Abstract
Introduction
Hepatocellular carcinoma (HCC) is associated with an increased risk of thrombosis, including venous thromboembolism (VTE), 1 acute cerebral infarction (ACI), 2 acute coronary syndrome (ACS) 3 and disseminated intravascular coagulation. 4 VTE includes pulmonary embolism, 5 deep vein thrombosis (DVT), 6 and portal vein thrombosis (PVT) which is the most frequent site of thrombosis, with a reported incidence of 20%-40%. 7 Both local and systemic factors can foster the development of site-specific thrombosis, particularly PVT.8,9 Cancer- related ACI has received increased attention in recent years, 2 such that it now has its own specific clinical features, and most of its pathogenesis is known to be related to hypercoagulability.10,11
A hypercoagulable state is also a cause of thrombosis in patients with HCC. 12 Approximately 20% of all VTE cases may be related to cancer, which confers a 4- to 7-fold higher risk of developing VTE. 13 VTE in cancer patients is associated with plasma hypercoagulability, which might be triggered either by the tissue factor (TF) from cancer cells or the host response to cancer. 14 In addition, patient and treatment-related factors, such as chemotherapy, bed rest, infection, and surgery, may play a role in increasing the risk.15-18
However, HCC is almost always a complication of liver cirrhosis (LC), 19 with the prevalence of LC among patients with HCC estimated to be 85% to 95%. 20 Patients with LC are usually considered to have hypocoagulability due to a prolonged prothrombin time (PT), 21 whereas the rebalanced and unstable hemostatic status of LC can be easily tipped towards thrombotic complications by superimposed conditions, including HCC. 22
The PT and activated partial thromboplastin time (APTT) are useful biomarkers for detecting bleeding tendency but are not adequate for evaluating hypercoagulability.23,24 The PT-international normalized ratio (INR) and APTT are useful for monitoring warfarin and heparin, respectively. A clot waveform analysis (CWA) can enhance the ability of APTT and PT, 25 and a CWA-APTT is useful for analyzing hemostatic abnormalities and monitoring anticoagulant therapy,26-28 while a small amount of TF-induced FIX activation assay (sTF/FIXa) using platelet-rich plasma (PRP) can evaluate hemostatic abnormalities and hypercoagulability. 28 A thrombin generation test (TGT) 29 and thromboelastogram (TEG) 30 can also help visualize the clotting process and provide more information than conventional clotting assays; however, these assays are expensive and are generally used only in research. Compared to the TGT and TEG,29,30 the recently developed CWA using a fully automated optical coagulation analyzer can easily analyze hemostatic abnormalities.
In the present study, the hypercoagulable state was evaluated using a CWA-APTT and CWA-sTF/FIXa to examine the relationship between hypercoagulability and thrombosis in 86, 48, and 153 samples from patients with HCC, LC, and chronic liver disease (CH), respectively.
Material and Methods
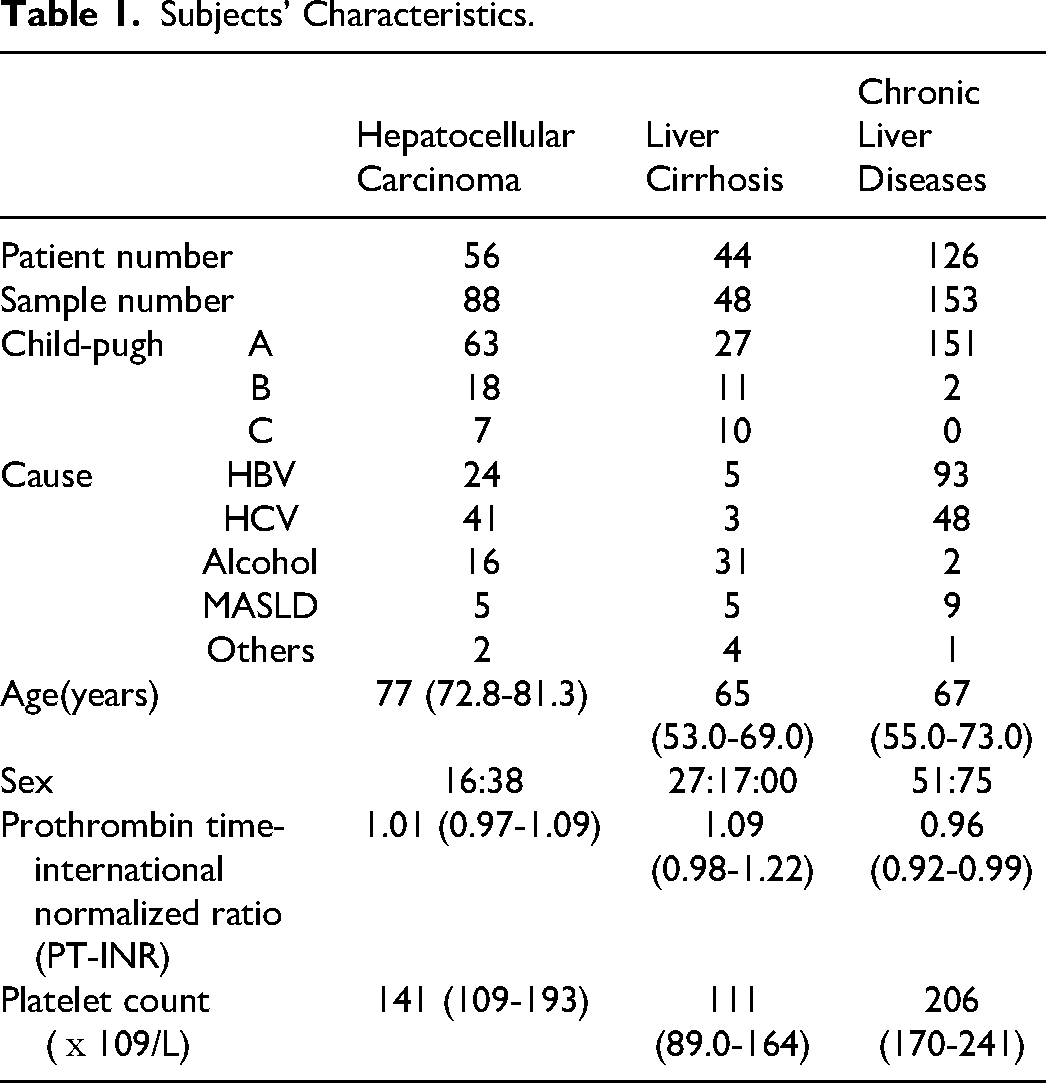
This study included 289 samples from 226 consecutive patients with liver disease who attended our hospital or were hospitalized between August 2020 and August 2023. Thrombotic complications and outcome of HCC were followed until December 31, 2023. Patients were classified into CH, LC, HCC, and HCC post-treatment groups. Patients with transient liver injury such as drug-induced hepatitis; congenital liver disease, such as congenital biliary dilatation; and multiple possible causes, such as hepatitis C virus infection and alcohol use, were excluded. Patients treated with anticoagulant agents were also excluded from CWA. CH (126 patients, 153 samples), LC (44 patients, 48 samples), HCC (29 patients, 48 samples), and HCC post-treatment (27 patients, 40 samples) cases were included in the study.
CH is a condition in which an abnormal liver function persists for more than 6 months. This category included patients retrospectively registered with chronic hepatitis B, hepatitis C, alcoholic liver disease, nonalcoholic steatohepatitis, autoimmune hepatitis, and primary cholestatic cholangitis.
