Abstract
Introduction
Essential thrombocythemia (ET) involves the proliferation of megakaryocytes and platelets and is associated with an increased risk of thrombosis. We aimed to evaluate thrombotic risks in patients with epigenetic regulator mutations and generate a model to predict thrombosis in ET.
Materials and Methods
This cohort study enrolled patients aged > 15 years diagnosed with ET at the Songklanakarind Hospital between January 2002 and December 2019. Twenty-five targeted gene mutations, including somatic driver mutations (JAK2, CALR, MPL), epigenetic regulator mutations (TET2, DNMT3A, IDH1, IDH2, TET2, ASXL1, EZH2, SF3B1, SRSF2) and other genes relevant to myeloid neoplasms, were identified using next-generation sequencing. Thrombotic events were confirmed based on clinical condition and imaging findings, and thrombotic risks were analyzed using five survival models with the recurrent event method.
Results
Ninety-six patients were enrolled with a median follow-up of 6.91 years. Of these, 15 patients experienced 17 arterial thrombotic events in total. Patients with JAK2 mutation and IDH1 mutation had the highest frequency of thrombotic events with somatic driver mutations (17.3%) and epigenetic regulator mutations (100%). The 10-year thrombosis-free survival rate was 81.3% (95% confidence interval: 72.0-91.8%). IDH1 mutation was a significant factor for thrombotic risk in the multivariate analysis for all models. The Prentice, William, and Peterson (PWP) gap-time model was the most appropriate prediction model.
Conclusions
The PWP gap-time model was a good predictive model for thrombotic risk in patients with ET. IDH1 mutation was significant risk factors for thrombosis; however, further studies with a larger sample size should confirm this and provide more insight.
Keywords
Introduction
Essential thrombocythemia (ET), a myeloproliferative neoplasm (MPN), is marked by increased megakaryocytes and platelets, enhancing the risk of thrombosis, hemorrhage, myelofibrosis (MF), and acute leukemia.1,2 Thrombosis in patients with ET adversely affects quality of life, leading to morbidity and fatal complications, making thrombotic prevention crucial. An effective thrombotic risk prediction model would guide clinicians in preventative decision-making.
ET pathogenesis involves somatic mutations of certain driver genes, including JAK2, CALR, or MPL, resulting in the activation of the Janus Kinase (JAK)/signal transducer and activator (STAT) signaling pathway. 3 Therefore, mutation in either of these three driver genes is included in the diagnostic criteria of ET. Additionally, with advancements in sequencing technology, co-mutations in certain genes that regulate epigenetics (so-called epigenetic regulator mutations) have been found to contribute to the pathogenesis and prognosis of MPNs, including ET. Epigenetic change is a reversible process that alters gene expression through DNA methylation or histone modification without changing the deoxyribonucleic acid (DNA) sequence. Mutations in DNA methylation regulators including TET2, IDH1/2 and DNMT3A, and histone modifiers EZH2 and ASXL1, are frequently found in MPN including ET. 4 Despite a decade of these findings, scant research has examined the relationship between epigenetic mutations and thrombotic risk in ET. Nie et al found that Chinese patients with ASXL1 mutation faced elevated thrombosis risk. 5 Conversely, Guglielmelli et al found higher arterial thrombosis risk in American and Italian patients lacking ASXL1 or RUNX1 mutations and higher venous thrombosis risk in those without EZH2 or SRSF2 mutations. 6 Notably, the two studies reported different types of epigenetic mutations.
JAK2 mutations are included in prediction models for thrombotic risk such as the International Prognostic Score of Thrombosis for Essential Thrombocythemia (IPSET)-thrombosis model or the revised IPSET-thrombosis model.7,8 However, no study yet has included epigenetic mutations in predictive models. Thus, we aimed to assess the mutation status of somatic driver, epigenetic regulator, and other genes relevant to myeloid neoplasms using a 25-gene panel to evaluate their association with thrombotic risk in ET. Based on our results, we further aimed to develop a model incorporating these mutations to predict thrombotic risk in patients with ET.
Materials and Methods
Study Population
This cohort study enrolled patients aged >15 years old, diagnosed with ET according to the 2008 or 2016 World Health Organization (WHO) criteria 9 and treated at Songklanakarind Hospital in southern Thailand from January 2002 to December 2019. ET diagnosis was confirmed through clinical characteristics, lab findings, and pathological reports of bone marrow studies. Patients who had hypertension, diabetes mellitus, dyslipidemia and/or who smoked were defined as patients with cardiovascular risk. All patients received antiplatelet agents for cardiovascular risk prevention; those aged > 60 years old or with thrombosis history additionally received hydroxyurea or anagrelide based on individual needs and physician judgement. Patients with <1 month follow-up or inadequate data were excluded. The cohort was part of a prior external validation study. 10
This study was approved by the institutional review board of our institution and conducted in accordance with the Declaration of Helsinki (EC approval number: REC. 63-311-14-1). All patients provided written informed consent before enrollment.
