Abstract
Direct oral anticoagulants (DOACs) have been compared with standard therapy in large phase III studies to assess their safety and efficacy in the treatment of deep vein thrombosis and/or pulmonary embolism and in the secondary prevention of recurrent venous thromboembolism. Although the mean population age and the gross inclusion and exclusion criteria were similar across these studies, they differed in other aspects such as overall study design and acute treatment strategies. The 4 DOACs examined in phase III trials (apixaban, edoxaban, rivaroxaban, and dabigatran) showed noninferiority compared with standard therapy for the treatment of deep vein thrombosis and/or pulmonary embolism and for the prevention of recurrent venous thromboembolism. Furthermore, these DOACs exhibited a similar safety profile to standard therapy, with the risk of major bleeding significantly reduced in some of these studies. Rivaroxaban and apixaban were tested as a single-drug approach, whereas in the dabigatran and edoxaban studies, initial bridging with parenteral agents was employed. The purpose of this review is to compare the phase III studies of DOACs in this indication, to highlight the differences, and to discuss a series of clinically relevant issues, including the management of key patient subgroups (eg, fragile patients, those with cancer or renal impairment), extended treatment, use of comedications, heparin pretreatment versus a single-drug approach, and the bleeding profiles of the DOACs.
Introduction
Venous thromboembolism (VTE), comprising both deep vein thrombosis (DVT) and pulmonary embolism (PE), is associated with high rates of morbidity and mortality. 1,2 The economic burden associated with the treatment of the initial venous thromboembolic event and subsequent complications, such as recurrence, is substantial. 3 Standard anticoagulation therapy is effective when initiated with unfractionated heparin (UFH), low-molecular-weight heparin (LMWH), or fondaparinux and bridged with and followed by an oral vitamin K antagonist (VKA) for long-term treatment. 4,5 However, standard anticoagulation has limitations: heparins require parenteral administration and weight-based dose adjustments, 4 –6 whereas VKAs have a slow onset of action, narrow therapeutic window, and unpredictable pharmacological responses with extensive drug and food interactions, and require frequent coagulation monitoring and dose adjustment. 7 To overcome the disadvantages of heparins and VKAs, direct oral anticoagulants (DOACs) have been developed.
Three direct factor Xa inhibitors—apixaban, edoxaban, and rivaroxaban—and one direct thrombin inhibitor—dabigatran—have been compared with standard therapy in large phase III studies to assess their efficacy and safety in the treatment of DVT and/or PE, in the prevention of recurrence 8 –13 and in the extended prevention of recurrent VTE. 10,14,15 For the physician, comparing the results of these studies to draw clinically meaningful conclusions is problematic, because different trials varied in study design, patient recruitment, and length of follow-up. It is instructive, in the absence of head-to-head comparisons, to compare the design, patient populations, and key outcomes to understand the strengths and limitations of each study, and the advantages and disadvantages of each agent as well as to avoid making invalid conclusions regarding specific efficacy and safety outcomes. This review will compare phase III studies of DOACs for the acute treatment of DVT/PE and the secondary prevention of VTE (Apixaban for the Initial Management of Pulmonary Embolism and Deep-Vein Thrombosis as First-Line Therapy [AMPLIFY], 8 Hokusai-VTE, 9 Efficacy and Safety of Dabigatran Compared to Warfarin for 6 Month Treatment of Acute Symptomatic Venous Thromboembolism [RE-COVER] and Phase III Study Testing Efficacy & Safety of Oral Dabigatran Etexilate vs Warfarin for 6 m Treatment for Acute Symp Venous Thromboembolism (VTE) [RE-COVER II], 12,13 and EINSTEIN DVT and EINSTEIN PE 10,11 ) as well as studies of extended anticoagulation treatment (Apixaban after the Initial Management of Pulmonary Embolism and Deep Vein Thrombosis with First-Line Therapy–Extended Treatment [AMPLIFY-EXT], 14 EINSTEIN EXT, 10 Twice-daily Oral Direct Thrombin Inhibitor Dabigatran Etexilate in the Long Term Prevention of Recurrent Symptomatic VTE [RE-SONATE], and Secondary Prevention of Venous Thrombo Embolism (VTE) [RE-MEDY] 15 ) by addressing a series of questions of clinical relevance.
Study Design: Is Double-Blind Better Than Open-Label?
Four studies on the acute treatment of symptomatic DVT/PE with apixaban (AMPLIFY 8 ), dabigatran (RE-COVER and RE-COVER II 12,13 ), and edoxaban (Hokusai-VTE 9 ) were double blinded. By contrast, the EINSTEIN DVT 10 and EINSTEIN PE 11 studies with rivaroxaban were open-label with a blinded-end point adjudication design. 16 Blinded studies are regarded as the most rigorous methodologically for hypothesis testing but are unrepresentative of daily practice because they test different regimens that involve more complex logistics, as these require greater complexity of care and monitoring. 17 Open-label studies are generally viewed as having a potential for bias. 17 Nevertheless, an open-label study design is more representative of real-world clinical practice and can, therefore, provide a more realistic assessment of patient-reported outcomes, which are increasingly important in assessing patient satisfaction, adherence to therapy, and health care burden. In conclusion, double-blind and open-label trial designs have different pros and cons and both should be considered suitable for the purposes of these studies. The design and key study parameters of the 6 acute VTE treatment phase III studies are shown in supplementary Table S1.
Study Populations: Who Were the Patients and How Did They Differ?
In total, over 27 000 patients were included across all phase III studies investigating DOACs in the treatment of acute VTE. 8 –13 Although broadly similar inclusion and exclusion criteria were used in each trial (Table S2), the study populations differed in some aspects.
Treatment Duration
Current American College of Chest Physicians guidelines recommend anticoagulant therapy beyond 3 months for patients with a first unprovoked proximal DVT of the leg or first unprovoked PE and who have a low or moderate risk of bleeding; however, this is not recommended in patients with a high risk of bleeding. 4 Therefore, the type of patients recruited in each of the phase III VTE treatment studies was influenced by the treatment durations assessed. Patients who were diagnosed with DVT or PE and had a high risk of bleeding could not be included in the RE-COVER, 12 RE-COVER II, 13 or AMPLIFY 8 studies, because the recommended treatment duration for these patients is only 3 months. By contrast, these patients could be included in the EINSTEIN DVT, 10 EINSTEIN PE, 11 and Hokusai-VTE 9 studies but were less likely to be included owing to the conservative exclusion criteria.
Treatment duration in the Hokusai-VTE study (3-12 months) and in the EINSTEIN DVT and EINSTEIN PE studies (3, 6, and 12 months) dictated the exclusion of patients with a life expectancy of less than 3 months, 9 –11 whereas in RE-COVER, RE-COVER II, and AMPLIFY, patients with a life expectancy of less than 6 months were excluded (treatment duration 6 months). 8,12,13
Age
The average age across all studies ranged from 55 to 58 years, which is a younger cohort than seen in routine daily practice (>60 years old). 8 –13,18 Most patients were <65 years old in the AMPLIFY (64.8%), 8 RE-COVER (68.9%), 12 and EINSTEIN DVT and EINSTEIN PE pooled (62.7%) populations (no data were available for RE-COVER II). 19
Patients aged from 65 to <75 years in AMPLIFY and RE-COVER or 65 to 75 years in the EINSTEIN DVT and EINSTEIN PE pooled population comprised 20.9%, 19.7%, and 21.8%, respectively, of these populations. 8 ,12 ,19 In AMPLIFY and RE-COVER, patients aged ≥75 years accounted for 14.3% and 11.4% of the respective populations, 8,12 whereas in the Hokusai-VTE and in the EINSTEIN DVT and EINSTEIN PE pooled populations, those aged ≥75 years comprised 15.5% and 13.4%, respectively. 9 ,19
Unprovoked VTE
The proportion of patients with unprovoked VTE also varied among studies and was approximately 90% in AMPLIFY 8 compared with 62% to 66% in the EINSTEIN DVT, 10 EINSTEIN PE, 11 and Hokusai-VTE 9 studies (Table 1). These proportions reflect the respective inclusion criteria, which for AMPLIFY required a minimum of 6 months’ therapy. The proportion of patients with provoked/unprovoked VTE can also influence expected outcomes and duration of therapy, because the risk of recurrence after an unprovoked event is higher than in the case of provoked VTE due to a reversible risk factor. 4,20
Comparison of Population Characteristics of Venous Thromboembolism Studies That Compared Direct Oral Anticoagulants With Standard Therapy.
