Abstract
Inflammation is pivotal in the pathogenesis and development of cerebral venous thrombosis (CVT). Herein, we aimed to assess the anti-inflammatory effects of batroxobin combined with anticoagulation in CVT. Participants were categorized into the batroxobin group (batroxobin combined with anticoagulation) and the control group (anticoagulation only). Regression analysis was employed to explore the association between the number of episodes of batroxobin administration and the fluctuation of inflammatory indicators, as well as the proportion of patients with inflammatory indicators that were reduced after batroxobin use. Twenty-three cases (age: 39.9 ± 13.8 years, female: 39.1%) in the batroxobin group and 36 cases (40.3 ± 9.6 years, 52.8%) in the control group were analyzed. Compared to the control group, batroxobin combined with anticoagulation significantly decreased fibrinogen (P < .001), platelet–lymphocyte ratio (PLR) (P = .016) and systemic immune–inflammation index (SII) (P = .008), and increased the proportion of the patients with lower fibrinogen (P < .001), neutrophil–lymphocyte ratio (NLR) (P = .005), PLR (P = .026), and SII (P = .006). Linear analysis showed that as the number of episodes of batroxobin administration increased, the fibrinogen (P < .001), the PLR (P = .001), and the SII (P = .020) significantly decreased. Logistic regression analysis showed as the number of episodes of batroxobin administration increased, the ratio of the patients with decreased NLR (P = .008) and PLR (P = .015), as well as SII (P = .013), significantly increased. Batroxobin could decrease NLR, PLR, and SII in CVT. The effect was related to the number of episodes of batroxobin administration. Besides reducing fibrinogen and indirect thrombolysis effects, this may be another critical benefit of batroxobin for CVT.
Keywords
Introduction
Cerebral venous thrombosis (CVT) represents a rare manifestation of cerebrovascular disease that detrimentally impacts the well-being and quality of life.1,2 Insights gleaned from large international registries in recent years have significantly advanced our understanding of the pivotal role of inflammation in the initiation and progression of CVT. Clinical investigations documenting the pronounced activation of inflammatory biomarkers in serum and cerebrospinal fluid during CVT provide empirical support for inflammation's causal involvement in the onset of CVT.3,4 Blood biomarkers associated with inflammations also exhibit correlations with the severity and prognosis of CVT.5–8 However, the definitive conclusion regarding the potential improvement in patient outcomes through anti-inflammatory therapy remains elusive. Consequently, anti-inflammatory therapy is not presently integrated into the standard treatment regimen for CVT patients, underscoring the pressing need to explore the therapeutic efficacy of anti-inflammatory drugs in this population.
Batroxobin, functioning as a fibrinogen-lowering agent, has demonstrated a commendable recanalization effect in managing CVT when combined with anticoagulation.9,10 Although previous researches had noticed its anti-inflammatory effect,11–13 batroxobin has not been adopted as a mainstream antithrombotic and anti-inflammatory agent in clinical setting; mainly it is commonly used as an adjunctive agent in CVT treatment. Fibrinogen, a key player in peripheral inflammation, was reduced significantly during batroxobin use.14,15 The mechanism of batroxobin in correcting CVT may not be limited to its reduction of fibrinogen and indirect thrombolysis but also its remarkable anti-inflammatory effects. Herein, we aimed to substantiate the hypothesis that the anti-inflammatory effect of batroxobin also plays a vital role in CVT correction except for its approved well-reducing fibrinogen and indirect thrombolysis effects.
Method
Study Population
Patients aged ≥18 years with confirmed CVT through computerized tomography venography or magnetic resonance venography in Xuanwu Hospital, Capital Medical University, were retrospectively enrolled from July 2017 through July 2023. Detailed diagnostic criteria were reported previously. 1 The inclusion and exclusion criteria were as follows:
Inclusion criteria:
Patients meeting the diagnostic criteria of CVT. Age ranged from 18 to 80. Anticoagulation initiated immediately upon CVT confirmation. Insufficient inflammatory indicators assays (less than 2). Batroxobin and anticoagulation were administered before the initial laboratory test. Use anti-inflammatory medications with effects (eg, antibiotics, glucocorticoids) during hospitalization.
Exclusion criteria:
Intervention and Grouping
All patients underwent standard anticoagulation when CVT was confirmed. We did not make a deliberate decision for the patients to undergo batroxobin. We recommend all eligible patients (the level of fibrinogen is more than 1.0 g/L) take batroxobin, and the treatment is carried out based on the individual's personal choice. Patients underwent intravenous infusion immediately after signing an informed consent [Batroxobin injection (produced by Beijing Tuobixi Pharmaceutical Co., Ltd, the approval number was H20031074) 10 BU for the first time, followed by 5 BU every other day, for a total of 2 times or more, unless the level of fibrinogen dropped to 1.0 g/L or less] during treatment were enrolled in the batroxobin group. Patients who underwent standard anticoagulation alone were in the control group.
