Abstract
Background
Sepsis-Induced Coagulopathy (SIC) is common complications of sepsis in intensive care unit (ICU) patients. It has a significant impact on the prognosis of patients with sepsis and is an important cause of death. However, there is no evidence for the use of Argatroban in SIC anticoagulant therapy.
Method
A retrospective study was conducted based on data collected from patients with sepsis from the Medical Information Mart for Intensive Care IV (MIMIC-IV) database. SIC patients were screened according to SIC scores formulated by the International Society of Thrombosis and Hemostasis. Propensity score matching was used to match patients who received Argatroban with patients who did not receive Argatroban. Cox regression analysis was used to explore the correlation between Argatroban and SIC prognosis, and the hazard ratio was calculated. Finally, Kaplan-Meier curve was used to compare the survival rate between the two groups.
Outcome
After PSM, 108 patients who received Argatroban were matched with 108 patients who did not receive Argatroban. In terms of outcomes, Argatroban admission to ICU was associated with a higher risk of death 30 days after ICU admission for SIC patients (HR = 1.800; 95%CI: 1.064∼3.043; P = .043) correlation.
Conclusion
Argatroban is not recommended for anticoagulant therapy in SIC patients.
Keywords
Background
Sepsis-Induced Coagulopathy (SIC) is a common complication of sepsis. SIC usually manifests as abnormal blood clotting function, and in severe cases can lead to multiple organ dysfunction and even death, 1 it can increase the risk of death.2,3 The incidence of SIC may vary in different regions, but the incidence is higher in patients with sepsis. 4 According to the survey, about 30% to 60% of patients with sepsis will have coagulation dysfunction. This means that in people with sepsis, about one-third of cases will develop SIC. If SIC management is not effective, it can further develop into disseminated intravascular coagulation (DIC).5,6
The treatment of SIC mainly includes infection control, fluid resuscitation, anticoagulation therapy and so on. In terms of anticoagulant therapy, heparin is the first choice of anticoagulant drugs for SIC patients.7,8 There are two types of anticoagulation indications in SIC patients. One is to initiate anticoagulation when sepsis patients meet SIC diagnostic criteria. 9 The purpose of initiating anticoagulation at this time is to protect vascular endothelium, reduce clotting substrate consumption, inhibit thrombosis and improve organ blood supply. Second, anticoagulation should be initiated when sepsis patients have definite thrombotic complications. 10 A multicenter retrospective analysis in Japan found that anticoagulant therapy only improved the prognosis of SIC patients and did not benefit all sepsis patients. 11
In view of the differences in the effectiveness of different anticoagulants,12–14 and the optimal treatment timing, dosage and duration of different drugs are different. 15 Therefore, it is necessary to further study the application of anticoagulant drugs in SIC. At present, there are no reports on Argatroban in the treatment of SIC.16,17 In order to fill this gap, this study aimed to explore whether Argatroban can improve the prognosis of SIC patients through MIMIC database.
Database
Medical Information Mart for Intensive Care IV Version 3.0 (MIMIC-IV -V3.0) is collected at Beth Israel Deaconess Medical Center between 2008 and 2022 (BIDMC) Patient information, which includes personnel information, laboratory tests, and other records, can be used to evaluate medical procedures, 18 and evaluate the influence of different factors on the prognosis of patients. 19 Data were entered and extracted according to the ninth International Classification of Diseases (ICD-9) and tenth Revised code (ICD-10). 20 One of the authors of this study (Jiaqi Tang) completed the CITI Data or modules Only Research course (Record ID: 12467279) and gained access to the MIMIC database. Since the database records patients’ personal information in an anonymous manner, there is no requirement for ethical consent. 21
Study Population
Identify patients with sepsis in the database using the diagnostic criteria for Sepsis 3.0; According to the SIC scoring system formulated by the International Society of Thrombosis and Hemostasis (ISTH), SIC patients were screened according to the total score of SIC scoring system ≥4 points. 22 Because of its simplicity and practicality, SIC scoring systems are widely used in retrospective studies.23–27
Inclusion criteria: 1. Sepsis patients admitted to ICU for the first time; 2. ISTH-SIC>= “4 point.
