Abstract
Background
Dual antiplatelet therapy (DAPT) with aspirin and a P2Y12 inhibitor is a standard therapy in patients with ischemic vascular diseases (IVD) including coronary artery, cerebrovascular and peripheral arterial diseases, although the optimal duration of this treatment is still debated. Previous meta-analyses reported conflicting results about the effects of long-term and short-term as well as non-DAPT use in various clinical settings. Herein, we conducted a comprehensive meta-analysis to assess the efficacy and safety of different durations of DAPT.
Methods
We reviewed relevant articles and references from database, which were published prior to April 2023. Data from prospective studies were processed using RevMan5.0 software, provided by Cochrane Collaboration and transformed using relevant formulas. The inclusion criteria involved randomization to long-term versus short-term or no DAPT; the endpoints included at least one of total or cardiovascular (CV) mortalities, IVD recurrence, and bleeding.
Results
A total of 34 randomized studies involving 141 455 patients were finally included. In comparison with no or short-term DAPT, long-term DAPT reduced MI and stroke, but did not reduce the total and CV mortalities. Meanwhile, bleeding events were increased, even though intracranial and fatal bleedings were not affected. Besides, the reduction of MI and stroke recurrence showed no statistical significance between long-term and short-term DAPT groups.
Conclusion
Long-term DAPT may not reduce the mortality of IVD besides increasing bleeding events, although reduced the incidences of MI and stroke early recurrence to a certain extent and did not increase the risk of fatal intracranial bleeding.
Keywords
Introduction
Increasingly, various ischemic vascular diseases (IVD) are prevented and treated with dual antiplatelet therapy (DAPT), which includes aspirin and a P2Y12 inhibitor (clopidogrel, ticagrelor, or prasugrel). Acute coronary syndromes (ACS) should be treated for a minimum of 12 months in accordance with European guidelines. 1 Concerning antiplatelet therapy following percutaneous transluminal coronary intervention (PCI), the use of DAPT is crucial in preventing coronary stent thrombosis and is currently recommended for 12 months after implantation of either drug-eluting stents (DES) or bare metal stents (BMS).1,2 For patients with ACS following coronary artery bypass grafting (CABG), the use of DAPT consisting of ASA and ticagrelor is supported by guidelines. 3 In patients with stable ischemic heart disease, DAPT should be administered for at least 1 or 6 months following the implantation of BMS or DES.1,2 For patients undergoing carotid artery stenting for cerebrovascular diseases, DAPT is necessary to prevent stent-related ischemic complications, but the optimal duration of DAPT remains unknown.4,5
Previous meta-analyses on the safety and effects of varying duration of DAPT had yielded controversial results. In ACS patients who underwent DES treatment, a network meta-analysis categorized the duration of DAPT into three groups, with short-term DAPT (≤6 months) as the reference group. The analysis provided evidence that most patients could be considered for short-term DAPT after DES implantation. 6 In contrast, a recent meta-analysis conducted by Gianluigi et al reported that long-term DAPT significantly reduces the incidence of myocardial infarction (MI) and stroke recurrence in patients with IVD, without increasing the risk of fatal intracranial bleeding. 7
Given these conflicting results, this study analyzed all randomized controlled trials (RCTs) to compare the effects of long-term and short-term as well as non-DAPT used in patients with MI and ischemic stroke on the aspects of IVD recurrence, bleeding, and mortality.
Methods
We performed a meta-analysis of RCTs based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 8 The review protocol was registered in the PROSPERO International Prospective Register of Systematic Reviews (ID CRD42023399895).
Search Strategy for Identification of Studies
PubMed, Medline, Embase, Cochrane, and Clincialtrials.gov were searched for relevant articles and references prior to April 2023 using the following terms: “randomized controlled trial,” “dual antiplatelet therapy,” “clopidogrel,” “prasugrel,” and “ticagrelor.” No limitation was set on language, but an English abstract must be provided for non-English articles.