Data Collection
Comprehensive clinical data, including demographics, symptoms, imaging, and CVT risk factors, were recorded. Peripheral blood was collected twice to assess inflammatory indicators, including neutrophil count (standard reference value: 1.8-6.4 × 109/L), lymphocyte count (1-3.3 × 109/L), monocyte count (0.2-0.7 × 109/L), platelet count (100-300 × 109/L), fibrinogen (2-4 g/L), hypersensitive C-reactive (0-3 mg/L), and interleukin-6 (<7 pg/mL). The derivative indicators of peripheral inflammatory indicators were also included in the study, including neutrophil–lymphocyte ratio (NLR), lymphocyte–monocyte ratio, platelet–lymphocyte ratio (PLR), and systemic immune–inflammation index (SII). Neutrophil–lymphocyte ratio was calculated by dividing the neutrophil count by the lymphocyte count. Platelet–lymphocyte ratio was calculated by dividing the platelet count by the lymphocyte count. Lymphocyte–monocyte ratio was calculated by dividing the lymphocyte count by the monocyte count. SII was derived from platelet count × neutrophil/lymphocyte count.4,6 The first collection of inflammatory markers occurred after admission and before the initiation of batroxobin and anticoagulation therapy. The second collection of inflammatory markers occurred at least one week after the start of treatment and before discharge. The interval between the 2 laboratory tests was also recorded. All tests of the same type were conducted in the same laboratory at our center using the same equipment model and the same testing methods. Inflammatory indicator variation was defined as the difference between the second and first values. The inflammatory indicator variation < 0 was defined as the decreased inflammatory indicator.
Statistical Analysis
SPSS (version 27) performed statistical analysis. Continuous variables were described using mean ± standard deviation (SD). Categorical variables were expressed as numbers (percentages) and analyzed using a chi-square test. Independent sample t-test was used to compare the variation of inflammatory indicators between the batroxobin group and the control group after treatment. Paired sample t-test was used to compare the variation of inflammatory indicators before and after treatment in the batroxobin group. Linear regression analyzed