Cirrhosis is a condition that causes decreased liver parenchymal cells, impaired blood flow due to fibrosis and structural remodeling, portal hypertension due to the formation of portal vein-major circulation shunt, ascites, and hepatic encephalopathy. 29 LC included cases diagnosed with cirrhosis on a liver biopsy or cases of clinical cirrhosis with findings of portal hypertension, such as esophageal or gastric varices or a platelet count ≤ 100,000, no other triggers, and recent abdominal ultrasound showing morphology, liver stiffness, portal venous blood flow, and hepatic venous blood flow waves suggestive of LC.
HCC is the most common type of primary liver cancer. HCC was defined according to the Clinical Practice Guidelines for HCC 2021, 31 and patients were those with diagnosis of HCC by dynamic computed tomography, gadoxetate sodium-enhanced magnetic resonance imaging or a liver tumor biopsy. The Barcelona Clinic Liver Cancer (BCLC) staging system is the most widely accepted staging system for staging HCC based on the performance states, Child-Pugh classification, number of tumors, number of nodes, and portal vein invasion.32-34 We divided HCC patients into groups according to the BCLC staging system, and those who underwent radiofrequency ablation or transcatheter arterial chemoembolization and had no recurrence were assigned to the post-treatment group, while those who underwent surgery were excluded from the post-treatment group. Thirteen patients with HCC were associated thrombosis (ACI in 8, PVT in 4, ACS in 4, and disseminated intravascular coagulation in one) and no patients without HCC were associated with thrombosis. Blood sampling was performed at the onset in 9 patients with thrombosis and 4 patients with HCC.
The study protocol (2019-K9) was approved by the Human Ethics Review Committee of the Mie Prefectural General Medical Center, and informed consent was obtained from each participant. This study was conducted in accordance with the principles of the Declaration of Helsinki.
The PT-INR was measured using a Thromborel S (Sysmex Co.) and an automatic coagulation analyzer CS-5100 (Sysmex Co.). Platelet counts were measured using a fully automatic blood cell counter XN-3000 (Sysmex Co.). The CWA-APTT was performed using an APTT-SP®, which uses silica as an activator of FXII and synthetic phospholipids (Werfen, Bedford, MA, USA) and platelet-poor plasma (PPP), with an ACL-TOP® system (Werfen) as previously reported.25-28 The CWA- sTF/FIXa assay was performed using PRP and 2000-fold diluted HemosIL RecombiPlasTin 2G (TF concentration <0.1 pg/ml; Werfen). 28 One curve and 2 derivative peaks (DPs) are shown on the monitor of this system25,26; one shows the changes in the absorbance observed while measuring the APTT, corresponding to the fibrin formation curve (FFC); the second (first derivative peak firstDP) corresponds to the coagulation velocity; and the third (second derivative peak second DP) corresponds to the coagulation acceleration. The height and time of the first DP, second DP, and fibrin formation are called the first DP height (first DPH) and first DP time (first DPT), second DPH and second DPT, and fibrin formation height (FFH) and fibrin formation time (FFT), respectively (Figure 1). PRP was prepared by centrifugation at 900 r/min for 15 min (platelet count, 40 x1010 /L), and PPP was prepared by centrifugation at 3000 r/min for 15 min (platelet count, < 0.5 × 1010 /L). 28
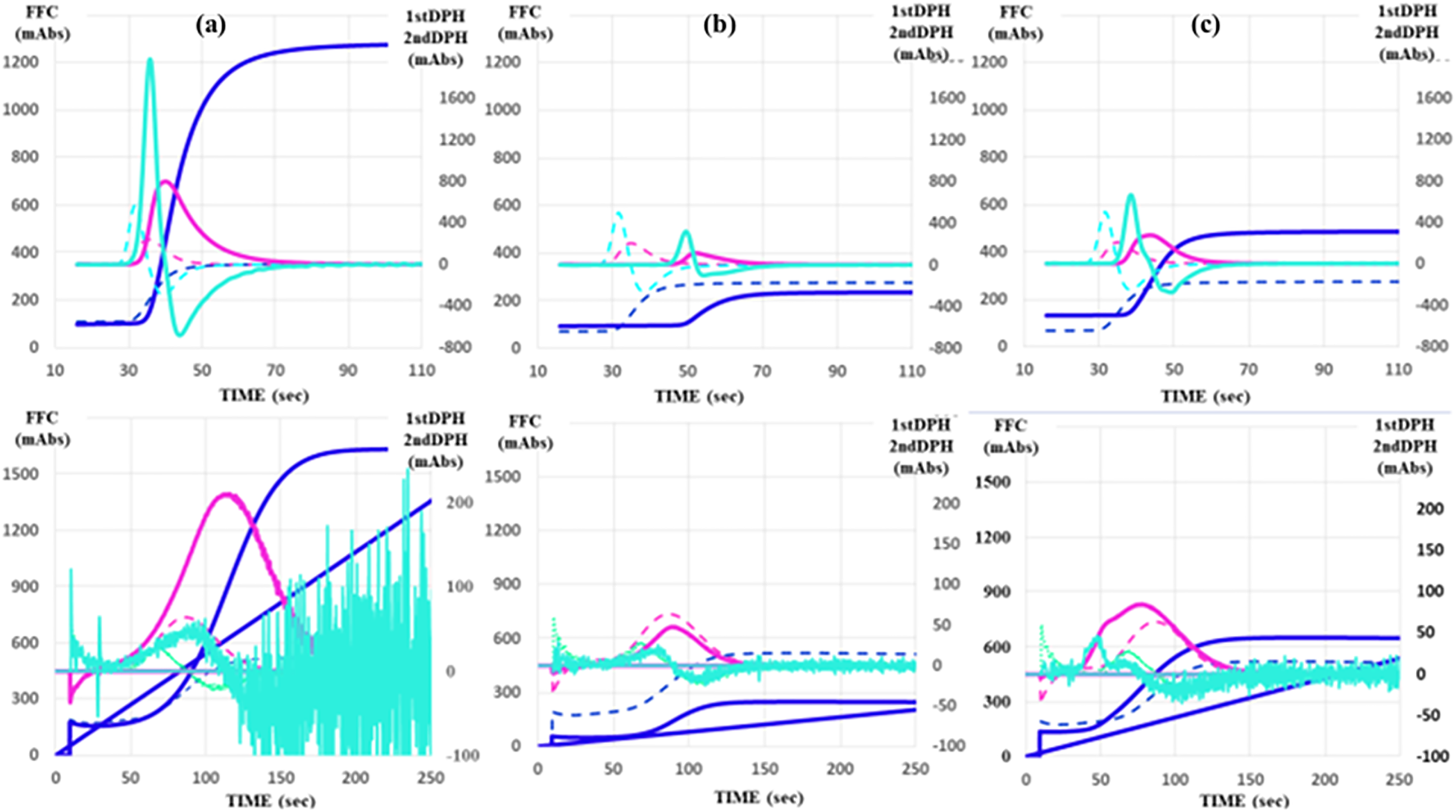
CWA-APTT (upper figure) and CWA-sTF/FIXa (lower figure) in patient with hepatocellular carcinoma (a), liver cirrhosis (b) and chronic liver disease (c).
Statistical Analyses
Data are expressed as the median (range). The significance of the differences between groups was examined using the Mann-Whitney U-test. The cutoff values, determined as the point at which the sensitivity and specificity curves intersected, were examined using a receiver operating characteristic (ROC) analysis. Significance was set at P < .05. All statistical analyses were performed using the Stat-Flex software program (version 6; Artec Co. Ltd).
