Abstract
Purpose
To investigate the differences in survival after venous thromboembolism (VTE) and anticoagulation efficacy and safety between catheter (CRVTE) and non-catheter-related VTE (NCRVTE) in cancer patients.
Methods
A retrospective research was conducted, and consecutive cancer (digestive, respiratory, genitourinary, blood and lymphatic, and the other cancers) patients with VTE were enrolled. The anticoagulation therapies included low-molecular-weight heparin (LMWH), warfarin, new type of direct oral anticoagulants (NDOACs), LMWH combined with warfarin, and LMWH combined with NDOACs. Data were collected from the electronic medical record database of our hospital and were analyzed accordingly by Kruskal-Wallis H Test, Chi-square test, Fisher's exact test, Logistic regressions, Kaplan-Meier analysis, and Cox regressions.
Results
263 patients were included, median age in years (interquartile range) was 64(56-71) and 60.5% were male. VTE recurrence rate was 16.7% in CRVTE group which was significantly lower than 34.8% in NCRVTE group (P = .032). Heart diseases were independently associated with VTE recurrence (P = .025). Kaplan-Meier survival estimates at 1, 2, and 3 years for CRVTE group were 62.5%, 60.0%, and 47.5%, respectively, compared with 47.9% (P = .130), 38.7% (P = .028), and 30.1% (P = .046), respectively, for NCRVTE group. Cox regression showed surgery (P = .003), anticoagulation therapy types (P = .009), VTE types (P = .006) and cancer types (P = .039) were independent prognostic factors for 3-year survival after VTE. Nonmajor and major bleeding were not significantly different (P = .417). Anticoagulation therapy types were independently associated with the bleeding events (P = .030).
Conclusions
Cancer patients with CRVTE potentially have a better anticoagulation efficacy and survival compared to NCRVTE, and the anticoagulation safety seems no significant difference.
Keywords
Introduction
Venous thromboembolism (VTE), comprising both deep vein thrombosis (DVT) and pulmonary embolism (PE), is a chronic illness that affects nearly 10 million people every year worldwide. 1 Cancer is a strong provoking risk factor for VTE, cancer patients have a 4–7 times higher risk of VTE compared to non-cancer patients, 2 and 20% of the overall incidence of VTE is accounted by active cancer. 3 A prospective, multicenter cohort study of 1836 cancer patients in China showed that the incidence of VTE events was 11.2% (95% CI 9.8-12.7), including DVT (11.0%, 95% CI 9.6-12.5) and PE (0.2%, 95% CI 0-0.5), 4 which is extremely high. From another prospective cohort study in China, the incidence of VTE in cancer patients was as high as 36.8%. 5 Apart from high incidence, the occurrence of VTE is a prognostic factor in people with cancer. VTE can cause serious negative effects which is a major cause of both morbidity and mortality in patients with cancer, 6 in addition, cancer patients with VTE have a poorer prognosis than those without. A retrospective study evaluated the survival of 668 cancer patients with and without a diagnosis of VTE, and demonstrated significantly inferior survival rates in patients with VTE compared to patients without VTE (12% vs 36%, P < .001). 7 The anticoagulation efficacy and safety management of VTE in population with cancer is challenging because of the high risk of VTE recurrence and inherent risk of bleeding. 8 Patients with an active cancer have up to nine-fold increased risk for recurrent VTE, 9 and in a current real-world study of 3027 consecutive acute symptomatic VTE patients, the cumulative 5-year incidences of recurrent VTE and major bleeding were significantly higher in patients with active cancer compared to patients without cancer (recurrent VTE: 17.7% vs 8.6%, P < .001; major bleeding: 26.6% vs 9.3%, P < .001). 10 Central venous access devices are a critical instrument in the treatment and supportive care delivery for cancer patients. Central venous catheters (CVCs) are more frequently used in cancer patients as cancer patient care, and chemotherapy administration is being increasingly shifted to the outpatient setting.11–12 CVCs raise the risk of catheter-related thrombosis in patients with cancer, 13 and catheter-related thrombosis is a common complication of central venous access devices in cancer patients. 14 With increasing use of CVCs, the incidence of catheter-related thrombosis has been on the rise, and catheter-related thrombosis constitutes 10% of all DVT in adults and 50%–80% of all DVT among children. 15 In China, a retrospective study showed almost 8% of the 748 lung cancer patients had peripherally inserted central catheter-related thrombosis. 16 Moreover, a prospective, observational study of 233 cancer patients in China found catheter-related thrombosis occurred in 15.9% of the patients. 17 Catheter-related thrombosis may lead to pulmonary embolism and infection, as well as catheter failure and potential delays in treatment in cancer patients. In hospitalized patients, CVC correlated more significantly with upper extremity deep venous thrombosis (UEDVT), and patients with UEDVT had a higher prevalence of significant medical comorbidities and higher all-cause mortality on the index hospital admission. 18 CVC-VTEs are associated with an increase of bloodstream infections19–23 and thus cause additional morbidity and a substantial increase in health care costs. 24 Study of clinical course and complications of catheter and non-catheter-related UEDVT in patients with cancer showed that complication rates and PE were significantly lower in patients with catheter-related thrombosis compared to patients with non-catheter-related thrombosis (27.7%vs55%, P < .05). 25 However, for cancer patients, fewer study is available on catheter-related venous thromboembolism (CRVTE) compared to non-catheter-related venous thromboembolism (NCRVTE), and whether there were differences in survival after VTE and anticoagulation efficacy and safety between CRVTE and NCRVT are unknown. Accordingly, we investigated and compared the VTE recurrence, bleeding events during anticoagulation therapies and survival rates at 1 year, 2 years and 3 years after VTE between CRVTE and NCRVTE in cancer patients, and the risk factors of the anticoagulation efficacy and safety, and survival after VTE were also analyzed, which is of great meaning for the VTE prevention and treatment in cancer patients.
Materials and Methods
After appropriate approval of the ethics committee, clinical data between 1 January 2010 and1 January 2020 were extracted from the electronic medical record database of our hospital including demographics, diagnosis, medical history, radiology reports, cancer-related treatments (chemotherapy, radiotherapy and surgery), catheterization and catheters, and anticoagulation therapies of VTE. Consecutive cancer patients with a diagnosis of VTE including DVT and PE were eligible for our study. All the patients were unique. However, splanchnic thrombosis, cancer history or combined with other cancers, tumor thrombus, venous disease such as varicose veins, PE alone, and incomplete clinical data were excluded. VTE recurrence, bleeding events (major or nonmajor bleeding), and survival at 1, 2, and 3 years after VTE were also recorded.
