Abstract
The pathological consequences of decreased protein Z (PZ) and/or Z-dependent protease inhibitor (ZPI) levels remain as yet unclear, despite a growing body of evidence which supports their involvement in an increased thrombotic risk. The purpose of the present study was 2-fold: to evaluate plasma concentrations of protein Z and ZPI in patients with essential thrombocythemia (ET) and to determine their significance in thrombotic complications. The median (range) plasma concentrations of PZ in our patients with ET were lower, but not significantly, than in healthy individuals: PZ (1.42 µg/mL, 0.36-3.14 µg/mL vs 1.6 µg/mL, 0.75-2.56 µg/mL, P = .08). On the other hand, the median (range) plasma concentrations of ZPI in the said patients with ET were meaningfully lower than in the reference group: ZPI (3.22 µg/mL, 0.85-6.97 µg/mL vs 4.41 µg/mL, 1.63-7.83 µg/mL, P = .0004). More importantly, the study revealed a statistically significant lower concentration of PZ and ZPI in patients with the presence of the JAK2V617F mutation relative to patients without the mutation, for PZ: 1.38 µg/mL, 0.36-2.6 µg/mL versus 1.63 µg/mL, 0.88-3.14 µg/mL, P = .03, and ZPI 2.89 µg/mL, 0.85-5.91 µg/mL versus 3.61 µg/mL, 1.53-6.97 µg/mL, P = .002. Additionally, significant differences between the concentrations of PZ and ZPI were found in patients with venous thrombotic episodes compared to healthy individuals, for PZ: 1.23 µg/mL, 0.82-1.99 µg/mL versus 1.6 µg/mL, 0.75-2.56 µg/mL, P = .043, and ZPI: 2.42 µg/mL, 0.85-4.21 µg/mL versus 4.41 µg/mL, 1.63-7.83 µg/mL, P < .0001. To recapitulate, our results suggest that the deficiency of PZ may increase tendency to thrombosis in patients with ET.
Introduction
Essential thrombocythemia (ET) is a myeloproliferative disorder associated with an increased tendency to thrombosis. 1 Multiple factors are likely to contribute to the pathogenesis of thrombosis, including platelet and leukocyte number, activation of platelets and leukocytes, and their interaction to form platelet–leukocyte aggregates, not to mention prothrombotic circulating and endothelial factors. 2 Even in the absence of manifest thrombosis, patients with ET present with a hypercoagulable state characterized by an increased concentration of several plasma markers of hemostatic system activation. Reduction in the concentrations of natural anticoagulants may also contribute to determining thrombotic episodes in these patients. 3
Evaluation of the thrombotic risk in individual patients with ET is still approximate and heavily reliant on a limited number of variables. Consequently, a concerted scientific effort has been recently directed toward studies testing new diagnostic and prognostic tools or aimed at clarifying the pathogenetic mechanism of myeloproliferative neoplasms-associated thrombophilia. Identification of plasma markers changing the hemostatic balance in patients with ET could prove particularly useful in patients with a clinically significant risk of thrombosis.
Protein Z (PZ) is a vitamin K-dependent coagulation glycoprotein, synthesized in liver 4 and, presumably, by the human endothelial cells. 5 Being structurally similar to other vitamin K-dependent factors (factors VII, IX, X, protein C), it could act as a cofactor of another protein which downregulates coagulation, 6 in the same manner as protein S. This hypothesis was confirmed by the isolation of PZ-dependent protease inhibitor (ZPI), which inhibits activated factor Xa in a process that requires the presence of protein Z, calcium ions, and phospholipids. Consecutive studies revealed a new inhibitory function of this molecule that is also relevant to the hemostatic system: ZPI was demonstrated to efficiently and rapidly inhibit FXIa in a mechanism that does not require PZ, calcium, or phospholipids. 7 Protein Z and ZPI form a complex and in the pooled normal plasma, which contains excess ZPI, all the PZ appear to be bound to ZPI. 8 In line with this hypothesis, other clinical studies showed a significant correlation between PZ and ZPI levels in patients with vascular complications. 9,10
The potential role of alterations in PZ and/or ZPI levels in the pathogenesis of thrombotic and/or hemorrhagic diseases has been investigated in a handful of studies which, however, yielded inconsistent results. Some of these analyses reported low PZ levels connected with the occurrence and progression of several types of ischemic vascular diseases, 11 whereas others neither observed this correlation nor mentioned the association between low levels of PZ and hemorrhagic diseases. 12,13 There is no published data evaluating PZ–ZPI complex in patients with ET.