Predictors and Outcome
Baseline characteristics and patient laboratory results, including symptoms, underlying diseases, smoking, complete blood count, and bone marrow testing, were gathered at diagnosis. Thrombotic events before diagnosis were determined via clinical condition or imaging methods like ultrasound, CT, or MRI. Blood samples for gene sequencing were obtained post-informed consent.
Thrombosis occurrence data were collected during follow-up and confirmed through clinical condition and imaging, including ultrasound, CT, or MRI.
Detection of Gene Mutation
We performed targeted gene sequencing using the QIAact Myeloid DNA UMI Panel (Qiagen, Hilden, Germany), which comprises 25 genes associated with myeloid neoplasms, including somatic driver genes (JAK2, CALR, MPL), epigenetic regulator genes (TET2, DNMT3A, IDH1, IDH2, TET2, ASXL1, EZH2, SF3B1, SRSF2) genes related to RNA splicing (ZRSR2, U2AF1), signaling pathway (CBL, FLT3, KIT, KRAS, NRAS, SH2B3), transcription factors (CEBPA, NPM1, RUNX1, SETBP1), the tumor suppressor gene (TP53), and the CSF3R gene. First, DNA was extracted from peripheral blood using a QIAamp DNA Mini Kit (Qiagen, Hilden, Germany). DNA concentration was measured using a Qubit dsDNA HS Assay (Thermo Fisher, Waltham, MA, USA). Target enrichment, library preparation, and clonal amplification were performed using the GeneRead DNA Library Q Kit and GeneRead Clonal Amp Q Kit (QIAGEN), according to the manufacturer's instructions. The libraries were sequenced using the QIAGEN GeneReader Next-Generation Sequencing System. Mutation analysis and interpretation were performed using the QIAGEN Clinical Insight Software (QCIA and QCII).
Statistical Analysis
Data were analyzed using R 4.0.2. Missing values, such as two patients’ CBC at diagnosis, were addressed via regression imputation. Maximally selected log-rank statistics determined continuous variable cutoffs. Kaplan–Meier curves depicted thrombosis-free survival (TFS). Recurrent thrombotic events violated Cox model assumptions of independent outcomes, prompting us to employ five models for recurrent TFS analysis 11 : Andersen Gill (AG), Prentice, William, and Peterson (PWP) total-time, PWP gap-time, Marginal, and Frailty models. Variables statistically significant in univariate analysis (p < 0.05) and known thrombotic risk factors (age > 60 years old, history of thrombosis, JAK2 mutation, cardiovascular risk) were included in each model's multivariate analysis.
For prediction model generation, variables significant in univariate analysis (p < 0.05) and those associated with high thrombotic risk were analyzed using backward stepwise regression. Models with the lowest log-likelihood, Akaike Information Criterion (AIC), and Bayesian Information Criterion (BIC) were selected as the best fit. Internal validation of the top model was conducted using bootstrap resampling with 500 resamplings. A calibration plot was used to assess the model.
Results
During the 18-year study period, 245 patients were diagnosed with ET at our institution. Sixty-nine patients were excluded due to not meeting WHO diagnostic criteria. Eight and 19 patients were excluded due to short follow-up time (less than 1 month) and inadequate data, respectively. Blood samples could not be obtained from 44 patients who died and nine patients who were lost to follow-up. Hence, 96 patients were included in the study (

Flow chart of patient selection.
The clinical and hematological characteristics of the patients are shown in Table 1. The median age was 55.4 years. The majority of patients (60%) were women, and 56% of patients had cardiovascular risk. Approximately 24% of the patients had a history of thrombosis before or at diagnosis of ET. Transient ischemic stroke (TIA) or ischemic stroke was the most common type of thrombosis. The median platelet count at ET diagnosis was 1,042,000 cells/µL.
Baseline characteristics of patients with essential thrombocythemia.

Thrombosed arteriovenous malformation, **94 patients.
The total number of patients was 96.
Abbreviations: CBC, complete blood count; Hb, hemoglobin; Hct, hematocrit; IQR, interquartile range; WBC, white blood cell.