Results
Figure 1 shows the CWA-APTT and CWA-sTF/FIXa findings in a typical case of HCC, LC, or CH. The CWA-APTT and CWA-sTF/FIXa showed elevated peak heights and prolonged peak times in a patient with HCC before the onset of ACI, and decreased peak heights and prolonged peak times in a patient with LC. The CWA-sTF/FIXa showed slightly higher peak height and shortened peak times in a patient with CH in compared to healthy volunteers. The platelet count was significantly lower in patients with LC than in those with HCC or CH (P < .001 and P < .05, respectively). Although the PT-INR was higher in patients with LC than in those with CH or HCC, the difference was not substantial (Table 1).
Subjects’ Characteristics.
There were no significant differences in the peak times of the CWA-APTT between healthy volunteers and patients with HCC, LC, or CH, whereas the peak heights of the CWA-APTT were significantly higher in patients with HCC or CH than in healthy volunteers (Table 2). The peak times of the CWA-APTT were significantly longer in patients with HCC than in those with CH (P < .001). The peak heights of the CWA-APTT were significantly lower in patients with LC than in those with HCC and CH. Regarding the CWA-sTF/FIXa, the FFT in patients with LC or CH and the first DPT in those with HCC, LC, or CH, as well as the second DPT in those with LC or CH were significantly shorter than those in healthy volunteers. The FFH in patients with LC was significantly lower than that in healthy volunteers, and the first DPH in patients with HCC or CH was significantly higher than that in healthy volunteers (Table 2). The peak time×peak height of the FFC, first DP, and second DP on the CWA-APTT in HCC patients and those without LC were significantly longer than HCC patients with LC or CH (Figure 2). The peak time×peak height of the FFC and first DP on the CWA-sTF/FIXa in patients with HCC were significantly higher than those in patients with LC or CH and that of the FFC on the CWA-sTF/FIXa in patients with LC was significantly lower than that in patients with CH (Figure 3).
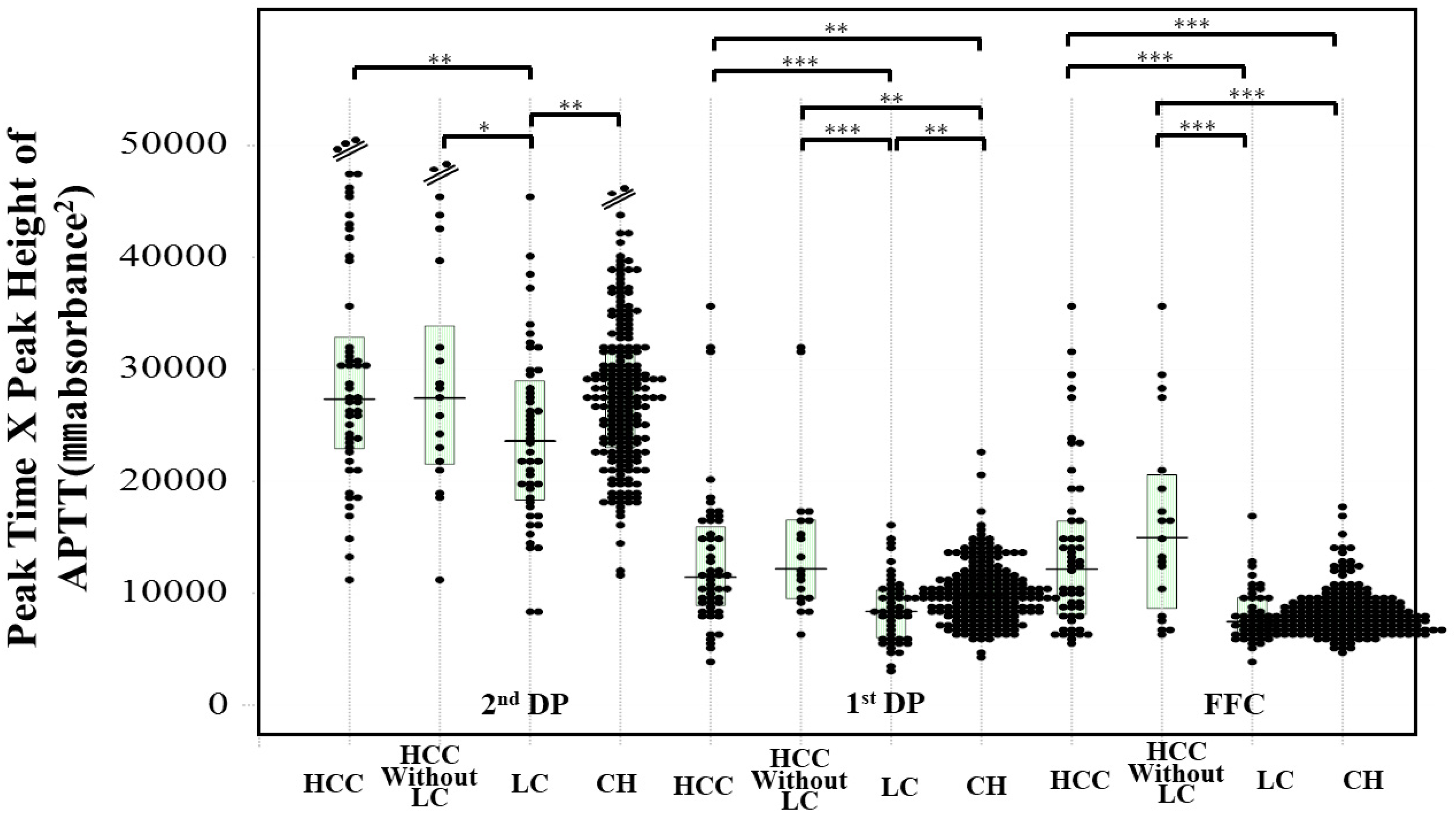
Peak time×peak height of CWA-APTT in patients with HCC, HCC without LC, LC and CH. Abbreviations: CWA, clot waveform analysis; APTT, activated partial thromboplastin time; DP, derivative peak; second DP, second derivative peak, first DP, first derivative peak; FFC, fibrin formation curve; HCC, hepatocellular carcinoma; LC, liver cirrhosis; CH, chronic liver diseases. *, P < .05; **, P < .01+; ***, P < .001.
