Abstract
Background
Oral Factor Xa Inhibitor (FXaIs) are standard of care for anticoagulation and now account for nearly 60% of all anticoagulation prescriptions. 1 In the event that the effects of an FXaI must be urgently reversed, prothrombin complex concentrate (PCC) is a fast-acting option in the acute care setting.2,3 PCC can be prescribed as a fixed dosage or a weight-based dose. Fixed doses are intended to streamline the order verification and medication preparation processes while maintaining similar efficacy outcomes. PCC is FDA-approved for vitamin K antagonist (VKA) reversal and has been used off-label to reverse other anticoagulants. PCC has demonstrated efficacy in promoting hemostasis in patients taking FXaIs who present with life-threatening bleeds. 4 A fixed-dose approach to reverse VKA effects demonstrated a 22-min reduction in time from PCC order entry to administration with no difference in achieving hemostasis. 5 There are limited data however comparing fixed and weight-based strategies for reversing FXaIs.
The cost of each strategy should be considered as well. Outside research showed a potential for cost-savings using the fixed dosing method. Utilizing the actual wholesale price of 2.90 USD per unit, their estimated annual cost savings from the use of Kcentra® fixed-dosing was 148,348 USD. 6
The purpose of this retrospective cohort study is to compare the administration time and efficacy between fixed-dose and weight-based PCC for the reversal of FXaI effects.
Methods
This retrospective cohort study was conducted in compliance with Institutional Review Board/Human Subjects Research Committee requirements.
In April 2022, the protocol for urgent reversal of FXaIs at our institution was transitioned from a weight-based approach (50 units/kg) to a fixed-dose strategy: 5000 units for intracranial hemorrhage (ICH) and 2000 units for non-ICH. After the fixed dose went live, prescribers still had the option of ordering weight-based PCC. Patients receiving PCC between April 26, 2021 to December 31, 2022 were included in the weight-based cohort if they received weight-based dosing, while patients receiving PCC between April 26, 2022 to December 31, 2022 were included in the fixed-dose group if they received fixed dosing. Protocol administration was identified from chart review.
Patients were included if they were aged ≥ 18 years and received PCC to reverse hemorrhage associated with either apixaban or rivaroxaban. Subjects receiving PCC for hemorrhage unassociated with apixaban or rivaroxaban were excluded.
The primary outcome was the time difference between PCC order entry and medication administration. Secondary outcomes included: average dose of PCC, repeat PCC doses needed, post-administration surgeries or interventions, hemostasis, 30-day mortality, thromboembolic events within 30 days, hospital length of stay, discharge destination, and nonthrombotic adverse drug events. For patients discharged earlier than 30 days, the researchers relied on electronic health records to screen for venous thromboembolism (VTE) genesis and determine mortality status.
Data collection was performed through chart review with FXaI home dosages obtained using third-party fill histories and patient-reported medication histories.
To be consistent with the anticoagulation reversal guidelines at the study site, intracerebral, subdural, and subarachnoid bleeding were all classified as ICH.
Length of stay was measured from the first point of medical contact at the study site to the time of discharge. Researchers utilized a combination of imaging reports repeated within 48 h of initial scan, stable hemoglobin, and hematocrit levels, and the need for repeat doses of reversal agents to determine if hemostasis was achieved. For ICH, hemostasis was determined primarily based on repeat imaging within 48 h. For non-ICH, assessments from gastrointestinal and surgery physicians, repeat doses of reversal agents, and hemoglobin changes were reviewed. If the hemoglobin decreased by 2 g/dL, then hemostasis was documented as not achieved.
Mortality was assessed 30 days from the administration date of PCC. Any patient who opted for hospice at discharge was counted as a death for the purposes of 30-day mortality. This decision was based on a systematic review of studies reporting the interval from initiation of palliative care to death which found that the median number of days was 19. 7
Statistical analyses were completed using JMP Pro 15.2.1. Means, medians, standard deviations, and interquartile ranges were calculated for continuous variables and proportions were calculated for nominal variables. Continuous variables were analyzed with t-tests or Mann-Whitney U tests based on the assumed distribution. Nominal variables were analyzed using Chi-square tests or Fischer Exact tests if the number of observations were small (ie < 5 in any cell).
