Abstract
Background
Currently, no universally accepted standardized VTE risk assessment model (RAM) is specifically designed for critically ill patients. Although the ICU-venous thromboembolism (ICU-VTE) RAM was initially developed in 2020, it lacks prospective external validation.
Objectives
To evaluate the predictive performance of the ICU-VTE RAM in terms of VTE occurrence in mixed medical-surgical ICU patients.
Methods
We prospectively enrolled adult patients in the ICU. The ICU-VTE score and Caprini or Padua score were calculated at admission, and the incidence of in-hospital VTE was investigated. The performance of the ICU-VTE RAM was evaluated and compared with that of Caprini or Padua RAM using the receiver operating curve.
Results
We included 269 patients (median age: 70 years; 62.5% male). Eighty-three (30.9%) patients experienced inpatient VTE. The AUC of the ICU-VTE RAM was 0.743 (95% CI, 0.682-0.804, P < 0.001) for mixed medical-surgical ICU patients. Comparatively, the performance of the ICU-VTE RAM was superior to that of the Pauda RAM (AUC: 0.727 vs 0.583, P < 0.001) in critically ill medical patients and the Caprini RAM (AUC: 0.774 vs 0.617, P = 0.128) in critically ill surgical patients, although the latter comparison was not statistically significant.
Conclusions
The ICU-VTE RAM may be a practical and valuable tool for identifying and stratifying VTE risk in mixed medical-surgical critically ill patients, aiding in managing and preventing VTE complications.
Introduction
Venous thromboembolism (VTE), including pulmonary embolism (PE) and deep vein thrombosis (DVT), ranks as the third leading vascular disease following acute myocardial infarction and stroke. 1 It represents a significant global disease burden, with approximately 10 million cases reported annually. 2 Critically ill patients face a heightened risk of VTE due to a combination of general risk factors and specific risk factors related to the intensive care unit (ICU), such as sedation, immobilization, vasopressor use, or central venous catheters. The incidence of VTE in ICU patients ranges from 25.0% to 37.2%, nearly doubling that of general medical patients.3–5 In addition, VTE is associated with an increased risk of mortality.6–8 Therefore, accurate identification of individuals at high risk of VTE in the ICU, coupled with the implementation of multiple preventive measures, may help prevent or delay the progression of venous thrombosis and improve prognosis. Consequently, early assessment of VTE risk in ICU patients appears to be particularly important.
The Caprini and Padua risk assessment models (RAMs) are the most widely used RAMs in medical9,10 and surgical patients,11,12 respectively. The ninth edition of the American College of Chest Physicians Antithrombotic Therapy and Prevention of Thrombosis guidelines (AT9) acknowledges the Caprini RAM for nonorthopedic surgical populations and the Padua RAM for medical patients.13,14 However, to date, there is no universally accepted standardized VTE RAM is designed specifically for critically ill patients. Viarasilpa et al 15 developed the ICU-VTE RAM, a predictive model based on six independent risk factors: central venous catheterization (5 points), immobilization ≥ 4 days (4 points), prior VTE (4 points), mechanical ventilation (2 points), lowest hemoglobin during hospitalization ≥ 90 g/L (2 points), and baseline platelet count > 250,000/μL (1 point). This model was derived from the data of 37,050 critically ill patients during hospitalization at the Henry Ford Health System. 15 It was utilized to predict the risk of venous thrombosis in critically ill patients and categorize them into three grades: low risk (0-8 points), intermediate risk (9-14 points), and high risk (15-18 points).
However, the ICU-VTE RAM has not undergone prospective evaluation, and its clinical significance requires further verification. Notably, the original study focused solely on symptomatic VTE and did not consider asymptomatic VTE. It would be reasonable to assess the accuracy of the ICU-VTE RAM in predicting both symptomatic and asymptomatic VTE. Therefore, we aimed to perform an external validation to assess the predictive performance of the ICU-VTE RAM for VTE occurrence and its ability to stratify VTE risk in mixed medical-surgical ICU patients.