the correlation between the number of episodes of batroxobin administration and inflammatory indicator variations. Logistic regression analysis examined the correlation between the number of episodes of batroxobin administration and the number of patients with decreased inflammatory indicators. A receiver operating characteristic (ROC) analysis was conducted to demonstrate the sensitivity and specificity of the number of episodes of batroxobin administration to decrease inflammatory indicators. The area under the curve (AUC) reflecting the accuracy of diagnosis was also evaluated. The optimal cutoff values were considered with maximal sensitivity and specificity. Propensity score matching (PSM) was used to rule out confounding factors. It was determined that P < .05 was statistically significant.
Results
Baseline Characteristics
In this study, 59 patients were included based on the inclusion and exclusion criteria. In which 23 cases (mean age, 39.9 ± 13.8 years, female, 39.1%) were enrolled in the batroxobin group and 36 cases (mean age, 40.3 ± 9.6 years, female, 52.8%) in the control group. There were no significant differences in baseline data between the 2 groups. SPM successfully matched 21 pairs of patients to rule out confounding factors (sex and age), and there were also no significant differences in baseline characteristics among them. Comparison of specific baseline data can be seen in Table 1.
Characteristics of the Enrolled Patients.
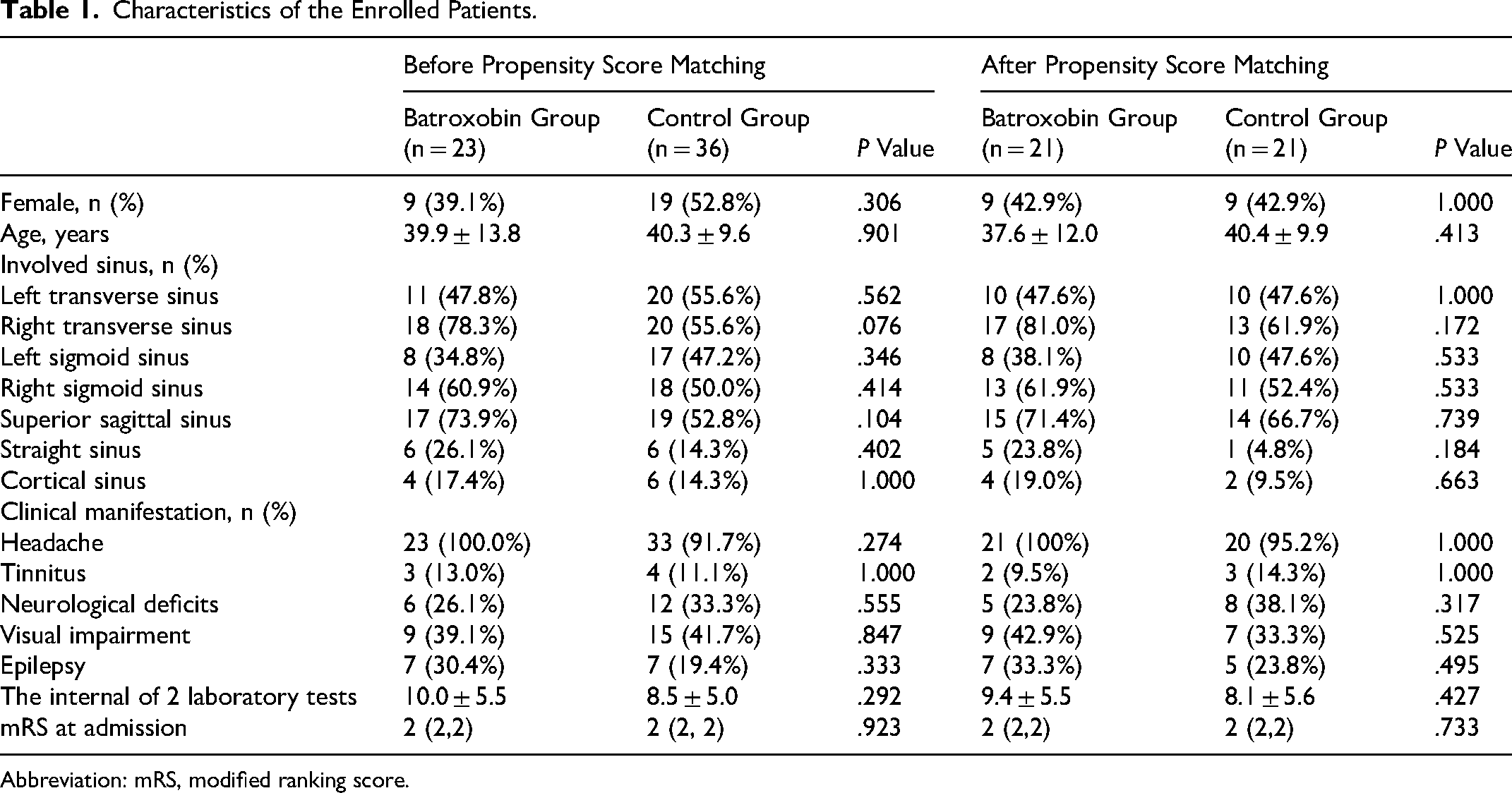
Abbreviation: mRS, modified ranking score.
The Difference in the Variations of Inflammatory Indicators Between the Batroxobin and Control Group
By comparing the variations of inflammatory indicators before and after treatment between the batroxobin and control group, we found that the reduction of PLR (mean [SD]: −22.9 [42.0] vs 7.4 [44.3]; P = .013), SII (−133.3 [213.9] vs 33.8 [276.1]; P = .019), and fibrinogen (−1.7 [1.1] vs −0.6. [1.2]; P < .001) in batroxobin group was more remarkable than that in the control group. After PSM, the extent of reduction in PLR (−22.9 [43.1] vs 10.1 [44.5]; P = .019), SII (−133.6 [219.2] vs 7.0 [294.6]; P = .037), and fibrinogen (−1.8 [1.1] vs −0.6. [1.1]; P < .001) remained a significant difference between the 2 groups (Table 2). Paired sample t-test results also showed that batroxobin combined with anticoagulation significantly reduced platelet count (mean difference [SD]: −30.5 [67.0]; P = .040), PLR (−22.4 [41.1]; P = .016), SII (−128.6 [210.2]; P = .008), and fibrinogen (−0.9 [1.2]; P < .001) compared to pretreatment (Supplemental Table 1).