Inclusion and Exclusion Criteria
The inclusion criteria involved randomization to long-term versus short-term or no DAPT; the endpoints included at least one of total or cardiovascular (CV) mortality, IVD recurrence, and bleeding. A “long-term DAPT” refers to any combination therapy that includes aspirin and a P2Y12 inhibitor lasting more than 6 months, while “short-term or no DAPT” refers to the duration lasting not more than 6 months or mono antiplatelet therapy (aspirin, clopidogrel, ticagrelor, or prasugrel). Studies failing to meet these definitions will be excluded. These studies were also excluded if their patients enrolled were undergoing anticoagulant therapy, or were with bleeding diathesis, such as coagulation disorder, as well as with venous thrombus embolism (VTE) and pulmonary embolism (PE).
Study Selection Process
Two investigators (XZ and HX) screened and assessed trial eligibility independently and in parallel. After the initial selection of titles and abstracts, relevant articles and references were subjected to full-text reviews to determine their eligibility. Any discrepancies were resolved through discussion between the two reviewers.
Data Extraction
From each study, data were extracted from the full text of articles by one study author (XZ) using a standardized form, including the inclusion criteria, patient demographics, medical history, baseline drugs, number of patients enrolled, and duration of follow-up. The endpoints assessed included: total or CV mortality, IVD recurrence, and bleeding.
Risk of Bias Assessment
The Risk of Bias Assessment Tool developed by The Cochrane Collaboration was used to evaluate the methodological quality of trials. 9
Data Synthesis and Analysis
Statistical analyses were conducted using RevMan5.0 software, with relative risks (RRs) employed to measure the association between studies. We derived aggregated risk estimates expressed with a 95% confidence interval (CI). We employed random effect models to account for the heterogeneity of aggregate estimates and utilized the Cochran Q test to evaluate heterogeneity between studies, quantified using Cochran Q and I² statistics. Study heterogeneity was assessed based on recommendations from the Cochrane Handbook for Systematic Reviews of Interventions, where I² values ranging from 0% to 50%, 50% to 75%, and 75% to 100% represent low, substantial, and considerable heterogeneity, respectively. 9
Trials enrolled stable cardiovascular diseases (CVD) and unstable CVD patients were analyzed in subgroups. Patients with stable CVD or those who did not initiate study treatments during an acute CV event were classified as the stable patient subgroup. Conversely, patients with acute CVD or those who started study treatments during an acute CV event were classified as another subgroup of unstable patients. Subgroup analyses were performed not only for the two aforementioned subgroups but also for studies comparing long-term DAPT groups with short-term and non-DAPT groups.
Results
Results of the Research
We initially identified 3933 papers and retrieved 88 for full-text review. After excluding 54 trials, we eventually included 34 RCTs in our analysis (Figure 1).10–43 The local ethics committees approved all studies enrolled and all patients signed the informed consents. Additionally, we summarized the baseline characteristics of these 34 RCTs incorporated into our meta-analysis (Table 1).