Methods
Study Design and Participants
This single-center prospective study was conducted in the ICU of the Central Hospital of Wuhan. The trial is registered on ClinicalTrials.gov ((NCT05487066). Between May 2022 and October 2023, a total of 332 patients fulfilled the following inclusion criteria: aged 18 years or older, expected to stay in the ICU for more than 48 h, and volunteered and signed an informed consent form. Patients who met any of the following conditions were excluded: (1) had a diagnosis of VTE upon admission or within 24 h of ICU admission; (2) used therapeutic anticoagulation before or within 24 h of ICU admission; or (3) had a diagnosis of VTE without imaging confirmation. Ultimately, 269 patients were included in this study (
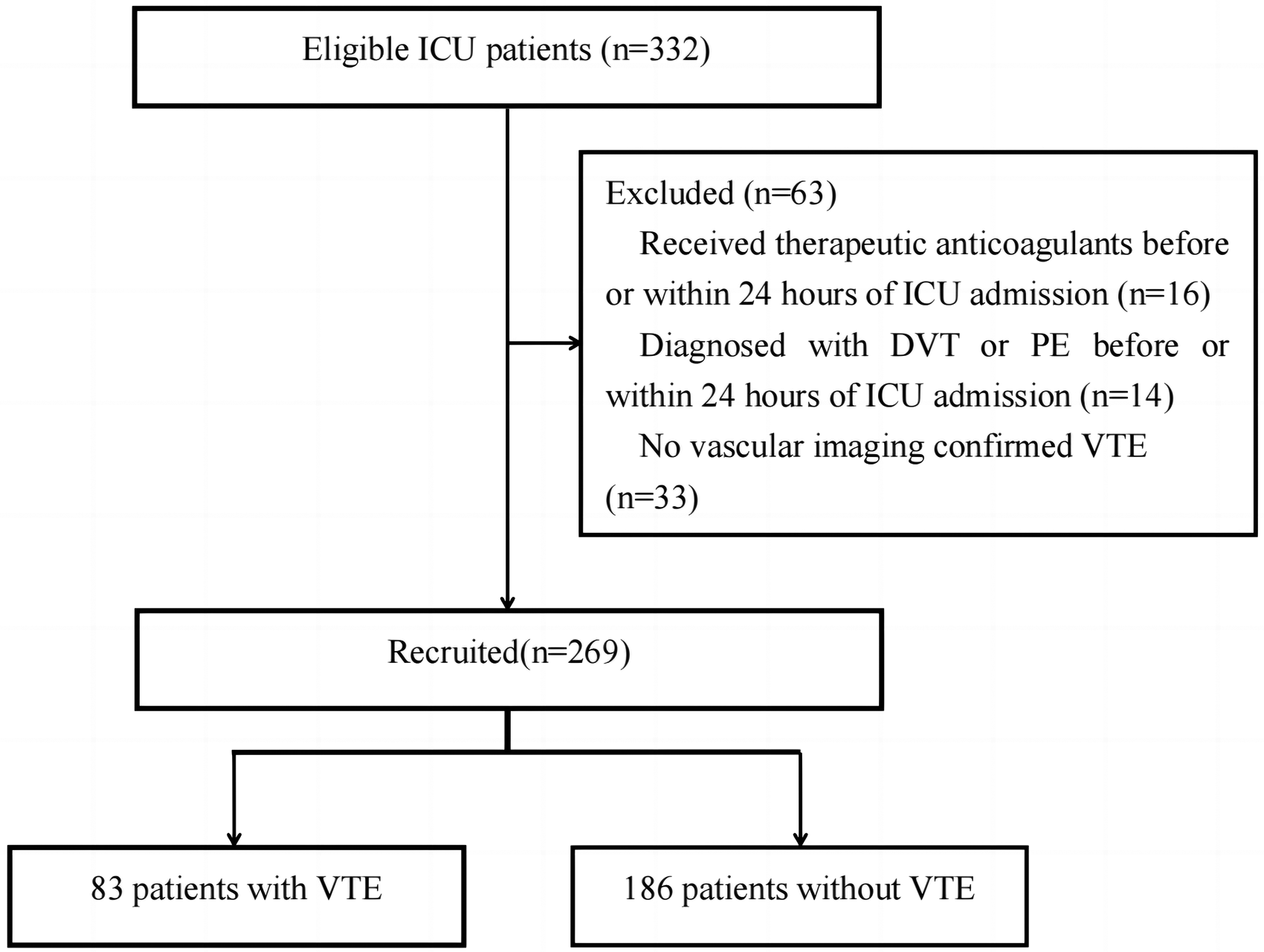
Flowchart of this study.