Frequency of thrombotic events during follow-up
With a median follow-up of 6.91 years (interquartile range [IQR] 4.67-10.25), 17 thrombotic events occurred in 15 patients, with two patients having two events each. All thrombotic events were associated with arterial thrombosis. TIA and ischemic strokes comprised most events (8/17, 47.1%). (Supplementary Table S1). The median time to first thrombosis was 4.85 years (IQR: 2.26-7.33).
Prevalence of Somatic Driver Mutations and Epigenetic Regulator Mutations
Mutational profiling of 25 genes by next-generation sequencing was successfully performed for all samples. Ninety patients (93.8%) had at least one somatic driver or epigenetic regulator mutation, and 44 patients (45.8%) had both types of mutations. The prevalence of mutations in each gene is presented in Table 2. The prevalences of JAK2, CALR, and MPL mutations were 54.2%, 27.1%, and 15.6%, respectively. Thirteen patients (16.2%) had two somatic driver mutations (JAK2 and CALR in one patient, JAK2 and MPL in six patients, and CARL and MPL in six patients). Fifty-four patients (56.2%) had epigenetic regulator mutations. TET2 mutations (34.4%) were the most common epigenetic regulator mutation, followed by DNMT3A (11.5%), and EZH2 (10.4%) mutations.
Prevalence of thrombotic events according to each mutation type in 96 patients with essential thrombocythemia.
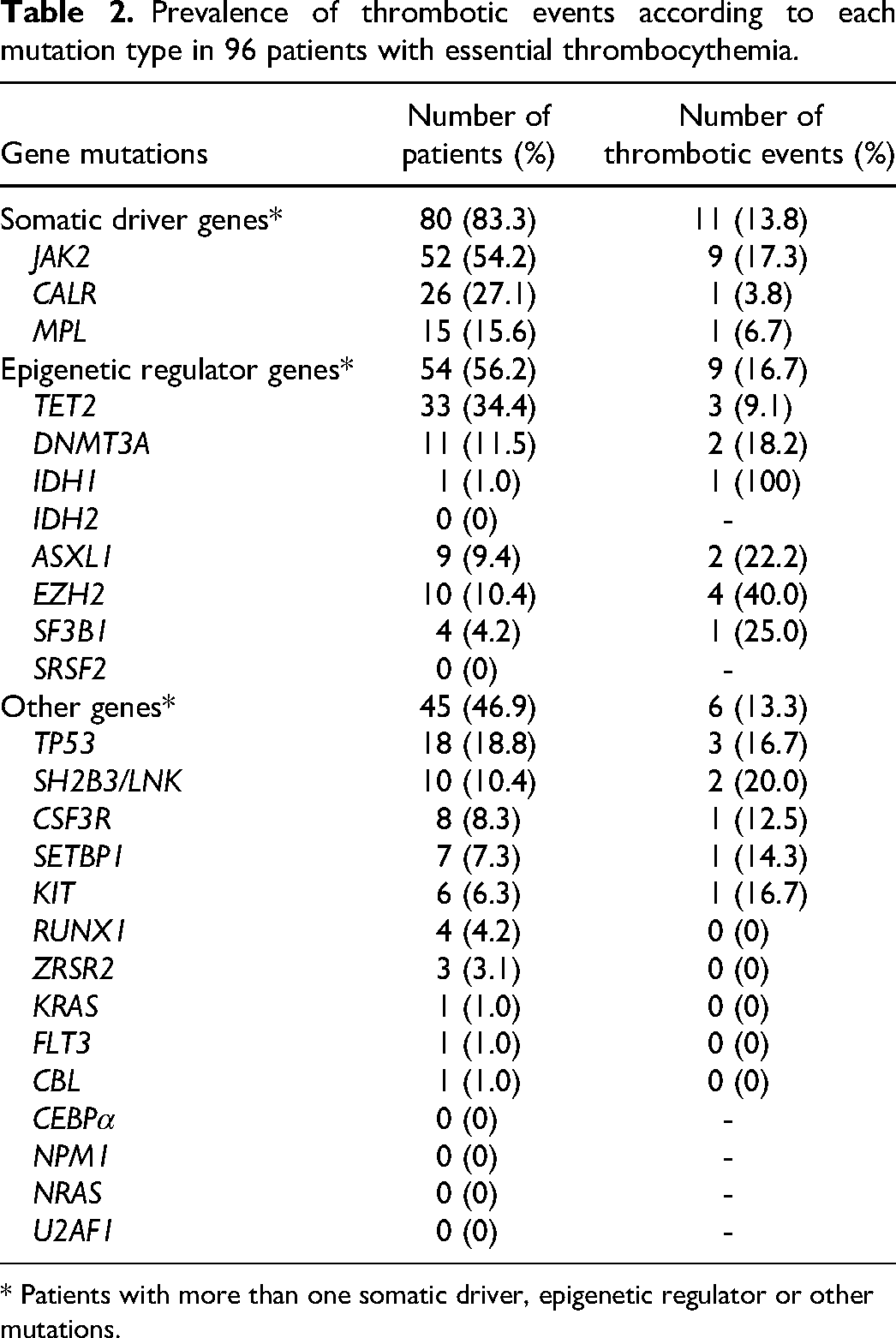
* Patients with more than one somatic driver, epigenetic regulator or other mutations.
