Abstract
Metastatic colorectal cancer (mCRC) patients are predisposed to venous thromboembolism (VTE). This study aimed to (1) evaluate the efficacy of 4 existing cancer-specific VTE models in predicting VTE incidence among hospitalized mCRC patients, and (2) examine the influence of incorporating mCRC molecular subtypes into these models. We conducted an evaluation of 4 cancer-specific VTE models, including Khorana, Vienna CATS, Protecht, and CONKO in a dataset involving 1392 mCRC patients. To evaluate the predictive performance, we utilized receiver operating characteristic (ROC) curves for both the original models and the modified models that incorporated microsatellite instability status or KRAS/NRAS/BRAF mutations. Moreover, we computed the net reclassification improvement (NRI) to quantify the enhancements made to the modified VTE risk models. All models demonstrated a moderate area under the ROC curve (ROC-AUC) when predicting the occurrence of VTE: Khorana (0.550), Vienna CATS (0.671), Protecht (0.652), and CONKO (0.578). The incorporation of KRAS and BRAF mutations significantly improved the ROC-AUC of all 4 existing models (modified Khorana: 0.796, modified Vienna CATS: 0.832, modified Protecht: 0.834, and modified CONKO: 0.809). After dichotomizing the risk using a threshold of 3 points and comparing them with the original models, NRI values for the 4 modified models were 0.97, 0.95, 1.11, and 0.98, respectively. All 4 cancer-specific VTE models exhibit moderate performance when identifying mCRC patients at high risk of VTE. Incorporating KRAS and BRAF mutations may enhance the prediction of VTE in hospitalized mCRC patients.
Introduction
Venous thromboembolism (VTE) is a common complication of patients with malignancies, especially in those with metastatic disease. Also, VTE is a significant cause of morbidity, including postthrombotic complications and bleeding events associated with anticoagulation therapy. The impact of colorectal cancer (CRC) on VTE has been studied for years.1,2 However, there is a lack of a large body of retrospective data regarding occurrence rate of thrombosis in metastatic CRC hospitalized patients.
The clinical benefit of VTE prophylaxis in cancer patients primarily relies on accurate and individualized risk assessment models (RAMs); however, no consensus exists regarding the optimal model for predicting VTE occurrence in metastatic CRC (mCRC).3,4 To determine the risk of venous thromboembolism for cancer patients, the Khorana score is widely used. The score is based on 5 parameters: site of the cancer, obesity, platelet count, hemoglobin, and white blood cell count; CRC is scored as 0 for “site of cancer.” In the original Khorana model derivation cohort, high-risk scores (≥ 3) were associated with increased VTE risk (6.7% at 2.5 months), while low scores (≤ 2) corresponded to a VTE risk below 1%. 5 However, there are no reports of concordance of statistical differences in Khorana score between thrombosis and nonthrombosis groups in the validation studies of patients with gastrointestinal cancer. 6 Three RAMs, Vienna CATS, Protecht, and CONKO, modified from the Khorana score by addition or replacement of predictors, are based on discharge/ambulatory patients.7–9 The Vienna CATS model incorporates soluble P-selectin and D-dimer as predictors to enhance performance. 7 Subsequently, the Protecht score integrates platinum and gemcitabine-based chemotherapy into the Khorana model, while the CONKO score adds the World Health Organization (WHO) performance status to Khorana's predictive values.8,9 The development of these models highlights the need to further personalize VTE prophylaxis by identifying novel predictors in patient subgroups, including those with mCRC.
CRC is a heterogeneous group of malignancies distinguished by distinct clinical, biological, and genetic features. Approximately 55% of mCRC patients exhibit mutations in the KRAS or NRAS genes, and 5% to 10% present with BRAF mutations. 10 Notably, around 15% of colorectal carcinomas develop via the microsatellite instability pathway. 10 Although the molecular subtype of CRC has a prognostic significance for patients, the association of these subtypes with VTE risk is yet to be clarified.11,12 A recent study suggested that KRAS mutations in mCRC patients might elevate the risk of VTE 13 ; however, conflicting findings were reported in another study. 14 The role of other molecular subtypes in VTE has been scarcely investigated.