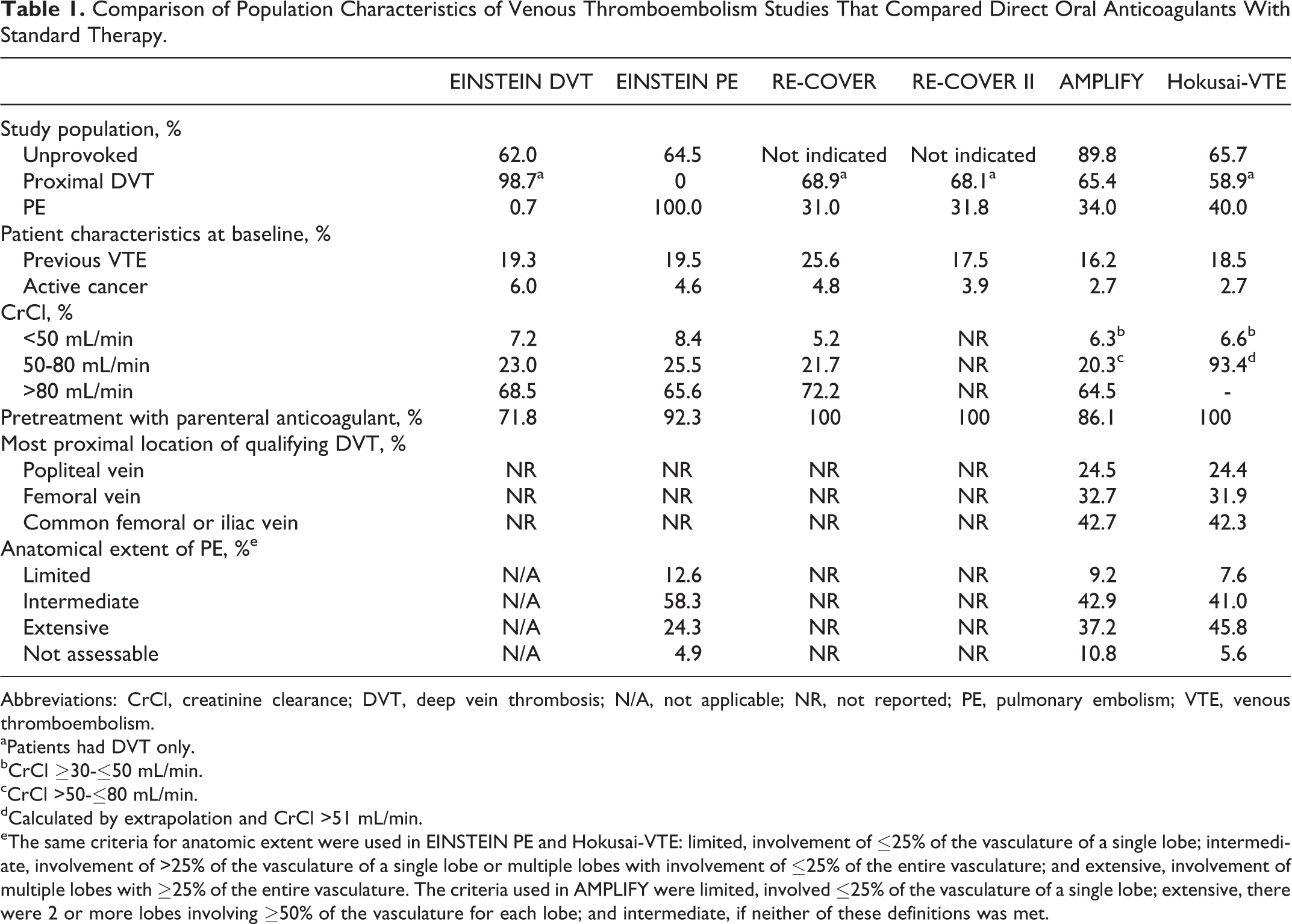
Abbreviations: CrCl, creatinine clearance; DVT, deep vein thrombosis; N/A, not applicable; NR, not reported; PE, pulmonary embolism; VTE, venous thromboembolism.
aPatients had DVT only.
bCrCl ≥30-≤50 mL/min.
cCrCl >50-≤80 mL/min.
dCalculated by extrapolation and CrCl >51 mL/min.
eThe same criteria for anatomic extent were used in EINSTEIN PE and Hokusai-VTE: limited, involvement of ≤25% of the vasculature of a single lobe; intermediate, involvement of >25% of the vasculature of a single lobe or multiple lobes with involvement of ≤25% of the entire vasculature; and extensive, involvement of multiple lobes with ≥25% of the entire vasculature. The criteria used in AMPLIFY were limited, involved ≤25% of the vasculature of a single lobe; extensive, there were 2 or more lobes involving ≥50% of the vasculature for each lobe; and intermediate, if neither of these definitions was met.
Extent of Anatomic Disease
The numbers of patients with DVT and PE and the extent of anatomic disease are also important distinguishing factors. A predefined pooled analysis of the EINSTEIN DVT and EINSTEIN PE studies 19 allows for these parameters to be compared with AMPLIFY, 8 RE-COVER, 12 RE-COVER II, 13 and Hokusai-VTE, 9 which recruited mixed VTE populations. The AMPLIFY, RE-COVER, and RE-COVER II studies included a higher number of patients diagnosed with DVT only (65.5%, 68.9%, and 68.1%, respectively) versus PE only (25.2%, 21.3%, and 23.2%, respectively). 8,12,13 This difference was less striking in Hokusai-VTE (59.7% DVT only and 40.3% PE only), 9 and in the EINSTEIN DVT and EINSTEIN PE pooled population (41.1% confirmed DVT only and 44.2% confirmed PE only). 19
Study Populations: Which Patients Were Not Included?
Patients with DVT events that were considered distal, splanchnic, cerebral, or upper limb located were not included in any of the recent phase III studies for acute VTE treatment. 8 –13 This is particularly relevant for physicians when considering that distal DVT can represent approximately 25% to 50% of all DVT cases seen in secondary care, 21 with some studies reporting an incidence of distal DVT as high as 60% to 70%. 22
Patients with malignancy were generally underrepresented in phase III studies, 23 –26 and patients with liver and severe renal impairment were excluded from the trials altogether. 8 –13 Further data are needed on the safety of DOACs in patients with these frequent comorbidities.
The rates of pregnancy-related and postpartum VTE are about 1 to 2/1000 pregnancies 27,28 ; however, the use of DOACs is contraindicated during pregnancy or breast-feeding, and thus there are no data on the efficacy and safety of these agents for the treatment of pregnancy-related DVT/PE. 29 –32 Furthermore, there are still no data on DOACs in pediatric patients. 29 –32
Variations between patient populations involved in phase III studies and those encountered in daily practice must be noted because these may lead to differences regarding the efficacy end points and safety outcomes of the DOACs obtained from real-world or phase III studies.
Were Possible Interactions With Frequently Prescribed Comedications Assessed?