The aim of the present study was to evaluate plasma concentrations of PZ and ZPI in patients with ET and to elucidate the participation of these anticoagulant plasma system proteins in the pathogenesis of thrombotic complications associated with this disease. Furthermore, we investigated the relationship between plasma concentrations of anticoagulant proteins (PZ and ZPI) and the occurrence of JAK2V617F mutation, a molecular marker of these diseases and the potential risk factor for thrombotic complications.
Patients
Seventy-one patients with newly diagnosed ET were included in the study. Their median age at the time of sample collection was 63 years and the range was 23 to 82 years. In all, 46 patients were females and 25 were males. Patients with acute or chronic infections, inflammatory processes, and liver or kidney diseases have been excluded from the study. None of the female patients had undergone oral contraception. Our patients did not receive cytoreductive therapy or anticoagulants (except aspirin 75 mg/day in 52 patients) prior to or at the time of the study, either. None of the patients included in the study had venous or arterial thromboembolic complications at the time of sample collection.
The diagnosis was based on clinical and laboratory features specified in the World Health Organization (WHO) criteria. 14 Of our patients, 49 (69%) were positive for JAK2V617F mutation. In addition, bone marrow aspiration and biopsy samples were examined immunohistochemically to confirm the diagnosis of ET. Patients without the mutation were diagnosed with ET if no evidence for reactive thrombocytosis was confirmed and if other WHO criteria were met. Complete blood count, tests of hemostasis as well as liver and renal function tests were assayed in all cases. Patient characteristics are listed in Table 1.
Characteristic of Patients With Essential Thrombocythemia.
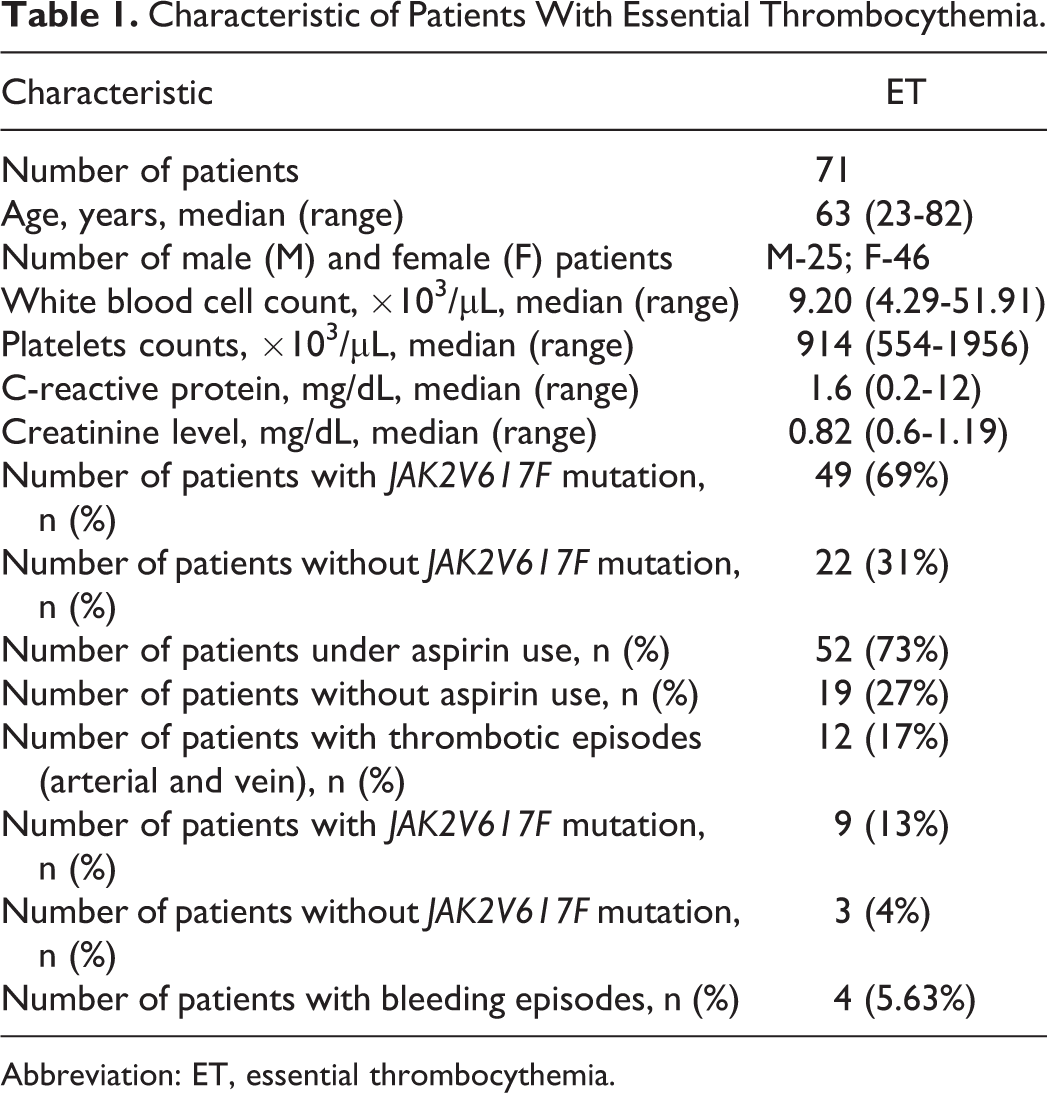
Abbreviation: ET, essential thrombocythemia.