Prevalence of Thrombotic Events According to Type of Mutation
The prevalence of thrombotic events related to each mutation type is shown in Table 2. Somatic driver and epigenetic regulator mutations had similar prevalence of thrombotic events: 13.8% and 16.7% in the first event and 2.5% and 3.7% in the second event, respectively. Among somatic driver mutations, patients with JAK2 mutations had the highest frequency of thrombotic events (17.3%). Regarding epigenetic regulators, one patient with IDH1 mutation had recurrent thrombotic events, and patients with EZH2 mutations experienced the second most frequent number of thrombotic events. Moreover, regarding the two patients who experienced recurrent thrombotic events, one had JAK2 and IDH1 mutations, and the other had JAK2, EZH2, SH2B3/LNK, and TP50 mutations.
Prognostic Factors of Thrombosis by Survival Analysis in Recurrent Events
The 10-year TFS was 81.3% (95% confidence interval (CI): 72.0–91.8%), while the median TFS was not reached within the follow-up period (Supplementary Figure S1). In the univariate analysis, age, neutrophil and eosinophil percentages, EZH2 mutation, and IDH1 mutation were significant factors in the AG, PWP total-time, and frailty models. Age, eosinophil percentage, EZH2 mutation, and IDH1 mutation were significant factors in the PWP gap-time and marginal models (Supplementary Table S2). Furthermore, IDH1 mutation was a risk factor for thrombotic risk in the multivariate analysis for all models (Supplementary Table S3).