Procedures
The demographic characteristics, causes of ICU admission, comorbidities, and baseline laboratory results of patients who were included in the routine workup at ICU admission were collected. The Acute Physiology and Chronic Health Evaluation II (APACHE II) score, Padua score for medical patients, or Caprini score for surgical patients and the ICU-VTE score for all patients were determined within 24 h of admission. It should be noted that we collected hemoglobin values and evaluated for expected the immobilization ≥ 4 days at ICU admission to calculate the ICU-VTE score in our cohort. Enrolled patients were stratified into different risk levels based on their Caprini RAM score or Padua RAM score and received a nursing protocol for VTE prophylaxis. Patients classified as very low-risk according to the Caprini RAM and low-risk according to the Padua RAM were encouraged to ambulate early. In contrast, low-risk patients, according to the Caprini RAM, received mechanical prophylaxis, including graduated compression stockings (GCS) or intermittent pneumatic compression (IPC). For patients with moderate to high risk according to the Caprini RAM and high risk according to the Padua RAM, the physician determined the appropriate prophylaxis, whether mechanical, pharmacologic, or a combination of both, based on an assessment of bleeding risk using tools published by the National Programme for Prevention and Management of Pulmonary Embolism and Deep Venous Thrombosis. 16 Pharmacological thromboprophylaxis was considered adequate if initiated within 48 h of admission and covered >80% of the ICU stay. Routine duplex ultrasonography was performed for DVT surveillance, and CT pulmonary angiogram (CTPA) was performed to confirm the diagnosis of pulmonary embolism (PE). All VTE events were verified by a team of expert radiologists. We further followed the enrolled patients until they were discharged from the hospital. Additional clinical information on the treatments related to VTE risk was collected. During the follow-up period, the occurrence of VTE (including timing and type) and clinical outcomes were documented. The primary outcome was the occurrence of VTE during hospitalization. The secondary outcomes included all-cause mortality during hospitalization, ICU length of stay, and hospital length of stay. All the data were cross-checked and centrally validated.
Statistics Analysis
Statistical analysis was performed using the SPSS software package (version 26.0). The normality of continuous variables was assessed using the Kolmogorov-Smirnov test. Normally distributed continuous variables are reported as the means ± standard deviations (SD), and group comparisons were performed using the Student's t-test. Skewed distribution continuous variables are presented as medians and interquartile ranges (IQRs), and group comparisons were conducted using the Mann-Whitney U test. Categorical variables are presented as numbers and percentages and were compared using the Chi-squared test or Fisher's exact test. No variables were missing and no imputation methods were used in the analysis. The rates of in-hospital VTE, stratified by the ICU-VTE RAM, were analyzed. Forward stepwise logistic regression analysis was performed to identify risk factors for in-hospital VTE, and the results were expressed as odds ratios (ORs) with corresponding 95% confidence intervals (95% CIs). Considering that the limited number of VTE patients (n = 83) in our study might have resulted in overfitting in the model, only variables that were not included as factors in the ICU-VTE RAM and had a P value < 0.05 in our univariate regression model were entered into the multivariate logistic regression analysis. Receiver operating characteristic (ROC) curves were constructed to assess sensitivity and specificity, and the area under the ROC curve (AUC) was calculated using the GraphPad Prism statistical software (version 8). The AUCs were compared using the Delong–Delong test. P values < 0.05 (two-tailed) were considered to indicate statistical significance.
Results
Baseline Characteristics of the Study Subjects
The patients’ demographic and clinical characteristics at baseline are presented in
Baseline Characteristics of the Study.

Abbreviations: DVT, deep vein thrombosis; PE, pulmonary embolus; VTE, venous thromboembolism.
Predictors of in-hospital VTE
According to the univariate analysis (
Comparison of Clinical Characteristics Between Patients with VTE and Without VTE.