CRVTE was defined as Venous thrombosis that occurred after catheterization and around the catheters. 26 NCRVTE mainly included lower extremity DVT (LEDVT) and upper extremity DVT (UEDVT). VTE recurrence was defined as an increase in thrombosis or embolism at a new site. Major bleeding event was defined as bleeding event that (1) was related to death, (2) took place at a fatal site (intracranial, intraocular, retroperitoneal, intraspinal, or pericardial), or (3) required a transfusion of at least 2 units of packed red blood cells or led to a hemoglobin decrease of at least 2.0 g/dL. 27 Nonmajor bleeding was defined as any overt bleeding that did not fulfill the criteria for major bleeding but resulted in medical attention, unappointed visits, a discontinuance of anticoagulants, or a decrease in daily activities. 28
Statistical Analysis
Patients included in our study were divided into CRVTE and NCRVTE groups, and the baseline characteristics of the 2 groups including gender, age, cancer types, hypertension, diabetes, heart diseases, chemotherapy, radiotherapy, surgery and anticoagulation therapies were analyzed. Age of the CRVTE and NCRVTE groups was shown as median and quartiles (Q1 and Q3) and was analyzed by Kruskal-Wallis H Test, and the frequency of other baseline characteristics were calculated and analyzed by Chi-square test or Fisher's exact test.
The frequency of the VTE recurrence, bleeding events of the 2 groups were analyzed by Chi-square test or Fisher's exact test. Logistic regressions were performed to determine the independent risk factors for VTE recurrence and bleeding events of the VTE anticoagulation treatment . Furtherly, for CRVTE, the VTE recurrence was compared between the catheter removal and not by Chi-square test or Fisher's exact test. The independent risk factors for VTE recurrence was also determined by Logistic regressions. The odds ratios (OR) and 95% confidence intervals (CI) were estimated from the regression coefficients. The most clinically relevant variables and those with P values < .1 in the univariate analyses of every subgroup were introduced in the logistic regression analyses.
Survival rates at 1 year, 2 years and 3 years after VTE were estimated using standard Kaplan-Meier methods and compared by log-rank significance test between the CRVTE group and NCRVTE group. Multivariable Cox regressions were performed to determine the independent prognostic factors for 3-year survival after VTE in cancer patients.
All statistical analyses were performed using Statistical Package for Social Science (SPSS) version 22.0 (IBM Corp., Armonk, NY, USA), and an alpha level of 0.05 was considered significant.
Results
Totally, 263 patients were enrolled (see Figure 1 for the flow diagram) and the patients’ characteristics were shown in Table 1, and the cancer types included in our study were shown in Table 2. There were 5 types of anticoagulation therapies: low-molecular-weight heparin (LMWH), warfarin, new type of direct oral anticoagulants (NDOACs), LMWH combined with warfarin, and LMWH combined with NDOACs.
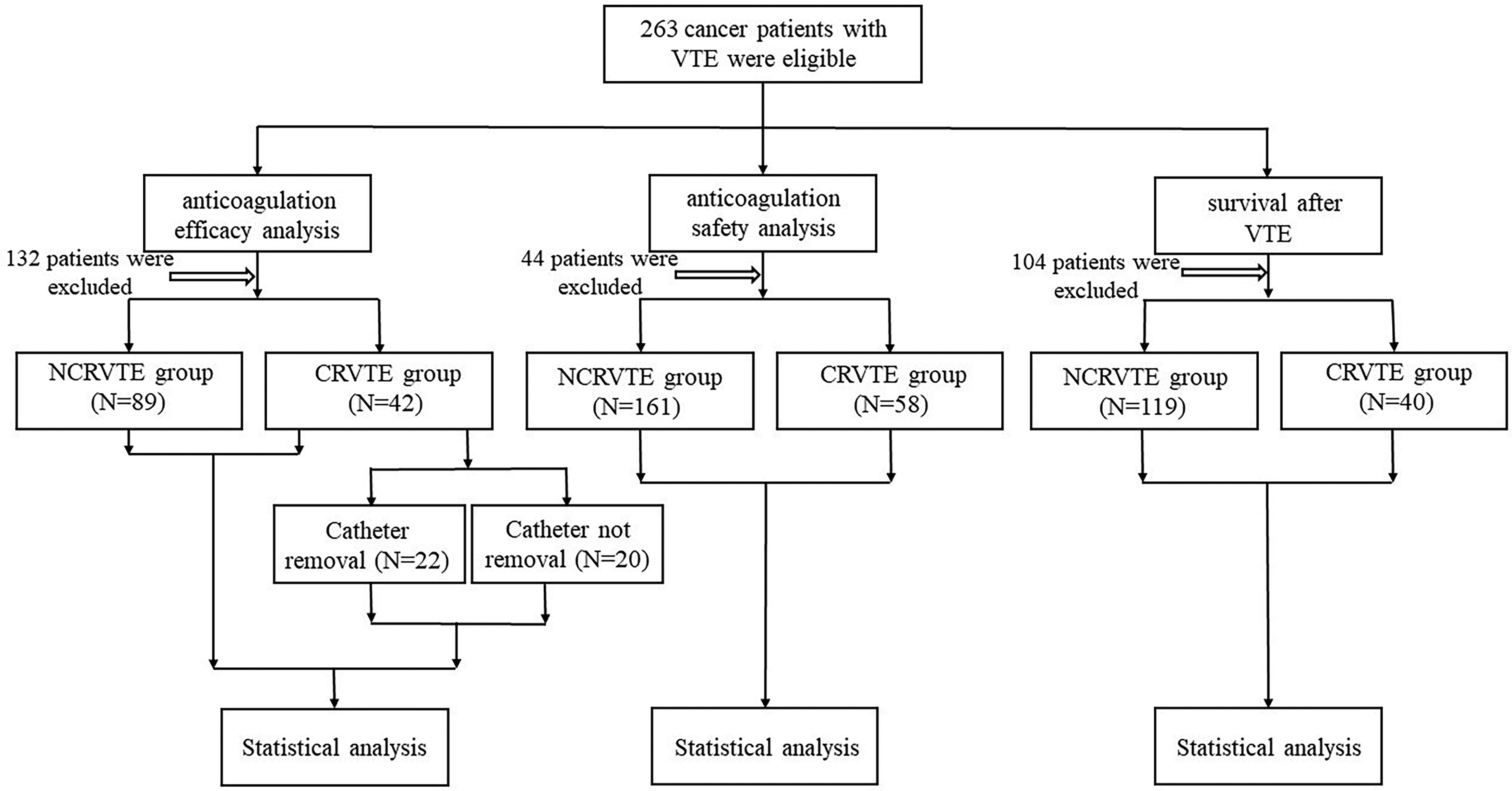
Patients’ flow diagram.
The Baseline Characteristics of the Patients Enrolled.
