvic pain. It is characterized by endometrial-like tissue outside the uterine cavity, which causes local immune and inflammatory responses associated with a hypercoagulable state in the advanced stages. 37 Medical treatment is most often started in patients over 30 years of age owing to the delay in diagnosis. 26 However, because aging is a well-known risk factor for VTE,31–33 clarifying the effects of COCs on blood coagulation and fibrinolysis parameters is important for safe use in patients with endometriosis.
Since guidelines recommend that oral contraceptive pills be used for the treatment of endometriosis,1,2,29 the same dosage and regimen, ie, E4 15 mg/DRSP 3 mg daily for 24 days, followed by a hormone-free interval for 4 days, was adopted in the study.26,27
Dominant anticoagulant proteins (total PS antigen and activity, free PS antigen, and free TFPI antigen) were decreased by EE/DRSP, resulting in the lack of a compensatory mechanism for the increased production and activation of coagulation. Although COCs are generally known to affect lipid metabolism, 38 neither E4/DRSP nor EE/DRSP increased levels of LDL-cholesterol (data not shown), which is a dominant component binding to TFPI, contributing to its free-form fraction. 39 Ali et al recently reported that COCs downregulated TFPI at the transcriptional level via the classical (genomic) pathway using the ERα-expressing breast cancer cell line MCF-7.40,41 Furthermore, Dahm et al reported that both E2 and EE reduced TFPI production by −30% to −39% in human coronary arterial endothelial cells. 42 A low plasma TFPI level is probably a weak risk factor for VTE, 43 but it remains unclear whether a decreased TFPI level during COC use is associated with an increased VTE risk.
More importantly, EE/DRSP significantly reduced the levels of PS antigens (free and total) and total PS activity, which are associated with decreased free TFPI levels and increased procoagulant factors, and further increased the prothrombotic state induced by the COC. Conversely, E4/DRSP did not affect and even slightly increased the levels of PS antigens, thus having a low impact on the ETP-based APC resistance assay, confirming the results of previous investigations. 18 Estrogen downregulates PROS1 at the transcription level through the non-classical ERα pathway. The ERα-Sp1-RIP140 interaction regulates PROS1 expression by recruiting the NCoR-SMRT corepressor complex and HDAC3, which induces histone hypoacetylation and less permissive transcription of the PROS1 gene. 44
Membrane ER-mediated, non-genomic signaling can be integrated with nuclear ER-mediated gene transcription via protein kinase-mediated phosphorylation of ER and other ER-interacting transcription factors, such as AP-1, SP-1, and cAMP response element-binding protein (CREB), to regulate latent gene expression in target cells. Therefore, the increase in PS antigen levels may be due to the antagonistic activity of E4 on MISS. However, further studies are needed to confirm this mechanism of action.
PS is a key protein that enhances the binding affinity of free TFPI to FXa independently of PC,45–48 and the TFPI-FXa complex efficiently suppresses the tissue factor-FVIIa complex. Then, reduction of PS associated with TFPI causes less inactivation of the extrinsic coagulation pathway and the amplification phase of the coagulation cascade. The complex formed by APC and PS also inhibits FVa and FVIIIa. This downregulates thrombin via the intrinsic and common pathways of the coagulation cascade.49,50 Any impairment of this process increases thrombin generation. 51
Although EE is widely used as an estrogenic component in COCs, the effects of COCs on hemostasis cannot be attributed only to the estrogenic component. Hughes et al found that PROS1 transcription levels are upregulated via progesterone receptor isoform B in MCF-7 cells, a triple-positive breast cancer cell line. 52 Kozuka et al recently showed that progestins facilitate PS transcription elongation by enhancing de novo PS mRNA expression modulated by RNA polymerase II (Pol II), but not by PROS1 promoter activity. They also reported that DRSP displayed a lesser extent of transcriptional elongation than LNG in HepG2 cells. 53 Miyoshi et al found that free PS levels were significantly lower in healthy Japanese women who started using cyclic COCs. This reduction is more important in EE/DSG and EE/DRSP users than in EE/norethisterone (NET) and EE/LNG users, even after the first treatment cycle. 54 Therefore, knowing the extent to which progestins offset the reduction in PS antigen levels induced by EE is mandatory.