Thus, we conducted a comprehensive retrospective cohort study to evaluate the incidence of VTE among hospitalized patients with mCRC and compare the performance of 4 cancer-specific VTE RAMs (Khorana score, Vienna CATS score, Protecht score, and CONKO score). Furthermore, we investigated whether the incorporation of mCRC molecular subtypes (microsatellite instability status or KRAS/NRAS/BRAF mutations), routinely tested in this population, would enhance VTE risk stratification based on the 4 existing models.
Materials and Methods
Study Design and Patients
This study was a retrospective analysis of a prospectively maintained CRC database of The Sixth Affiliated Hospital, Sun Yat-sen University. Our Institutional Review Board (the Institutional Review Board of the Sixth Affiliated Hospital of Sun Yat-sen University) approved this retrospective study with a waiver of informed consent due to its retrospective nature (approval no. 2023ZSLYEC-261). We identified all patients with mCRC that were hospitalized in the center between 2019 and 2022. The approaches used to identify VTE cases in patients with CRC have been reported. 15 Baseline characteristic data encompassed patient-level factors (sex, age at diagnosis, body mass index, comorbidities, cardiovascular, and thromboembolic risk factors), cancer-level factors (molecular subtypes and primary tumor location), treatment-level factors, and laboratory test-level factors. Formalin-fixed, paraffin-embedded (FFPE) blocks of primary cancer tissue and matching metastatic lesions were prepared for immunohistochemistry (IHC) and real-time polymerase chain reaction (RT-PCR). Four primary antibodies, including MLH1 (Abcam, UK), MSH2 (Cell Signaling Technology, USA), MSH6 (Cell Signaling Technology, USA), PMS2 (Abcam, UK), were applied for staining. Visualization was performed using a DAB kit and counterstaining was done using hematoxylin. Tumors were considered as microsatellite instability (MSI) when showing high-frequency MSI (MSI-H; 2 or more of the 4 markers showing instability) and low-frequency MSI (MSI-L; only one of the 4 markers showing instability), while the rest (no instabilities were detected) were classified as microsatellite stability (MSS). A KRAS/NRAS/BRAF Mutation Analysis kit (EntroGen, Tarzana, CA, USA) was used for determination of KRAS, NRAS, and BRAF mutation status through RT-PCR. The TNM stage was defined according to the eighth TNM AJCC/UICC guidelines. 16 The primary outcome of interest was VTE, which occurred during the patient's initial hospitalization. Patients diagnosed with pulmonary embolism (PE) and/or deep vein thromboembolism (DVT) through any radiologic imaging method, such as CT or ultrasound, were considered to have VTE. There was no formal predefined protocol or prospective screening plan for occult VTE. Patients with elevated D-dimer levels would accept imaging examinations during hospitalization. Both symptomatic individuals, diagnosed through symptom-prompted diagnostic imaging, and asymptomatic individuals, identified via imaging studies conducted for other medical purposes (eg, cancer restaging), were included. This study aimed to (1) evaluate the efficacy of different cancer-specific VTE models in predicting VTE incidence among hospitalized mCRC patients, and (2) examine the influence of incorporating mCRC molecular subtypes into these models.
Validation of Existing RAMs
Each included patient was assessed retrospectively by 4 RAMs for VTE risk, including Khorana, Vienna CATS, Protecht, and CONKO, based on the information available at admission.5,7–9 The stratification of VTE risk was based on the cutoff points recommended in the corresponding derivation cohorts of different RAMs. The risk factors identified by the 4 RAMs and the point assigned for each factor are shown in Supplemental Table S1. In the Khorana, Vienna CATS, Protecht, and CONKO models, patients are categorized into 2 risk groups based on the score: low to intermediate (score 0–2), and high (score ≥ 3).5,7–9 The classification of high risk and low/moderate risk groups in these 4 models was also performed based on optimal cut-offs (maximum of Youden index) calculated by receiver–operating characteristic. Predicted abilities of the existing risk models were evaluated using discrimination and goodness of fit. Discrimination was assessed by obtaining C-index (the value of the C-index is the same as that of ROC-AUC in the logistic regression model). Goodness of fit was reported using the Akaike information criterion (AIC) for all included VTE-RAMs. Sensitivity (SEN), specificity (SPE), positive predictive value (PPV), and negative predictive value (NPV) of the various risk models were also calculated with 95% confidence intervals (95% CIs) to evaluate prediction performance. The cumulative VTE rates of the low/moderate risk and high-risk groups, as stratified by different models, were illustrated using Kaplan–Meier curves and compared using the log-rank test. Due to the binary nature of the classification with incidence of VTE, a calibration plot would not provide informative results. Therefore, we summarized the distribution of VTE and non VTE patients across different cumulative scores in the 4 existing models and calculated the corresponding odds ratios (ORs) for VTE occurrence.