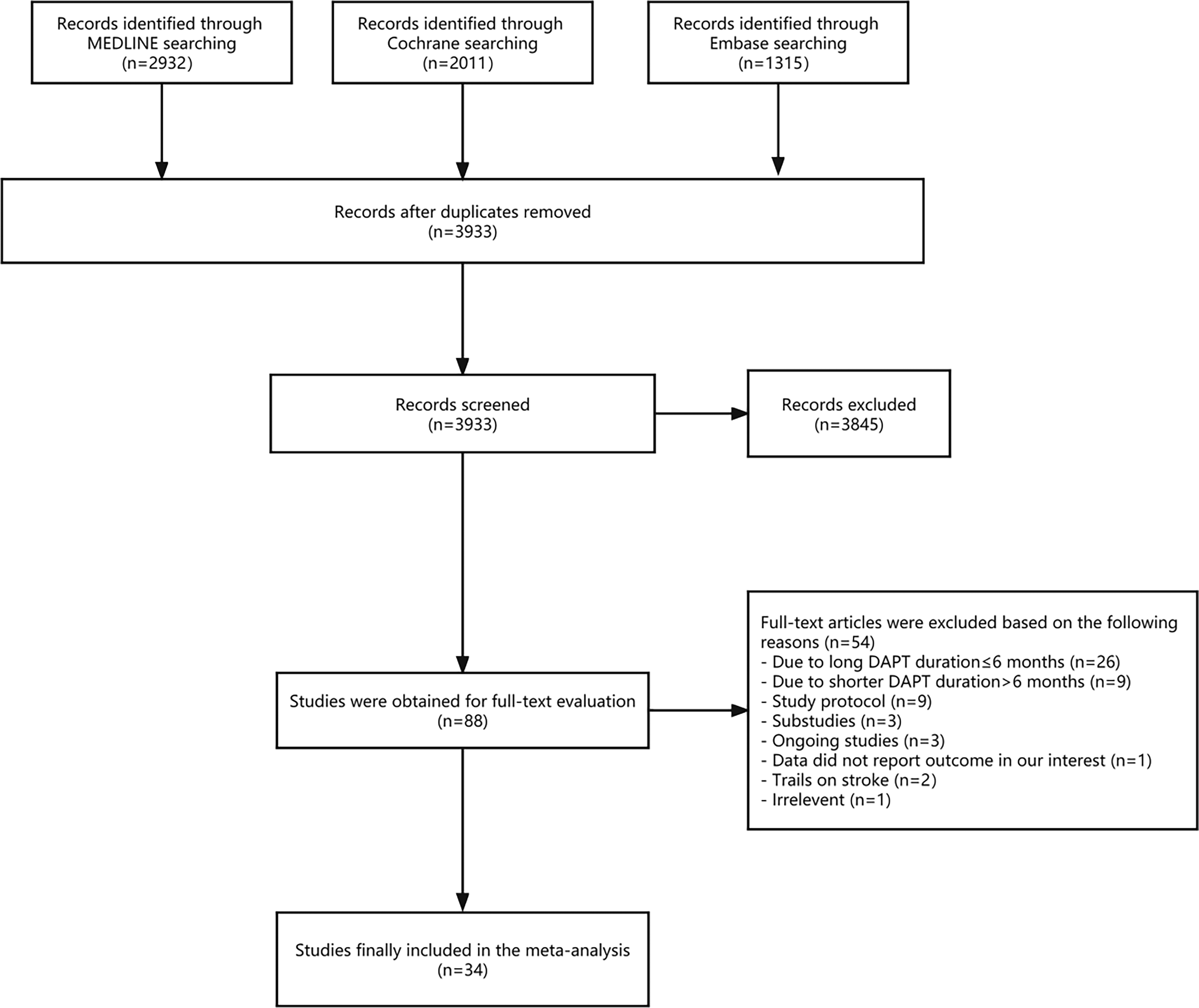
Meta-analysis flow chart. Fifty-four studies were subsequently excluded and 34 trials were eventually included in the analysis.
Demographic Characteristics of the Included Studies.