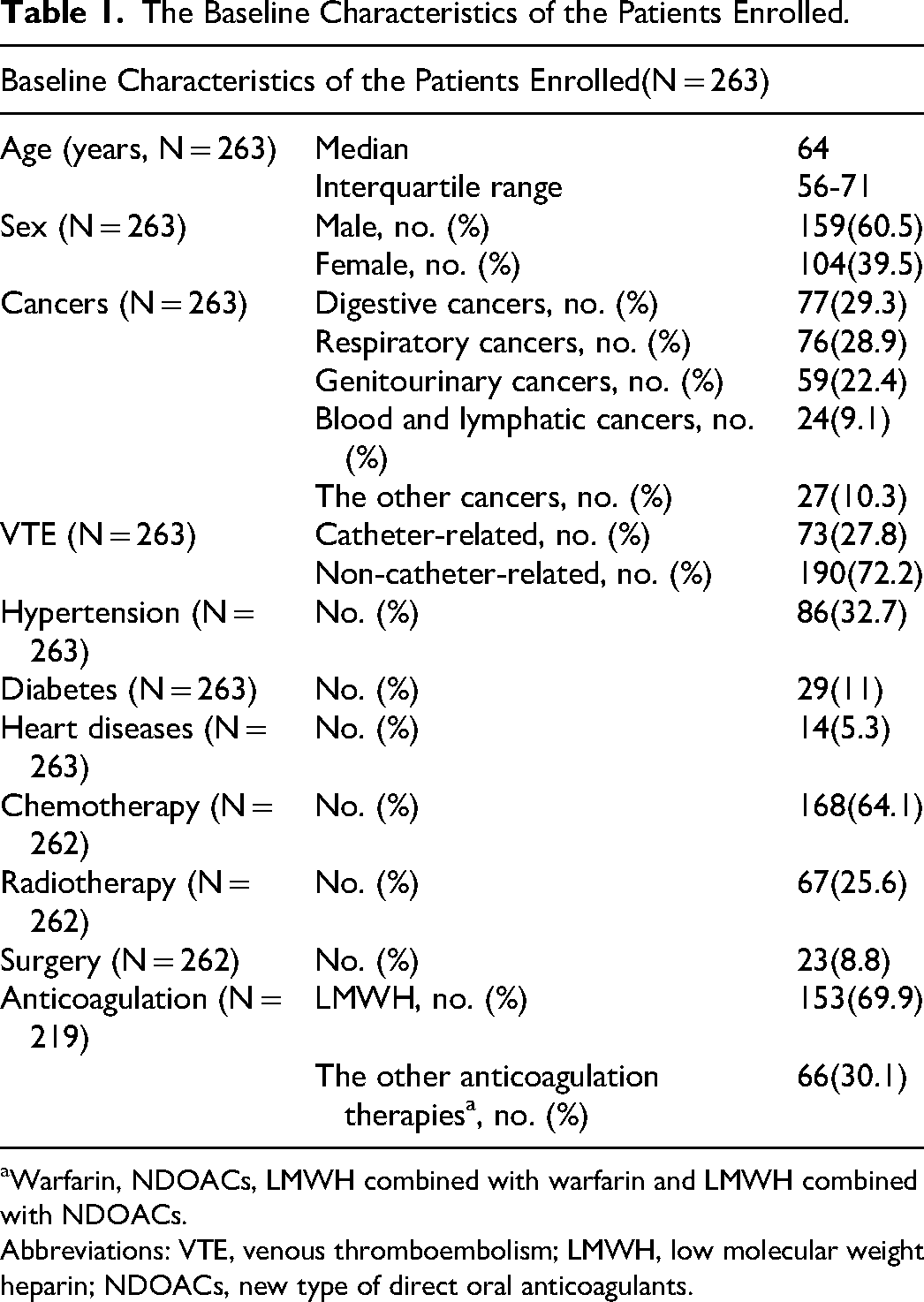
Warfarin, NDOACs, LMWH combined with warfarin and LMWH combined with NDOACs.
Abbreviations: VTE, venous thromboembolism; LMWH, low molecular weight heparin; NDOACs, new type of direct oral anticoagulants.
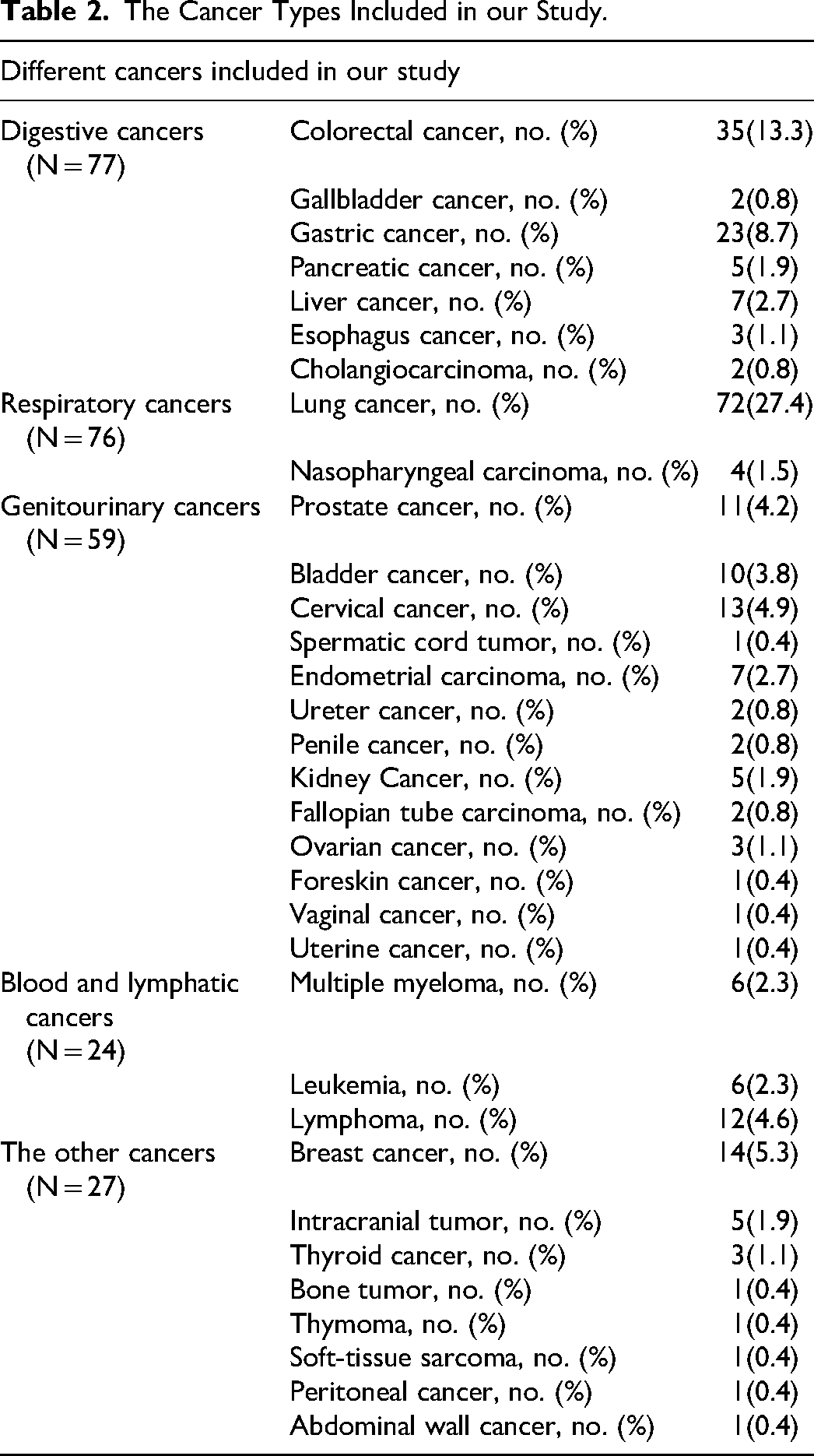
The Cancer Types Included in our Study.