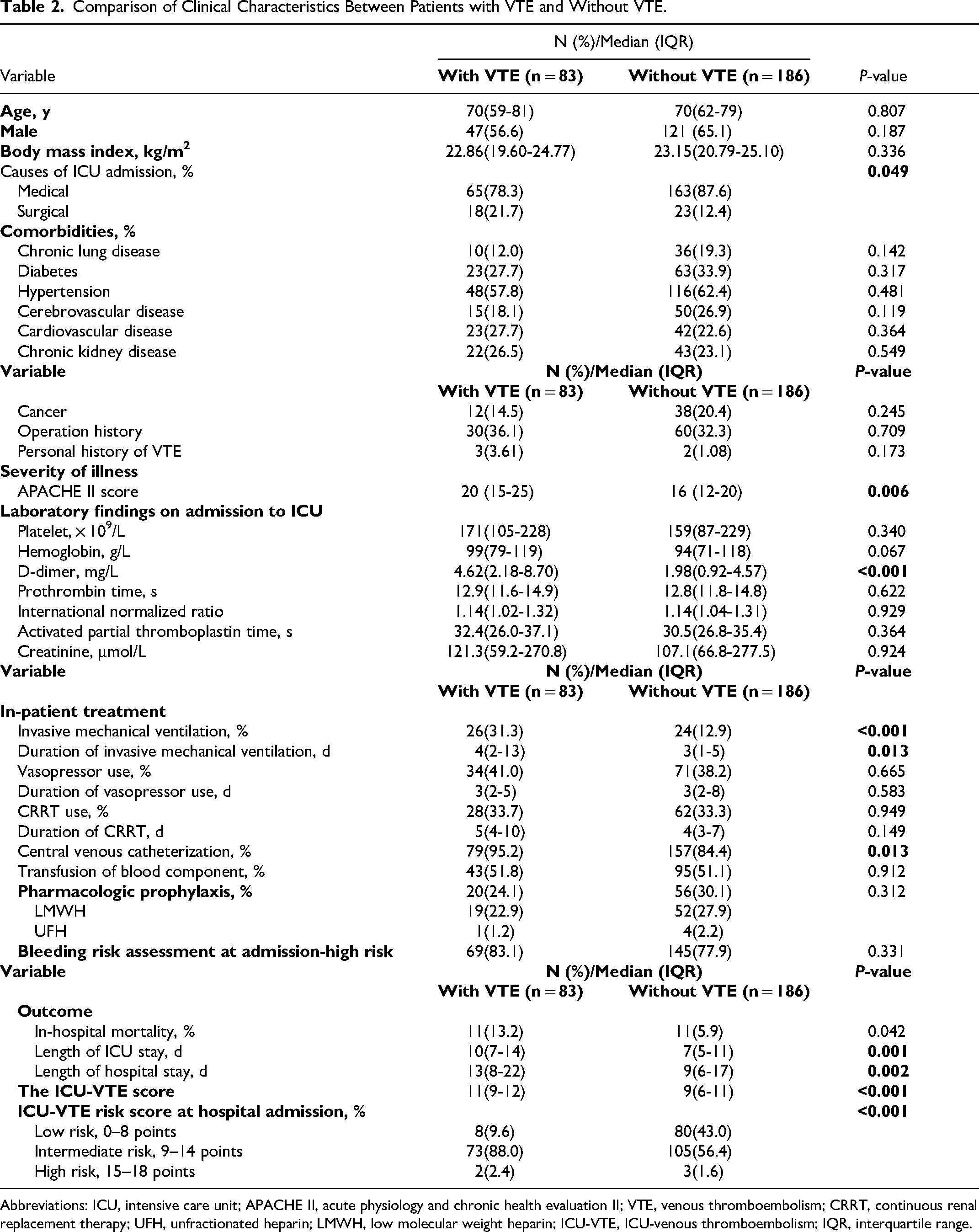
Abbreviations: ICU, intensive care unit; APACHE II, acute physiology and chronic health evaluation II; VTE, venous thromboembolism; CRRT, continuous renal replacement therapy; UFH, unfractionated heparin; LMWH, low molecular weight heparin; ICU-VTE, ICU-venous thromboembolism; IQR, interquartile range.
Risk Stratification by the ICU-VTE RAM
Patients were classified into different risk categories based on their ICU-VTE scores upon admission to the ICU (
Odds for Inpatient Venous Thromboembolism Stratified by ICU-VTE RAM.