New Models Development
Initially, ORs for 6 different mCRC molecular subtypes (microsatellite instability status and KRAS/NRAS/BRAF mutations) regarding VTE were calculated through regression analysis, while adjusting for the confounding effects of bevacizumab use and regorafenib/fruquintinib use, 17 which were documented as treatment-related factors associated with VTE. Secondly, molecular subtypes with an OR > 3.0 (or < 0.3) were integrated into the 4 existing models. 18 The training of new model was conducted on the training dataset (50% of the total). The total cohort was used as the validation dataset to compare the performance of all models. The assigned points for each factor in the modified models are presented in Supplemental Table S4. After incorporation of molecular subtypes, ROC-AUCs and cumulative risk curves of new models were calculated with a threshold set at 3 points to further examine the model performance.
Net Reclassification Improvement
Net reclassification improvement (NRI) assesses changes in the 4 domains of the confusion matrix. It is calculated by summing the change in sensitivity (the percentage of VTE patients correctly predicted solely by the new model, subtracted by the percentage of VTE patients incorrectly predicted solely by the new model) and the change in specificity (the percentage of non VTE patients correctly predicted solely by the new model, subtracted by the percentage of non-VTE patients incorrectly predicted solely by the new model). We calculated NRI of 4 new models and changes in sensitivity and specificity when comparing against their original models.
Statistical Analysis
Continuous variables were described using mean values with standard deviations, and group comparisons were conducted using the Student's t-test for normally distributed data. Median values with interquartile ranges were presented for continuous variables with skewed distributions, and group comparisons were performed using nonparametric tests. Discrete variables were summarized using frequencies and percentages, with group comparisons conducted via chi-squared tests. To calculate the OR for VTE, univariate and multivariate analyzes were performed based on different risk factors. Missing data were imputed using multivariate imputation by chained equations method in Python. All reported P-values in this study are two-tailed. P < .05 was considered to represent statistical significance. Data were analyzed using SPSS v20.0 (IBM, Armonk, NY, USA) and R statistical package (https://cran.r-project.org).
Results
Patient Characteristics at Baseline
In this study, we screened 2086 medical inpatients with mCRC for inclusion. A total of 434 patients were excluded for the following reasons: (1) unconfirmed VTE (n = 8), (2) nonactive cancer (n = 274), (3) hospital admission primarily due to VTE (n = 128), and (4) age below 18 years (n = 24). Additionally, 260 patients were excluded due to missing information regarding cancer-related factors, such as molecular subtypes and primary tumor location. Ultimately, 1392 patients were enrolled (Supplemental Figure S1).
Table 1 presents the baseline patient characteristics. The average patient age was 61 years, with 36.0% being female. A history of major bleeding was present in 50 patients (3.6%), while 30 patients (2.2%) experienced major bleeding during their hospital stay. The overall VTE rate within the study population was 35.6%. Within the study population, 115 patients (8.3%) exhibited MSI-L status, 52 patients (3.7%) had MSI-H status, 531 patients (38.1%) possessed KRAS mutations, 47 patients (3.4%) had NRAS mutations, and 136 patients (9.8%) displayed BRAF mutations. The most common primary tumor site for CRC was the rectum (737, 52.9%). Right colon cancer, transverse colon cancer, left colon cancer, and sigmoid colon cancer constituted 17.4%, 1.9%, 8.5%, and 18.5% of the population, respectively. Appendix/caecum cancer was relatively rare, with only 11 cases observed. Univariate analysis results from logistic and Cox regression is presented in Supplemental Table S2. Follow-up duration for all patients was comparable between the VTE and non-VTE groups: VTE group—75th quantile (39 days), 50th quantile (18 days), and 25th quantile (7 days); non-VTE group—75th quantile (35 days), 50th quantile (10 days), and 25th quantile (5 days).
Patient Characteristics at Baseline.