Differences of the Variations of Inflammatory Indicators Between the Batroxobin and Control Group After Treatment (Independent Sample T Test).
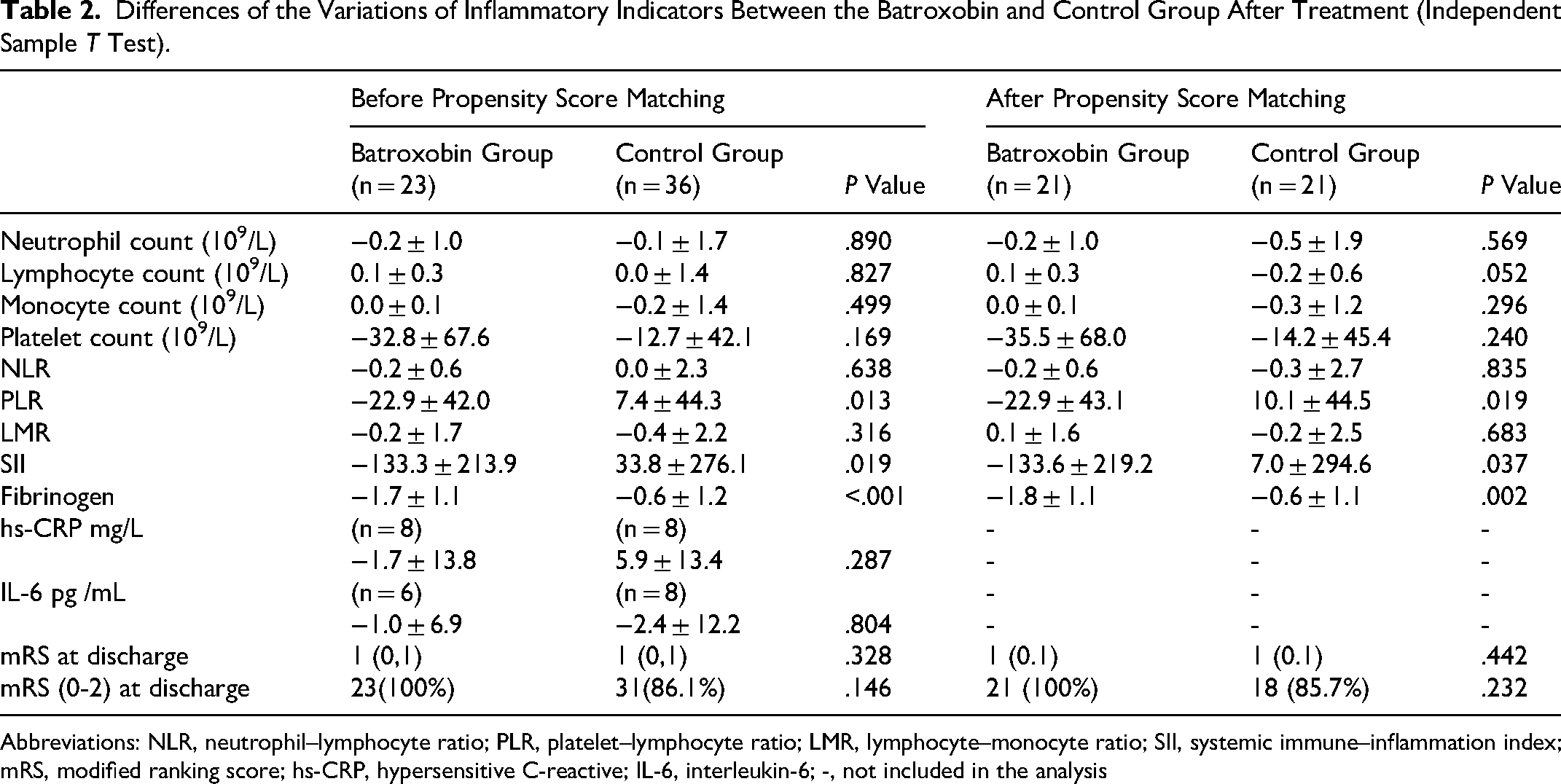
Abbreviations: NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio; LMR, lymphocyte–monocyte ratio; SII, systemic immune–inflammation index; mRS, modified ranking score; hs-CRP, hypersensitive C-reactive; IL-6, interleukin-6; -, not included in the analysis
The Association of the Number of Episodes of Batroxobin Administration With the Variations of Inflammatory Indicators
Univariate linear regression analysis evaluated the association between the number of episodes of batroxobin administration and the variations of inflammatory indicators. The results showed that the platelet count (β [95% CI]: −6.583 [−13.052, −0.114]; P = .046), NLR (−8.791 [−13.963, −3.619]; P = .001), SII (−45.811 [−76.513, −15.109]; P = .004), and fibrinogen (−0.285 [−0.424, −0.147]; P < .001) levels decreased significantly with the increased number of episodes of batroxobin administration. After adjusting for the confounding factors of age, sex, and the interval between 2 laboratory tests, the significant decrease remained in PLR (−9.256 [−14.803, −3.709]; P = .001), SII (−38.943 [−71.423, −6.463]; P = .020), and fibrinogen (−0.284 [−0.421, −0.146]; P < .001) levels with the number of episodes of batroxobin administration (Table 3).
The Association of the Number of Episodes of Batroxobin Administration With the Variations of Inflammatory Indicators (General Linear Model).a
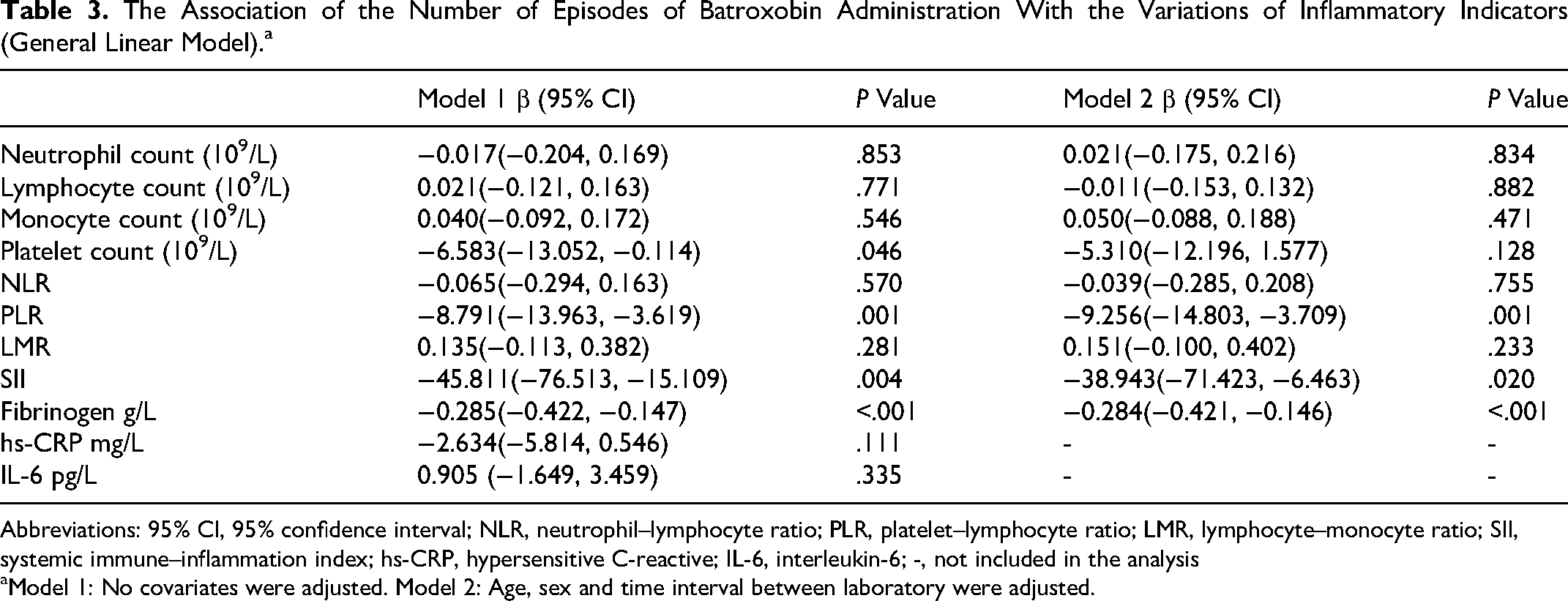
Abbreviations: 95% CI, 95% confidence interval; NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio; LMR, lymphocyte–monocyte ratio; SII, systemic immune–inflammation index; hs-CRP, hypersensitive C-reactive; IL-6, interleukin-6; -, not included in the analysis
Model 1: No covariates were adjusted. Model 2: Age, sex and time interval between laboratory were adjusted.