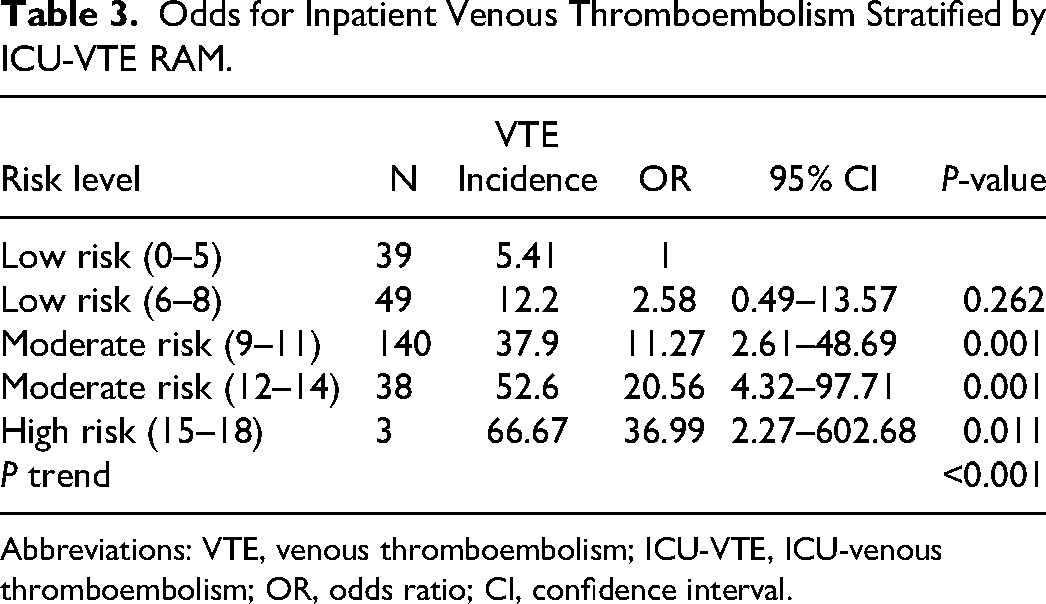
Abbreviations: VTE, venous thromboembolism; ICU-VTE, ICU-venous thromboembolism; OR, odds ratio; CI, confidence interval.
ROC Curve Analysis of the Predictive Value of the ICU-VTE RAM for in-hospital VTE
The ROC curve for predicting all VTEs in critically ill patients is showed in
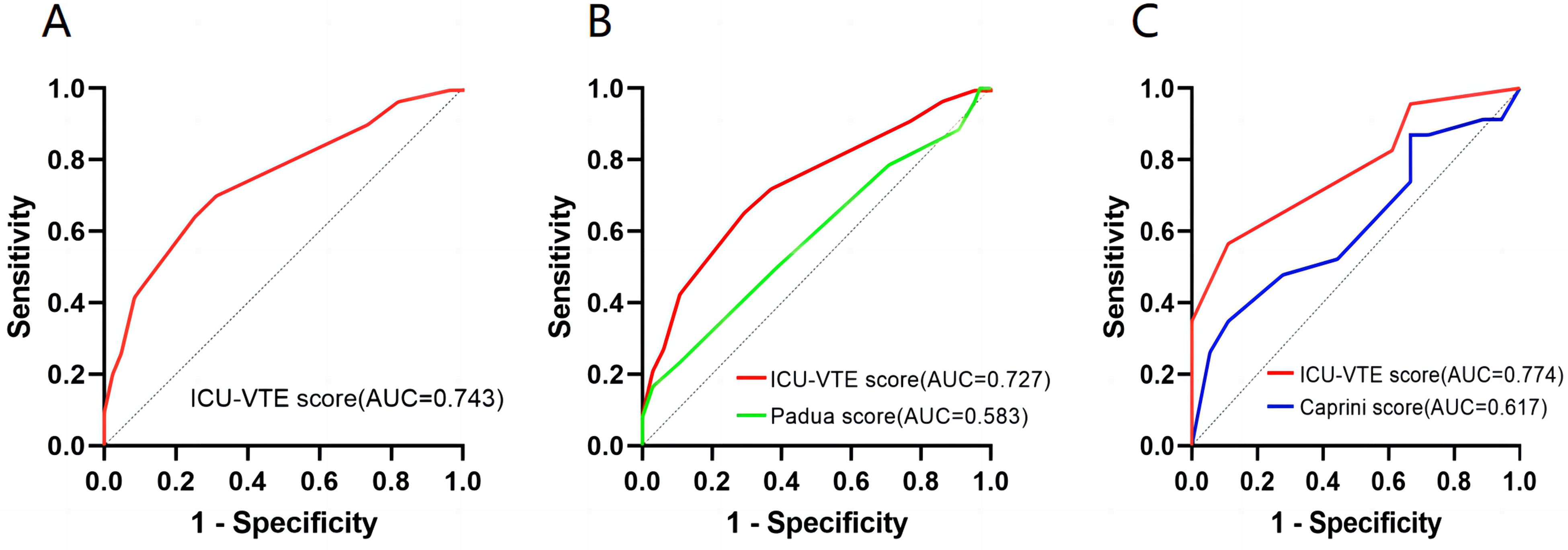
Receiver operating characteristic (ROC) curve of the ICU-VTE risk assessment model (RAM) for the prediction of venous thromboembolism in critically ill patients. A. ROC curve of the ICU-VTE RAM in mixed medical-surgical critically ill patients. B. ROC curve of the ICU-VTE and Pauda RAM in critically ill medical patients. C. ROC curve of the ICU-VTE and Caprini RAM in critically ill surgical patients.