Exclusion criteria: 1. Patients who were not admitted to ICU for treatment; 2. Patients not entering ICU for the first time; 3. Not a patient with sepsis; 4. ISTH-SIC < 4 points; 5. Age <18 years old
Data Extraction and Processing
In this study, Navacit tool was used for data extraction and management, and the data of SIC patients were screened according to the above inclusion and exclusion criteria, and the use of agatriban during the hospitalization of patients was extracted. The outcome is the patient's 30-day mortality after ICU admission (“ prognosis” below refers to the patient's 30-day prognosis after ICU admission). Subsequently, the extracted data was cleaned and encoded, and imported into IBM SPSS statistical software version 25.0 for analysis (Figure 1).
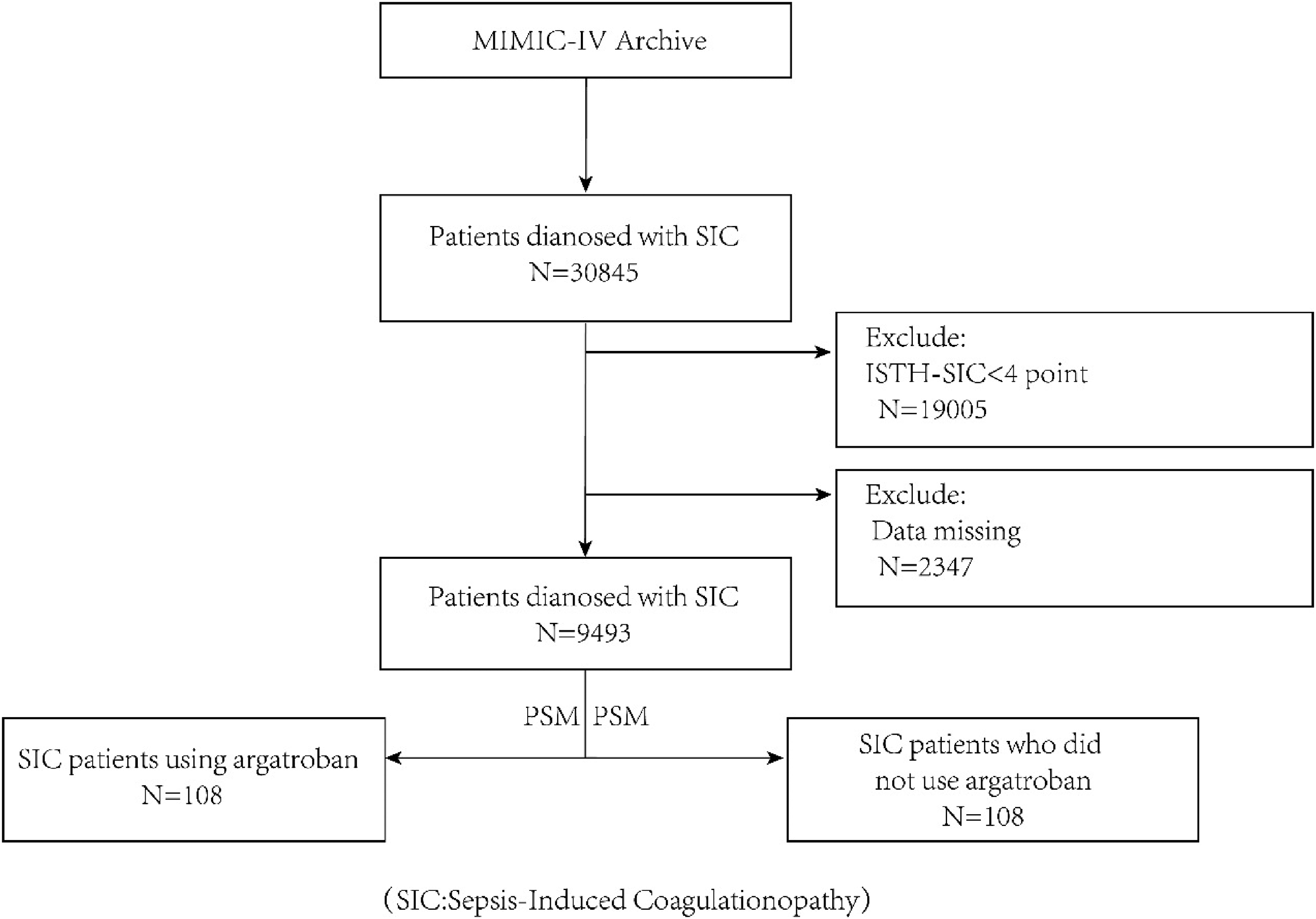
Depicts the extraction, cleaning, and screening process of research data.