Van Vliet et al reported that the ETP-based assay (nAPCsr) was elevated by COCs, and that the increase in nAPC resistance was correlated with the reductions of free PS and free TFPI antigens. They showed that nAPC resistance was greater for EE combined with third and fourth-generation progestins (ie, GSD, GTD, and DRSP) than for EE combined with LNG. 55 In the present study, EE/DRSP accounted for an approximately 4-fold greater increase in nAPCsr than E4/DRSP (154.4% vs 41.5%). These results are comparable to those previously obtained for E4/DRSP and EE/DRSP, 18 and the nAPCsr values of E4/DRSP, EE/LNG, and EE/DRSP after three cycles were estimated to be 2.31, 3.61, and 4.36, respectively. This is in line with the present results and confirms the lower impact of E4 on APC resistance than EE-containing products.
The APTT-based assay (APCsr) showed only a slight increase in APCsr because it shows only the initiation phase of thrombin generation, unlike the ETP-based assay that depends on the entire phase of thrombin generation.56,57 Altogether, the present data demonstrate a significant difference in changes in coagulation and fibrinolysis markers and in global evaluation of hemostasis by measuring thrombin generation by APCsr between E4/DRSP and EE/DRSP. This further confirms the superior safety profile of E4/DRSP compared with EE/DRSP.
The activity of AT, another anticoagulation protein, was not affected by E4/DRSP or EE/DRSP treatment. PC activity increased slightly from baseline in the EE/DRSP groups, but it did not counterbalance the reduced levels of protein S, which is a limiting factor in the anticoagulant properties of the protein C-protein S complex.58–60
Collectively, these results suggest a more prothrombotic switch with EE/DRSP than with E4/DRSP, which seems to remain neutral in the hemostatic system. This was further confirmed by the measurement of prothrombin F 1 + 2. These peptides are released from prothrombin during its conversion to thrombin, reflecting coagulation activation. This sensitive marker was significantly elevated by 47.2% after 12 weeks of treatment in the EE/DRSP group compared with no change in the E4/DRSP group.
SHBG levels were increased by less than one-fourth in the E4/DRSP group compared with those in the EE/DRSP group, consistent with the previous investigations. 18
Disturbance of the coagulation cascade had complementary effects on the fibrinolysis cascade. This included increased plasminogen, total PAI-1 (tPA-PAI-1 complex), and SFMC levels, but no significant differences in total PAI-1 and SFMC levels. However, all these factors tended to be more elevated in the EE/DRSP group than in the E4/DRSP group. The proportion of participants with SFMC ≥ 3 µg/mL (lower limit of the reportable range) was approximately 7-fold higher in the EE/DRSP group than in the E4/DRSP group. This showed that the fibrinolysis cascade was firmly triggered in the EE/DRSP group. In fact, D-dimer levels increased by 79.1% from baseline in the EE/DRSP group, with only a very slight, clinically irrelevant increase (16.5% from baseline) in D-dimer levels in the E4/DRSP group.
DRSP is a spironolactone derivative that has anti-androgenic properties. 61 Although it is necessary to further investigate whether the androgenicity of progestins regulates PS upregulation at the transcriptional level, the choice of estrogen must be closely investigated when developing new COCs. 20
This study was performed for relative short term period (12 weeks), which might be temporal modifications. However, it was reported that a 2-year use of COCs translated into steadily reduced protein S antigen, a key co-factor of protein C anti-coagulation, as well as enhanced fibrinolysis. 62
There are several limitations in this study. First, the open-label design may have introduced potential biases due to lack of blindness; however, blood sample anonymization prior to measurements likely excluded the biases as much as possible. Second, no formal sample size estimation was performed due to the exploratory nature of this study, but the sample size was considered large enough to detect a medium effect size, ie, 0.5. Finally, this study suggests that E4/DRSP is a safe, new option for medical treatment. However, though the risk of VTE was potentially lower with E4/DRSP than with EE/DRSP, this needs to be interpreted carefully, since no biological endpoints such as APC resistance confirm VTE occurrence reliably, although increasing evidence suggests a link between VTE risk and specific coagulation markers such as ETP-based APC resistance. 22 Thus, it is hoped that the ongoing outcome study will be able to provide valid conclusions. 63