Thrombotic Prediction Models by Survival Analysis in Recurrent Events
We generated prediction models for all five survival analysis models for recurrent events, with all final models presented in Table 3. PWP gap-time was the most appropriate prediction model because it had the lowest log-likelihood, AIC, and BIC values. This model consisted of the percentage of eosinophils (hazard ratio (HR): 0.25; 95% CI: 0.10–0.67), EZH2 mutations (HR: 2.90; 95% CI: 1.10-7.65), and IDH1 mutations (HR: 10.18; 95% CI: 5.17-20.06). For the application of the prediction model, risk scores were assigned according to the HR of each prediction factor: eosinophil percentage <0.6% (3 points), presence of EZH2 mutation (2 points), and presence of IDH1 mutations (5 points). Patients were divided into two groups based on the sum of risk scores (low risk <3 points; high risk ≥3 points). This prediction model demonstrated a significant difference in 10-year TFS at the first event among the risk groups (84.9% in low risk and 47.6% in high risk, P = 0.003) (
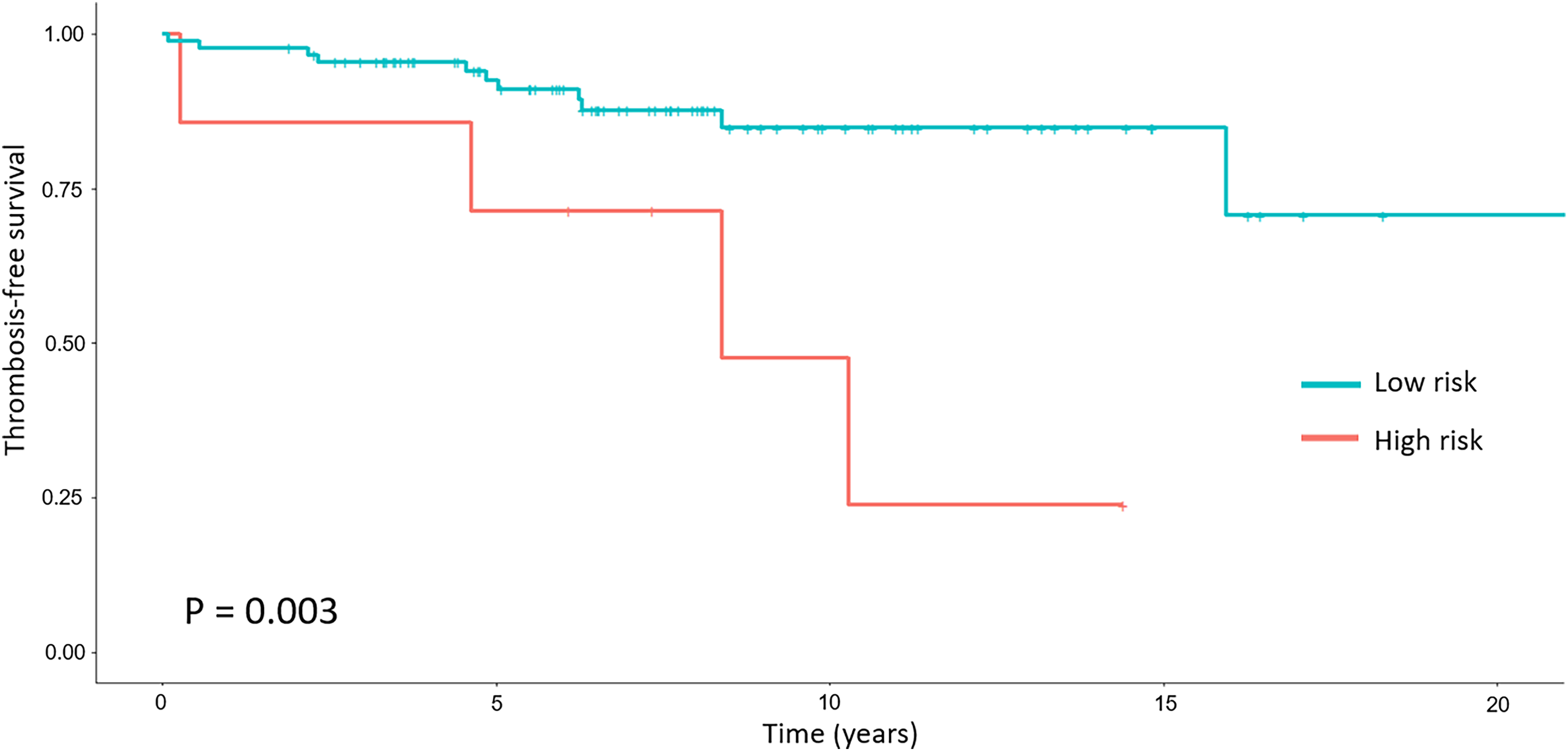
Thrombosis-free survival among risk groups according to the prediction model, which includes percentage of eosinophils, presence of EZH2 mutation, and presence of IDH1 mutation.
Prediction model in five models of survival analysis in recurrent events.
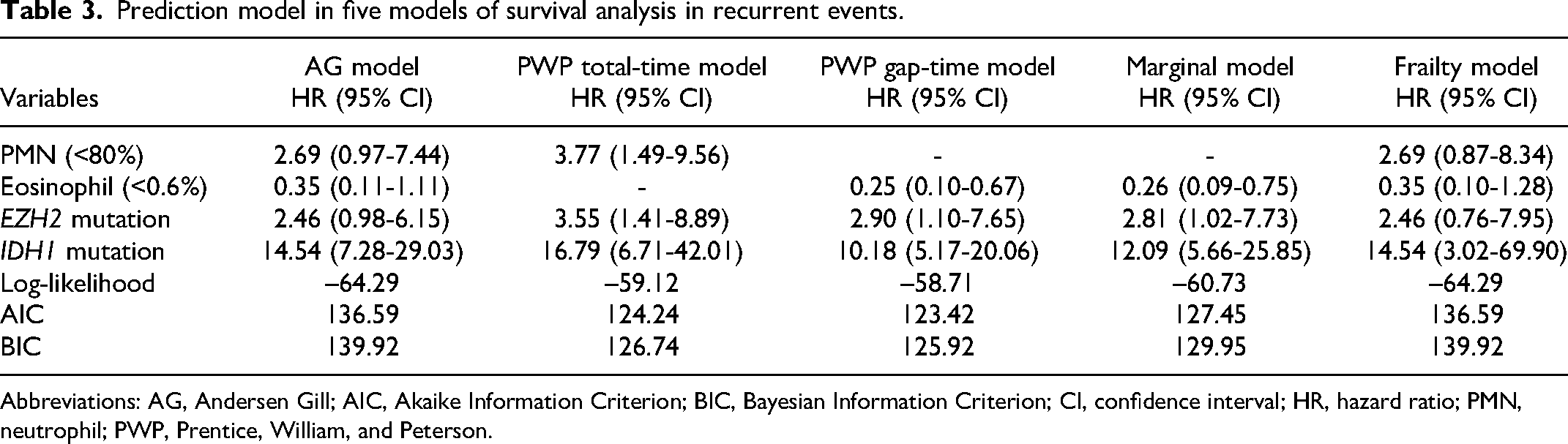
Abbreviations: AG, Andersen Gill; AIC, Akaike Information Criterion; BIC, Bayesian Information Criterion; CI, confidence interval; HR, hazard ratio; PMN, neutrophil; PWP, Prentice, William, and Peterson.