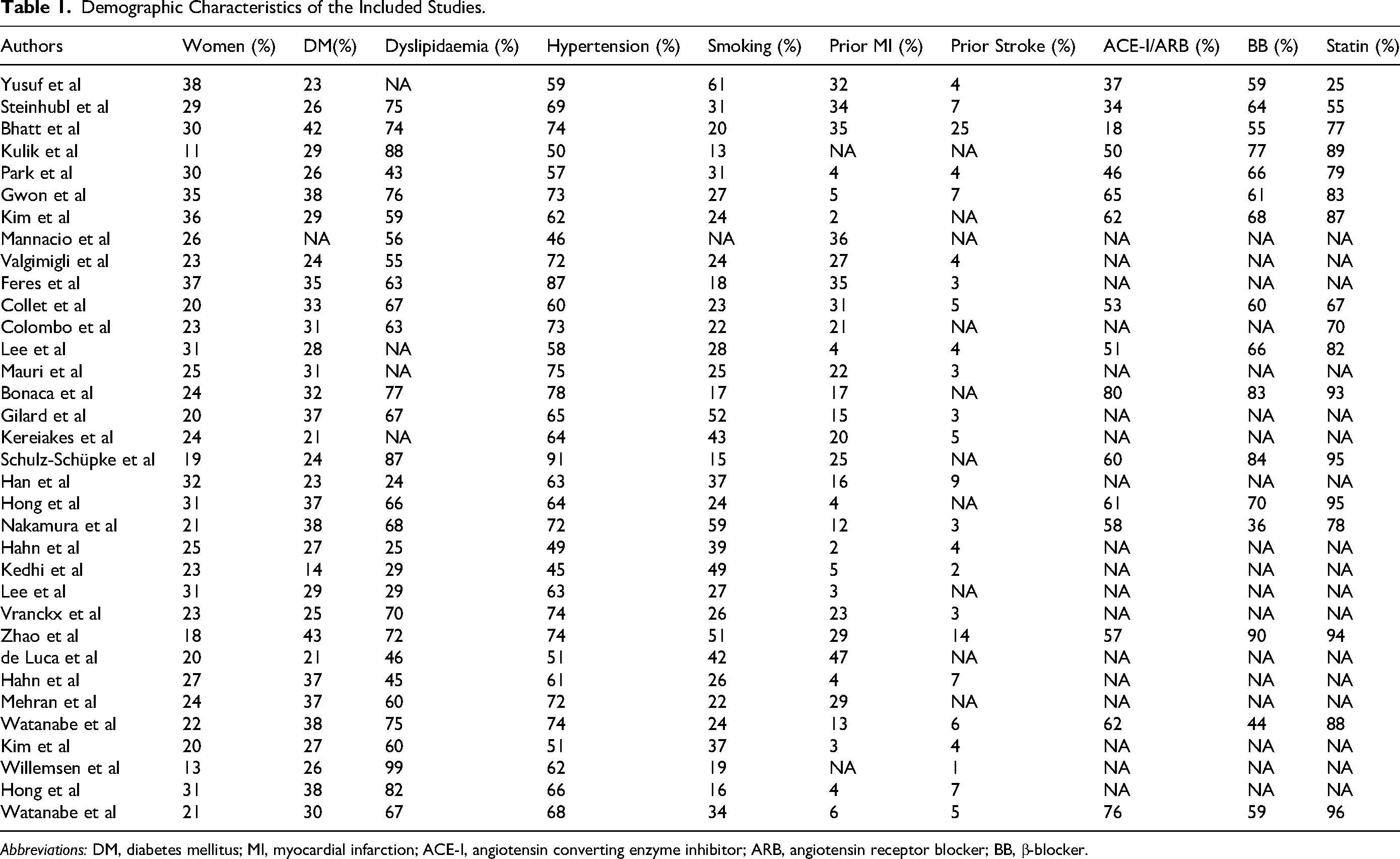
Abbreviations: DM, diabetes mellitus; MI, myocardial infarction; ACE-I, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; BB, β-blocker.
Risk of Bias in Included Studies
The risk of bias within all included RCTs was assessed by two researchers and the primary source of bias was related to incomplete outcome data, while other domains generally exhibited low risks of bias (Figure 2).