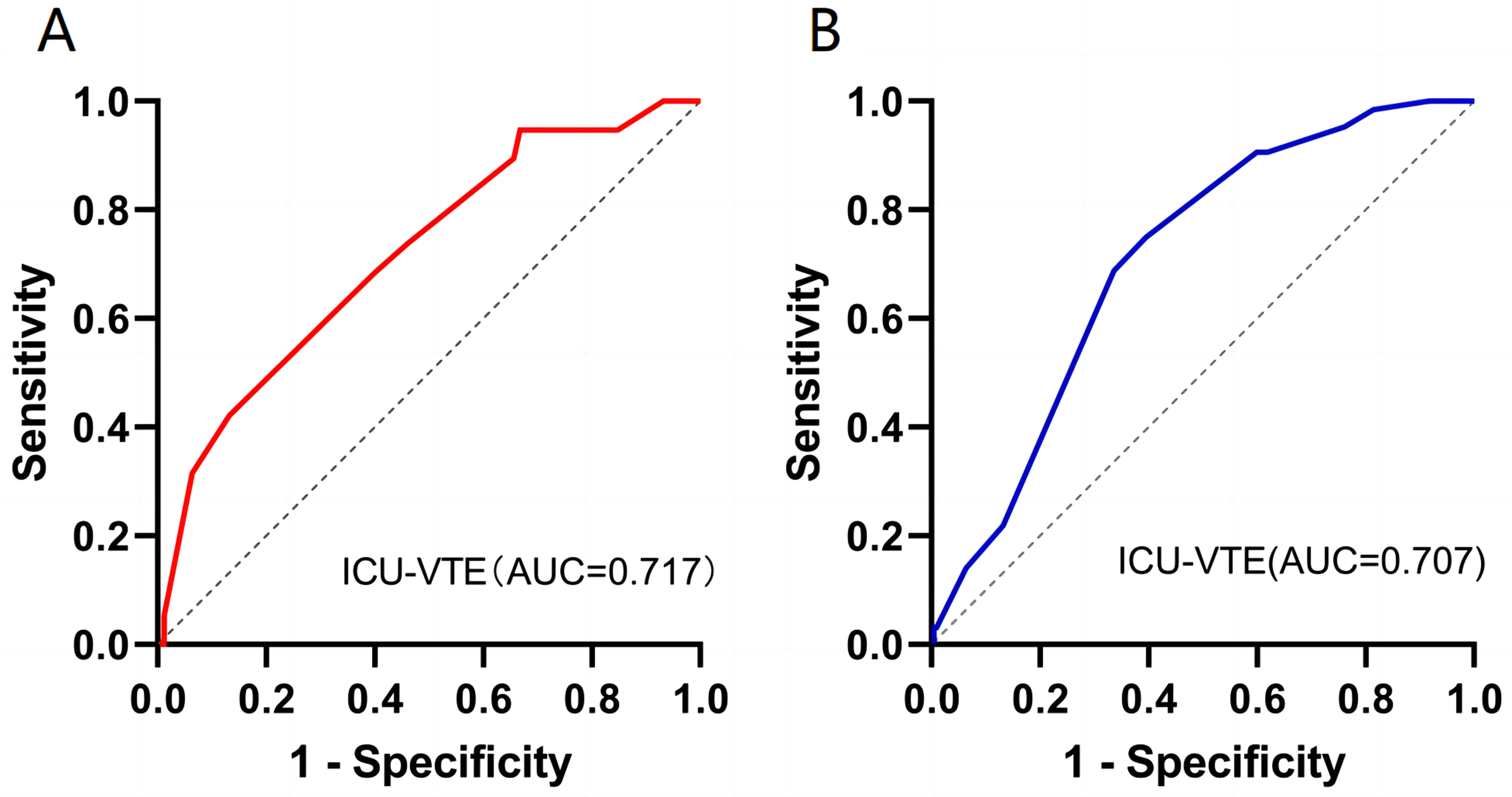
Receiver operating characteristic (ROC) curve of the ICU-VTE risk assessment model (RAM) for predicting symptomatic or asymptomatic venous thromboembolism in critically ill patients. A. ROC curve of the ICU-VTE RAM for the prediction of symptomatic venous thromboembolism. B. ROC curve of the ICU-VTE RAM for the prediction of asymptomatic venous thromboembolism.