The control group comprised 56 age- and sex-matched healthy volunteers. All patient samples, along those from normal controls, were collected with the approval of the Ethics Committee at the Medical University of Bialystok—approved protocol with written informed consent (Agreement No. R-I-002/330/2011).
Methods
Blood samples were collected without stasis from the antecubital vein with butterfly needle, after an overnight fast, between 8.00
Quantitative assessment of PZ and ZPI in the plasma was performed using commercial tests—Asserachrom Protein Z Elisa KIT (Diagnostica Stago, France) and Enzyme-linked Immunosorbent Assay kit for ZPI (USCN Life Science Inc, China), respectively. Plasma concentration of interleukin 6 (IL-6; Human IL-6 ELISA KIT Diaclone, SAS, France) was also evaluated using commercial tests.
Statistical Analyses
The results are presented as median (range). Protein Z, ZPI, and IL-6 were compared using the Mann-Whitney U test for nonparametric samples. Spearman (ρ) test was applied to correlate the parameters for nonparametric distribution. The program Statistica 10.0 PL (StatSoft, Poland) was used to perform statistical analyses. P values ≤.05 were considered statistically significant.
Results
Median and range of concentrations of PZ, ZPI, and IL-6 in the plasma of our healthy volunteers and patients with ET are listed in Table 2. The median concentration of PZ in the patients with ET we studied was lower, at the limit of statistical significance, compared to healthy individuals (1.42 µg/mL, 0.36-3.14 µg/mL vs 1.6 µg/mL, 0.75-2.56 µg/mL, P = .08). On the other hand, the study established meaningfully lower concentration of ZPI in patients with ET compared to healthy participants (3.22 µg/mL, 0.85-6.97 µg/mL vs 4.41 µg/mL, 1.63-7.83 µg/mL, P = .0004). Moreover, in the subgroup of patients with ET positive for JAK2V617F mutation, median plasma concentrations of PZ and ZPI were significantly lower compared to both the group of patients negative for the mutation and our healthy individuals, for PZ (1.38 µg/mL, 0.36-2.6 µg/mL vs 1.63 µg/mL, 0.88-3.14 µg/mL, P = .03 and 1.38 µg/mL, 0.36-2.6 µg/mL vs 1.6 µg/mL, 0.75-2.56 µg/mL, P = .016), for ZPI (2.89 µg/mL, 0.85-5.91 µg/mL vs 3.61 µg/mL, 1.53-6.97 µg/mL, P = .002 and 2.89 µg/mL, 0.85-5.91 µg/mL vs 4.41 µg/mL, 1.63-7.83 µg/mL, P < .0001). In addition, the study did not show any differences between the median concentration of PZ and ZPI in the subgroups of patients with ET without the mutation and healthy volunteers, P = .88 and P = .12, respectively.
Characteristic of Healthy Individuals and Patients With Essential Thrombocythemia.

Abbreviations: PZ, protein Z; ZPI, protein Z-dependent protease inhibitor; n, number of patients or healthy volunteers; ET, essential thrombocythemia.
aResults are expressed as median (range).
b P < .05 between patients having ET with JAK2V617F mutation and healthy volunteers.
c P < .05 between patients having ET with and without JAK2V617F mutation.
d P < .05 between patients with ET and healthy volunteers.
No significant differences in the concentration of protein Z and ZPI were noted either in our healthy participants or patients with ET, either male or female (for all, P > .05). Also, no differences in PZ or ZPI concentrations dependent on aspirin reception were observed in the patients with ET, P = .35 and P = .98, respectively. Moreover, our analyses demonstrated a positive statistically significant correlation between PZ and ZPI concentrations only in patients with a JAK2V617F mutation having ET (ρ = .31, P = .043).