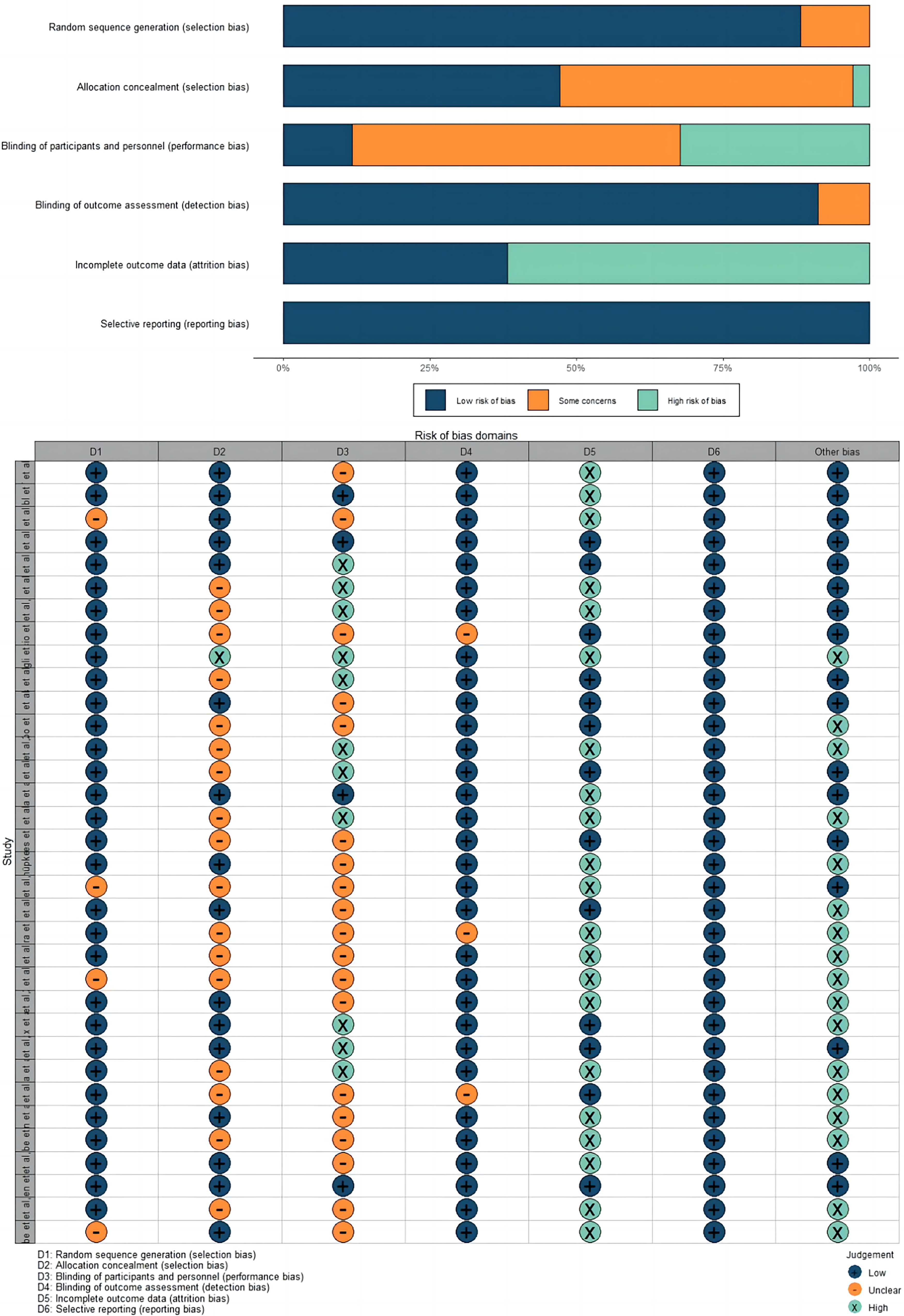
The risk of bias within all included RCTs.
Outcomes Analysis
Overall Analysis
The incidence of MI and stroke was 2.6% and 1.1% in long-term DAPT groups compared to 3.0% and 1.2% in short-term or no DAPT groups. Therefore, in comparison with short-term or no DAPT, long-term DAPT reduced MI risk by 17% (P = 0.02; I² = 38%) and stroke risk by 10% (P = 0.72; I² = 0%) (Supplemental material online, eFigures 1 and 2). Total and CV mortalities were 3.0% and 2.0% in long-term DAPT groups, respectively, compared to 2.8% and 2.0% in short-term or no DAPT groups. Accordingly, there was no difference in total (P = 0.56; I² = 0%) or CV mortality (P = 0.88; I² = 0%) between the two groups (Supplemental material online, eFigures 3 and 4). Major bleeding events were 1.9% of patients assigned to long-term DAPT and 1.3% of those assigned to short-term or no DAPT, showing a significant 44% increase in bleeding risk in long-term DAPT groups (P = 0.06; I² = 30%; Supplemental material online, eFigure 5). However, intracranial and fatal bleeding events were 0.3% and 0.3% in patients receiving long-term DAPT compared to 0.2% and 0.2% in those receiving short-term or no DAPT, showing no statistic difference in the risk of intracranial (P = 0.81; I² = 0%) and fatal (P = 0.63; I² = 0%) bleeding between two groups (Supplemental material online, eFigure 6).