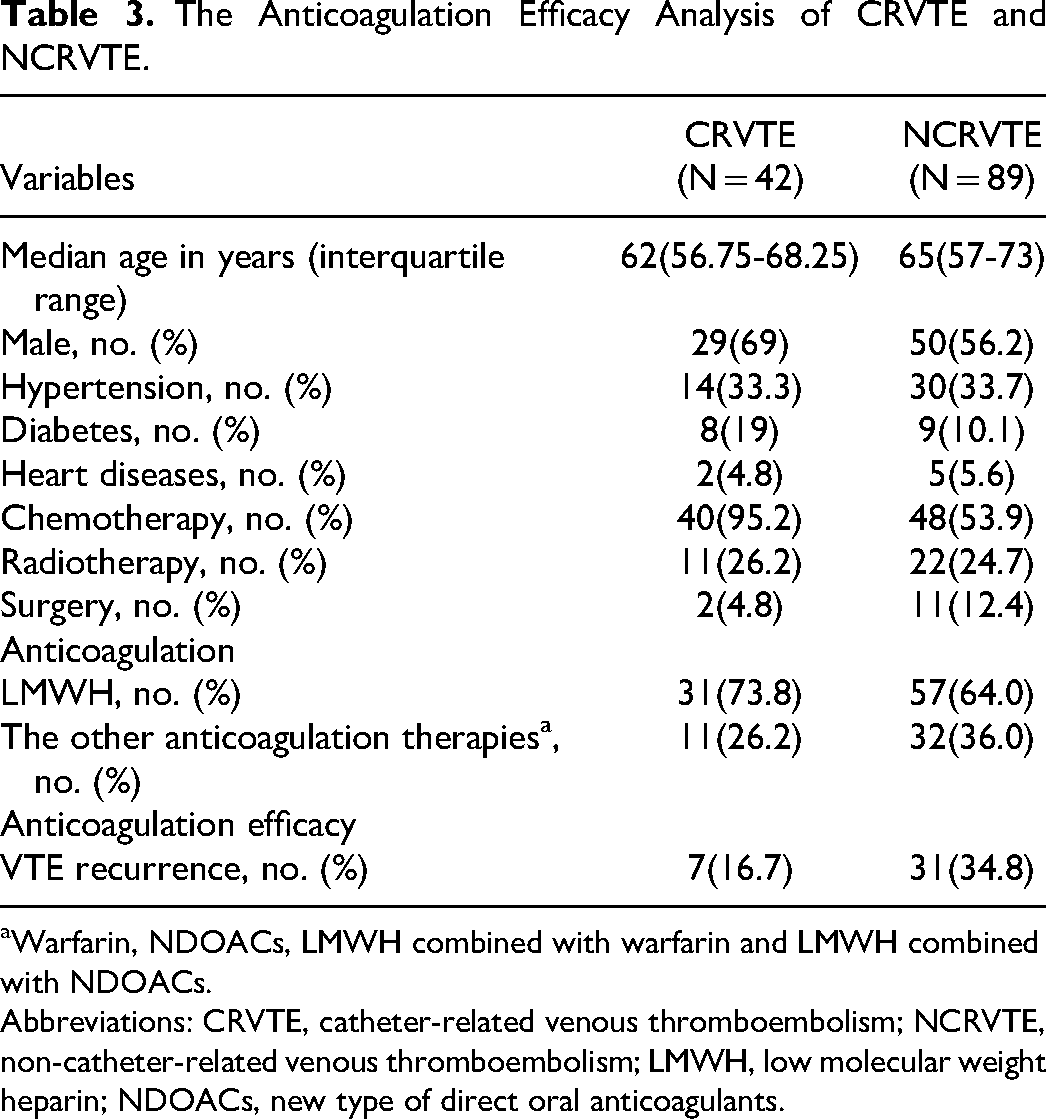
In the anticoagulation efficacy analysis, 132 patients were excluded because of the incomplete clinical data of anticoagulation therapies and VTE recurrence, 131 patients were included (Table 3) and there were no significant differences (P > .05) in the baseline characteristics between CRVTE group (N = 42) and NCRVTE group (N = 89), except for that the frequency of chemotherapy in CRVTE group was significantly higher than that in NCRVTE group(P < .01). The VTE recurrence of CRVTE was significantly lower compared to NCRVTE(P = .032). Logistic regression analysis using age, sex, hypertension, diabetes, heart diseases, chemotherapy, radiotherapy, surgery and anticoagulation therapies as variables showed that only heart diseases were independently associated with the VTE recurrence (P = .025). Patients without heart diseases were more likely to have lower VTE recurrence, compared to patients with heart diseases (OR 6.894, 95%CI 1.275-37.268).
The Anticoagulation Efficacy Analysis of CRVTE and NCRVTE.
Warfarin, NDOACs, LMWH combined with warfarin and LMWH combined with NDOACs.
Abbreviations: CRVTE, catheter-related venous thromboembolism; NCRVTE, non-catheter-related venous thromboembolism; LMWH, low molecular weight heparin; NDOACs, new type of direct oral anticoagulants.
The baseline characteristics of the catheter removal and catheter not removal groups were not significantly different, and there was no significant difference in the VTE recurrence between catheter removal and catheter not removal groups (P = .414) (Table 4). Logistic regression analysis using age, sex, hypertension, diabetes, heart diseases, chemotherapy, radiotherapy, anticoagulation therapies and catheter removal or not as variables showed that none of the variables were independently associated with the VTE recurrence.
The anticoagulation efficacy analysis of CRVTE between the catheter removal and not.