Conclusions
E4/DRSP had less effect on hemostasis than EE/DRSP. Notably, this study enrolled Japanese participants with endometriosis, with an average age of 34.9 years, and the incidence rate of VTE increases with age. Since nAPCsr is a sensitive index of thrombogenesis, E4/DRSP is expected to be much safer than EE/DRSP by mitigating the risk of VTE.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241286514 - Supplemental material for Impact of Estetrol Combined with Drospirenone on Blood Coagulation and Fibrinolysis in Patients with Endometriosis: A Multicenter, Randomized, Open-Label, Active-Controlled, Parallel-Group Study
Supplemental material, sj-docx-1-cat-10.1177_10760296241286514 for Impact of Estetrol Combined with Drospirenone on Blood Coagulation and Fibrinolysis in Patients with Endometriosis: A Multicenter, Randomized, Open-Label, Active-Controlled, Parallel-Group Study by Takao Kobayashi, Masashi Hirayama, Masayoshi Nogami, Kanna Meguro, Masato Iiduka, Jean-Michel Foidart, Jonathan Douxfils and Tasuku Harada in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296241286514 - Supplemental material for Impact of Estetrol Combined with Drospirenone on Blood Coagulation and Fibrinolysis in Patients with Endometriosis: A Multicenter, Randomized, Open-Label, Active-Controlled, Parallel-Group Study
Supplemental material, sj-docx-2-cat-10.1177_10760296241286514 for Impact of Estetrol Combined with Drospirenone on Blood Coagulation and Fibrinolysis in Patients with Endometriosis: A Multicenter, Randomized, Open-Label, Active-Controlled, Parallel-Group Study by Takao Kobayashi, Masashi Hirayama, Masayoshi Nogami, Kanna Meguro, Masato Iiduka, Jean-Michel Foidart, Jonathan Douxfils and Tasuku Harada in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-3-cat-10.1177_10760296241286514 - Supplemental material for Impact of Estetrol Combined with Drospirenone on Blood Coagulation and Fibrinolysis in Patients with Endometriosis: A Multicenter, Randomized, Open-Label, Active-Controlled, Parallel-Group Study
Supplemental material, sj-docx-3-cat-10.1177_10760296241286514 for Impact of Estetrol Combined with Drospirenone on Blood Coagulation and Fibrinolysis in Patients with Endometriosis: A Multicenter, Randomized, Open-Label, Active-Controlled, Parallel-Group Study by Takao Kobayashi, Masashi Hirayama, Masayoshi Nogami, Kanna Meguro, Masato Iiduka, Jean-Michel Foidart, Jonathan Douxfils and Tasuku Harada in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors would like to thank the investigators who participated in this multicenter study: Motoyasu Furuya, MD (Machida Higashimachi Clinic); Hideki Hanashi, MD (New Medical Research System Clinic); Masaski Hashimoto, MD (Hashimoto Clinic); Kouzo Hirai, MD (Minami-Morimachi Ladies Clinic); Yuji Kashiwazaki, MD (Kashiwazaki Ladies Clinic); Toshiro Mizutani, MD (Aiiku Ladies Clinic); Hiroko Mouri, MD (Rinkan Clinic); Sonoe Nishiguchi, MD (Asahi Clinic); Akira Nishikawa, MD (Nishikawa Women's Health Clinic); Seiji Ogawa, MD (Chayamachi Ladies Clinic Honmachi); Hisato Oku, MD (Chayamachi Ladies Clinic); Kaori Suenaga, MD (Parkside Hiroo Ladies Clinic); Yukoku Tamaoka (Ikebukuro Metropolitan Clinic); Akiko Tanabe, MD (Tanabe Ladies Clinic); Rikako Uchida, MD (Rikako Ladies Clinic); Minoru Yaegashi, MD (Sapporo Maternity Women's Hospital); Teruko Yasuda, MD (Yoshio Clinic); Tsuneo Yokokura, MD (Yokokura Clinic).
Declaration of Conflicting Interests
Takao Kobayashi and Tasuku Harada received consulting fees from Fuji Pharma Co., Ltd. The COIs of the author and co-authors were submitted as supplemental material files (ICMJE disclosure forms).
Ethics Approval
Ethical approval for this study was obtained from the following institutional review boards: Sapporo City Medical Association Institutional Review Board: 284 Specified non-profit organization National Clinical Research Council Institutional Review Board: 55-348, 94-348, 57-348, 45-348, 59-348, 29-348, 46-348, 78-348 Medical Corporation IHL Shinagawa East One Medical Clinic Institutional Review Board: S0342 Fujikeikai Medical Corporation Kitamachi Clinic Institutional Review Board: FJP06971
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Fuji Pharma Co., Ltd. Fuji Pharma Co., Ltd designed and conducted the study and was involved in the analysis and interpretation of the data obtained.
Informed Consent
Written, informed consent was obtained from the patients for their anonymized information to be published in this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