In the included trials, bleeding was defined differently. TIMI criteria were used in 16 trials, BARC criteria in 11, GUSTO criteria in 4, and STEPPLE criteria in 1. However, no statistic differences were observed among the different criteria used in the trials (Supplemental material online, eTable 1).
Stable Versus Unstable Cardiovascular Diseases
18 trials enrolled patients with stable CVD, 8 trials enrolled patients with unstable CVD, and 4 enrolled both types of patients.
In the stable and unstable CVD patients, the incidence of MI was 2.5% and 2.7% in long-term DAPT groups versus 2.7% and 3.6% in short-term or no DAPT groups, demonstrating a reduction of MI risk by 19% and 24% using long-term DAPT in the stable (P = 0.04) and unstable (P = 0.23) CVD patients.
In the stable and unstable CVD patients, stroke recurrence was 1.1% and 0.9% in long-term DAPT groups versus 1.2% and 1% in short-term or no DAPT groups. Therefore, long-term DAPT reduced stroke recurrence by 15% in stable CVD patients (P = 0.47) and 8% in unstable (P = 0.82) CVD patients.
In stable CVD patients, total and CV mortalities were 2.9% and 1.9% in long-term DAPT groups versus 2.5% and 1.7% in short-term or no DAPT groups, respectively. In unstable CVD patients, total and CV mortalities were 3.3% and 2.7% in long-term DAPT groups versus 3.5% and 2.8% in short-term or no DAPT groups. Thus, long-term DAPT did not influence total and CV mortalities in the stable (P = 0.67 and P = 0.59, respectively) and unstable (P = 0.32 and P = 0.62, respectively) CVD patients.
In the stable and unstable CVD patients, major bleeding events were reported in 1.7% and 2.4% in long-term DAPT groups versus 1.1% and 1.6% in short-term or no DAPT groups, showing a significant 59% and 46% increase of bleeding risk using long-term DAPT with stable (P = 0.20) and unstable (P = 0.83) CVD.
In stable CVD patients, the incidence of fatal and intracranial bleeding was 0.3% and 0.4% in long-term DAPT groups versus 0.2% and 0.3% in short-term or no DAPT groups. In unstable CVD patients, the incidence of fatal and intracranial bleeding was 0.1% and 0.1% in long-term DAPT groups versus 0.2% and 0.1% in short-term or no DAPT groups. Therefore, long-term DAPT did not influence the incidence of fatal and intracranial bleeding in stable (P = 0.40 and P = 0.66, respectively) and unstable (P = 0.65 and P = 0.35, respectively) CVD patients.
Long-Term DAPT Versus Short-Term DAPT or Non-DAPT
Twenty-one trials enrolled patients receiving long-term DAPT versus short-term DAPT, while 13 trials enrolled patients receiving long-term DAPT versus non-DAPT.
The incidence of MI was 2.2 and 3.0% in long-term DAPT groups versus 2.4 and 3.5% in short-term and non-DAPT groups. Stroke recurrence was 0.8 and 1.2% in long-term DAPT groups versus 0.8 and 1.5% in short-term DAPT and non-DAPT groups. Thus, long-term DAPT, when compared to short-term DAPT, reduced by 9% the risk of MI (P = 0.51) and resulted in no statistic impact on the risk of stroke (P = 0.71). Compared to non-DAPT, long-term DAPT reduced by 22% the risk of MI (P < 0.01) and 17% the risk of stroke (P = 0.88).