Physicians frequently encounter comedication use in routine clinical practice, especially in the elderly patient population, which may interfere with the efficacy and safety of the DOACs. Dabigatran has direct drug–drug interactions with strong inhibitors or inducers of P-glycoprotein (P-gp), such as amiodarone and rifampicin, and HIV protease inhibitors are not recommended for concomitant treatment with this anticoagulant. 29 Direct factor Xa inhibitors are contraindicated when used in combination with drugs that strongly inhibit both cytochrome P4503A4 and P-gp, such as azole-antimycotics, and require caution when used in combination with strong inhibitors of only one of these pathways. 30 –32 Important drug interactions of the DOACs may also occur with nonsteroidal anti-inflammatory drugs (NSAIDs) and antiplatelet drugs, such as acetylsalicylic acid (ASA) or clopidogrel. 29 –32
The AMPLIFY, EINSTEIN DVT, and EINSTEIN PE studies excluded patients receiving potent cytochrome P4503A4 inhibitors, whereas the Hokusai-VTE study excluded patients receiving potent P-gp inhibitors. 8 –11 The RE-COVER and RE-COVER II studies did not exclude patients receiving these drugs. 12,13 The use of NSAIDs or ASA/clopidogrel differed among the phase III studies for the acute treatment of VTE; in AMPLIFY, up to 165 mg daily of ASA was permitted, whereas in the other studies, the maximum dose was 100 mg/d.
Heparin Pretreatment Versus Single-Drug Approach: What Are the Advantages and Disadvantages?
Two different approaches were adopted in phase III studies for the treatment of acute symptomatic DVT and/or PE. A dual-drug approach was used in dabigatran and edoxaban studies by actively including an initial parenteral treatment step with heparin (usually subcutaneous LMWH) before commencing the DOAC. 9,12,13 In the standard of care (SOC) arm, parenteral anticoagulation was stopped, and treatment with warfarin only was continued after achieving a therapeutic international normalized ratio. 9,12,13 All patients in the RE-COVER study received parenteral anticoagulation with UFH, LMWH, or fondaparinux for a median of 3.0 days (interquartile range of 2.0-4.0 days) prior to randomization and a median of a further 6.0 days (interquartile range of 5.0-8.0 days) after randomization. 12 In the RE-COVER II study, patients in the dabigatran and warfarin groups received parenteral anticoagulation for a total duration (mean ± standard deviation) of 9.4 ± 3.8 and 9.6 ± 4.1 days, respectively (treatment duration after randomization of 6.8 ± 3.4 and 7.1 ± 3.0 days, respectively). 13 All patients in the Hokusai-VTE study received parenteral treatment postrandomization with open-label UFH or an LMWH (enoxaparin) for a median duration of 7 days. 9
In the EINSTEIN DVT, EINSTEIN PE, and AMPLIFY studies, a single-drug approach was investigated with an initial intensified regimen followed by a lower dose after the acute phase. Rivaroxaban was evaluated as an initial intensified 15 mg twice-daily dosing regimen for 21 days followed by 20 mg once-daily dosing for long-term and extended treatment. 10,11 The intensified treatment period in AMPLIFY was limited to 1 week of apixaban 10 mg twice daily, followed by apixaban 5 mg twice daily. 8 The decision to use an oral single-drug approach from the start simplifies the management of symptomatic VTE by avoiding bridging with a parenteral anticoagulant. However, it is common clinical practice for patients with a suspected DVT or PE to receive heparin treatment while waiting for diagnosis; therefore, a high proportion of patients received a parenteral anticoagulant before being randomized to one of the treatment arms in EINSTEIN DVT (72%), EINSTEIN PE (92%), and AMPLIFY (86%). 8,10,11 Prerandomization treatment was usually for 1 or 2 days only, and patients in the EINSTEIN studies were considered ineligible if they received LMWH, fondaparinux, or UFH for longer than 2 days. 10,11 Patients who had received more than 2 doses of a once-daily LMWH regimen, fondaparinux, or a VKA, or more than 3 doses of a twice-daily LMWH regimen, or more than 36 hours of continuous intravenous heparin, were excluded from the AMPLIFY study. 8 A subgroup analysis of the EINSTEIN DVT and EINSTEIN PE studies showed that no significant difference existed in the treatment effect of rivaroxaban versus enoxaparin/VKA in patients who did or did not receive prestudy heparin. 32 This supports the conclusion that initial heparin treatment is, in fact, unnecessary when treating patients with symptomatic VTE with rivaroxaban. 33
A single-drug approach may reduce the requirement for hospitalization and its duration, which will ultimately reduce VTE-related treatment costs. For example, rivaroxaban treatment was associated with a significantly reduced (P < .0001) length of hospital stay compared with standard therapy in both the EINSTEIN DVT and EINSTEIN PE studies: 54% and 45% of rivaroxaban-treated patients diagnosed with DVT or PE, respectively, were hospitalized for less than 6 days, compared with 31% and 33% of those receiving standard care. 34 A higher proportion of patients receiving standard therapy were hospitalized for 6 to 10 days compared with rivaroxaban: 29% in the rivaroxaban group versus 45% in the standard therapy group in EINSTEIN DVT, and 39% and 46%, respectively, in EINSTEIN PE. 34
Apixaban was associated with a significant reduction in all-cause hospitalizations after the index event (hazard ratio [HR] 0.807; 95% confidence interval [CI] 0.652-0.998; P = .048) compared with standard therapy in the AMPLIFY study. The rates of all-cause hospitalizations within the first 30 days after the index event were 2.3% with apixaban and 3.4% with standard therapy (HR = 0.679; 95% CI 0.490-0.939; P = .019). The median time to first hospitalization was longer for apixaban-treated patients compared with those receiving standard therapy (63.0 vs 34.5 days, respectively). 35
The large amount of evidence and experience with the use of heparin, the mainstay of acute VTE treatment for decades, prompts physicians to feel more comfortable treating VTE with these parenteral agents, particularly those patients who are less stable or who have known comorbidities. However, the need for initial in-hospital treatment requiring monitoring of heparin dosage and the inconvenience of parenteral administration are limitations that could be overcome by DOACs with a single-drug regimen, reducing the need for hospitalization and lowering costs.
Recurrent VTE: How Is it Defined and What Is the Risk Reduction?
The primary efficacy end point in all 6 acute VTE treatment studies was symptomatic, recurrent VTE defined as the composite of DVT and nonfatal or fatal PE. 8 –13 All studies demonstrated noninferiority to standard therapy for the reduction of recurrent venous thromboembolic events using noninferiority limits that were compatible with the a priori-calculated noninferiority margin. 36 These results support the use of DOACs as an effective alternative to current standard therapy (Table 2).
Comparison of Outcomes of Venous Thromboembolism Studies That Compared Direct Oral Anticoagulants With Standard Therapy.