Results
A total of 227 subjects were reviewed for inclusion. Of these, 54 patients were excluded for reversal of anticoagulants other than apixaban and rivaroxaban. Of note, 17 patients received a weight-based dose during the fixed-dose time frame. These subjects were included in the final analyses but placed in the weight-based group. This resulted in a final study cohort of 173 subjects, having 72 subjects in the fixed-dose and 101 subjects in the weight-based groups. There were no significant differences between the groups (Table 1). The majority of the population was male with a median age of 78 years (IQR = 71-86). Apixaban was the most common FXaI, and atrial fibrillation was the most common FXaI indication. At baseline, there was moderate risk of thrombosis and low risk of bleeding in people who had atrial fibrillation, assessed using CHA2DS2 VASc and HAS BLED scoring systems, respectively. ICH was the most common presentation in the fixed-dose and weight-based groups, 68.1% and 64.4%, respectively.
Baseline Characteristics.
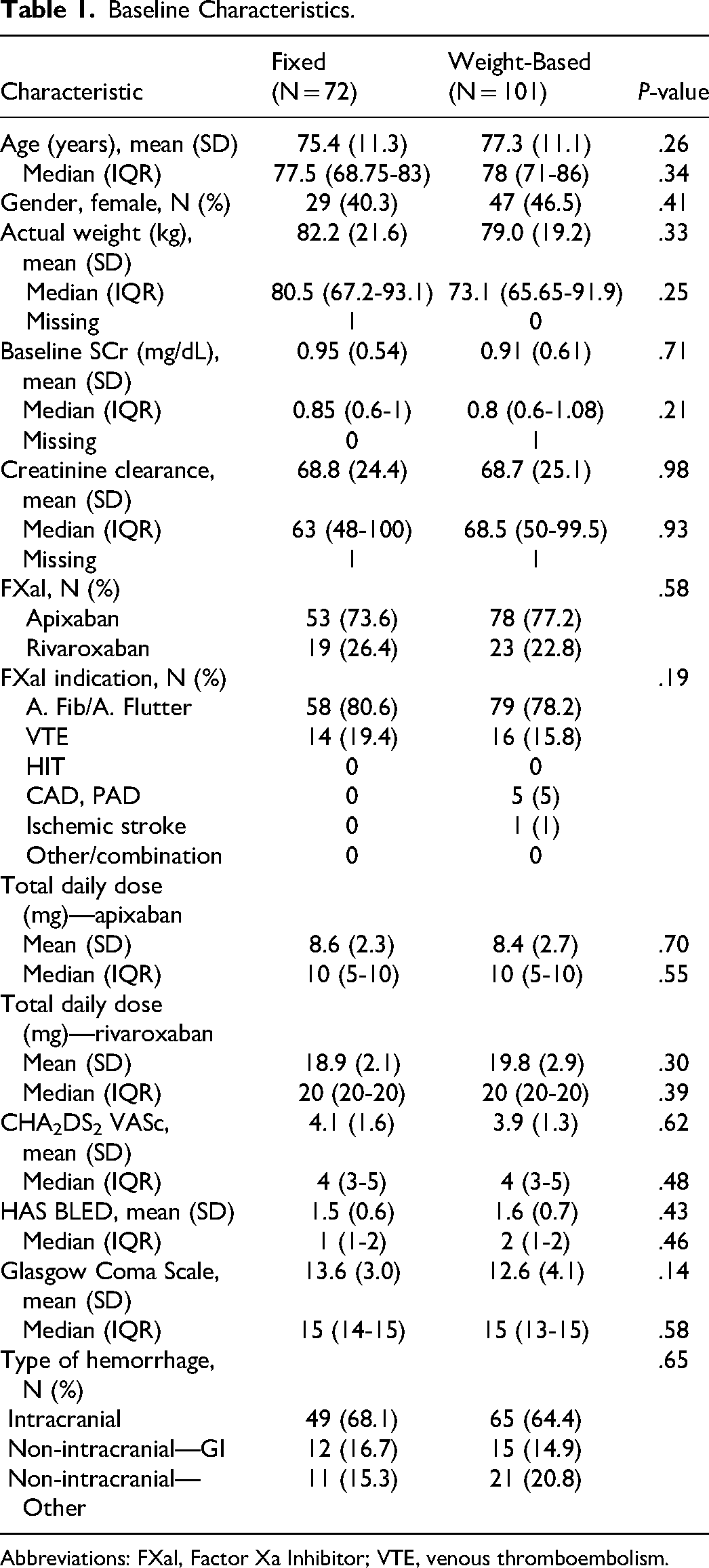
Abbreviations: FXaI, Factor Xa Inhibitor; VTE, venous thromboembolism.