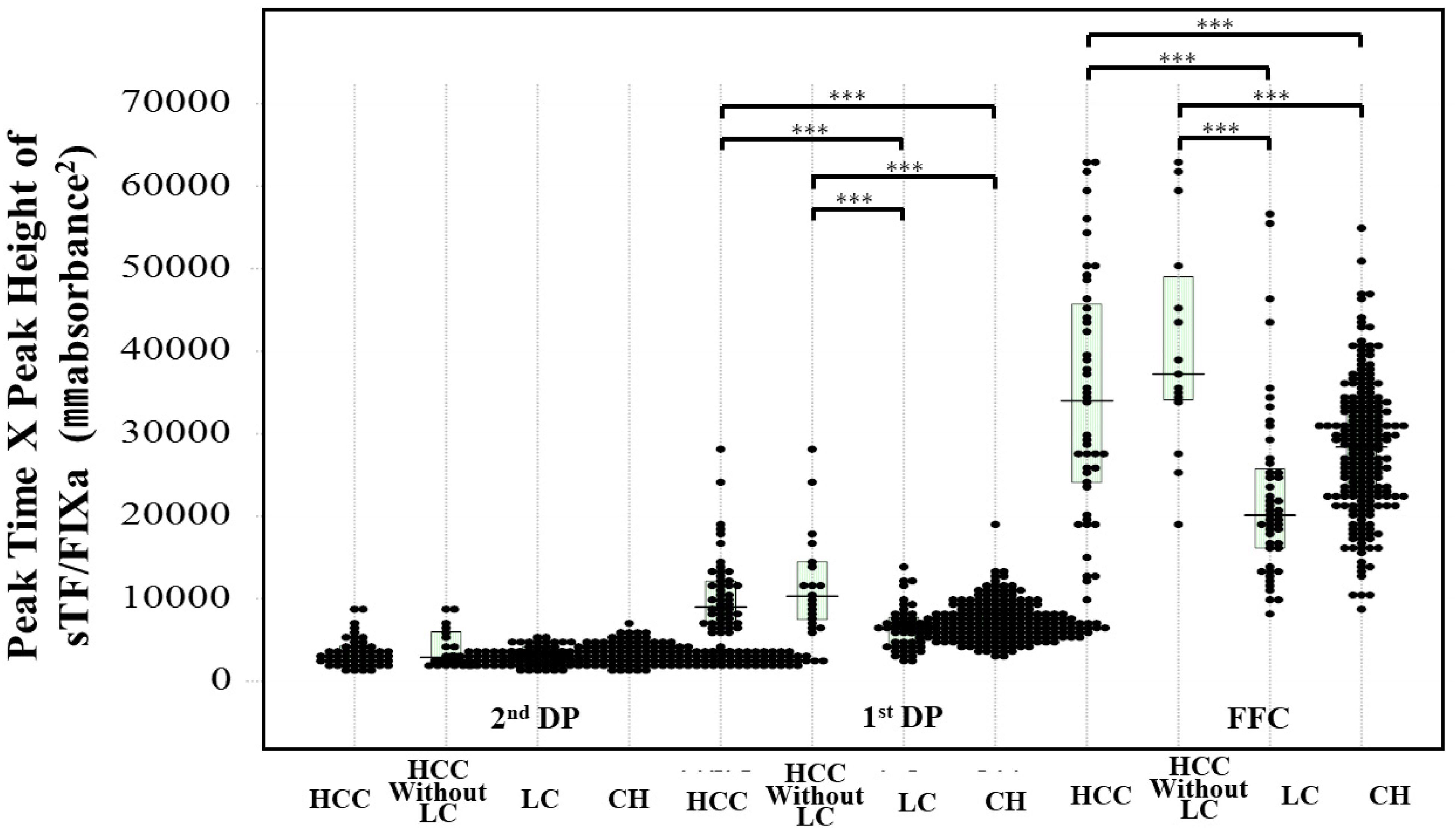
Peak time×peak height of CWA-sTF/FIXa in patients with HCC, HCC without LC, LC and CH. Abbreviations: CWA, clot waveform analysis; sTF/FIXa, small amount of tissue factor/factor IX activation assay; DP, derivative peak; second DP, second derivative peak, first DP, first derivative peak; FFC, fibrin formation curve; HCC, hepatocellular carcinoma; LC, liver cirrhosis; CH, chronic liver diseases. *, P < .05; **, P < .01+; ***, P < .001.
CWA-APTT and CWA-sTF/FIXa in Healthy Volunteers and Patients with HCC, LC, and CH.
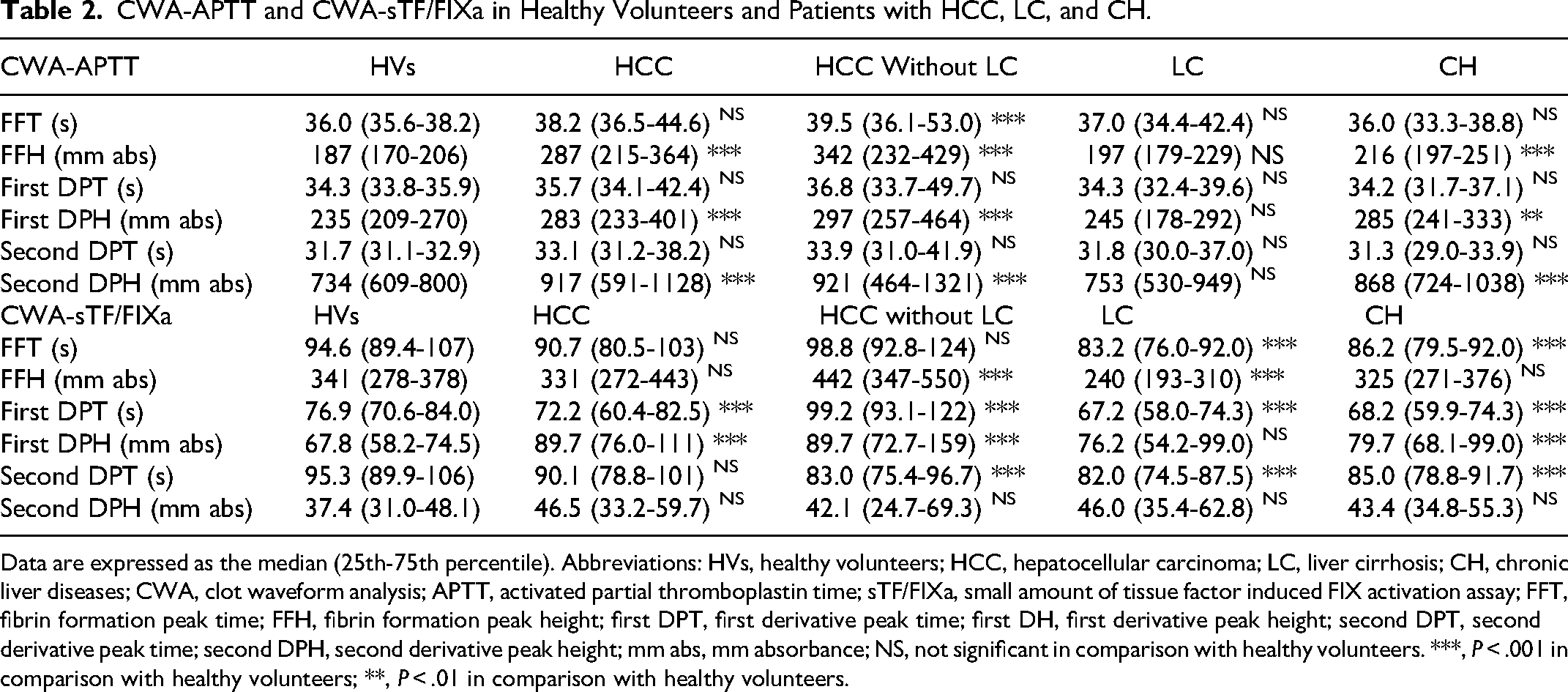
Data are expressed as the median (25th-75th percentile). Abbreviations: HVs, healthy volunteers; HCC, hepatocellular carcinoma; LC, liver cirrhosis; CH, chronic liver diseases; CWA, clot waveform analysis; APTT, activated partial thromboplastin time; sTF/FIXa, small amount of tissue factor induced FIX activation assay; FFT, fibrin formation peak time; FFH, fibrin formation peak height; first DPT, first derivative peak time; first DH, first derivative peak height; second DPT, second derivative peak time; second DPH, second derivative peak height; mm abs, mm absorbance; NS, not significant in comparison with healthy volunteers. ***, P < .001 in comparison with healthy volunteers; **, P < .01 in comparison with healthy volunteers.
In the ROC analysis, the area under the curve (AUC) for HCC without LC versus CH was the highest in the FFH of CWA-APTT, and that for LC versus CH was the highest in the FFH of the CWA-sTF/FIXa (Table 3). The peak times of the CWA-APTT were significantly longer in stages B-D than in stage A and cases with a response (Figure 4). There were no significant differences in peak heights of CWA-APTT, and peak times and heights of CWA-sTF/FIXa among stage A-D and response groups of HCC. Arterial thrombosis such as ACI and ACS was frequently associated with HCC patients without LC, and PVT was frequently associated with HCC patients with LC (Tables 4 and 5).