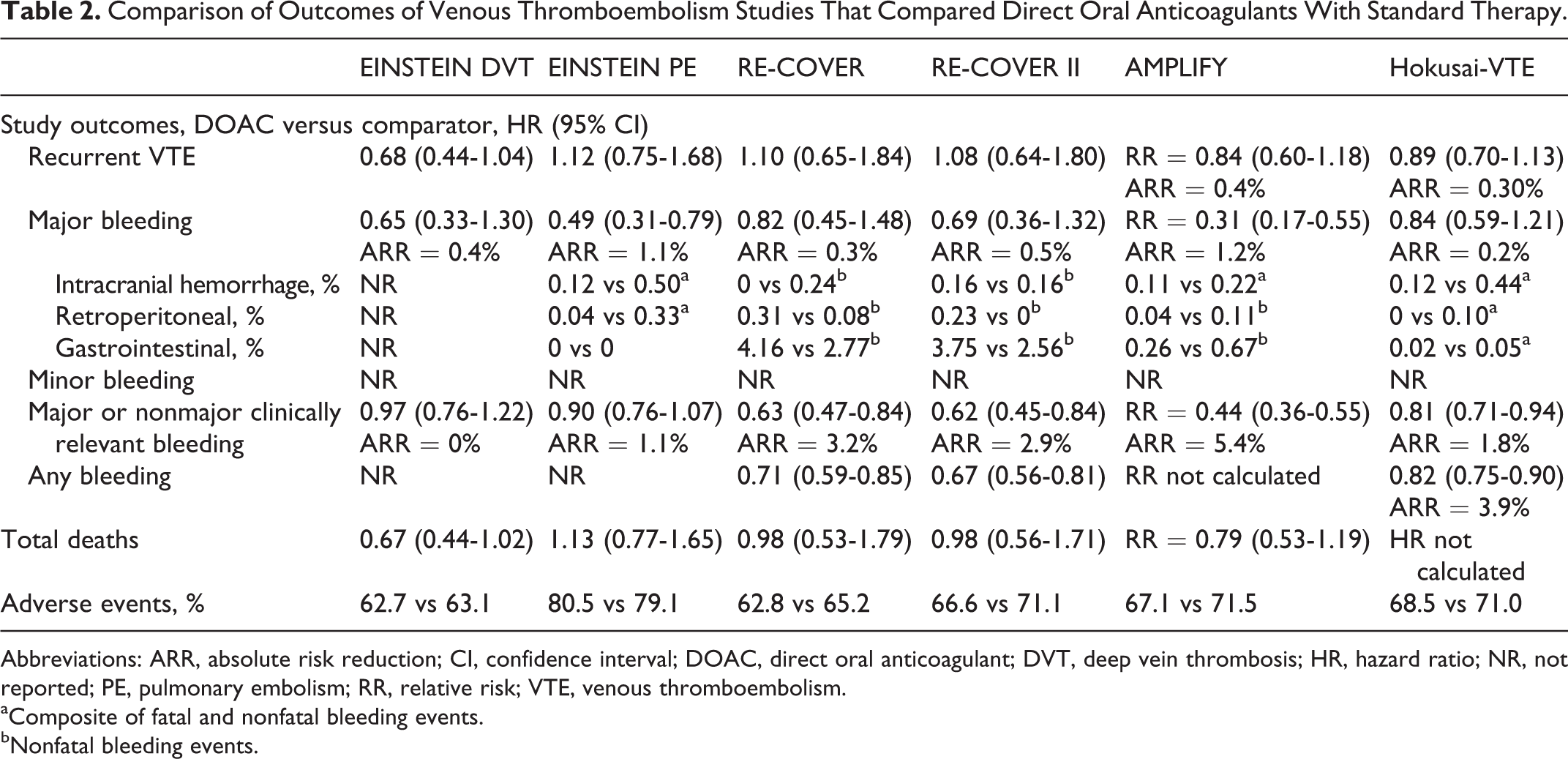
Abbreviations: ARR, absolute risk reduction; CI, confidence interval; DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; HR, hazard ratio; NR, not reported; PE, pulmonary embolism; RR, relative risk; VTE, venous thromboembolism.
aComposite of fatal and nonfatal bleeding events.
bNonfatal bleeding events.
Recurrent, fatal PE occurred at a rate of ≤0.2% in all study arms, with the exception of the RE-COVER study, for which this outcome was not provided. 8 –13 Rates of nonfatal PE in the pooled EINSTEIN studies and RE-COVER studies and rates of nonfatal PE with or without DVT in the AMPLIFY and Hokusai-VTE studies were approximately 0.6% to 1.5% in both arms and were similar to the proportion of recurrent DVT events, although recurrent DVT was more frequent in the comparator group across studies. 8,9,12,13,19
By the end of the intensified treatment regimen period with rivaroxaban (15 mg twice-daily for 21 days) in the EINSTEIN pooled analysis, the primary efficacy end point had occurred in 39 (0.9%) rivaroxaban-treated patients compared with 50 (1.2%) patients in the enoxaparin/VKA group. 19 These events accounted for 45.3% and 52.6% of all recurrent venous thromboembolic events recorded in each group (86 [2.1%] patients and 95 [2.3%] patients in the rivaroxaban and enoxaparin/VKA therapy arms, respectively) for the entire duration of the study. 19 These results highlight the importance of the initial phase of treatment, during which a substantial proportion of recurrences occur. 19 Similar data are not yet available for other DOACs.
Deep Vein Thrombosis: Do Clot Size and Location Affect Recurrent VTE?
The location and size of a DVT are important because these relate to the likelihood of embolism. Thrombosis of the deep calf vein is less serious than proximal vein thrombosis (popliteal veins, the superficial femoral, common femoral, or iliac veins) because it is less likely to embolize. 37,38 The assessment of a thrombus varied, however, between the phase III studies of the DOACs. In AMPLIFY, only 0.2% of the DVT events were later adjudicated as distal vein DVT, and these patients were included in the standard therapy group. 8 A higher proportion of patients had a DVT in the common femoral or iliac vein (43.1% in the apixaban group vs 42.3% in the standard therapy group) than in the femoral vein (32.6% vs 32.8%) or the popliteal vein (24.4% vs 24.7%). 8 In Hokusai-VTE, in patients with DVT only, the anatomic extent of the qualifying event was defined as “limited” if the thrombus was located in the popliteal vein, “intermediate” if located in the superficial femoral vein, and “extensive” if located in the common femoral or iliac vein. 9 A total of 24.4% versus 24.3% of patients treated with edoxaban or warfarin, respectively, had limited DVT, and 32.3% versus 31.5% had intermediate DVT, whereas a higher proportion of patients, 41.9% versus 42.8%, had DVT classified as extensive. 9 In the EINSTEIN pooled analysis, DVT or PE defined as of limited extent (ie, if thrombosis was not above the popliteal vein or if PE was confined to a single lobe involving ≤25% of the vasculature of that lobe) occurred in 19.5% of patients. 19 Extensive clot burden, when thrombosis involved the common femoral and/or iliac vein or if PE involved multiple lobes and affected >25% of the entire pulmonary vasculature, was reported in 32.5% of patients. 19 All other cases were classified as intermediate clot burden (45.3% of patients). 19 Rivaroxaban was similarly effective as standard therapy in patients presenting with limited (1.3% vs 2.3% with standard therapy; HR = 0.51, 95% CI 0.24-1.10), intermediate (2.2% vs 2.6%; HR = 0.82, 95% CI 0.54-1.24), or extensive (2.6% vs 2.0%; HR = 1.29, 95% CI 0.78-2.15) DVT or PE. 19
Pulmonary Embolism: Extent and Severity, What Is Important?
The European Society of Cardiology guidelines state that the severity of PE should be considered as the PE-related early mortality risk of an individual. 5 This risk is to be estimated on the basis of the presence of risk markers and not on the anatomic extent of the intrapulmonary emboli. 39 It is now recognized that anatomic severity may not translate into functional severity and this should be adequately and separately assessed. However, a classification of severity based on the anatomic extent of PE was used in several of the phase III studies of acute VTE treatment. 8,9,11 Patients with PE classified as intermediate in severity, based on the anatomic extent of the intrapulmonary emboli, were enrolled in higher proportions in EINSTEIN PE, 11 AMPLIFY, 8 and Hokusai-VTE 9 than those with limited or extensive PE. The proportions of patients in EINSTEIN PE with limited or extensive PE were higher and lower, 11 respectively, than in AMPLIFY 8 and Hokusai-VTE, 9 in which a relatively high number of patients were classified as having extensive PE. The anatomic extent of PE was not reported in the RE-COVER and RE-COVER II studies. 12,13 For example, in the Hokusai-VTE study, a subgroup of patients with severe PE and right ventricular dysfunction (RVD; identified as having N-terminal probrain natriuretic peptide levels ≥500 pg/mL and by computed tomography) was a priori identified for an exploratory analysis. 9 A reduction in rates of recurrent venous thromboembolic events was shown in the edoxaban group compared with the warfarin group (3.3% vs 6.2%; HR = 0.52, 95% CI 0.28-0.98) in this context. 9 The persistence of RVD after an acute PE is associated with higher PE-related mortality and is an independent predictor of recurrent VTE after hospital discharge compared with the absence of RVD 40 —this may partially account for the higher rate of VTE recurrence seen with warfarin treatment in this subgroup of patients (6.2%) compared with the overall population that underwent the same treatment (3.5%). 9 A subgroup analysis of AMPLIFY suggested noninferiority for apixaban in patients with extensive PE on the basis of rates of recurrent VTE, compared with standard therapy (1.4% vs 3.8%), and apixaban was associated with a reduction in the risk of major bleeding in these patients (0.8% vs 3.7%). 8 The EINSTEIN PE study did not include markers of PE severity or data on RVD or damage; however, indirect markers of PE severity, such as length of stay in intensive care units in 12% of included patients, were reported. A subgroup analysis of the rates of primary efficacy end point events for PE classified by anatomic severity consistently showed noninferiority with rivaroxaban treatment. The rates of recurrent VTE among patients for whom PE had been classified as first tercile, second tercile, or third tercile were 2.1% (16 of 751 patients), 2.2% (16 of 737), and 2.2% (18 of 810), respectively, in the rivaroxaban group and 1.5% (11 of 723), 2.1% (16 of 756), and 2.0% (16 of 820), respectively, in the standard therapy group. 11
Bleeding: Is it Reduced and Does it Contribute Significantly to the Net Clinical Benefit of DOACs?