Abbreviations: BMI, body mass index; BRAF, v–raf murine sarcoma viral oncogene homolog B; 5-FU, 5-fluorouracil; IQR, interquartile ranges; i.v., intravenous; KRAS, Kirsten rat sarcoma viral oncogene; LMWH, low molecular weight heparin; MSI-H, microsatellite instability-high; MSI-L, microsatellite instability-low; MSS, microsatellite stable; NRAS, neuroblastoma rat sarcoma viral oncogene; p.o., per oral; VTE, venous thromboembolism; WHO, world health organization.
A cycle of CAPEOX consists of the following: capecitabine 1000 mg/m2 p.o. twice daily for 14 out of 21 days.
A cycle of FOLFOX consists of the following: Day 1: i.v. oxaliplatin 85 mg/m2 in 120 min; i.v. leucovorin 200 mg/m2 in 2 h; 5-FU 400 mg/m2 bolus i.v. then 2400 mg/m2 perfusion i.v. over 46 h; the next chemotherapy cycle was repeated on the 15th day.
A cycle of FOLFOXIRI consists of the following: Day 1: i.v. irinotecan 165 mg/m2 in 90 min; i.v. oxaliplatin 85 mg/m2 in 120 min; i.v. leucovorin 200 mg/m2 in 2 h; 5-FU 400 mg/m2 bolus i.v. then 2400 mg/m2 perfusion i.v. over 46 h; the next chemotherapy cycle was repeated on the 15th day.
Validation of VTE-RAMs in Patients With mCRC
The median cumulative VTE risk score of all 4 RAMs was significantly greater than the median score for the non VTE group (Supplemental Table S3). The risk of VTE increased almost linearly with the increase of cumulative Khorana score after adjustment for VTE prophylaxis. Similar trends were observed with the other RAMs (Supplemental Table S3). In 3 out of 4 risk cancer-specific VTE-RAMs, more than 80% of patients were classified as low/moderate VTE risk, including (Khorana, 94.4%, Protecht, 88.9%, and CONKO, 93.3%). The Vienna CATS model identified the lowest number of patients as low/moderate-risk group (74.4%). Table 2 shows that all 4 cancer-specific VTE-RAMs (Khorana, Vienna CATS, Protecht, and CONKO) showed an moderate accuracy (AUC ≤ 0.8) in predicting VTE occurrence. The Khorana model exhibited the lowest AUC (0.550) and demonstrated the poorest prediction of VTE occurrence in mCRC patients, whereas the Vienna CATS model had the highest AUC (0.671). Furthermore, we calculated the sensitivity and specificity of 4 VTE models using both recommended high-risk cut-offs from derivation studies and optimized cut-offs determined by ROC curves (Table 2). Sensitivities improved for all 4 models while specificities simultaneously decreased using the calculated threshold values. Kaplan–Meier curves showed a higher cumulative VTE incidence in hospitalized mCRC patients with Khorana score ≥ 3, Vienna CATS score ≥ 3, Protecht score ≥ 3, and CONKO score ≥ 3 (Supplemental Figure S2).
VTE Occurrence Prediction Performance of VTE-RAMs in CRC Patients.

Abbreviations: ROC, receiver-operating characteristic; AUCs, areas under the curves; CI, confidence interval; CRC, colorectal cancer; RAM, risk assessment model; VTE, venous thromboembolism.
Data are ROC-AUCs and (95% CI).
The value of C-index is as same as that of AUC in logistic regression model.
Recmmonded cutoff points based on derivation studies.
Calculated cutoff points based on ROC curves.