Analysis Process
Based on the above inclusion and exclusion criteria, SIC patients and laboratory test information meeting our requirements were selected. Given that the ratio of patients using Argatroban to those not using Argatroban was too large (1:94), this study used propensity score matching (PSM) to narrow the difference in baseline data between the two groups of patients and obtain an approximate RCT effect.28–30
Subsequently, we conducted univariate Cox regression to preliminarily explore whether “use of Argatroban during ICU” had an impact on the prognosis of SIC patients. Multivariate Cox regression analysis was performed for significant variables, and a two-tailed P < .05 was considered statistically significant, and the hazard ratio (HR) was calculated. Finally, Kaplan-Meier curve was used to clarify the effect of Argatroban on the prognosis of SIC patients in different use contexts.
Results
Population Characteristics
A total of 9493 SIC patients (without PSM) met ISTH-SIC score 4 by screening and cleaning. There were 5894 males (60.1%); There were 6510 (68.6%) patients aged over 60 years. There were 6354 white Americans (66.9%), 283 Asian Americans (3.0%), and 763 black Americans (8.0%). In terms of complications, 94 SIC patients had pulmonary embolism (<1%); 160 patients had DIC (1.7%); There are 5955 patients (62.7%) have a coagulation system score greater than or equal to 1 point on the SOFA score scale, that is, a platelet count less than 150*103/ µl.
COX Regression Analysis
After PSM, 108 patients who used Argatroban during ICU were matched with 108 patients who did not receive Argatroban. Univariate and multivariate COX regression showed that Argatroban use during ICU increased 30-day mortality in patients with SIC (Table 1, Figure 2). Due to the small number of “the first day of ICU use of argatroban” and “the first day of hospital use of argatroban”, the correlation between “the first day of ICU use of argatroban” and “the first day of hospital use of argatroban” and the prognosis of SIC patients was not studied.

Shows the forest map and Hazard ratio for “using argatroban during ICU”.
Results of Univariate and Multivariate cox Regression Analyses (Association of Argatroban use During ICU with 30-day Mortality in SIC Patients).

Abbreviations: HR, Hazard ratio; CI, confidence interval.
Kaplan-Meier Curve Reveals Survival Differences
In this study, Kaplan-Meier curve was drawn to further clarify the survival difference between Argatroban and SIC patients. 31 As shown in Figure 3 it can be concluded that “use of Argatroban during ICU” (HR:1.800, 95%CI:1.064-3.043, P = .025) was associated with 30-day survival after admission to ICU.

Difference in survival of SIC patients with or without argatroban during ICU.
Discussion
SIC is an important cause of endothelial injury and coagulation disorder caused by sepsis, and also an important cause of death in sepsis patients. Early identification and intervention of SIC can prevent SIC from progressing to DIC and multiple organ dysfunction, and anticoagulation therapy plays an important role in blocking this process. At present, the anticoagulant drug therapy for SIC is still controversial.32,33 In addition, there is no correlation between Argatroban and SIC at present. This study fills this gap with data from MIMIC-IV.