The principal safety outcome varied between studies and was defined as major bleeding (according to the International Society on Thrombosis and Haemostasis definition) in AMPLIFY 8 and clinically relevant bleeding (composite of major and nonmajor clinically relevant bleeding) in EINSTEIN DVT, EINSTEIN PE, 10,11 and Hokusai-VTE. 9 Both of these outcomes were reported in the RE-COVER and RE-COVER II studies without designation of a principal safety outcome. 12,13 The definitions of major bleeding and clinically relevant bleeding were nonetheless similar between trials, allowing the results to be compared (Table 2). Major bleeding is an obvious concern for physicians. In the standard therapy arms of AMPLIFY, RE-COVER, RE-COVER II, Hokusai-VTE, and the EINSTEIN pooled study, rates of major bleeding events with VKA therapy were approximately 1.6% to 1.9%, 8,9,12,13,19 and thus consistent with the rates of 1% to 2% per year previously reported with VKA therapy. 41 Rates of major bleeding events for the DOACs varied from 0.6% to 1.6%, 8 –13 with significant reductions in major bleeding compared with standard therapy seen in AMPLIFY and the pooled EINSTEIN analysis (0.6% vs 1.8%; P < .001 and 1.0% vs 1.7%; P = .002, respectively). 8,19
Numerical reductions in the rates of major bleeding events at critical sites were seen with all agents when compared with VKA therapy. The most important reductions were in intracranial hemorrhage, seen with all 4 agents (but not consistently for dabigatran between the RE-COVER and RE-COVER II studies) and retroperitoneal bleeding events, seen with apixaban, edoxaban, and rivaroxaban. 8 –13 Rates of major or nonmajor clinically relevant bleeding events were similar (∼10%) in the heparin/VKA arms in the different studies, 8 –11 with the exception of a lower rate in RE-COVER and RE-COVER II (∼9% and ∼8%, respectively). 12,13 Significant reductions in rates of major or nonmajor clinically relevant bleeding events were seen in the DOAC arm in AMPLIFY, 8 Hokusai-VTE, 9 RE-COVER, 12 and RE-COVER II, 13 but not in the EINSTEIN pooled analysis, 19 possibly owing to the open-label design of the EINSTEIN DVT and EINSTEIN PE studies. 10,11 Apixaban and rivaroxaban treatment resulted in a significant net clinical benefit defined as the composite of recurrent VTE, death, and major bleeding outcomes in AMPLIFY, 8 and the composite of the rates of primary efficacy end point and major bleeding events over standard treatment in the EINSTEIN pooled analysis. 19 This result was not seen with edoxaban (determined on the basis of the composite of symptomatic recurrent VTE and major bleeding), mainly because of a nonsignificant reduction in major bleeding with this agent (2.9% vs 3.5% with warfarin; HR = 0.83; 95% CI 0.65-1.06). 9 Data from the EINSTEIN studies were analyzed further, with regard to the clinical impact of major bleeding events, by ranking the events in order of severity from 1 to 4 (least to most severe). Major bleeding events associated with rivaroxaban often had a milder presentation compared with major bleeding events linked to SOC. Although 82% of patients treated with rivaroxaban presented as categories 1 and 2, only 65% in the SOC group were associated with the same categories. Furthermore, rivaroxaban-associated major bleeding events took a milder clinical course than SOC-induced major bleeding events. 42
Although the exclusion criteria for the phase III clinical studies were consistent across some key criteria, there was variation between the complete lists of criteria between the AMPLIFY, EINSTEIN DVT and EINSTEIN PE, Hokusai-VTE, and RE-COVER and RE-COVER II studies. 8 –13 In AMPLIFY, for instance, patients with cancer who required treatment for >6 months with LMWH therapy and patients on dual antiplatelet therapy were excluded. 8 In the EINSTEIN DVT and EINSTEIN PE studies, concomitant use of ASA and clopidogrel at certain doses was allowed, and this may have been responsible for the observed increase in clinically relevant bleeding. 10,11
Key Patient Subgroups of Clinical Interest
“Fragile” Patients
For VTE treatment, the efficacy and safety of each DOAC have been shown to be similar to standard therapy in important subgroups. 8,9,12,13,19 A total of 19% of patients in the EINSTEIN pooled analysis were defined as fragile (age >75 years, calculated creatinine clearance [CrCl] <50 mL/min, or low body weight [≤50 kg]); these patients are known to have high rates of recurrent VTE. 19 Treatment of these patients with rivaroxaban was shown to be as efficient as standard treatment in reducing VTE recurrence (2.7% vs 3.8%; HR = 0.68; 95% CI 0.39-1.18), with a significant reduction in major bleeding (1.3% vs 4.5%; HR = 0.27; 95% CI 0.13-0.54), translating to a clear net clinical benefit (4.6% vs 8.4%; HR = 0.51; 95% CI 0.34-0.77) in comparison with standard treatment. 19 Edoxaban was more efficacious in fragile patients (defined as age ≥75 years, body weight ≤50 kg, or CrCl 30-50 mL/min) than treatment with warfarin (2.5% vs 5.0%). 9 Although no data were reported for major bleeding, no difference was seen between the rates of major and nonmajor clinically relevant bleeding events in each treatment arm (11.0% vs 13.7%) in this subgroup, which represented 17.2% of patients in Hokusai-VTE. 9 No data are currently available on the efficacy and safety of dabigatran or apixaban in this patient subgroup.
Malignancy
Patients with underlying malignancy are an important group in which clinical information on the effectiveness of the DOACs was obtained, because approximately 20% of all cases with VTE occur in patients with cancer. 43,44 However, in clinical practice, many patients with active cancer receive LMWH for 3 months or longer 4 and, therefore, the inclusion of patients with active malignancy in phase III studies was limited, resulting in a lack of data regarding the efficacy and safety of DOACs in those patients.