For internal validation, using the bootstrapping method with 500 resamplings, this model showed good calibration at three years (
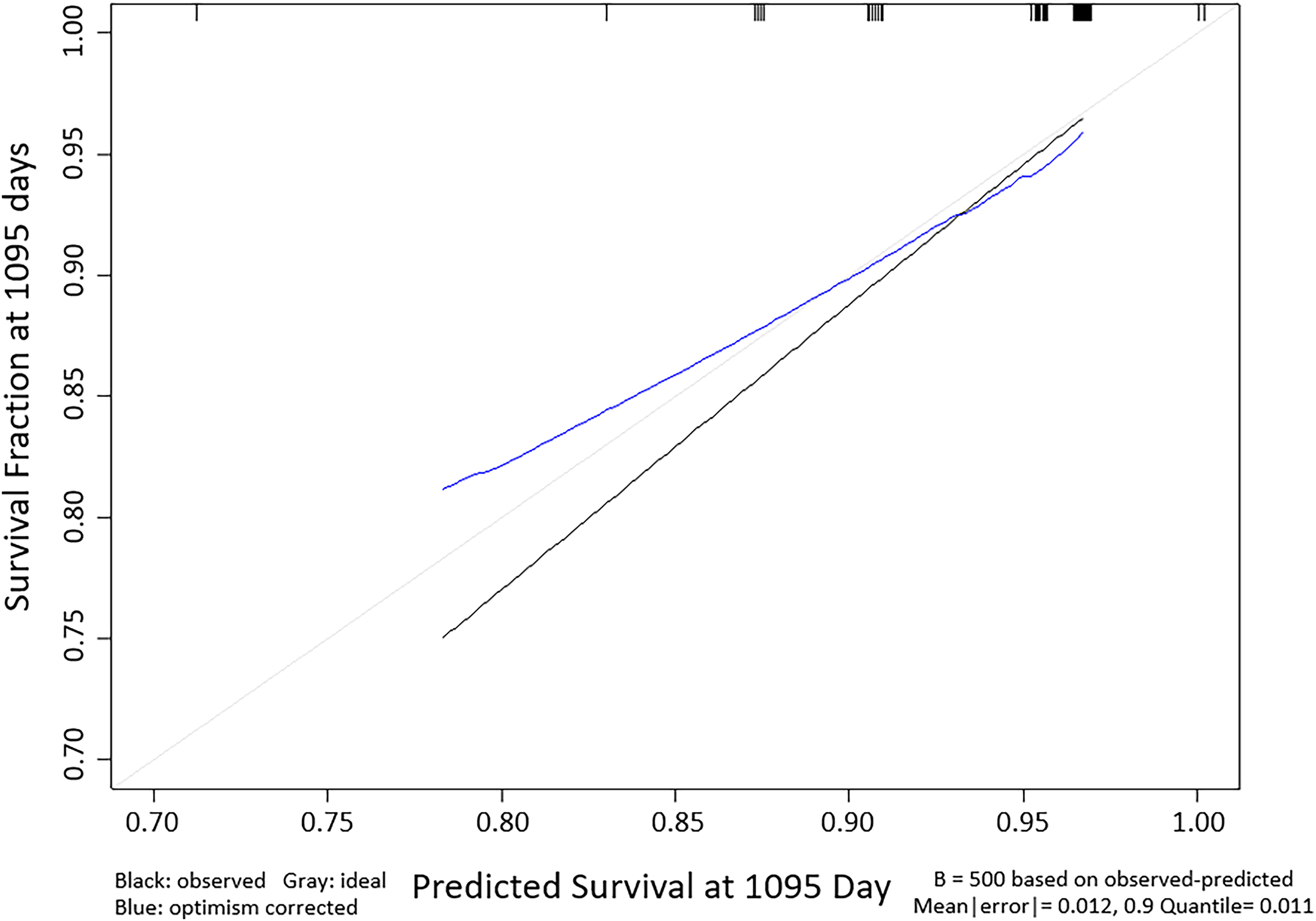
Calibration plot of prediction models to predict the thrombotic risk of patients with essential thrombocythemia at 3 years. Black line: apparent performance for calibration. Blue line: bias-corrected for calibration. Grey line: ideal.