Discussion
To our knowledge, this is the first study to validate the ICU-VTE RAM externally. We aimed to externally validate its predictive performance for VTE risk in critically ill patients, assess its role in VTE risk stratification, and compare it with that of the Pauda and Caprini RAM. Our findings highlight the importance of adopting this simple and effective ICU-VTE RAM for critical patients. Intensive surveillance and preventive measures should be implemented when patients exhibit a high ICU-VTE score.
Multivariate logistic regression analysis revealed that a higher ICU-VTE score was a reliable independent risk factor for in-hospital VTE. This confirmed the ability of the ICU-VTE RAM to predict VTE risk among critically ill patients. By categorizing risks, we found a statistically significant difference in VTE incidence among the five groups (P < 0.001). There was an evident increase in the odds ratio for VTE in patients with higher cumulative risk scores, indicating the effective risk stratification potential of the ICU-VTE RAM in critically inpatients. In the current study, the AUC of the ICU-VTE RAM was 0.743 for the entire study population. This is comparable to the AUC of 0.714 reported in a retrospective study of critically ill patients with tumors, 17 but lower than the AUC of 0.87 reported in the original study. 15 These differences in AUC values may be attributed to the lack of external validation of the original study, which focused only on symptomatic VTE, as well as variations in sample sizes between studies. Furthermore, similar AUC values of the ICU-VTE RAM for the prediction of symptomatic and asymptomatic venous thromboembolism were found, indicating that the model can more comprehensively reflect all types of VTE risk.
We compared the ICU-VTE RAM with other RAMs. The ICU-VTE RAM demonstrated superior effectiveness compared to the Pauda RAM (AUC 0.727 vs AUC 0.583, P < 0.001) in assessing VTE risk among medically critically ill patients. In our cohort, the AUC for the Pauda RAM was 0.583, consistent with a prospective study involving medical patients with sepsis (AUC 0.58). 18 Other retrospective studies have reported AUC values of 0.716 or 0.64 or lower for the Padua RAM in general medical patients.19,20 The Padua RAM exhibited unstable predictive performance in internal medicine patients. It was rarely used in critically ill patients, as evidenced by our study's poor predictive performance in critically ill medical patients.
In the case of critically ill surgical patients, the AUC of the ICU-VTE RAM was not significantly greater than that of the Caprini RAM (AUC: 0.774 vs 0.617, P = 0.128), indicating that the ICU-VTE and Caprini RAMs have similar predictive abilities. Lobastov et al reported an AUC for the Caprini RAM was 0.87 when standard prophylaxis was employed. 21 Hewes et al found an AUC of 0.818 (95% CI, 0.711-0.908) in esophagectomy patients. 22 In other studies, the AUC for the Caprini RAM ranged from 0.53 to 0.754.23–27 The Caprini RAM is complex, and some of the risk factors only pertain to surgical conditions, making it unsuitable for medical patients. Conversely, the ICU-VTE RAM is simple and intuitive, with only six risk factors related to the ICU. In a recent large-scale, nationwide cohort comprising both surgical and non-surgical patients and encompassing a large number of VTE events (n = 35,557), the predictive ability of both RAMs remained low for both surgical patients (Caprini 0.54, Padua 0.56) and non-surgical patients (Caprini 0.59, Padua 0.59). 28
The variability in AUC results for the Caprini and Padua models may stem from limitations in implementation heterogeneity. In our patient cohort, the Padua RAM proved to be inefficient in predicting VTE. The Caprini RAM, although comprehensive, lacks specific predictors for critically ill patients, limiting its ability to assess the risk of VTE in this population. Given the observed unstable performance and limited net benefit, the currently used RAMs are insufficient for patient stratification and determining VTE prophylaxis. The higher AUCs of the ICU-VTE RAM (AUC, 0.743) indicate its superior ability to predict VTE risk in critically ill patients. Our ROC analysis showed that a cut-off value of 9.5 for the ICU-VTE RAM aligned closely with the original study conducted by Viarasilpa et al and another study. 17 Therefore, we recommend defining high-risk patients with a score ≥10 points when utilizing the ICU⁃VTE rating scale. An ICU-VTE score of ≥10 can identify a subgroup of patients at a heightened high risk of VTE, necessitating a more robust prophylactic regimen.