The number of patients with active cancer enrolled in the phase III studies decreased from the first (EINSTEIN DVT, 6.0%) to the most recent trial published (Hokusai-VTE, 2.5%; Table 1). As shown in Figure 1, in the Hokusai-VTE study (which included 208 patients with active cancer), the primary efficacy end point occurred in 3.7% of patients with active cancer who were treated with edoxaban, compared with 7.1% of patients receiving standard treatment, with rates of clinically relevant bleeding of 18.3% versus 25.3%, respectively. 9 Data from the RE-COVER studies are limited: 3.5% versus 4.7% with standard treatment for the primary efficacy end point in a pooled analysis of RE-COVER and RE-COVER II; however, there are no accompanying safety data for this subgroup. 13 A subgroup analysis of the pooled data from EINSTEIN DVT and EINSTEIN PE showed that rivaroxaban has been found to be as effective as standard treatment in reducing VTE recurrence (5.1% vs 7.1% for enoxaparin/VKA; HR = 0.69, 95% CI 0.36-1.33) and major bleeding (2.8% vs 5.0% for enoxaparin/VKA; HR = 0.53, 95% CI 0.23-1.23) in patients with cancer, with a significant net clinical benefit statistically favoring rivaroxaban (8.2% vs 13.2%; HR = 0.60, 95% CI 0.36-0.99). A total of 430 patients had active cancer at baseline, with a further 167 patients diagnosed during the study. 19 Prins and colleagues conducted a comprehensive analysis of efficacy, safety, and survival of patients with VTE and active cancer. 45 Of 354 patients treated with rivaroxaban and 301 patients treated with SOC, recurrent venous thromboembolic events occurred in 4.5% and 6.6% of patients, respectively (HR = 0.67, 95% CI 0.35-1.30), and major bleeding events were seen in 2.3% and 5.0% of patients, respectively (HR = 0.42, 95% CI 0.18-0.99). A 16.4% mortality rate was observed in the rivaroxaban group, whereas the SOC group showed a rate of 17.6% (HR = 0.93, 95% CI 0.64-1.35). 45
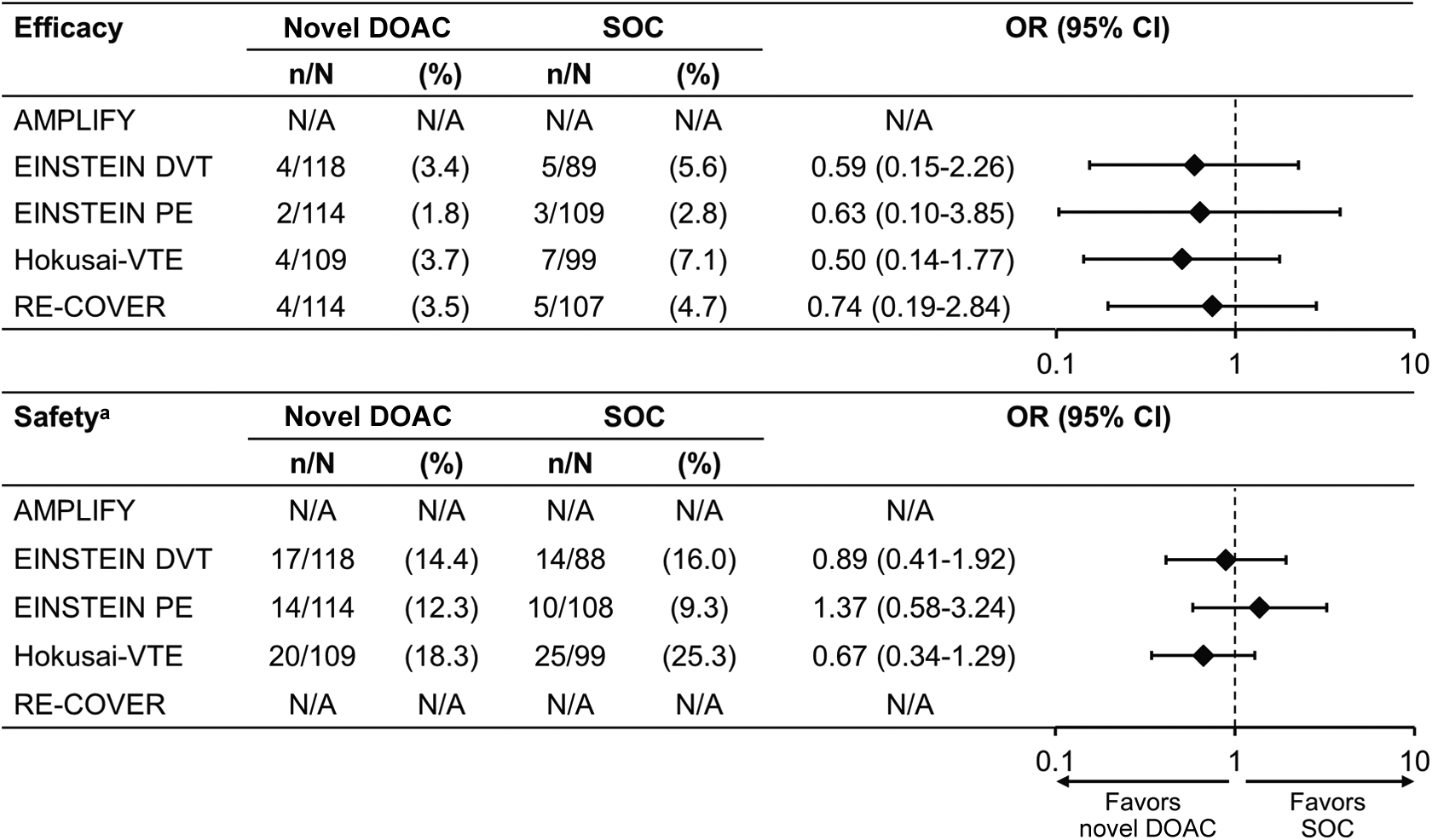
Relative efficacy and safety of direct oral anticoagulants compared with standard of care in the cancer subgroup in the specified phase III clinical studies in venous thromboembolism treatment. 8 –12 All OR calculations are based on the relevant publications. aORs for safety were calculated based on the rates of major bleeding and nonmajor clinically relevant bleeding events for the EINSTEIN and Hokusai-VTE studies and major bleeding events for AMPLIFY. CI indicates confidence interval; DOAC, direct oral anticoagulant; N/A, not available; OR, odds ratio; SOC, standard of care.
Data have only recently been presented in this subgroup of patients for the AMPLIFY study. 46 Of the 5395 patients assessed for efficacy and safety, 3.1% (n = 169) presented with active cancer. A 3.7% rate of recurrent VTE was seen in the apixaban group compared with 6.4% in the warfarin group (relative risk [RR] 0.56; 95% CI 0.13-2.37). Major bleeding was observed in 2.3% and 5.0% of patients with cancer for apixaban and warfarin, respectively (RR = 0.45; 95% CI 0.08-2.46). For patients who did not present with cancer at entry of the study, rates of recurrent VTE were 2.2% and 2.6% in the apixaban and warfarin groups (RR = 0.86; 95% CI 0.60-1.22), respectively, and 0.5% and 1.7% for major bleeding rates (RR = 0.29; 95% CI 0.16-0.54).