Total mortality was 2.0 and 3.8% in long-term DAPT groups versus 1.9 and 3.7% in short-term and non-DAPT groups. CV mortality was 1.0 and 2.7% in long-term DAPT groups versus 0.8 and 3.0% in short-term and non-DAPT groups. Therefore, no statistical significance in risk of total (P = 0.64) and CV mortalities (P = 0.96) was shown between long-term DAPT and short-term DAPT groups. Also, no statistical significance in risk of total (P = 0.39) and CV mortalities (P = 0.57) was shown between long-term DAPT and non-DAPT groups.
Major bleeding events were 1.7% and 2.0% of patients in long-term DAPT groups versus 1.3% and 1.3% in short-term and non-DAPT groups, showing a significant 33% and 53% increase of risk in long-term DAPT groups (P = 0.09; P = 0.24).
Intracranial bleeding events were 0.4% and 0.3% in long-term DAPT groups versus 0.2 and 0.2% in short-term and non-DAPT groups. Fatal bleeding events were 0.3% and 0.3% in long-term DAPT groups versus 0.2% and 0.2% in short-term and non-DAPT groups. Thus, no statistical significance in risk of intracranial (P = 0.43; P = 0.87) and fatal (P = 0.63; P = 0.42) bleeding was shown between groups.
Publication Bias and Sensitive Analysis
Funnel plots and the trim-and-fill method were utilized to assess and adjust for publication bias, with Supplemental materials provided. The I2 values of all included articles were less than 50%, indicating no significant heterogeneity (Supplemental material online, eFigure 7).
Discussion
Previous Meta-Analyses
The effects of long-term and short-term as well as non-DAPT use in different settings were conflicting in previous meta-analyses. In ACS patients receiving DES treatment, a network meta-analysis categorized the duration of DAPT into three groups, with short-term DAPT (≤6 months) as the reference group. The analysis provided evidence that most patients could be considered for short-term DAPT after DES implantation. 6 In contrast, long-term DAPT was shown to be associated with more bleeding events and mortalities. However, this study primarily evaluated the duration of DAPT using clopidogrel as a P2Y12 inhibitor, while other inhibitors, such as prasugrel and ticagrelor were not included, while the later may produce different results. 6 Conversely, a more recent network meta-analysis collecting 79 073 patients who underwent PCI with DES in 24 trials, showed long-term DAPT reduced the incidence of MI and stroke recurrence at the cost of more bleeding events and confirmed long-term DAPT is still warranted in patients with ACS or at a higher risk of IVD recurrence. 44 As for patients with ACS after CABG, a meta-analysis of 4 RCTs demonstrated that the combination of aspirin and ticagrelor was statistically associated with a reduced risk of vein graft failure, but the clinically significant bleeding risk remained. 45 Additionally, Udell et al clarified that continuation of DAPT beyond a year reduces IVD recurrence in the meta-analysis of 6 RCTs involving 33 435 MI patients. 46
As the most recent and comprehensive meta-analysis to investigate the optimal duration of DAPT in patients with IVD, our study reported 34 trials involving 141 455 patients, which was not restricted to trials of patients undergoing DES, but included all IVD patients using DAPT across different clinical settings. Distinguished from Gianluigi et al, 7 who collected 85 265 patients in 15 trials, we updated more encouraging results from recent large trials and included patients with ACS after CABG as well as those undergoing PCI for stable angina. Thus, we attribute the shining points of our meta-analysis to a diverse patient population, the inclusion of numerous RCTs, and subgroup analyses that were not previously explored.
Analysis of Long-Term Versus Short-Term or No DAPT in the Current Study
Overall, 17% of MI and 10% of stroke were reduced in long-term DAPT groups versus short-term or no DAPT groups, although a significant 44% increase in bleeding events.
MI and stroke reduction are considered the most clinically important benefits of long-term DAPT, although the absolute benefit appears modest, given the low overall incidence of these events during follow-up. Based on the available data, it is currently unfeasible to determine the extent to which the reduction in MI can be attributed to the prevention of stent thrombosis or non-stent-related MI. The meta-analysis included multiple trials, but only one of them collected data on both stent-related and non-stent-related MI, and DAPT was found to reduce the incidence of both types of MI in this study. 26 Therefore, long-term DAPT may not only prevent stent thrombosis but also reduce the risk of all types of MI.