Another significant finding of our study was the acquisition of additional data, which were unavailable in the original study, through prospective external validation. In our study, the incidence of VTE was 30.9%, which surpassed the rate reported in the original study that focused solely on symptomatic VTE. 15 Critically ill patients often do not manifest the typical signs and symptoms of DVT. 29 Therefore, it is reasonable for our study to assess DVT based on both symptomatic and asymptomatic cases, which, combined with the inclusion and exclusion criteria for patient selection, may have led to an overestimation of the incidence of DVT. Previous studies have demonstrated that the incidence of newly diagnosed DVT in the ICU ranges from 5% to 37%.3,5,30,31 Despite the high incidence of DVT in our study, only one PE patient was reported because of the lack of routine screening for PE, the unavailability of CTPA for extremely critically ill patients, and the small sample size of the study. Additionally, our univariate logistic regression analysis revealed that a high APACHE II score was a risk factor for VTE, consistent with previous studies.32,33 We also observed that critically ill patients with VTE had a longer duration of mechanical ventilation than did those who without VTE. However, the causal relationship between the duration of mechanical ventilation and VTE remains unclear. 34 Other studies shown that the using vasopressors is an independent risk factor for VTE.5,35 In our cohort, the use of a vasopressor did not appear to impact the risk of VTE, a finding not obtained in the original study. This discrepancy may be attributed to the low rate of anticoagulant prophylaxis, which potentially reduces the influence of vasopressors on VTE risk, as well as the small sample size of our study and the heterogeneity of the ICU population.
This study has several noteworthy limitations. First, the sample size was limited to a single center, leading to selection bias in the patient population and preventing comprehensive validation of the ICU-VTE RAM. Although this model is the first to use simple factors at admission to predict VTE risk, the findings must be replicated in an independent cohort. Further validation of the model's accuracy at other centers with a large external dataset is needed. Second, due to the observational nature of this study, residual confounding and potential bias cannot be entirely ruled out. Third, despite the extreme heterogeneity of the enrolled patients (surgical and non-surgical), we envisioned that the model will find application in all ICU patients, unlike traditional models that are based on specific medical or surgical disease categories.
Conclusion
The ICU-VTE RAM may be a practical and valuable tool for identifying and stratifying VTE risk in mixed medical-surgical critically ill patients, aiding in the management and prevention of VTE complications.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241271406 - Supplemental material for External Validation of the ICU-Venous Thromboembolism Risk Assessment Model in Adult Critically Ill Patients
Supplemental material, sj-docx-1-cat-10.1177_10760296241271406 for External Validation of the ICU-Venous Thromboembolism Risk Assessment Model in Adult Critically Ill Patients by Lijuan Zhang, Fuyang Chen, Su Hu, Yanxia Zhong, Bohua Wei, Xiaopin Wang and Ding Long in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-xlsx-2-cat-10.1177_10760296241271406 - Supplemental material for External Validation of the ICU-Venous Thromboembolism Risk Assessment Model in Adult Critically Ill Patients
Supplemental material, sj-xlsx-2-cat-10.1177_10760296241271406 for External Validation of the ICU-Venous Thromboembolism Risk Assessment Model in Adult Critically Ill Patients by Lijuan Zhang, Fuyang Chen, Su Hu, Yanxia Zhong, Bohua Wei, Xiaopin Wang and Ding Long in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-3-cat-10.1177_10760296241271406 - Supplemental material for External Validation of the ICU-Venous Thromboembolism Risk Assessment Model in Adult Critically Ill Patients
Supplemental material, sj-docx-3-cat-10.1177_10760296241271406 for External Validation of the ICU-Venous Thromboembolism Risk Assessment Model in Adult Critically Ill Patients by Lijuan Zhang, Fuyang Chen, Su Hu, Yanxia Zhong, Bohua Wei, Xiaopin Wang and Ding Long in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
We thank all the authors for their meticulous work.
Authors Contributions
LZ, FC and SH contributed equally and share first authorship. Concept and design: LZ, DL. Acquisition, analysis, or interpretation of data: LZ, FC, SH, YZ. Drafting of the manuscript: LZ. Critical revision of the manuscript for important intellectual content: all authors. Final approval of the manuscript: all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Administration of Traditional Chinese Medicine of Hubei Province, grant number ZY2023F059 and Wuhan Municipal Health Commission, grant number WZ22Q45.
Wuhan Municipal Health Commission, Administration of Traditional Chinese Medicine of Hubei Province, (grant number WZ22Q45, ZY2023F059).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