By analysis, we found that the risk of death on day 30 of ICU admission in patients who used Argatroban was 1.8 times higher than that of patients who did not use Argatroban. The risk of 30-day death was 5.1 times higher in patients who received Argatroban on the first day of hospitalization than in patients who did not receive Argatroban on the first day of hospitalization.
In animal models of organ dysfunction, unfractioned heparin (UFH), low molecular weight heparin (LMWH), and Argatroban have been reported to inhibit histone-induced leukopenia and thrombocytopenia, but only UFH and LMWH have been reported to reduce hepatic and renal dysfunction. Argatroban has no such effect. 34 Tanaka et al. confirmed that the use of Argatroban did not have an organ protective effect in a sepsis model. 35 It can be seen that Argatroban does not block or delay the process of SIC developing into multiple organ dysfunction. This may be the reason why Argatroban does not improve the prognosis of SIC patients.
Previous research confirmed that direct thrombin inhibitors (DTIs), such as Argatroban, promote thrombin production in the presence of thrombomodulin (TM).36,37 Studies have shown that the TM may be potential biomarkers of SIC. 38 Therefore, in order to improve the prognosis of SIC or DIC patients, it is necessary to inhibit thrombin production. 39 However, in SIC, vascular endothelial cells are damaged, leading to elevated TM levels.40,41 If used at this stage, Argatroban promotes thrombin formation, which leads to the progression of SIC and an increased risk of death.
From the above discussion, it can be found that in the presence of TM, Argatroban promotes the clotting process of SIC, leading to disease progression. Although anticoagulant therapy is an important part of the treatment of SIC patients, Argatroban is not suitable for the anticoagulant therapy of SIC patients. This suggests that the selection of appropriate anticoagulants is the key to improve the prognosis of SIC patients.
Although we have tried our best to improve our research protocol, due to certain objective conditions, our research still has the following limitations. First, the preliminary analysis of this study found that Argatroban decreased the 30-day survival rate of SIC patients after admission to ICU, so we did not conduct further clinical trials in our hospital for external validation. In addition, this study was a retrospective study based on MIMIC database, and the population was mostly white people in the United States. Therefore, the results of the study based on this database may not be applicable to the Eurasian and African populations, and further research is needed in other populations.
Conclusion
Although anticoagulant therapy is an important part of the treatment of SIC patients, our study shows that Argatroban does not improve the prognosis of SIC patients, but increases the risk of death. Therefore, this study does not recommend that clinicians use Argatroban for anticoagulant therapy in SIC patients.
Footnotes
Abbreviations
Acknowledgements
We appreciate the excellent work of MIMIC team (Massachusetts Institute of Technology Laboratory for Computational Physiology) to collect bedside data continuously and make the database available for every intensive care researcher.
Authors’ Contributions
JQT and ZWS participate in the data acquisition and revisions of data analysis and interpretation. HKD participate in study conception and design, data interpretation and revisions of the manuscript, HKZ supervise the whole study and provide financial support.
JQT and ZWS contributed equally to this article.
All authors provided their final approval for manuscript submission.
Availability of Data and Materials
Available upon request. Corresponding to Drs. HongKe Zeng (zenghongke@vip.163.com).
Consent for Publication
Not applicable
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The MIMIC-IV databases have received ethical approval from the Institutional Review Boards at Beth Israel Deaconess Medical Center and Massachusetts Institute of Technology. As the databases do not contain protected health information, a waiver of informed consent was included in the approval from the Institutional Review Boards at Beth Israel Deaconess Medical Center and Massachusetts Institute of Technology. Therefore, this manuscript does not involve a research protocol requiring approval by the relevant institutional review board or ethics committee. All methods in this study were carried out in accordance with relevant guidelines and regulations (declarations of Helsinki)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Guangdong Province (A1515012665).