The Difference in the Proportion of Decreased Inflammatory Indicators Between the Batroxobin and Control Group
Compared with anticoagulation alone, batroxobin combined with anticoagulation significantly increased the proportion of patients with the levels of inflammatory markers decreased, including monocyte count (56.5% vs 30.6%; P = .048), NLR (73.9% vs 13 36.1%; P = .005), PLR (69.6% vs 50.0%; P = .026), SII (78.3% vs 41.7%; P = .006), and fibrinogen (95.7% vs 69.4%; P = .015). After PSM, batroxobin combined with anticoagulation still significantly increased the proportion of patients with decreased PLR (71.4% vs 38.1%; P = .030) and SII (76.2% vs 42.9%; P = .028) (Table 4).
Proportion of Decreased Inflammatory Indicators After Treatment.
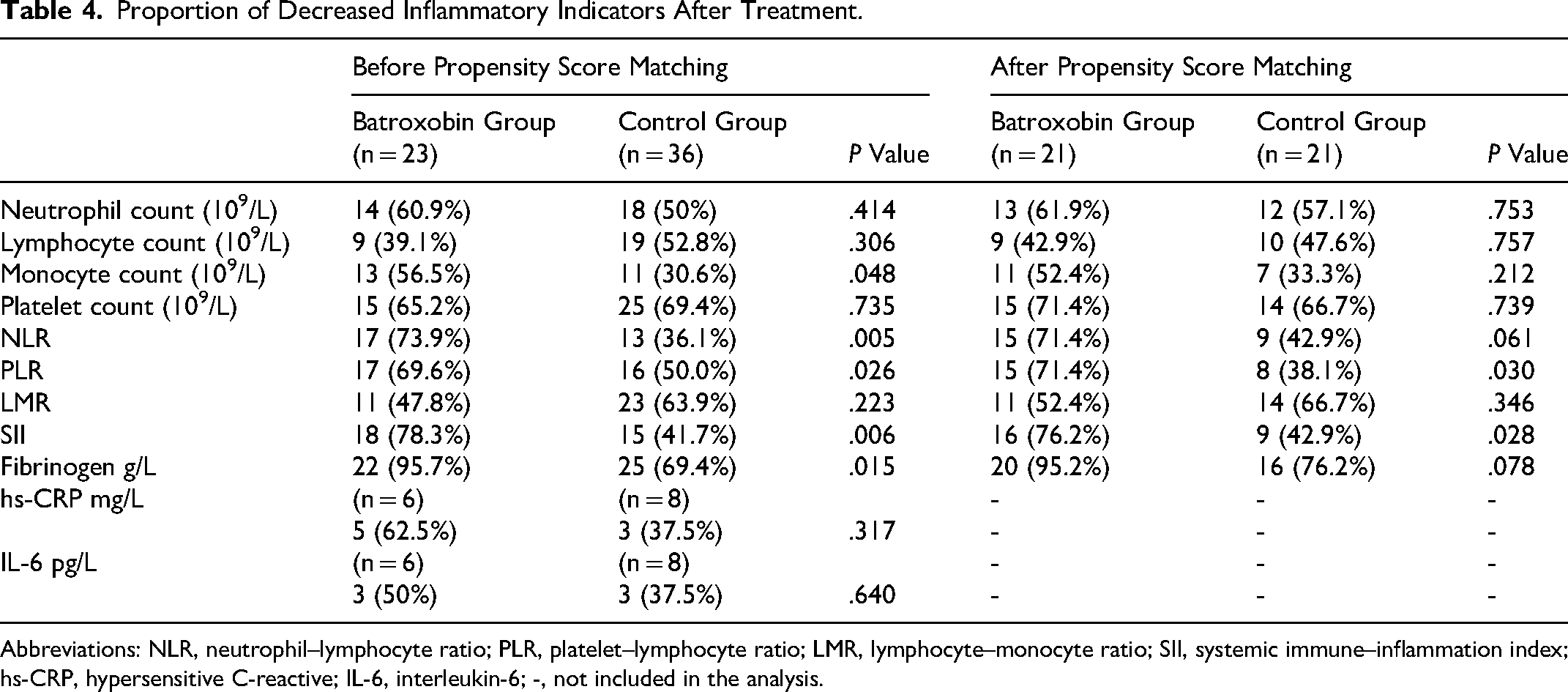
Abbreviations: NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio; LMR, lymphocyte–monocyte ratio; SII, systemic immune–inflammation index; hs-CRP, hypersensitive C-reactive; IL-6, interleukin-6; -, not included in the analysis.