In addition, we found that long-term DAPT does not confer a survival benefit over short-term or no DAPT in patients with IVD. Although the SMART-DATE trial reported a significantly higher risk of cardiac death in the 6-month DAPT group compared to the 12-month or longer DAPT group, our results are consistent with most previous studies indicating that long-term DAPT is not associated with increased mortality. 32
According to a large-scale registry study, a majority of bleeding occurs in the gastrointestinal tract, so physicians should be careful about gastrointestinal bleeding in patients receiving DAPT even if we concluded that major bleeding is nonfatal or intracranial. 47
Efficacy of Long-Term DAPT in Subgroup Analysis
Additional subgroup analyses in different groups were performed to assess the effects of long-term DAPT in certain clinical settings. A higher CV risk was observed in unstable CVD patients versus in stable CVD, so we further analyzed these two groups, separately. We found that the MI and stroke risk reduction and the major bleeding events increase associated with long-term DAPT also extended to the stable and unstable CVD patients, respectively.
In a separate analysis that was not analyzed in previous studies, we further analyzed the different influences on long-term DAPT versus short-term DAPT and non-DAPT, respectively. Long-term DAPT, when compared to non-DAPT, reduced the risk of MI by 22% and the risk of stroke by 17%, indicating a significant reduction of MI and stroke in long-term DAPT groups versus non-DAPT groups. However, the observed 9% reduction in MI rate did not retain statistical significance and there was no significant decrease in the risk of stroke when long-term DAPT versus short-term DAPT. This is clinically relevant since the benefits and risks of long-term DAPT versus short-term DAPT should be weighted more carefully.
Limitations
This meta-analysis has some limitations. Firstly, the definitions of some clinical endpoints vary slightly in different trials, and may potentially introduce effect modifiers. However, no statistic heterogeneity was found in our primary and secondary endpoints. Secondly, as a trial-level meta-analysis, we used published event rates instead of individual patient data for each trial. If individual patient data were accessible, it could be identified which patients would benefit more from long-term DAPT. Finally, we are unable to confidently recommend an optimal DAPT duration with certainty due to the varied DAPT durations in the included trials. Based on the current evidence, a 12-month DAPT for IVD patients seems reasonable given that the majority of long-term DAPT involved was 12 months.
Conclusion
Long-term DAPT may not reduce the mortality of IVD besides increasing bleeding events, although reduced the incidences of MI and stroke early recurrence to a certain extent and may increase the risk of fatal intracranial bleeding. Moreover, the MI and stroke risk reduction and the major bleeding events increase associated with long-term DAPT also extended to the stable and unstable CVD patients, respectively. Nevertheless, there is no statistical significance in the reduction of MI and stroke recurrence between long-term and short-term DAPT groups.
The results mentioned above revealed that it may be more appropriate to select the DAPT courses for the patients individually, based on the results of strict monitoring of the platelet aggregation status in peripheral blood dynamically, so as to reduce the incidence of ischemic events while avoiding the DAPT-related bleeding complications.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241244772 - Supplemental material for Efficacy and Safety of Long-Term Dual Antiplatelet Therapy: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296241244772 for Efficacy and Safety of Long-Term Dual Antiplatelet Therapy: A Systematic Review and Meta-Analysis by Xiaoming Zhang, Da Zhou, Siying Song, Xiangqian Huang, Yuchuan Ding and Ran Meng in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviations
Acknowledgments
The authors would like to thank all colleagues in this study for their cooperation.
Authors Contribution
XZ wrote the first draft of the manuscript; DZ, SS, and XH performed the material preparation, data collection and statistical analysis, RM and YD wrote sections of the manuscript and contributed to manuscript revision; RM takes full responsibility for the data, the analyses and interpretation, and the conduct of the research. All authors read and approved the submitted version.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Beijing Natural Science Foundation [grant number 7212047], and the National Natural Science Foundation of China [grant numbers 82171297, 82101390].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