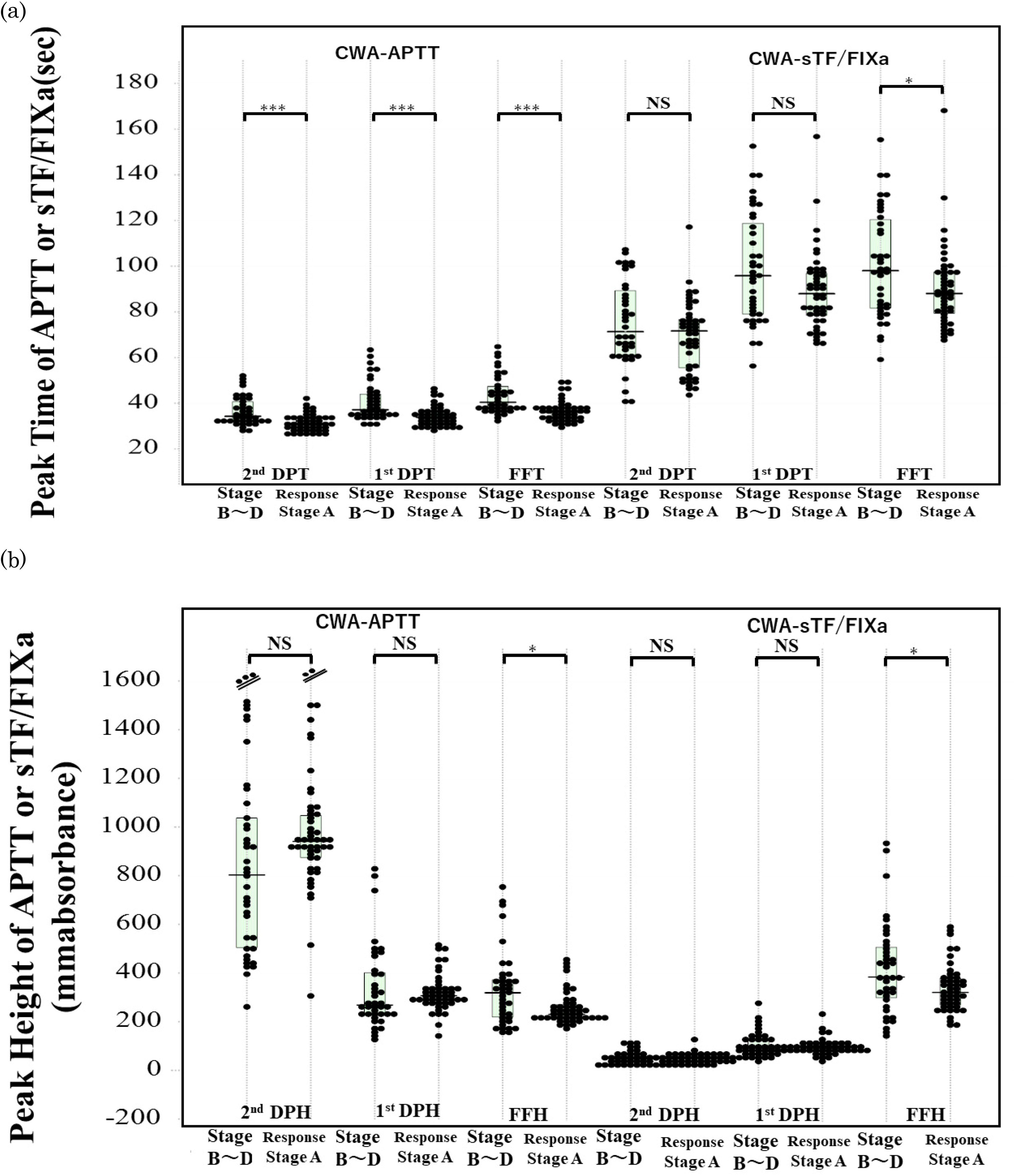
Peak time of CWA-APTT or CWA-sTF/FIXa in patients with 4 HCC stages. Abbreviations: APTT, activated partial thromboplastin time; CWA, clot waveform analysis; sTF/FIXa, small amount of tissue factor/factor IX activation assay; DP, derivative peak; second DPT, peak time of second derivative peak; first DPT, peak time of first derivative peak; FFT, fibrin formation time; second DPH, peak height of second derivative peak; first DPH, peak height of second derivative peak; FFH, fibrin formation height; HCC, hepatocellular carcinoma.
Results of ROC Analyses for HCC (HCC vs CH) or LC (LC vs CH).
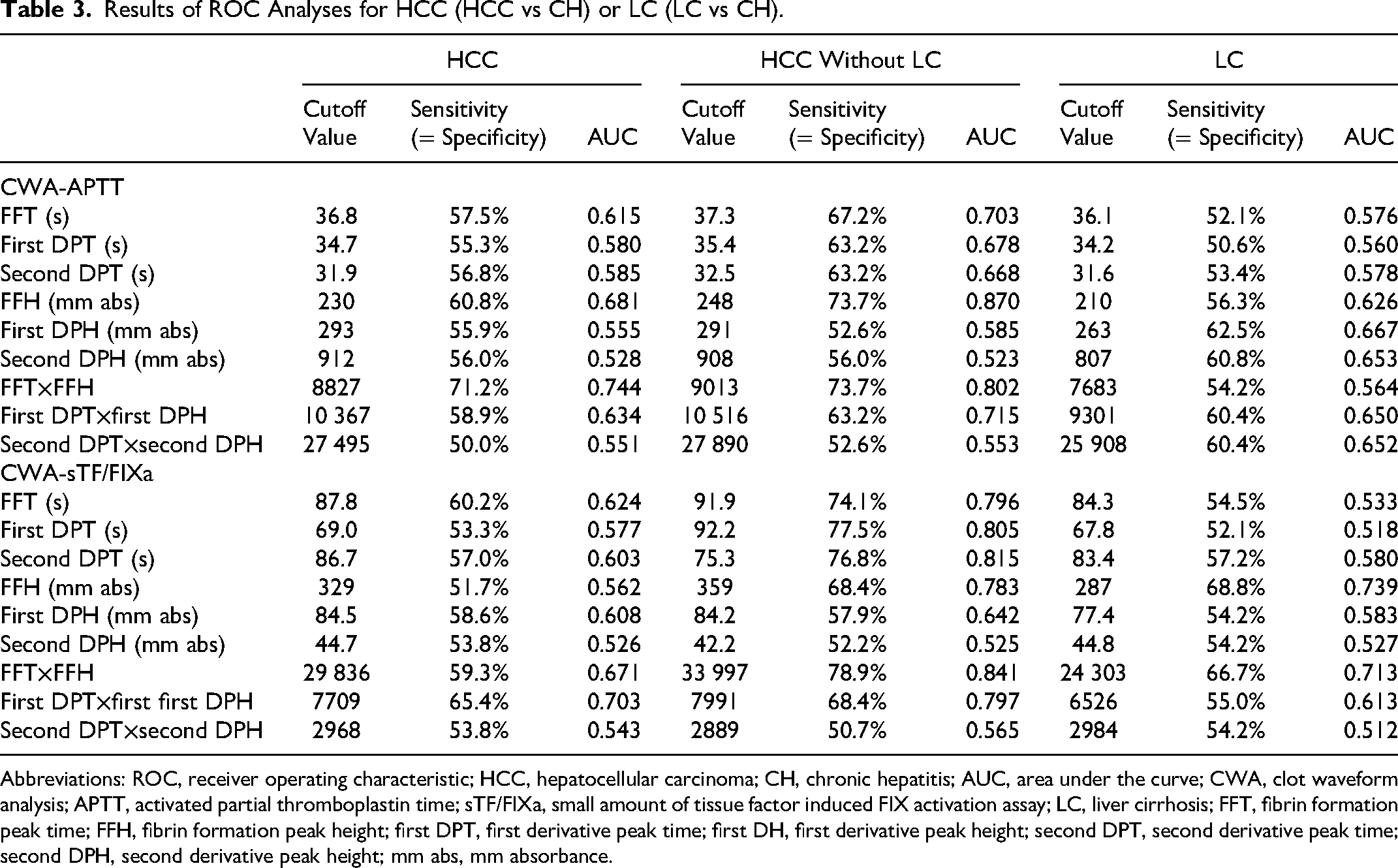
Abbreviations: ROC, receiver operating characteristic; HCC, hepatocellular carcinoma; CH, chronic hepatitis; AUC, area under the curve; CWA, clot waveform analysis; APTT, activated partial thromboplastin time; sTF/FIXa, small amount of tissue factor induced FIX activation assay; LC, liver cirrhosis; FFT, fibrin formation peak time; FFH, fibrin formation peak height; first DPT, first derivative peak time; first DH, first derivative peak height; second DPT, second derivative peak time; second DPH, second derivative peak height; mm abs, mm absorbance.