The Association of the Number of Episodes of Batroxobin Administration With the Number of Patients With Decreased Inflammatory Indicators
Univariate binary logistic regression analysis evaluated the association between the number of episodes of batroxobin administration and the number of patients with decreased inflammatory indicators. The results showed that the number of patients with decrease monocyte count (OR [95% CI]: 1.359 [1.036, 1.782]; P = .026), NLR (1.462 [1.085, 1.971]; P = .046), PLR (1.478 [1.080, 1.099]; P = .046), SII (1.517 [1.101, 2.092]; P = .011), and fibrinogen (1.961 [1.013, 3.794]; P = .046) increased significantly with the number of episodes of batroxobin administration. After adjusting for the confounding factors of age, sex, and the interval between 2 laboratory tests, the significant increase in the number of patients with decreased NLR (1.508 [1.113, 2.041]; P = .008), PLR (1.478 [1.080, 2.041]; P = .015), and SII (1.529 [1.092, 2.142]; P = .013) with the number of episodes of batroxobin administration remained (Table 5).
The Association of the Number of Episodes of Batroxobin Administration With Decreased Inflammatory Indicators (Binary Logistic Regression Analysis).a
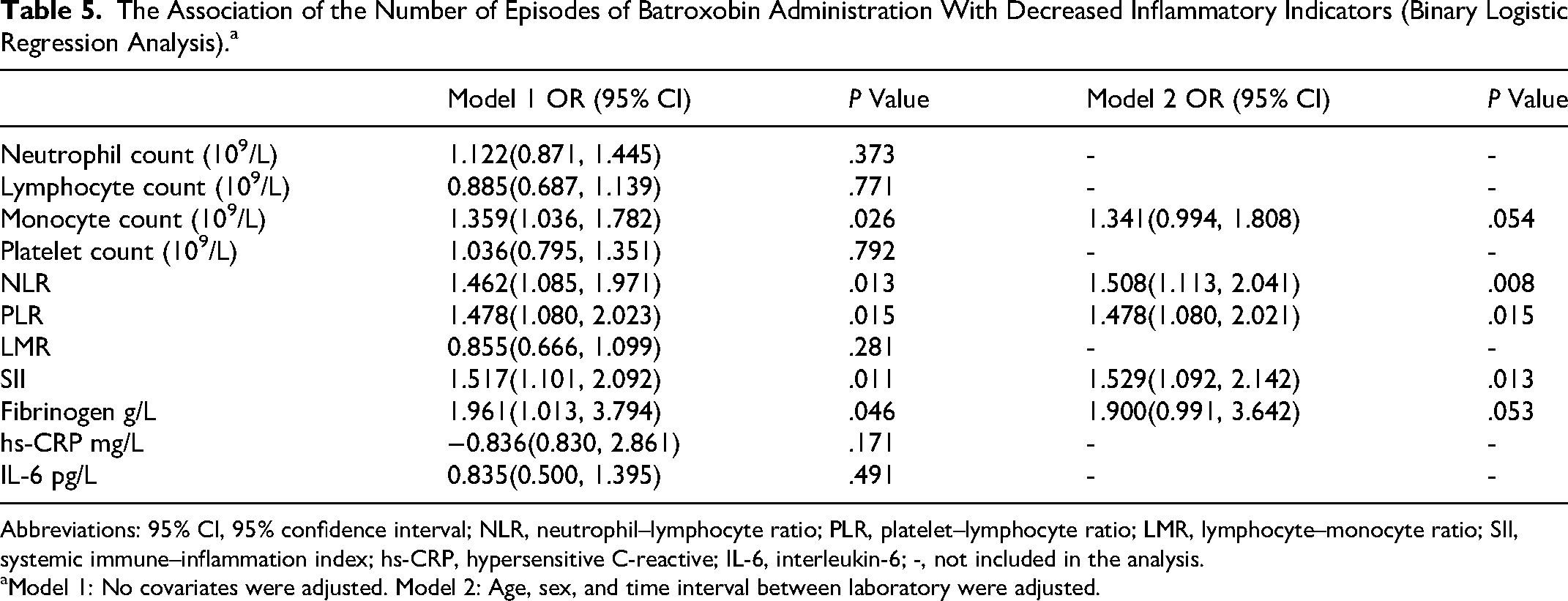
Abbreviations: 95% CI, 95% confidence interval; NLR, neutrophil–lymphocyte ratio; PLR, platelet–lymphocyte ratio; LMR, lymphocyte–monocyte ratio; SII, systemic immune–inflammation index; hs-CRP, hypersensitive C-reactive; IL-6, interleukin-6; -, not included in the analysis.