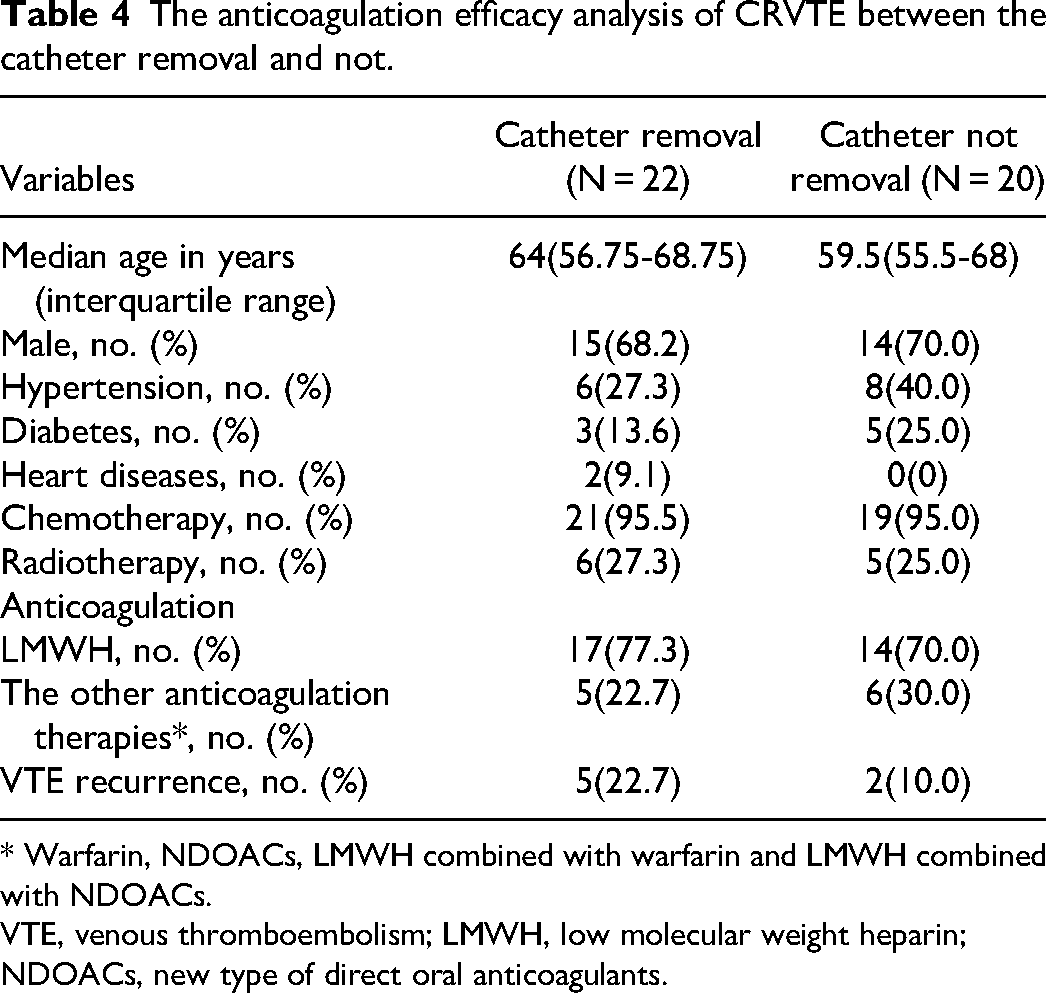
* Warfarin, NDOACs, LMWH combined with warfarin and LMWH combined with NDOACs.
VTE, venous thromboembolism; LMWH, low molecular weight heparin; NDOACs, new type of direct oral anticoagulants.
In the anticoagulation safety analysis, totally 219 patients were included (CRVTE group, N = 58; NCRVTE group, N = 161) (Table 5). No significant differences were found in the baseline characteristics between the 2 groups, except for age (P = .008), diabetes(P = .035) and chemotherapy(P < .01). The bleeding events were not significantly different between CRVTE and NCRVTE (P = .417), and ordinal logistic regression analysis using age, sex, hypertension, diabetes, heart diseases, chemotherapy, radiotherapy, surgery and anticoagulation therapies as variables showed that only anticoagulation therapy type was independently associated with the bleeding risk (P = .030), and compared to the other anticoagulation therapies in our study, LMWH was more likely to induce less bleeding events (OR 3.111, 95%CI 1.116-8.667).
The anticoagulation safety analysis of CRVTE and NCRVTE.
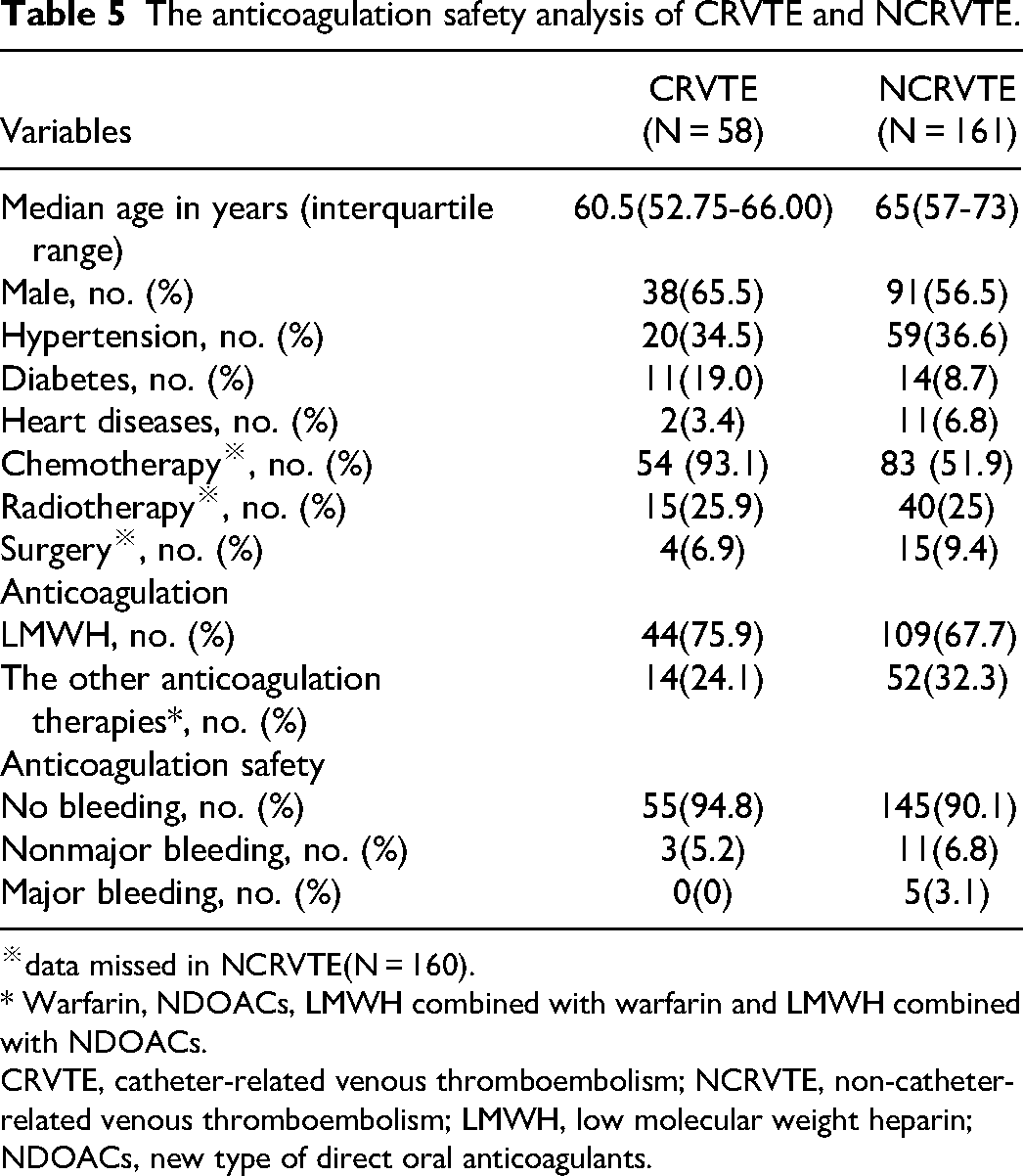
※data missed in NCRVTE(N = 160).