Characteristics of Patients with HCC.

Abbreviations: HCC, hepatocellular carcinoma; PT-INR, prothrombin time-international normalized ratio.
Thrombosis in Patients with HCC.
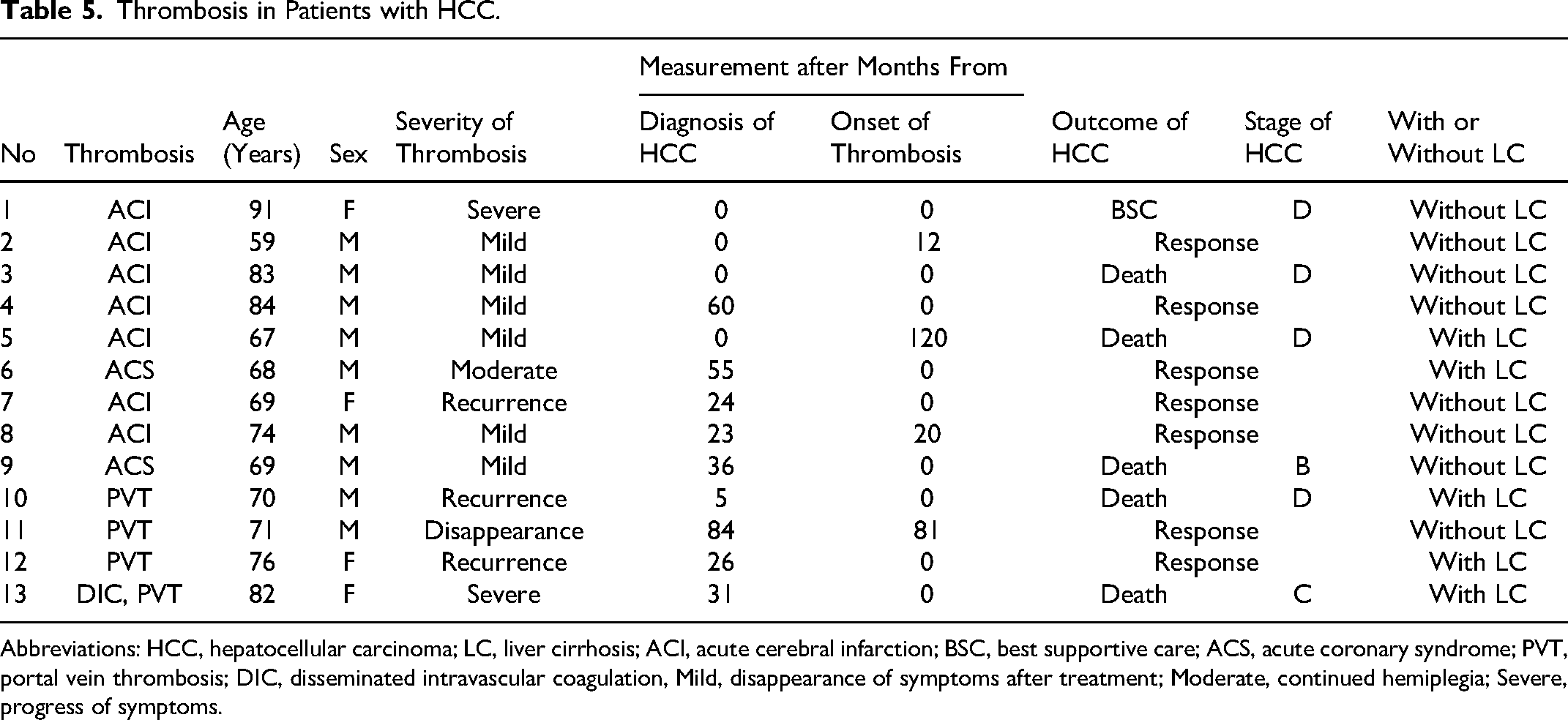
Abbreviations: HCC, hepatocellular carcinoma; LC, liver cirrhosis; ACI, acute cerebral infarction; BSC, best supportive care; ACS, acute coronary syndrome; PVT, portal vein thrombosis; DIC, disseminated intravascular coagulation, Mild, disappearance of symptoms after treatment; Moderate, continued hemiplegia; Severe, progress of symptoms.
The PT-INR, platelet count, FFH of the CWA-APTT, and FFH and second DPT of the CWA-sTF/FIXa were significantly different between HCC patients with and without LC. The platelet count in HCC patients with thrombosis was significantly higher than in those without thrombosis; however the platelet count was within the normal range. The FFH and first DPH of the CWA-APTT and CWA-sTF/FIXa were significantly higher in HCC patients with thrombosis than in those without thrombosis (Table 6). In the ROC analysis for patients with thrombosis versus patients with all liver diseases without thrombosis, an AUC of >0.800 was observed in FFH of the CWA-APTT, and first DPH and first DPT × first DPH of the CWA-sTF/FIXa (Figure 5 and Table 7).