Model 1: No covariates were adjusted. Model 2: Age, sex, and time interval between laboratory were adjusted.
Based on the results of logistic regression analysis, monocyte count, NLR, PLR, and SII were involved in the ROC analysis. Neutrophil–lymphocyte ratio (AUC [95% CI]: 0.669 [0.529, 0.809]; P = .026), PLR (0.667 [0.530, 0.804]; P = .029), and SII (0.836 [0.544, 0.828]; P = .018) with AUC > 0.6 were further calculated with optimal cutoff values. The optimal cutoff was 1.5 for NLR, PLR, and SII (Figure 1).
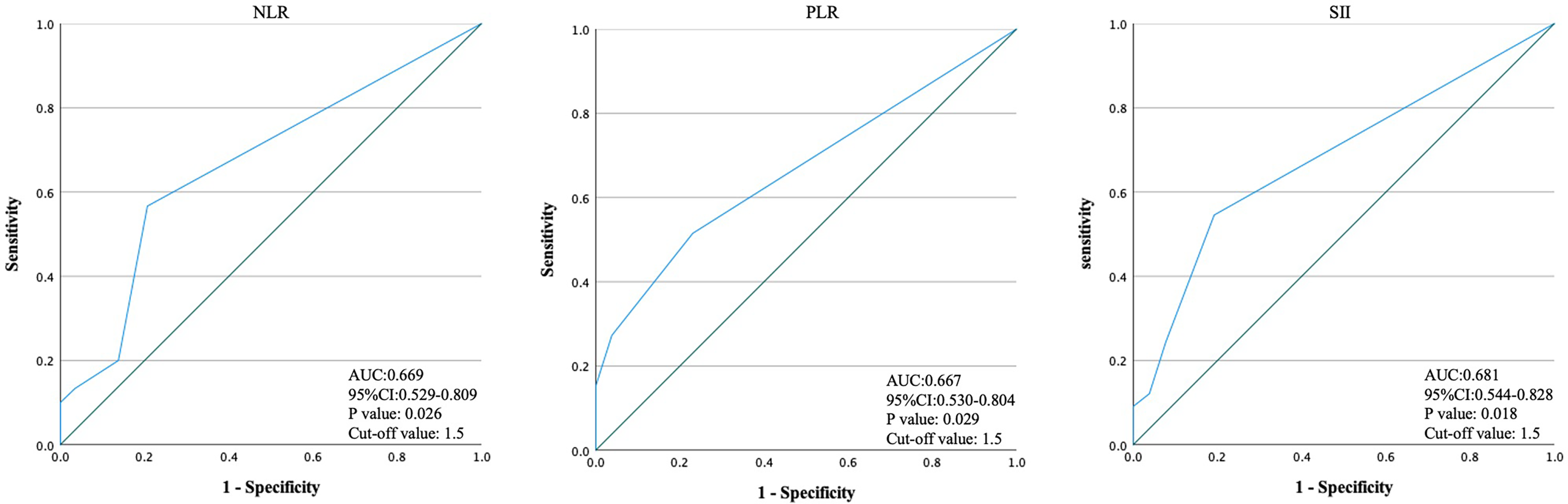
Receiver operating characteristic (ROC) analysis of the number of episodes of batroxobin administration for decreasing inflammatory indicators.
Discussion
Numerous studies have demonstrated that elevated inflammatory parameters can indicate severity and prognostic predictors in CVT.4–7,16–19 Batroxobin, recognized as an antithrombotic agent, has demonstrated the potential to promote CVT recanalization in combination with anticoagulation.9,10 Previous investigations noticed the anti-inflammatory effects of batroxobin in arterial diseases.11–13 In light of this, our study represents the first attempt to investigate the anti-inflammatory effects of batroxobin in CVT patients and revealed that batroxobin, when combined with anticoagulation, could significantly reduce the level of fibrinogen, PLR, and SII. Furthermore, linear dose–response relationships were observed between the number of episodes of batroxobin administration and the reduction in fibrinogen, NLR, and PLR. In addition, the ratio of patients with decreased fibrinogen, NLR, PLR, and SII increased significantly with the increased number of episodes of batroxobin administration.
Inflammatory biomarkers, including PLR, NLR, and SII, were investigated for their predictive value in thromboembolic pathologies and malignancies.20–23 Neutrophils, involved in innate immunity, secrete inflammatory mediators, while lymphocytes participate in specific immunity. NLR, a combination of the 2, reflects the balance and better mirrors the systemic inflammatory state.24,25 PLT indicates the severity of inflammation and more accurately reflects the state of systemic inflammation.26,27 Systemic immune–inflammation index comprehensively reflects the overall inflammation level in the body.28,29 In this study, batroxobin treatment increased the proportion of patients with reduced SII and decreased PLR and SII levels compared to the control group. The results suggest that batroxobin can reduce peripheral inflammation in patients with CVT.