Discussion
In this study, TET2 mutation was the most frequent epigenetic alteration. Survival analysis showed that IDH1 mutation was a risk factor for thrombosis across all five models. The PWP gap-time model, incorporating eosinophil percentage and the presence of EZH2 and IDH1 mutations, displayed good calibration.
The baseline characteristics of patients with ET in this study mirrored those in previous studies, with a median diagnosis age of 60 years12–15 and a predominance of women.12–17 Previous studies indicated that 50–60% had JAK2 V617F mutations,1,12,14,18 and about 40% were asymptomatic.13,19 Our study, however, reported a higher history of thrombosis before or at diagnosis (24.0% vs 13.0-18.4%).13,20 Despite all participants receiving thrombosis prevention medication, including antiplatelet and/or cytoreductive agents, 17 thrombotic events occurred. The post-diagnosis thrombosis incidence (25.6 events/1000 patients/year) was in line with previous findings (28.0-33.0 events/1000 patients/year).8,21,22 Although venous thromboembolism rates in Asian ET patients (8.6-18.0%) were lower than those in Western countries (31.9-40.5%),8,12,21,22 none were observed in our ET cohort.
We found that 79% of patients had epigenetic mutations, similar to the results of Asp et al. 23 However, this prevalence was higher than that reported in other studies (11-43%).24–28 This discrepancy may be due to various factors, including study population, number of epigenetic mutations examined, and updated type of epigenetic mutations in the next-generation sequencing program analysis database. Previous studies have evaluated epigenetic mutations in only 5–9 genes, compared with the 25 genes in our study.23–26 We used next-generation sequencing, which readily updates the database with new types of mutations, enabling the identification of mutations in 25 genes by 2022. Therefore, we identified a higher prevalence of epigenetic mutations than previous studies did. Similar to previous studies, TET2 mutations were ET's most common epigenetic mutations.23–27 The prevalence of IDH1 mutations (1.0%) was also similar to that in previous studies.27,28 However, the prevalence of EZH2 mutations was higher than that in previous studies (10.4% vs 1-6%, respectively).23–25,28
Regarding thrombosis frequency and mutations, JAK2 mutations yielded the highest rate (17.3%) among somatic mutations and were present in both patients experiencing a second thrombotic event post-ET diagnosis. This mutation is known to elevate thrombotic risk compared to CALR mutations. 29 In our study, the IDH1 mutation correlated with a 100% thrombotic rate, although it was found in just one patient. No prior study has reported this mutation's thrombotic rate. Guglielmelli et al studied 502 American and Italian ET patients, identifying high arterial thrombosis risk in those lacking ASXL1 or RUNX1 mutations and high venous thrombosis risk in those without EZH2 or SRSF2 mutations. Their study detected no IDH1 mutations. 6 Other studies that detected IDH1 mutations did not report associated thrombotic rates.23,27,28 Therefore, this may be the first study to report the high thrombotic risk associated with IDH1 mutations in patients with ET. The thrombosis rates among patients with EZH2, SF3B1, ASXL1, SH2B3, and TP53 mutations were 40.0%, 25.0%, 22.2%, 20.0%, and 16.7%, respectively. This study confirmed the high thrombosis rates of these mutations, which is consistent with previous studies.23,24
Analysis of recurrent events revealed IDH1 mutation to be a risk factor for thrombosis. IDH1 catalyzes the oxidative decarboxylation of isocitrate to-ketoglutarate in the cytoplasm, 30 and mutations decrease the level of IDH1 necessary for TET2 protein DNA methylation. 31 However, the pathogenesis of thrombosis in patients with ETs and IDH1 mutations remains poorly understood. To the best of our knowledge, this is the first report to show the association of IDH1 mutation with thrombotic risk in patients with ET. Few studies have reported patients’ prognoses with this mutation in other diseases. Yonal-Hindilerden et al reported the prognosis of primary myelofibrosis (PMF) in patients with IDH1 mutations, showing increased mortality, leukemic transformation, and risk of bleeding in these patients. 32 Unruh et al reported a low thrombotic risk of glioma in patients with IDH1 mutations, differing from the results of our study. Their study demonstrated 0% venous thromboembolism in patients with IDH1 mutations, compared with the 30% reported in those without. 33 However, the thrombotic risk associated with this mutation must be interpreted cautiously because IDH1 mutation was observed in only one patient with a second thrombotic event after the ET diagnosis. Further studies with larger sample sizes are needed to evaluate the role of IDH1 mutations in ET.