Renal Impairment
Renal function declines with age; therefore, elderly patients often present with some level of renal impairment. Severe renal impairment was an exclusion criterion for all phase III VTE treatment studies (CrCl <30 mL/min in Hokusai-VTE, EINSTEIN DVT, EINSTEIN PE, RE-COVER, and RE-COVER II, and CrCl <25 mL/min in AMPLIFY). 8 –13 In the EINSTEIN DVT and EINSTEIN PE pooled population, 7.6% and 24.4% of patients had moderate (CrCl 30-<50 mL/min) or mild (CrCl 50-79 mL/min) renal impairment, respectively. 19 In RE-COVER, 4.7% and 21.7% of patients had moderate or mild renal impairment, respectively. 12,13 In the AMPLIFY study, approximately 6% of the overall population had moderate renal impairment, and approximately 20% had mild renal impairment. 8 In the Hokusai-VTE study, 6.6% of patients had moderate renal impairment, with the remaining patients having either normal or mild renal impairment (CrCl >50 mL/min). No individual data were presented for patients with mild renal impairment. 9
Patients with renal impairment are at a higher risk of VTE but also have an increased risk of bleeding. In general, the available data show no significant difference in efficacy between any of the DOACs and standard treatment in subgroups defined by CrCl (Figure 2). In patients with renal impairment in the Hokusai-VTE study, the primary efficacy end point occurred in more patients on standard therapy compared with those receiving edoxaban (5.9% vs 3.0%, respectively), although this trend did not reach significance. 9
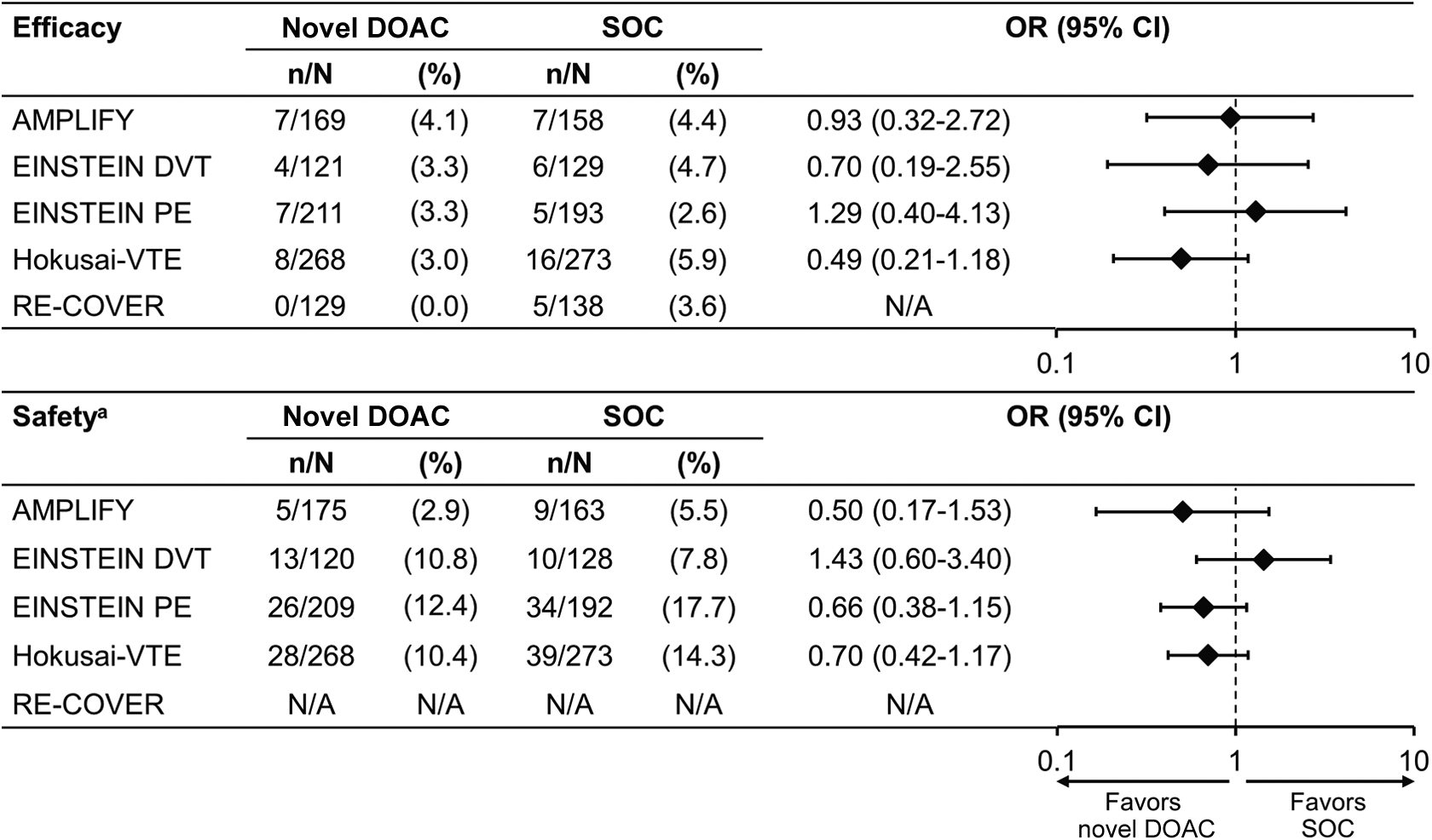
Relative efficacy and safety of direct oral anticoagulants compared with standard of care in the renal impairment subgroup in the specified phase III clinical studies in venous thromboembolism treatment. 8 –12 OR calculations are based on the relevant publications. aORs for safety were calculated based on rates of major bleeding and nonmajor clinically relevant bleeding events in the EINSTEIN and Hokusai-VTE studies and major bleeding events in AMPLIFY. CI indicates confidence interval; DOAC, direct oral anticoagulant; N/A, not available; OR, odds ratio; SOC, standard of care.
In the pooled EINSTEIN DVT and EINSTEIN PE analysis, rivaroxaban was shown to be as effective (P = .001) as standard treatment in preventing VTE recurrence in patients with mild (2.4% vs 3.1% with enoxaparin/VKA) or moderate (3.4% vs 3.2%) renal impairment as in those with normal renal function (1.8% vs 1.9%), but with a significant reduction in major bleeding events (patients with normal function, 0.8% vs 1.0%; mild renal impairment, 1.4% vs 3.0%; and moderate renal impairment, 0.9% vs 3.9%, respectively). 47
Nonsteroidal Anti-inflammatory Drugs and ASA
A total of 14.6% and 22.8% of patients (safety population) received concomitant ASA or NSAIDs in a prospective analysis of pooled data from the EINSTEIN DVT and EINSTEIN PE studies. An assessment of the risk of major bleeding and clinically relevant bleeding events was conducted for concomitant administration of NSAIDs or ASA with either standard therapy or rivaroxaban. 48 The rates of major bleeding events were 4.7/100 patient-years and 3.3/100 patient-years when rivaroxaban was combined with NSAIDs or ASA, respectively, versus 1.4/100 patient-years (HR = 2.56; 95% CI 1.21-5.39) and 1.6/100 patient-years (HR = 1.50; 95% CI 0.63-3.61) during nonuse. Major bleeding occurred at a rate of 8.4/100 patient-years for standard therapy with use of NSAIDs versus 2.7/100 patient-years during nonuse (HR = 2.28; 95% CI 1.28-4.04), and 6.9/100 patient-years for concomitant treatment with ASA compared with 2.9/100 patient-years during nonuse (HR = 1.50; 95% CI 0.74-3.05). The results showed an increase in the risk of major bleeding and clinically relevant bleeding events with concomitant administration of NSAIDs or ASA and were consistent across rivaroxaban, standard therapy, and pooled data. A similar trend was observed for clinically relevant bleeding. Patients with DVT and PE taking anticoagulants should avoid taking NSAIDs for prolonged periods without an important clinical indication for NSAID use. Additionally, HR values were calculated for rivaroxaban versus enoxaparin/VKA use. No significant differences in the rates of major bleeding or clinically relevant bleeding events were detected between the rivaroxaban and enoxaparin/VKA regimens with concomitant ASA or NSAID use (clinically relevant bleeding with concomitant use of ASA [HR = 0.99; 95% CI 0.67-1.46]; major bleeding with concomitant use of ASA [HR = 0.54; 95% CI 0.19-1.51]; clinically relevant bleeding with concomitant use of NSAIDs [HR = 1.04; 95% CI 0.73-1.47]; and major bleeding with concomitant use of NSAIDs [HR = 0.60; 95% CI 0.26-1.37]). In the corresponding comparator group without NSAID/ASA use, an increase in the rate of major bleeding events was observed in enoxaparin/VKA-treated patients compared with the rivaroxaban-treated patients, but no difference was seen for clinically relevant bleeding (clinically relevant bleeding/no ASA [HR = 0.91; 95% CI 0.78-1.06]; major bleeding/no ASA [HR = 0.54; 95% CI 0.36-0.82]; clinically relevant bleeding/no NSAID [HR = 0.91; 95% CI 0.79-1.06]; and major bleeding/no NSAID [HR = 0.54; 95% CI 0.35-0.83]). 48 In a prespecified subgroup analysis of pooled data from RE-COVER and RE-COVER II, dabigatran was compared with warfarin in patients concomitantly treated with NSAID or ASA. Therefore, a direct statistical conclusion cannot be drawn on the additional impact of NSAID/ASA use on the efficacy and safety of dabigatran and warfarin. In 21.9% and 9.3% of patients who received NSAIDs or ASA, respectively, a similar efficacy was determined (rates of VTE or VTE-related death) between anticoagulant treatments, irrespective of the concomitant use of NSAIDs (2.6% with dabigatran vs 2.5% with warfarin, and 2.8% vs 2.0%, without or with NSAIDs, respectively) or low-dose ASA (2.6% vs 2.4% and 3.1% vs 2.3%, without or with ASA, respectively). 49 Significantly lower rates of major bleeding (HR = 0.60; 95% CI 0.36-0.99) and major bleeding or clinically relevant bleeding (HR = 0.56; 95% CI 0.45-0.71) events were observed with dabigatran compared with warfarin, regardless of concomitant medication status. 49 No data have yet been made available regarding the effect of concomitant medication with NSAIDs and ASA on the efficacy or safety of apixaban and edoxaban.