Net Reclassification Improvement of New Models Compared With Original Models
Molecular subtypes significantly associated with VTE were shown in Figure 1 after adjusted for 2 important treatment-related confounding factors. The use of anti-angiogenic therapy in mCRC patient including bevacizumab, regorafenib, and fruquintinib is known to elevate the risk of both bleeding and thrombotic events. 17 We selected variables with a OR value larger than 3.0 or a OR lower than 0.3 (KRAS and BRAF mutations) as new predictors to avoid the potential risk of observational bias. 18 The ROC-AUCs demonstrated a significant improvement after adding new predictors to the original models (Table 3). Among these models, the “modified Protecht score” which incorporated KRAS and BRAF mutations, achieved the highest ROC-AUC of 0.834 (95% CI: 0.812-0.856). Due to the high OR values of KRAS and BRAF mutations, different point assignments of those mutations were evaluated in modified VTE models. No statistically significant improvement in those ROC-AUCs was observed when assigning an additional 1 point for those mutations (Supplemental Tables S5 and S6). Finally, 4 new models were developed by incorporating 2 predictors (KRAS and BRAF mutations) into their respective original models (Supplemental Table S4). When comparing against original models, NRI of 4 new models was 0.97 for modified Khorana score, 0.95 for modified Vienna CATS score, 1.11 for modified Protecht score, and 0.98 for modified CONKO score, and all P-value < .01. All NRIs were driven by improvement in sensitivity (Table 4). Kaplan–Meier curves showed a higher cumulative VTE incidence in hospitalized mCRC patients with modified Khorana score ≥ 3, modified Vienna CATS score ≥ 3, modified Protecht score ≥ 3, and modified CONKO score ≥ 3 (Supplemental Figure S3).
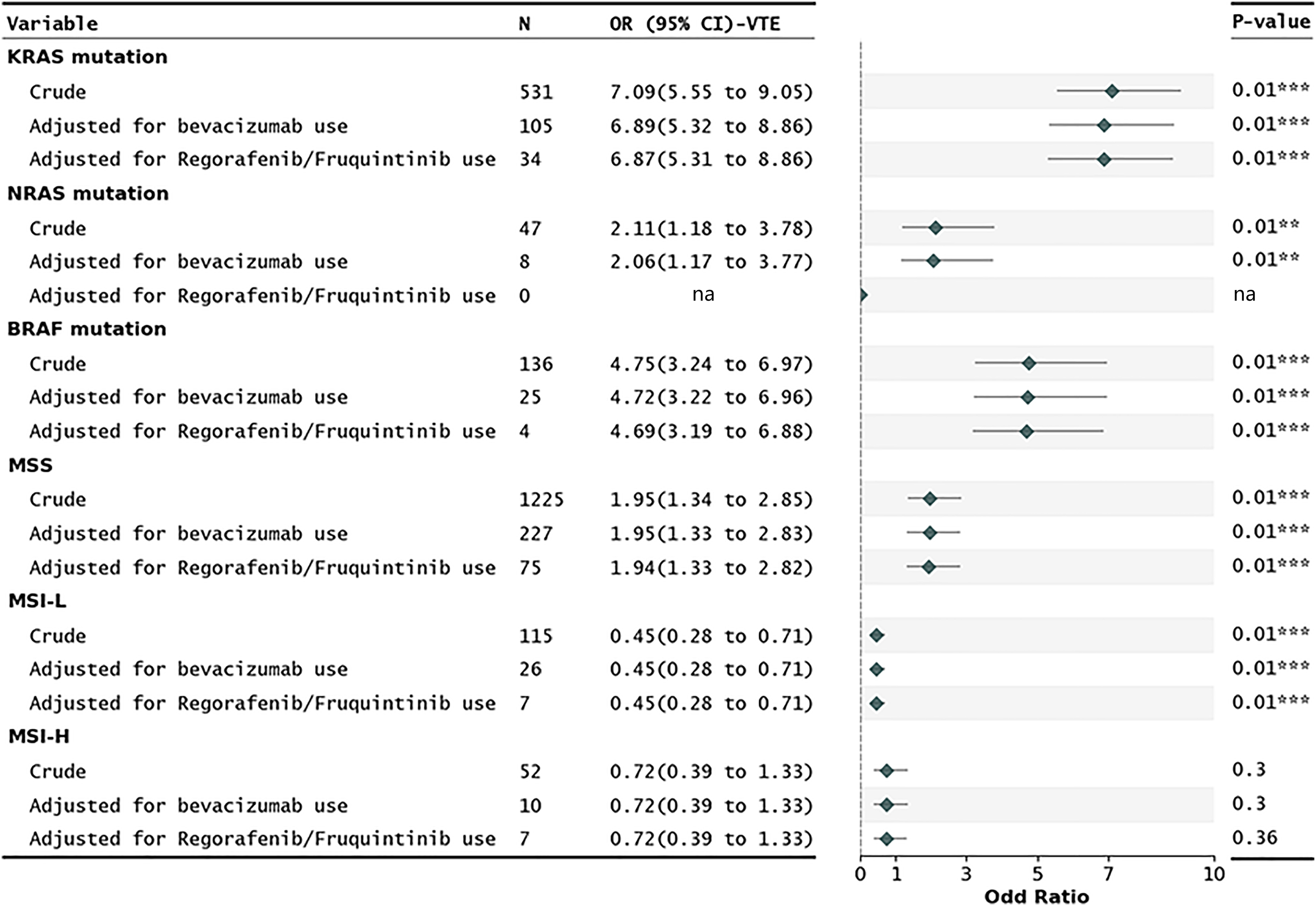
Risk of VTE in hospitalized mCRC patients with different molecular subtypes. Rates and ORs were calculated with data from VTE patients and non-VTE patients. ORs were adjusted for bevacizumab use and regorafenib/fruquintinib use. Abbreviations: BRAF, v–raf murine sarcoma viral oncogene homolog B; CI, confidence intervals; KRAS, Kirsten rat sarcoma viral oncogene; mCRC, metastatic colorectal cancer; MSI-H, microsatellite instability-high; MSI-L, microsatellite instability-low; MSS, microsatellite stable; NRAS, neuroblastoma rat sarcoma viral oncogene; ORs, odds ratios; VTE, venous thromboembolism. All ORs were adjusted for use of VTE prophylaxis.
VTE Occurrence Prediction Performance of VTE-RAMs After Addition of KRAS/BRAF Mutations.

Abbreviations: CI, confidence intervals; CRC, colorectal cancer patient; ROC-AUC, receiver-operating characteristic-areas under the curve; VTE, venous thromboembolism.