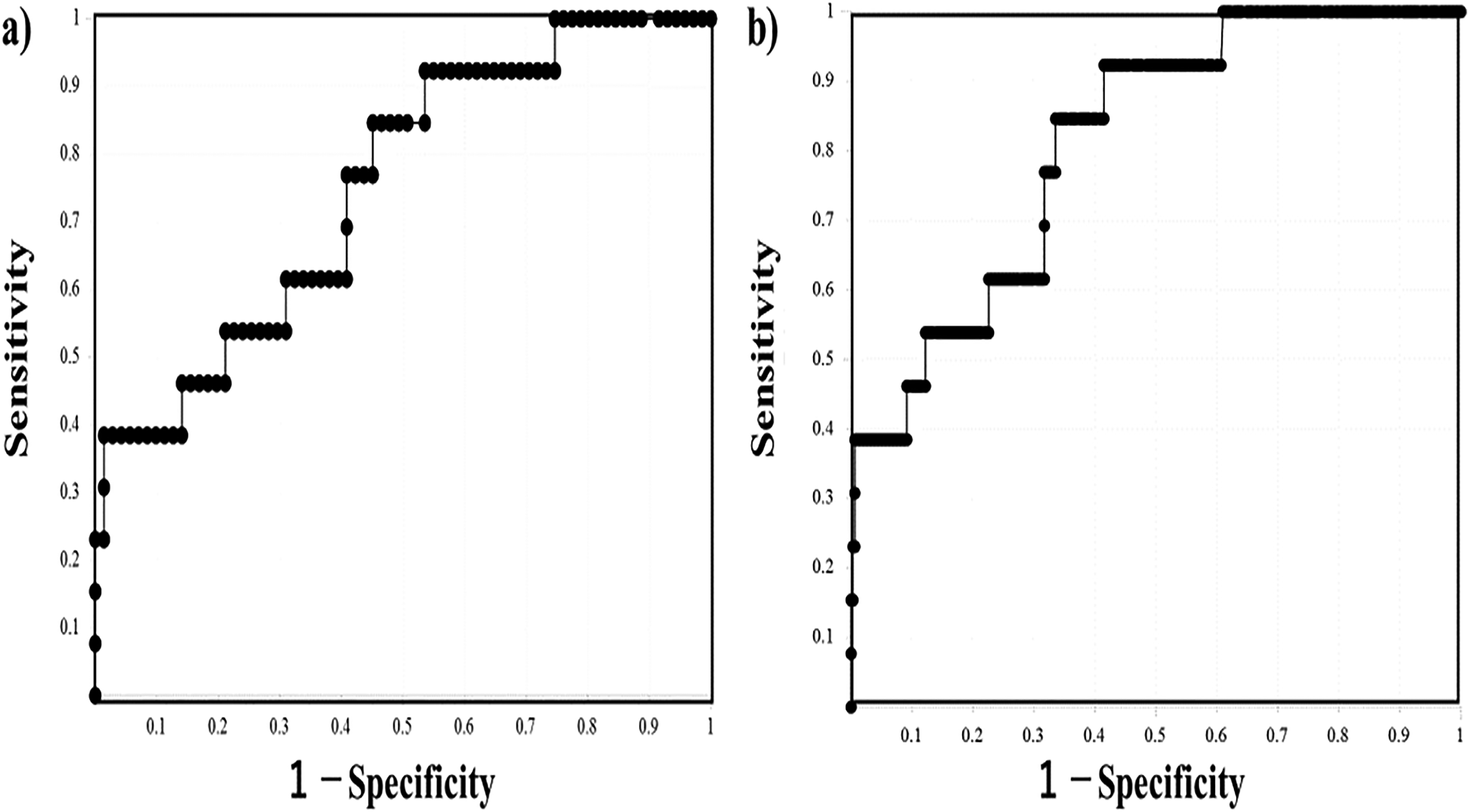
An ROC analysis of the second DPT CWA-APTT (a) and first DPT × first DPH on a sTF/FIXa (b) for HCC patients with thrombosis versus all patients without thrombosis. Abbreviations: ROC, receiver operating characteristic; CWA, clot waveform analysis; APTT, activated partial thromboplastin time; DP, derivative peak; first DPT, peak time of first derivative peak; first DPH, peak height of first derivative peak; HCC, hepatocellular carcinoma.
PT-INR, Platelet Count, CWA-APTT and CWA-sTF/FIXa in HCC Patients with and Without LC or Thrombosis.
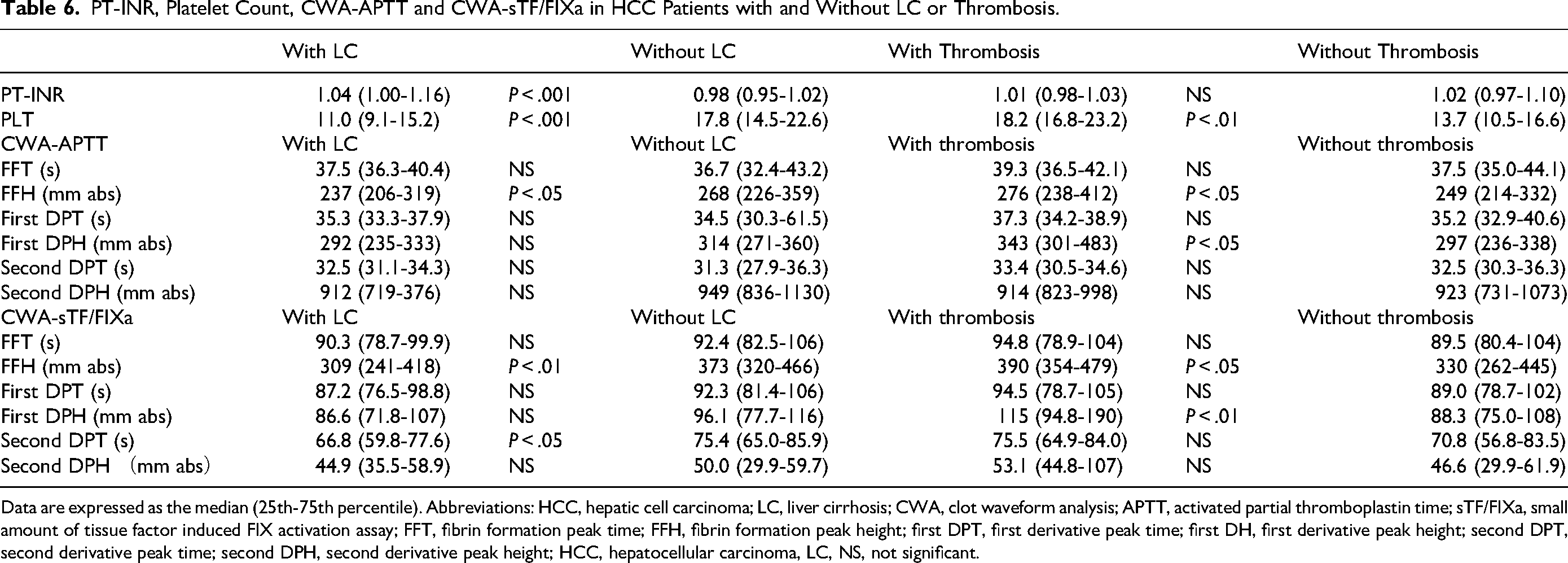
Data are expressed as the median (25th-75th percentile). Abbreviations: HCC, hepatic cell carcinoma; LC, liver cirrhosis; CWA, clot waveform analysis; APTT, activated partial thromboplastin time; sTF/FIXa, small amount of tissue factor induced FIX activation assay; FFT, fibrin formation peak time; FFH, fibrin formation peak height; first DPT, first derivative peak time; first DH, first derivative peak height; second DPT, second derivative peak time; second DPH, second derivative peak height; HCC, hepatocellular carcinoma, LC, NS, not significant.
Results of ROC Analyses for Thrombosis in Patients with all Liver Diseases.