* Warfarin, NDOACs, LMWH combined with warfarin and LMWH combined with NDOACs.
CRVTE, catheter-related venous thromboembolism; NCRVTE, non-catheter-related venous thromboembolism; LMWH, low molecular weight heparin; NDOACs, new type of direct oral anticoagulants.
159 patients were included in the survival comparation between CRVTE (N = 40) and NCRVTE groups (N = 119), (Table 6). There were no significant differences in the baseline characteristics between the 2 groups (P > .05). Survival estimates for the cancer patients with CRVTE and NCRVTE at 1 year after VTE were 62.5% (95% CI, 47.4% to 77.6%) and 47.9% (95% CI, 38.9% to 56.9%), respectively (P = .130). The survival rate at 2 years after VTE was 60.0% (95% CI, 44.9% to 75.1%) for the cancer patients with CRVTE versus 38.7% (95% CI, 29.9% to 47.5%) for the cancer patients with NCRVTE (P = .028). At 3 years, the survival rate was 47.5% (95% CI, 32.0% to 63.0%) for the cancer patients with CRVTE and 30.1% (95% CI, 21.9% to 38.3%) for the cancer patients with NCRVTE (P = .046) (Figure 2). Multivariable Cox regression analysis using age, sex, hypertension, diabetes, heart diseases, chemotherapy, radiotherapy, surgery, anticoagulation therapies (LMWH or the other anticoagulation therapies), VTE types (CRVTE or NCRVTE) and cancer types (digestive cancers, respiratory cancers, genitourinary cancers, blood and lymphatic cancers and the other cancers as variables showed that only surgery (P = .003), anticoagulation therapy types (P = .009), VTE types (P = .006) and cancer types (P = .039) were independent prognostic factors for 3-year survival after VTE in cancer patients (Table 7). Compared to no surgery, patients with surgery were more likely to have lower death rate (HR 0.171, 95%CI 0.053-0.549). Compared to LMWH, death rate with the other anticoagulation therapies was more likely to be lower (HR 0.521, 95%CI 0.319-0.851). 3-year death rate after NCRVTE was more likely to be higher compared to CRVTE (HR 2.177, 95%CI 1.255-3.775), and for cancer types, patients with the other cancers were more likely to have better 3-year survival after VTE.
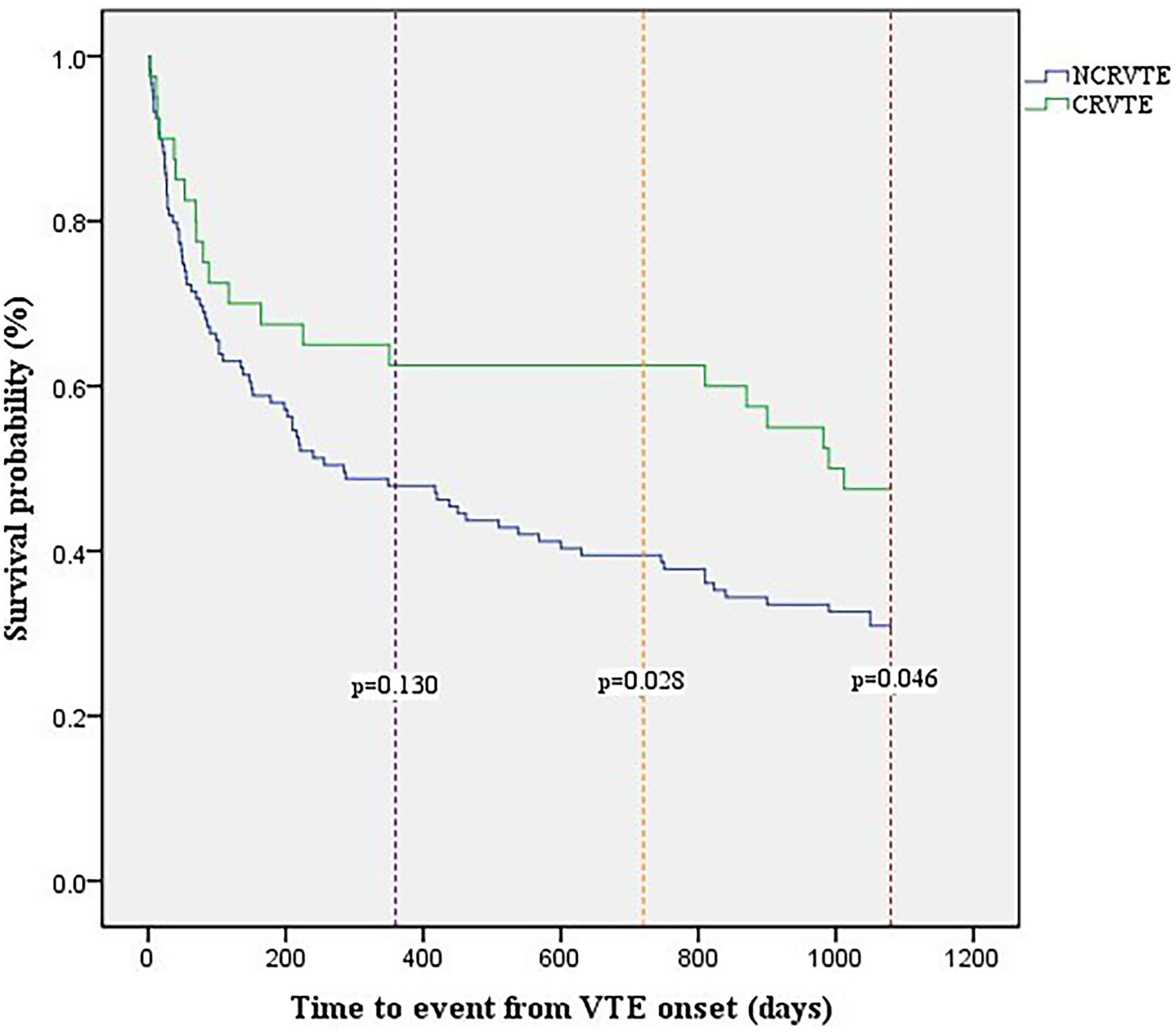
Kaplan-Meier survival curves for cancer patients with CRVTE and NCRVTE.