A medical history of thromboembolic episodes was reported in 12 patients (17% of the studied patients), and 4 patients had a history of bleeding (5.6%). Nine patients with a history of thrombosis were positive for JAK2V617F mutation. The median concentration of PZ and ZPI in the group of patients with ET having venous thromboembolic episodes and the JAK2V617F mutation was significantly lower compared to healthy volunteers, for PZ (1.23 µg/mL, 0.82-1.99 µg/mL vs 1.6 µg/mL, 0.75-2.56 µg/mL, P = .043) and for ZPI (2.42 µg/mL, 0.85-4.21 µg/mL vs 4.41 µg/mL, 1.63-7.83 µg/mL, P < .0001).
We subsequently evaluated the association of each protein under scrutiny with a series of hematological variables of well-known parameters which may be involved in the pathogenesis of ET and the thrombotic risk. The median concentration of IL-6 in the plasma of our patients with ET stood at 1.31 pg/mL (0.05-5.26 pg/mL), nearly twice as high as the median of the healthy control group (0.69 pg/mL; 0.31-1.62 pg/mL), which constituted a statistically significant difference (P = .008). The concentration of IL-6 was significantly higher in patients positive for JAK2V617F mutation (1.51 pg/mL, 0.25-5.26 pg/mL) than in the negative patients with ET (0.89 pg/mL, 0.05-2.73 pg/mL) and healthy volunteers, P = .029 and P = .021, respectively. There was no difference between the healthy volunteers and JAK2V617F negative patients with ET (P = .301). In addition, the study established an insignificant negative correlation between the concentration of IL-6 and the concentration of PZ and ZPI (ρ = −.15, P = .25 and ρ = −.1, P = .63). Even more importantly, it showed a significant negative correlation between concentrations of PZ and levels of IL-6 in the subgroup of JAK2V617F positive patients with ET (ρ = −.36, P = .04). There was no meaningful link in the negative subgroup (ρ = .02, P = .91). In addition, the study did not reveal a significant association between the concentrations of PZ and ZPI and age, platelets, or white blood cells counts and C-reactive protein (for all P > .05).
Discussion
Essential thrombocythemia is currently classified among some relatively benign diseases in which the main objective of the treatment strategy consists in preventing thrombotic events. The widespread use of routine hematologic screening and novel diagnostic tools greatly facilitates disease recognition and treatment. This helps to prevent a significant number of early vascular events, which still constitute the first disease manifestation in approximately one third of the patients. 16 Even in the absence of any thrombotic manifestations, patients with ET present with a hypercoagulable state, which is a laboratory finding of increased levels of plasma biomarkers of hemostatic system activation. 17,18 There are multiple factors believed to contribute to the pathogenesis of thrombosis in ET, some of which are host related whereas others disease related. Therefore, an acquired thrombophilic state develops in patients who are prone to vascular complications, but the mechanisms ultimately responsible for blood activation coagulation and the increased thrombotic tendency in ET have not yet been fully elucidated.
The potential role of PZ/ZPI levels and PZ polymorphisms in the pathogenesis of thrombotic diseases have been investigated in various clinical studies which, however, have produced conflicting results. Many of these studies reported low PZ levels to be associated with an increased risk of thrombosis in several types of vascular diseases, such as the ischemic stroke, 19 coronary heart disease, 20 venous thromboembolic disease, 21 and fetal loss. 22 Yet, other studies either did not observe this association or, on the contrary, reported a link between high levels of PZ and thrombotic events. As suggested by Martinelli et al, 23 if only a very low level of PZ could serve as an isolated risk factor for venous thrombosis (VT), moderate PZ deficiency would certainly increase the venous thromboembolic risk of other well-known prothrombotic risk factors. These observations were confirmed by Sofi et al 11 who performed a systematic meta-analysis and indicated that only a very low level of PZ could serve as an isolated risk factor, particularly in arterial thrombosis and venous thromboembolism. 11 In addition, procoagulant consequences of low PZ levels might be altered when combined with additional risk factors such as a hematological malignancy, which is the case especially in multiple myeloma and its antiangiogenic treatment. 24
Our study is the first to have established the concentration of PZ and ZPI in a group of patients with ET. We found the median plasma concentrations of PZ to be decreased, at the limit of statistical significance, compared to healthy individuals. Moreover, the median concentration of ZPI in our patients with ET was meaningfully lower than in the healthy individuals. More importantly, the study revealed statistically significant lower concentrations of PZ and ZPI in patients positive for JAK2V617F mutation relative to patients without the mutation. Finally, the study established a significantly lower concentration of PZ and ZPI, in a group of patients with VT (75% of them were JAK2V617F positive) compared to healthy individuals. Indeed, PZ and ZPI circulate as a complex, 6 and it has been shown that the plasma concentration of one partner affects the concentration of the other. 