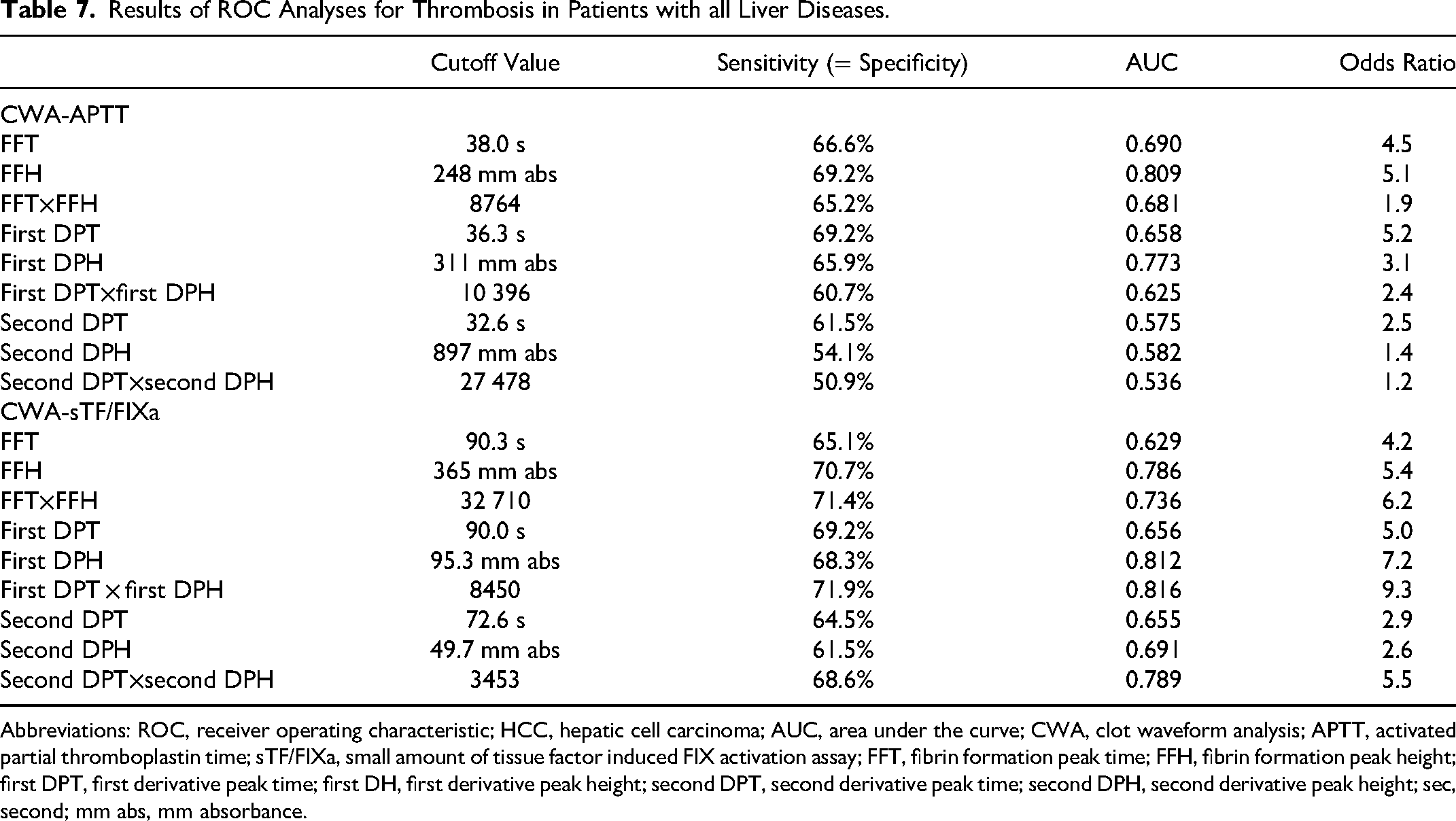
Abbreviations: ROC, receiver operating characteristic; HCC, hepatic cell carcinoma; AUC, area under the curve; CWA, clot waveform analysis; APTT, activated partial thromboplastin time; sTF/FIXa, small amount of tissue factor induced FIX activation assay; FFT, fibrin formation peak time; FFH, fibrin formation peak height; first DPT, first derivative peak time; first DH, first derivative peak height; second DPT, second derivative peak time; second DPH, second derivative peak height; sec, second; mm abs, mm absorbance.
Discussion
PVT is the most common thrombotic complication in HCC patients with LC,1,35-36 and the present study showed that 4 patients had PVT, while 9 had arterial thrombosis such as ACI or ACS. Our findings indicate a high incidence of thrombosis in patients with HCC, and the arterial thrombosis incidence in particular was high in HCC patients without LC, while that of PVT was high in those with LC. LC patients with PVT generally have low physiological anticoagulants such as antithrombin, protein C, and protein S 37 and a low platelet count; whereas HCC patients with arterial thrombosis had a normal platelet count, suggesting that arterial thrombosis in HCC might be caused by platelet activation. Of note, a high incidence of AMI or ACI was not noted in a previous study of cancer-associated thrombosis.38,39 The incidence of VTE is significantly higher in North America and Europe than in Japan. In addition, arterial thromboses may take a longer time to manifest the symptoms or onset than VTE. In the future, improving the survival in patients with cancer may result in an increased incidence of arterial thrombosis.
Cancer patients are generally considered to be in a hypercoagulable state due to increased fibrinogen levels, 40 thrombocytosis, 41 a high TF expression,42,43 increased microvesicles,44,45 neutrophil extracellular traps 46 and platelet activation.47,48 However, HCC is frequently associated with LC, and fibrinogen levels and platelet counts may not be significantly increased in patients with HCC. Although hepatoma cells can synthesize TF, hepatic dysfunction in HCC may not sufficiently synthesize the clotting factors that are activated by increased TF. Our findings indicated a marked prolongation of peak times for both the CWA-APTT and CWA-sTF/FIXa, suggesting that the coagulation system is not sufficiently activated in patients with HCC.
Activation of clotting factors is generally considered to be one of the causes of thrombosis in cancer.49,50 A previous report 51 on patients with other cancers showed prolonged peak times in the CWA-APTT but not in the CWA-sTF/FIXa. The peak times of the CWA-APTT were also prolonged in patients with advanced-stage HCC. The peak heights of both CWA-APTT and CWA-STF/FIXa were significantly higher in patients with HCC than in those with CH, suggesting that coagulation ability might be significantly higher in patients with HCC than in those with CH. Marked increases in the peak heights of the CWA-APTT and CWA-sTF/FIXa were also reported in patients with ACI. 52 The increase in the peak height of the CWA-APTT and CWA-sTF/FIXa is reported to be caused by a thrombin burst.53-56 Although the mechanism underlying the prolonged clotting time in patients with HCC may involve hepatic dysfunction, the concomitance of both prolonged peak times and increased peak heights cannot be sufficiently understood. These findings in the CWA of prolonged peak times and increased peak heights indicate that a strong coagulation process might continue for a long time, suggesting that a thrombotic risk may exist in patients with HCC.
Previous studies51,52 reported that an increased first DPH could be a useful predictor of thrombosis. However, the increase of first DPH was not sufficient for diagnosing thrombosis in the present study. Therefore, the AUC of first DPT × first DPH for HCC versus CH and thrombosis versus all liver diseases was the highest, while that for CH versus LC was not among the parameters of the CWA-APTT and CWA-sTF/FIXa, suggesting that the first DPT × first DPH may be a useful parameter for suggesting HCC or diagnosing thrombosis in HCC. However, in cases where the first DPT would be too long, such as with heparin treatment, the first DPT × first DPH would not be useful for diagnosing thrombosis.
Regarding the limitations of this study, as the CWA-sTF/FIXa is carried out in fresh platelet-rich plasm, these assays were performed during the day on a weekday. Patients treated with anticoagulant agents were excluded.
In conclusion, significantly prolonged peak times and increased peak heights of the CWA-APTT and CWA-STF/FIXa were observed in patients with HCC, and the first DPT × first DPH was shown to be useful for suspecting HCC in CH and diagnosing thrombosis in HCC.
Footnotes
Conflicts of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Ministry of Health, Labour and Welfare of Japan (grant number 21FC1008). CWA was partially supported by IL Japan
Ethical Approval
This study protocol (2019-K9) was approved by the Human Ethics Review Committee of the Mie Prefectural General Medical Center. This study was conducted in accordance with the principles of the Declaration of Helsinki.
Informed Consent
Informed consent was obtained from each participant.