The Survival Analysis of CRVTE and NCRVTE.
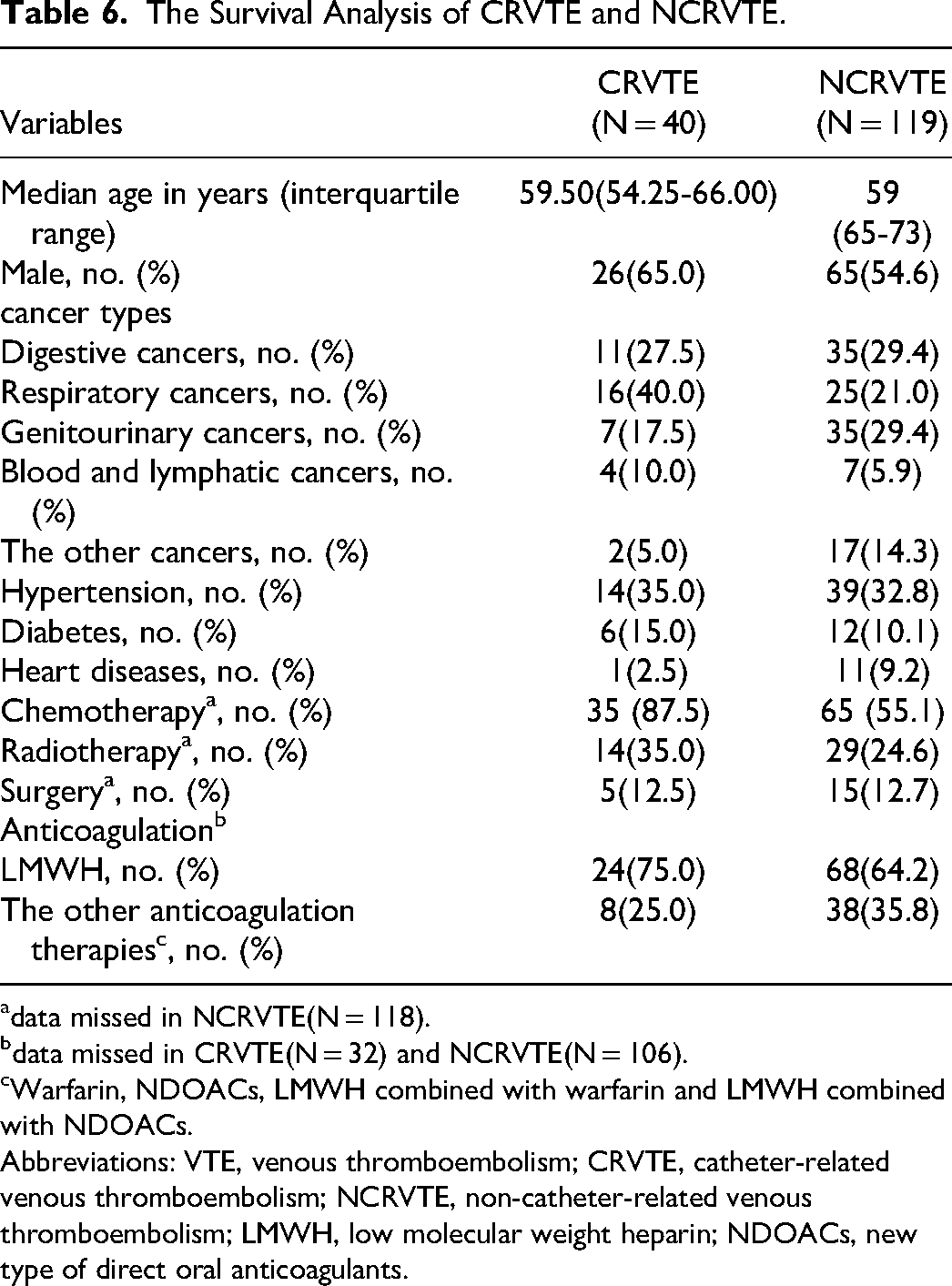
data missed in NCRVTE(N = 118).
data missed in CRVTE(N = 32) and NCRVTE(N = 106).
Warfarin, NDOACs, LMWH combined with warfarin and LMWH combined with NDOACs.
Abbreviations: VTE, venous thromboembolism; CRVTE, catheter-related venous thromboembolism; NCRVTE, non-catheter-related venous thromboembolism; LMWH, low molecular weight heparin; NDOACs, new type of direct oral anticoagulants.
Prognostic Factors for 3-Year Survival of Cancer Patient with VTE.
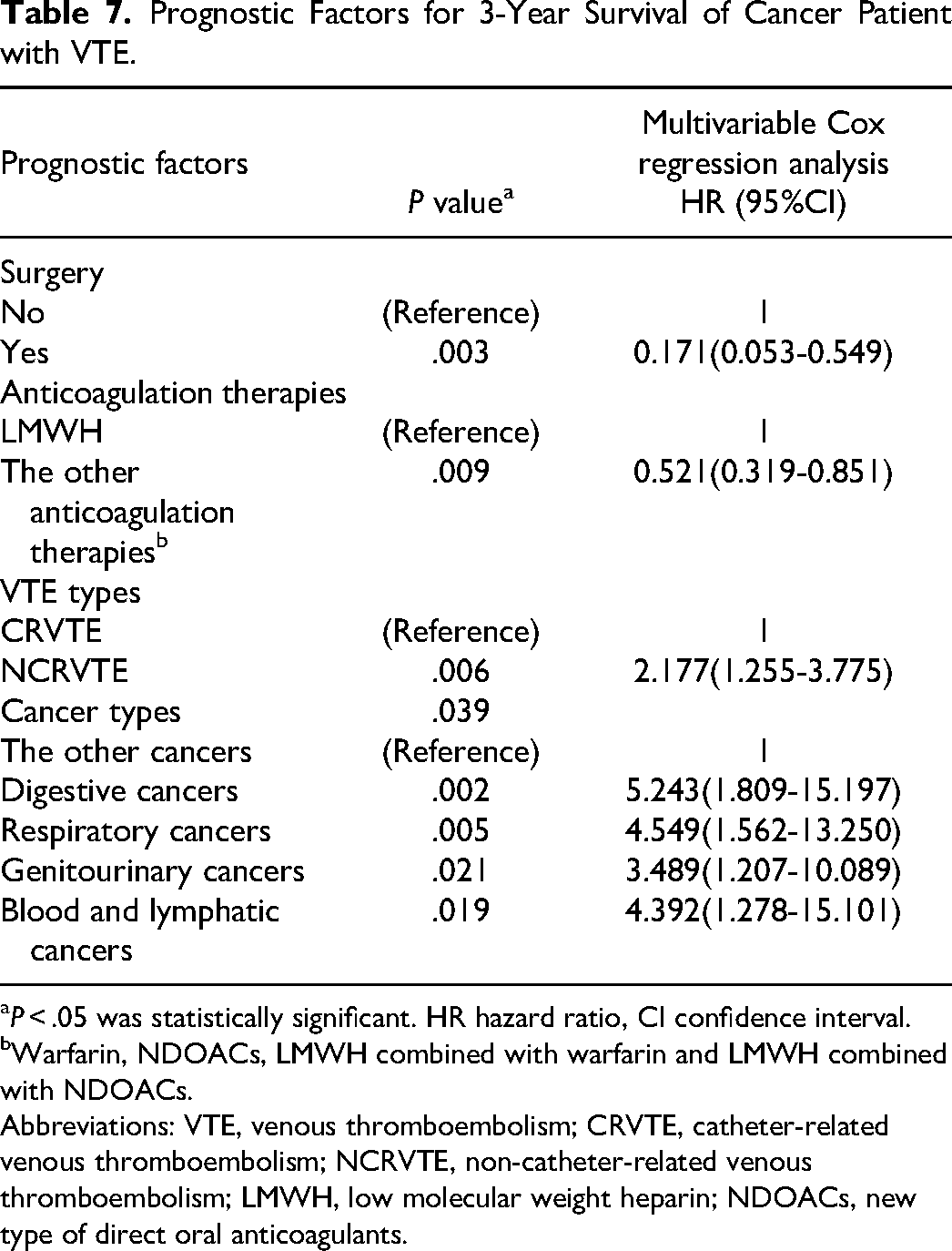
P < .05 was statistically significant. HR hazard ratio, CI confidence interval.