Derived from Bothrops moojeni, batroxobin acts as a defibrinogenating agent, lowering fibrinogen levels. 11 Using batroxobin can decrease fibrinogen value rapidly, 30 which may be the primary mechanism by which batroxobin exerts its anti-inflammatory effect. Several mechanisms of fibrinogen-dependent inflammatory cell migration have proved that fibrinogen could be a bridging molecule to facilitate cell-to-cell adhesion between inflammatory cells and the endothelium.31–33 In addition, fibrinogen can directly influence inflammatory cell activities and alter leukocyte function, leading to changes in cell movement, phagocytosis, NF-kB-mediated transcription, production of chemokines and cytokines, degranulation, and other processes.34–37 Therefore, fibrinogen clearance by batroxobin would reduce fibrinogen-driven inflammation. Fibrinogen, as a critical component of the coagulation/hemostasis pathway, means that batroxobin's fibrinogen-reducing effects could inevitably increase the risk of bleeding in patients, especially when combined with anticoagulation therapy. Although previous studies have shown that the combined use of batroxobin and anticoagulant drugs does not increase the risk of bleeding in patients, 9 strict monitoring of fibrinogen levels is essential during batroxobin combined with anticoagulation therapy.
Previous animal studies have also demonstrated additional anti-inflammatory effects of batroxobin, including promotion of the expression of growth-associated protein-43, inhibition of the excessive increase of tumor necrosis factor-alpha, increase of the superoxide dismutase activities, and reduction of oxygen-free damage.38–41 Aside from the fibrinogen pathway, these mechanisms may contribute to the decrease in inflammatory mediators observed in this study, necessitating further basic research.
Several studies have emphasized the prognostic significance of NLR and SII in CVT patients.5–7,17,18 As anti-inflammatory therapy is not a routine treatment for patients with CVT, no studies have investigated whether reducing inflammatory mediators induced by anti-inflammatory drugs can improve the prognosis of patients with CVT. Although we did not further analyze the effect of batroxobin's anti-inflammatory effect on the prognosis of CVT patients in this study, we are still optimistic that batroxobin combined with anticoagulant therapy can improve the clinical prognosis of CVT patients with high inflammation levels, such as severe CVT patients.
Infection or autoimmune-mediated inflammatory reactions lead to localized hypercoagulation and endothelial injury, potentially causing CVT. CVT-induced inflammation exacerbates brain tissue ischemic injury, resulting in poor clinical outcomes.2,42 Consequently, inflammation is a predisposing and exacerbating factor in CVT onset and development. The study results suggest anti-inflammatory therapy may represent a novel research direction in CVT treatment. Further investigations into anti-inflammatory drugs for CVT are eagerly anticipated.
Our findings hold crucial clinical implications for better comprehending the therapeutic target of CVT and refining therapeutic strategies. Patients with CVT exhibiting high inflammatory indicator levels may face an elevated risk of adverse clinical outcomes, warranting early intervention with optimal adjunctive medical therapy to mitigate this risk. Our study indicates that batroxobin showed promising results in achieving both anti-inflammatory and antithrombotic effects, resulting in satisfactory clinical outcomes during CVT treatment.
Conclusion
The combination of batroxobin and anticoagulation remarkably decreased inflammatory levels in peripheral blood in patients with CVT. The effect was related to the number of episodes of batroxobin administration, which may be another important benefit of batroxobin for patients with CVT besides its reduction of fibrinogen and indirect thrombolysis.
Limitation
Certain limitations exist: The primary constraint lies in the small sample size resulting from stringent inclusion and exclusion criteria, which hinders the inclusion of a broader range of confounding factors in the correlation analysis. We aspire to undertake a future study with a larger sample size to validate the findings presented in this paper. Secondly, due to the limited sample size, we could not conduct research related to prognosis. Concurrently, we anticipate future large-scale studies to ascertain further whether the anti-inflammatory effects of batroxobin can significantly improve patient outcomes.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241264516 - Supplemental material for Anti-inflammatory Effect of Batroxobin Combined With Anticoagulation in Patients With Cerebral Venous Thrombosis
Supplemental material, sj-docx-1-cat-10.1177_10760296241264516 for Anti-inflammatory Effect of Batroxobin Combined With Anticoagulation in Patients With Cerebral Venous Thrombosis by Duo Lan, Xiaoming Zhang, Xiangqian Huang, Jingrun Li, Jiahao Song, Da Zhou and Ran Meng in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
The authors want to thank all colleagues in this study for their cooperation.
Author Contributions
DL and XZ contributed equally to this work and should be considered cofirst authors. DL and XZ wrote the first draft of the manuscript; JL, JS, DZ, and XH performed the material preparation, data collection, and statistical analysis; RM wrote sections of the manuscript and contributed to manuscript revision; RM takes full responsibility for the data, the analyses and interpretation, and the conduct of the research. All authors read and approved the submitted version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Patient Consent Statement
The study was approved by the Institutional Ethic Committee of Xuanwu Hospital, Capital Medical University and was conducted according to the guidelines laid down in the Declaration of Helsinki. Before any study procedure, written informed consent was obtained from the participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant numbers 82171297, 82101390).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