Logistic regression analysis.
VTE occurrence prediction using recommended cutoff points based on derivation studies.
Changes to Reclassification Across VTE-RAMs After Incorporating

Abbreviations: CI, confidence intervals; RAM, risk assessment model; VTE, venous thromboembolism.
Discussion
We investigated the VTE incidence in a large cohort of hospitalized patients with mCRC treated at our institution. The performance of multiple VTE-RAMs for patients with different molecular subtypes was compared, accounting for important confounders. All 4 cancer-specific VTE models (Khorana, Vienna CATS, Protecht, and CONKO) demonstrated moderate performance (ROC-AUCs < 0.8) in identifying mCRC hospitalized patients at high VTE risk. KRAS and BRAF mutations were found to be significantly associated with an increased incidence of VTE compared to other molecular subtypes. Furthermore, the addition of KRAS and BRAF mutations as new predictors enhanced the predictive ability of all 4 VTE-RAMs.
Given the wide variability in VTE rates among cancer patients (ranging from 1.8% to 20%),19,20 existing risk scores for VTE are inadequate for the mCRC patient subpopulation, primarily because they were not specifically designed for this purpose. Both Vienna CATS and Protecht displayed acceptable sensitivity with their calculated cutoff points, likely due to the incorporation of hematological and chemotherapy-related risk factors. However, the validation of Vienna CATS and Protecht scores revealed relatively poor specificities (< 60.5% using calculated cutoff values). Similarly, low specificity of Khorana model has also been reported in several studies.21,22 Notably, despite satisfactory sensitivity at the calculated cutoff value, the Vienna CATS model classified over 50% of mCRC patients (> 700 patients) as high-risk, and its low positive predictive value (∼ 50%) indicated that a significant number of patients might be subjected to unnecessary risks associated with VTE prophylaxis. A similar trend was observed in the Protecht score with its calculated cutoff point. In fact, CRC patients with chronic portal or mesenteric vein thrombosis frequently exhibit cavernous transformation and/or signs of portal hypertension, which can lead to complications such as variceal bleeding. 23 All 4 existing models demonstrated moderate ROC-AUCs (< 0.8) for VTE prediction in hospitalized mCRC patients. This is consistent with that previously reported in a validation study of Khorana model and Vienna CATS model in patients with other cancer types. 24 The potential underestimation of VTE risk in these models for the mCRC subpopulation could be attributed to the absence of CRC-specific risk factors. Novel approaches are required to optimize VTE risk prediction for individual mCRC patients.