Warfarin, NDOACs, LMWH combined with warfarin and LMWH combined with NDOACs.
Abbreviations: VTE, venous thromboembolism; CRVTE, catheter-related venous thromboembolism; NCRVTE, non-catheter-related venous thromboembolism; LMWH, low molecular weight heparin; NDOACs, new type of direct oral anticoagulants.
Discussion
Catheter removal or not may be an important factor confounding anticoagulation efficacy, to address this, this study showed that there was no significant difference in the VTE recurrence between catheter removal and not. VTE recurrence of CRVTE was significantly lower compared to NCRVTE, and similar to our study, Mansour A et al 25 also found that VTE complications in cancer patients with NCRVTE were significantly higher compared to CRVTE. It probably because the in place of catheters and more proximal site make CRVTE more readily identified, which leads to earlier diagnoses of incidental VTE at an earlier stage. Moreover, maybe other pathophysiologic factors of the cancers play a more significant role in promoting VTE than the mere presence of a catheter does. Heart diseases were significant risk factor for VTE recurrence in cancer patients, which is in concordance with reported literatures.29–30 Hypercoagulability, immobilization, swelling caused by heart diseases, and co-existing risk factors like smoking and obesity increase VTE recurrence.
Bleeding is another extremely important outcome of VTE anticoagulation treatment in cancer patients. Currently, bleeding evaluation studies of VTE anticoagulation treatment in cancer patients mainly focus on the anticoagulation types,31–32 whether the VTE types (CRVTE and NCRVTE) make a difference is unknown. In our study, although nonmajor and major bleeding incidences of CRVTE during anticoagulation treatment were lower compared to NCRVTE (nonmajor bleeding: 5.2% vs 6.8%; major bleeding: 0% vs 3.1%), however, no significant difference was found (P = .417). A review of RIETE Registry also showed no significant difference in major bleeding between UEDVT including CRVTE and LEDVT, 33 which to some extent is consistent with our study. Logistic regressions showed a significant association between the anticoagulation type and bleeding events, and the bleeding events risk with LMWH was more likely to be lower, compared to the other anticoagulation therapies. Study of cancer patients with catheter-related thrombosis suggested a lower bleeding risk with LMWH compared to warfarin 34 which is in line with our study. Previous studies showed that the risk of bleeding was similar among LMWH, warfarin and NDOACs,35–38 which seemed conflict with our study, however, different from the reported studies, in our study more types of anticoagulation therapies were included, and combination of LMWH and warfarin or NDOACs may increase the bleeding risk compared to LMWH alone.
Although there was no significant difference in 1-year survival, 2-year and 3-year survival after CRVTE were both significantly better compared to NCRVTE. VTE complications including PE, superior vena cava syndrome and post-thrombotic syndrome are significant contributors to the morbidity and mortality. 39 From the reported literature, VTE complications including PE were significantly lower in CRVTE compared to NCRVTE (55% vs 27.7%), and overall survival was toward worse in patients with VTE complications compared to without, 25 and this provides an explanation for the survival results in our study. For the prognostic factors results for 3-year survival, the stages of cancers may be another important factor. Surgery is usually the initial therapy of cancers at early-stage. 40 And, it was found that most of the cancer patients with NCRVTE had advanced-stage cancer. 25 Anticoagulation therapies have antitumor effects, and evidence suggests that LMWH has antitumor effects,41–42 and can improve overall survival of cancer. 43 Warfarin was also found to be associated with an improvement in cancer survival and has potential as an anti-cancer agent. 44 Consequently, the other anticoagulation therapies including LMWH combined with warfarin may introduce better survival compared to LMWH alone.
Limitations
Although the differences in the baseline characteristics between CRVTE and NCRVTE groups during anticoagulation efficacy and safety analysis were addressed by logistic regressions, several limitations of our study should be noted. Most important is the relatively small number of patients. Selection bias may be caused between patients with CRVTE and NCRVTE, because catheterization or not is up to the differences of the disease conditions. Although patients enrolled all have a diagnosis of cancer, considerable heterogeneity may also exist related to different cancer stages, different cancer sites, and different anticancer treatment details administered. And, the dose of the anticoagulation treatment is not investigated, which may can also affect the anticoagulation efficacy and safety.
Conclusions
Cancer patients with CRVTE potentially have a better anticoagulation efficacy and survival compared to NCRVTE, and the anticoagulation safety seems no significant difference. Heart diseases are the significant risk factor for VTE recurrence, and anticoagulation therapy type is significantly associated with bleeding events, and, surgery, anticoagulation therapy type, VTE types, and cancer types are independent prognostic factors for 3-year survival after VTE in cancer patients.
Footnotes
Contributorship
Conceptualization, F.L., M.C. and X.Y.; methodology, F.L., K.S., H.W. and L.J.; Writing – original draft, F.L; Writing, review and editing, F.L. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of Zhejiang Provincial People's Hospital, Affiliated People's Hospital, Hangzhou Medical College (ZJPPHEC 2022O(306)).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Medical and Health Science and Technology Project of Zhejiang Province, Traditional Chinese Medicine Science and Technology Project of Zhejiang Province, (grant number 2023KY470, 2022ZB025).