Extension Studies
The risk of recurrence is known to be high after discontinuing anticoagulation therapy in patients with unprovoked acute proximal DVT or PE. The cumulative incidence of recurrent VTE reaches approximately 40% within 10 years, and more than 10% of these recurrences are fatal events with either confirmed PE or possible PE that cannot be excluded as the cause of death. 20 Nonetheless, the decision to provide extended anticoagulation treatment is always weighed against the higher risk of bleeding. For example, extended VKA treatment, although effective, increases the risk of major bleeding, with rates as high as 2% to 4%/year in patients with VTE receiving long-term treatment with VKAs. 50 –52 Three studies have compared the efficacy and safety of apixaban (AMPLIFY-EXT), 14 dabigatran (RE-SONATE), 15 or rivaroxaban (EINSTEIN EXT) 10 versus placebo for extended treatment of VTE after initial anticoagulation therapy and have reported broadly similar results (Table 3). Extended therapy differed among studies and was provided for an additional 12 months with apixaban after 6 to 12 months of initial treatment, 14 6 or 12 months with rivaroxaban after 6 to 12 months of initial treatment, 10 and for 6 to 18 months with dabigatran after at least 3 months of initial treatment. 15 Reductions in the risk of recurrent VTE ranging from 81% to 92% compared with placebo were observed with these agents. 10,14,15
Results of Extension Studies for Venous Thromboembolism Prevention With Direct Oral Anticoagulants.

Abbreviations: ARI, absolute risk increase; ARR, absolute risk reduction; CI, confidence interval; DOAC, direct oral anticoagulant; DVT, deep vein thrombosis; HR, hazard ratio; NR, not reported; PE, pulmonary embolism; RR, relative risk; VTE, venous thromboembolism.
Preliminary data for apixaban showed a reduced number of hospitalization events due to VTE recurrence in the context of extended treatment for secondary prevention of VTE, with 26% and 18% of hospitalizations recorded in the apixaban 2.5 mg twice-daily and 5 mg twice-daily groups, respectively, compared with 52% of hospitalizations in the placebo group. 35 Extended anticoagulation with apixaban also prolonged the average time to first hospitalization by 43 days for the 2.5 mg twice-daily regimens and 49 days for the 5 mg twice-daily regimens. 35
Patients receiving a DOAC had a higher rate of nonmajor clinically relevant bleeding events, 10,14,15 but these are more easily managed than major bleeding, for which no significant increases occurred compared with placebo, although a trend toward increased major bleeding was seen. 10,14,15 This numerical increase in major bleeding events was not observed with the apixaban 2.5 mg and 5 mg twice-daily regimens in the AMPLIFY-EXT study. 35 On the other hand, the placebo study arms showed rates of recurrent VTE ranging from 5.6%/year to 11.6%/year, 10,14,15 supporting the benefit of extended anticoagulation treatment and suggesting some differences in risk between placebo populations, possibly owing to study design differences.
Only one study has compared extended treatment with a DOAC with an active comparator such as a VKA. In the RE-MEDY study, patients receiving dabigatran for 6 to 36 months, after an initial 3 to 6 months of anticoagulant treatment, showed a trend toward a higher incidence of recurrent or fatal VTE (1.8% vs 1.3%; HR = 1.44; 95% CI 0.78-2.64; P = .01 for noninferiority) and acute coronary events (0.9% vs 0.2%; P = .02) than patients receiving warfarin. 15 However, dabigatran was associated with lower rates of major bleeding (0.9% vs 1.8%; HR = 0.52; 95% CI 0.27-1.02; P = .06), major or clinically relevant bleeding (5.6% vs 10.2%; HR = 0.54; 95% CI 0.41-0.71; P < .001), and any bleeding (19.4% vs 26.2%; HR = 0.71; 95% CI 0.61-0.83; P < .001) compared with warfarin. 15 These results are consistent with those of the RE-LY study of dabigatran for stroke prevention in patients with atrial fibrillation, in which an increased risk of myocardial infarction was seen with dabigatran compared with warfarin. A similar trend of increased myocardial infarction was seen in the RE-COVER pooled analysis and a recent meta-analysis. 13,53,54
Future Prospects for VTE Treatment With DOACs
All of the DOACs are now approved for the treatment of DVT and/or PE and the prevention of VTE in Europe, the United States, and other countries around the world. Treatment with LMWH is still the main option for pregnancy-related VTE. Although encouraging results in subgroup studies support a role for DOACs in managing VTE in patients with cancer and those with different levels of renal impairment, further data are required to clarify the efficacy and safety of all agents in patients with these conditions. Real-world studies in unselected patient populations may help to address evidence gaps on the use of DOACs, including in patients with underlying malignancy or renal impairment, as well as providing reassurance to clinicians on the applicability of clinical trial data to the patients they see in their daily practice. At the moment, there is only very limited published real-world data available on the use of the DOACs for treatment of VTE. Patients with VTE treated with rivaroxaban (n = 575) in the Dresden NOAC Registry were older than those enrolled in the EINSTEIN studies (median age 68 years vs mean age 57 years), and the rate of major bleeding in these patients was 4.1%/year. 19,55 Patients with VTE treated with rivaroxaban (n = 103) at a Spanish center were similar in age as those in the EINSTEIN studies. Results of an open follow-up study confirmed that rivaroxaban represented a therapeutic alternative to VKAs with practical advantages. 56 Lastly, a retrospective, propensity-score matched, US database analysis of patients with DVT treated with rivaroxaban or LMWH/warfarin reported that rivaroxaban treatment was associated with a 27% reduction in hospital admission rates. 57 In addition, numerous noninterventional and registry studies enrolling patients with VTE are ongoing or have been completed recently and expected to report results within the next few years, these include the XALIA noninterventional trial and the Prevention of Thromboembolic Events - European Registry in Venous Thromboembolism (PREFER in VTE), Global Anticoagulant Registry in the FIELD-VTE (GARFIELD-VTE), Follow-up In Rivaroxaban Patients in Setting of Thromboembolism (FIRST), and Dresden NOAC registries.
Conclusions
The DOACs have shown at least similar efficacy and similar or better safety for the treatment and prevention of VTE recurrence and all-cause mortality compared with standard treatment. These agents offer a more convenient option than VKAs for extended treatment in the outpatient setting. Moreover, rivaroxaban and apixaban have been tested as a single-drug approach and have been shown to provide rapid initial anticoagulation without the need for bridging anticoagulation with parenteral agents. The DOACs have not been compared head to head, and with minimal data existing to guide selection between these agents for a given patient, this overview of data from phase III studies seeks to assist physicians in making informed decisions by summarizing how the efficacy and safety of the DOACs are influenced by differences in study design. Anticoagulant therapy will continue to be the cornerstone of VTE treatment, and because DOACs may provide improved patient care with better benefit–risk profiles across a broad spectrum of patients with VTE compared with standard therapy, these agents have the potential to become the new SOC.
Footnotes
Acknowledgments
The authors would like to acknowledge Jasmina Saric for editorial support, with funding from Bayer HealthCare Pharmaceuticals and Janssen Scientific Affairs, LLC.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ATC is a medical consultant and has received consultancy and clinical trial funding from pharmaceutical companies including Astellas, AstraZeneca, Bayer, Boehringer-Ingelheim, BMS, Daiichi, GSK, Johnson & Johnson, Mitsubishi Pharma, Pfizer, Portola, Sanofi-aventis, Schering-Plough, and Takeda. ATC is an advisor to the UK Government Health Select Committee, the All-Party Working Group on Thrombosis, the Department of Health, and the NHS, on the prevention of VTE. ATC is also an advisor to Lifeblood, The Thrombosis Charity, and is the founder of the European educational charity the Coalition to Prevent VTE. SG and AH have no disclosures to make.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