6,8 Thus, a primary reduction in PZ would be anticipated to reduce the level of ZPI and vice versa. A significant association of PZ–ZPI complex in patients with peripheral arterial disease was detected in an analysis performed by Sofi et al. 11 In line with the previous data, our study showed a significant positive association of PZ–ZPI complex but only in JAK2V617F mutation-positive patients with ET, which suggests a possibility of a different mechanism of PZ decrease compared to isolated thromboembolic complication. First, this phenomenon can be associated with the activation of platelets and leukocytes observed mostly in JAK2V617F mutation-positive patients with ET, what may perturb the resting state of the endothelium, one of the sources of PZ, and turn it into a surface triggering a prothrombotic status. 25 In addition, in patients with ET, activated neutrophils release reactive oxygen species and intracellular proteases, which can act on endothelial cells modifying the hemostatic balance toward a prothrombotic state. Proteases can induce detachment or lysis of endothelial cells, affecting functions involved in thromboregulation, 26 which can cause a decrease in the PZ synthesis by endothelial cells. Indeed, reductions in protein C and protein S, vitamin K-dependent factors structured similar to PZ, were found in patients with ET having thrombosis. 3 However, not all of our patients with ET had a significantly decreased PZ concentration, but this phenomenon was mostly associated with JAK2V617F mutation. Thus, the lower concentration of PZ observed in patients with ET may be caused by its consumption in the process of coagulation-induced endothelium activation. However, not all patients carrying this mutation had a thromboembolic complication due to increased neutrophil–platelet aggregates. Additionally, Falanga et al confirmed that patients with ET on aspirin had a significantly reduced aggregate formation, decreasing the endothelium activation, 25 which should result in the normalization of PZ concentration. Yet, our study did not establish significant differences in PZ concentration according to aspirin use.
Second, the impact of inflammation on PZ or ZPI plasma levels is debatable. Some studies informed of lower levels of PZ in patients with high plasma IL-6 or fibrinogen levels, 27 whereas there was also a report of a significant increase in plasma PZ at 72 hours after a percutaneous coronary intervention, 28 strengthening the possible influence of inflammation on PZ levels during an acute vascular event. 29 In vitro studies did not support the role of inflammatory cytokines in PZ biosynthesis by cultured hepatocytes, 30 whereas Oncostatin M, a cytokine of the IL-6 family, increased PZ biosynthesis by microvascular endothelial cells, 5 suggesting a possible induction of PZ production by inflammatory mediators. A contrasting observation was made by Undar et al 27 ; similarly, a negative correlation between PZ concentration and IL-6 was reported in a group of patients with hematological malignances in our previously published study. 24 Undeniably, an increased inflammatory state in patients with ET can play a causative role in both arterial and venous thromboses. 31 In the study of Barbui et al, 31 the incidence of thromboembolic complications in ET was independently associated with high concentrations of C-reactive protein, doubling the risk. However, patients included in our study had confirmed the lack of a chronic as well as acute inflammation process, and their level of C-reactive protein was within the limit of normal.
Finally, IL-6 is a pleiotropic cytokine involved in a wide range of physiologic and pathologic states, including ET. 32 Indeed, IL-6 is one of the cytokines related to megakaryocytic development, which exerts its effect through binding to a cell surface receptor, IL-6Ra, and a signal transducing unit, gp130, especially in JAK2V617F-positive patients. 33 In addition, Cacciola et al 34 confirmed that IL-6 may facilitate clonal expansion in JAK2V617F mutated patients with ET and represent an unfavorable prognostic indicator. Indeed, our study confirmed significantly higher concentrations of IL-6 in all the patients with ET compared to healthy individuals and even more importantly, higher concentrations in the patients with ET carrying the mutation as opposed to those who do not, which falls in line with the previous reports. Above all, however, the negative correlation we established between PZ concentration and IL-6 existing only in JAK2V617F mutation-positive patients participating in our study may confirm the pathogenesis of PZ deficiency.
Our results suggest that the deficiency of PZ may increase thrombosis tendency in patients with ET. Further research is required in order to fully elucidate the hemostatic role of PZ–ZPI complexes, especially in patients under treatment and to evaluate its significance in thrombosis complication.
Footnotes
Acknowledgments
We would like to express our gratitude to the Laboratory of Molecular Biology at the Department of Haematology and Proliferative Disease of Hematopoietic System, Bone Marrow Transplantation Section, University Hospital of Lord’s Transfiguration in Poznan for carrying out the analysis of JAK2V617F mutation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