Our findings revealed that the differential VTE risk among molecular subtypes might be obscured when all CRCs were treated as a single entity. Patients with KRAS and BRAF mutations exhibited a significantly higher risk of VTE compared to those with other molecular types. KRAS and BRAF mutation status functions as a clinically validated predictive biomarker, currently employed to discern mCRC patients who are unlikely to benefit from anti-EGFR therapy. Laboratory evidence indicates that normal TF expression on CRC cell surfaces is augmented by the activation of KRAS oncogene mutations and TP53 tumor suppressor gene inactivation, both of which represent significant transformative events in cancer progression. 25 Given that TF serves as the primary cellular trigger for blood coagulation and a substantial procoagulant associated with systemic hypercoagulability in cancer patients, 25 we hypothesized that mCRC patients with KRAS/BRAF mutations would exhibit an elevated risk of VTE. Ades et al 13 identified a significant association between KRAS gene mutations and an elevated risk of VTE in mCRC patients. This relationship was found to be particularly pronounced in the low-risk group in Khorana score. However, conflicting evidence exists concerning the connection between KRAS mutation and VTE risk in lung cancer patients, as reported by a positive study 26 and a negative study. 27 In another study, Morán et al 28 performed a retrospective cohort of mCRC patients to analyze the incidence of VTE based on KRAS, NRAS, and BRAF mutations. Patients with KRAS/NRAS mutations exhibited a low incidence of VTE, while those with BRAF mutations showed a high incidence; however, the observed difference did not reach statistical significance, likely due to the limited sample size. Guha et al 29 reported an interesting finding that BRAF inhibitors were associated with an increased risk of VTE, as demonstrated in 2 independent real-world cohorts. Further research based on large cohort is required to determine the association between KRAS and BRAF mutations and VTE due to the existing conflicting evidence. Moreover, we observed a minimal impact of other molecular subtypes (microsatellite instability status or NRAS mutations) on VTE incidence.
Our study had several potential limitations: (1) the study was conducted in a single center, (2) our study involved a retrospective analysis of a consecutively collected CRC database. However, due to incorrect coding, some individuals may have been missed when identifying patients with VTE, (3) the routine screening of hospitalized patients for asymptomatic VTE was not performed in our hospital, which may have led to an underestimation of VTE prevalence in patients with mCRC, and (4) patients with poor prognosis may give up treatment due to the economic burden, which may lead to an underestimate of VTE rate. Given that all participants in our study underwent CT or ultrasound for cancer or other medical purposes at admission, the potential effect is likely negligible.
Conclusions
All 4 existing cancer-specific VTE models exhibit moderate performance in identifying mCRC inpatients with high VTE risk. If confirmed in additional cohorts, the inclusion of KRAS and BRAF status could become essential for assessing VTE risk in patients with mCRC, given the substantial effect size and the prevalence of KRAS and BRAF gene mutations in CRC. Notably, KRAS and BRAF status, as a routine test in mCRC patients, could aid in selecting patients who should receive VTE thromboprophylaxis during hospitalization. Clinicians should pay greater attention to VTE risk stratification in mCRC inpatients.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231196859 - Supplemental material for Estimating Venous Thromboembolism Risk in Metastatic Colorectal Cancer Inpatients: Validation of Existing Risk Scores and Development of New Risk Scores
Supplemental material, sj-docx-1-cat-10.1177_10760296231196859 for Estimating Venous Thromboembolism Risk in Metastatic Colorectal Cancer Inpatients: Validation of Existing Risk Scores and Development of New Risk Scores by Li Qin, Zhikun Liang, Jingwen Xie and Xiaoyan Li in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296231196859 - Supplemental material for Estimating Venous Thromboembolism Risk in Metastatic Colorectal Cancer Inpatients: Validation of Existing Risk Scores and Development of New Risk Scores
Supplemental material, sj-docx-2-cat-10.1177_10760296231196859 for Estimating Venous Thromboembolism Risk in Metastatic Colorectal Cancer Inpatients: Validation of Existing Risk Scores and Development of New Risk Scores by Li Qin, Zhikun Liang, Jingwen Xie and Xiaoyan Li in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The studies involving human participants were reviewed and approved by the Institutional Review Board of the Sixth Affiliated Hospital of Sun Yat-sen University (approval no. 2023ZSLYEC-261).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the program of Guangdong Provincial Clinical Research Center for Digestive Diseases (grant number 2020B1111170004).
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation. Please contact corresponding authors for more data information.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