The EZH2 mutation, another epigenetic regulator, significantly predicted thrombosis in our study. It encodes the catalytic polycomb repressive complex 2, responsible for activating H3K27me3 histone methyltransferase, leading to gene silencing. 34 This mutation correlates with higher mortality, myelofibrosis, and leukemic transformation in ET patients, 24 as well as poor overall survival in myelodysplastic syndrome and PMF. 35 Contrary to our findings, Guglielmelli et al reported increased venous thrombosis in ET patients without EZH2 mutations but did not provide thrombotic risk odds ratios. 6 The pathogenesis of EZH2-related thrombosis in ET is still unclear.
In this study, 42.6% of patients had eosinophilia at diagnosis, defined as an eosinophil count over 500 cells/µL, a common presentation in MPNs such as chronic myeloid leukemia. 36 No prior studies have linked eosinophil percentage to thrombotic risk in MPNs, including ET. Notably, a higher eosinophil percentage was a protective factor against thrombosis in our predictive model. Conversely, eosinophilia elevates thrombotic risk in diseases like hypereosinophilic syndrome. 37 The mechanism underlying eosinophils’ thrombosis prevention in ET remains unclear.
Our model, consisting of percentage of eosinophils, the presence of IDH1 and EZH2 mutation, showed a good performance at predicting the thrombotic risk in patients with ET. With the standard treatment, more than 50% of the patients classified as high-risk by this model developed thrombotic events within 10 years after ET diagnosis. Moreover, administration of once-daily low-dose aspirin with cytoreductive agents was not enough to prevent thrombotic events in these patients. The ARES study reported that a twice-daily aspirin administration was better than a once-daily aspirin administration at maintaining antiplatelet efficacy 38 . Therefore, we recommend a twice-daily low-dose aspirin with cytoreductive agents as standard treatment to enhance thrombotic prevention in these patients.
This study presents limitations. First, owing to the study's retrospective nature, some data could not be obtained; these data were imputed using a regression imputation method. Sensitivity analysis demonstrated the suitability of the PWP gap-time model in predicting thrombotic risk, and hemoglobin at diagnosis may enhance the performance of this model. (Supplementary Table S4–5) Second, blood samples for mutation detection were collected after enrolling patients; therefore, samples could only be collected from patients who were still alive and visited our hospital. Mutation frequency or type in patients who died from thrombosis varying from that in patients who survived may lead to selection bias and cause underestimation of the thrombotic risk associated with certain mutations. Third, our sample size was limited. A larger sample could reveal significant associations between known risk factors (age, history of thrombosis, JAK2 mutation, cardiovascular risks) and other epigenetic mutations with thrombosis. Lastly, while our prediction model's calibration was evaluated, its discrimination was not due to the absence of suitable packages for analyzing recurrent events in survival models. However, this study's analytical method is robust, as it accounts for multiple thrombotic events per patient through recurrent event survival analysis.
In conclusion, this inaugural study employs survival analysis to assess thrombotic risk in patients with recurrent events. IDH1 mutation emerged as a thrombosis risk factor in patients with ET. The PWP gap-time model, incorporating eosinophil percentage and the presence of IDH1 and EZH2 mutations, effectively predicts thrombotic risk. Subsequent larger-scale studies are needed to validate the predictive impact of epigenetic mutations in ET.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241263099 - Supplemental material for Thrombotic Prediction Model Based on Epigenetic Regulator Mutations in Essential Thrombocythemia Patients Using Survival Analysis in Recurrent Events
Supplemental material, sj-docx-1-cat-10.1177_10760296241263099 for Thrombotic Prediction Model Based on Epigenetic Regulator Mutations in Essential Thrombocythemia Patients Using Survival Analysis in Recurrent Events by Pirun Saelue, Patuma Sinthujaroen, Supaporn Suwiwat and Paramee Thongsuksai in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
We thank the officers of the Division of Digital Innovation and Data Analytics, Faculty of Medicine, Prince of Songkla University.
Author Contributions
Pirun Saelue and Paramee Thongsuksai contributed to the study design, data collection, data analysis, and manuscript preparation. Patuma Sinthujaroen contributed to experiments and manuscript preparation. Supaporn Suwiwat contributed to experiments.
All authors have read and approved the final version of this manuscript before its submission.
Data Sharing Statement
Data is available upon reasonable request to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Faculty of Medicine, Prince of Songkla University (grant number 64-003-3).
Faculty of Medicine, Prince of Songkla University, (grant number 64-003-3).
Previous Presentation
Oral presentation (some data) in 59th Annual Meeting of the Thai Society of Hematology, 17-20 March 2022 at Centara Grand Hotel, Bangkok, Thailand
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