The median time between order entry and drug administration, was 4.5 min shorter in the fixed-dose group compared to the weight-based group (34.5 min, IQR = 26.25-42 vs 39 min, IQR = 26.25-55.75, P = .10). A higher percentage of patients in the fixed-dose arm achieved hemostasis, but this difference was not statistically significant (79.2% vs 71.3%, RR = 1.11, 95% CI = 0.94-1.32). Recipients of fixed-dose PCC also required fewer subsequent surgeries (29.2% vs 36.7%, RR = 0.80, 95% CI = 0.51-1.24) and had no difference in mortality (26.4% vs 35.6%, RR = 0.73, 95% CI = 0.46-1.17) (Table 2).
Outcomes.
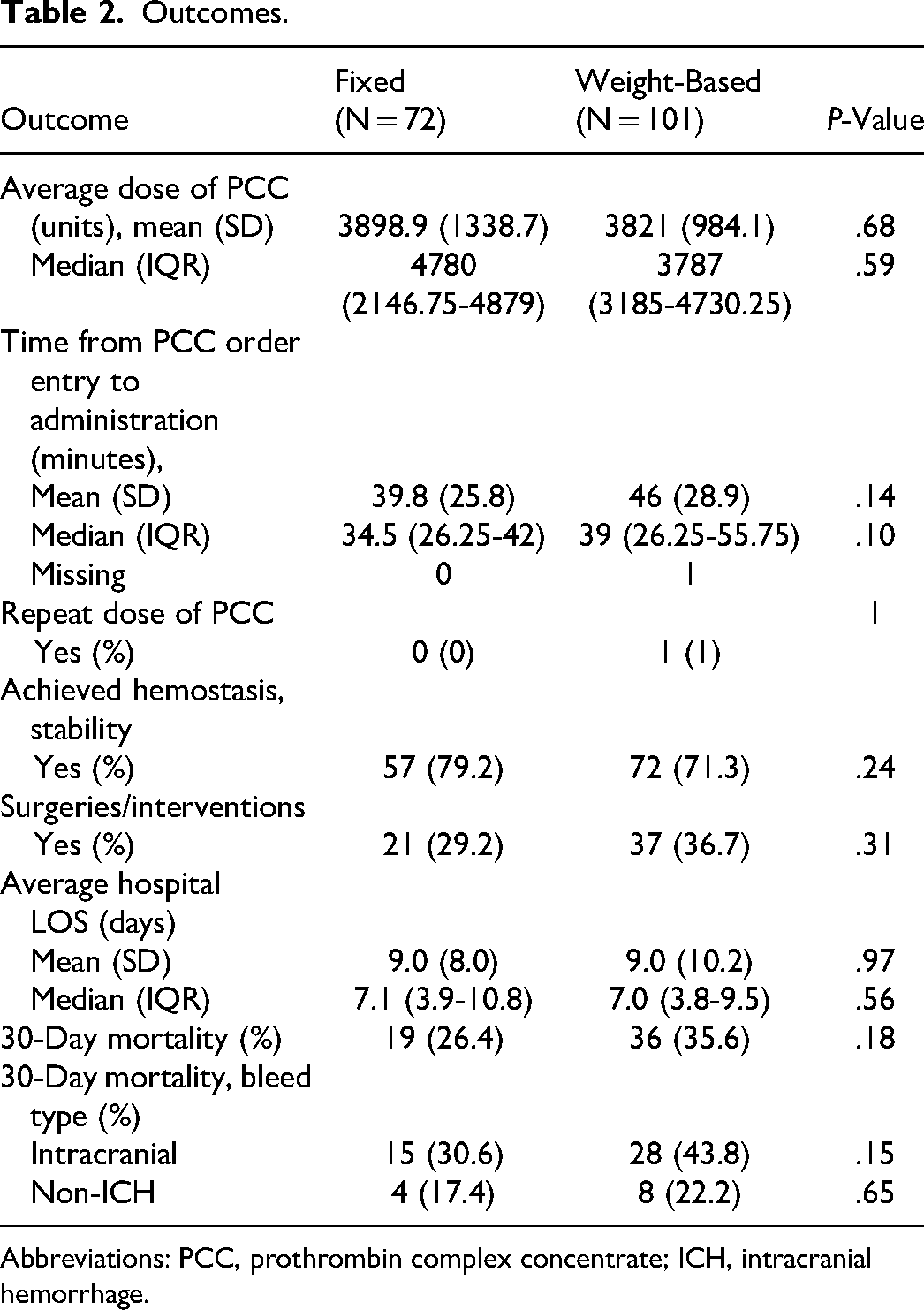
Abbreviations: PCC, prothrombin complex concentrate; ICH, intracranial hemorrhage.
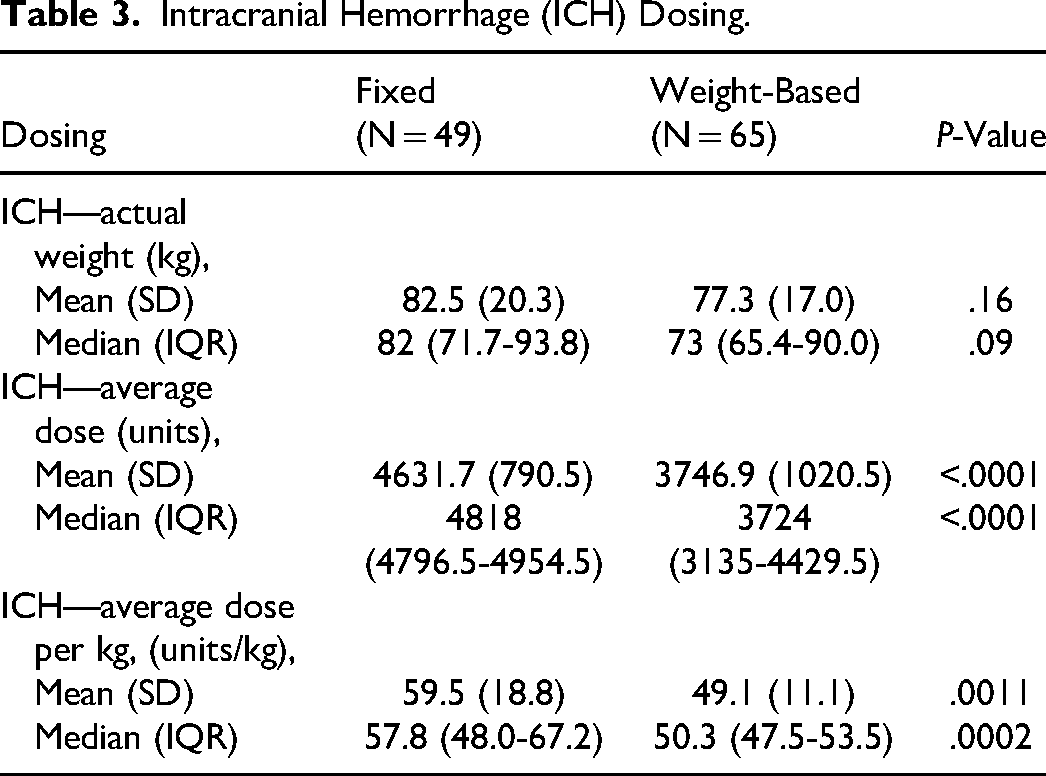
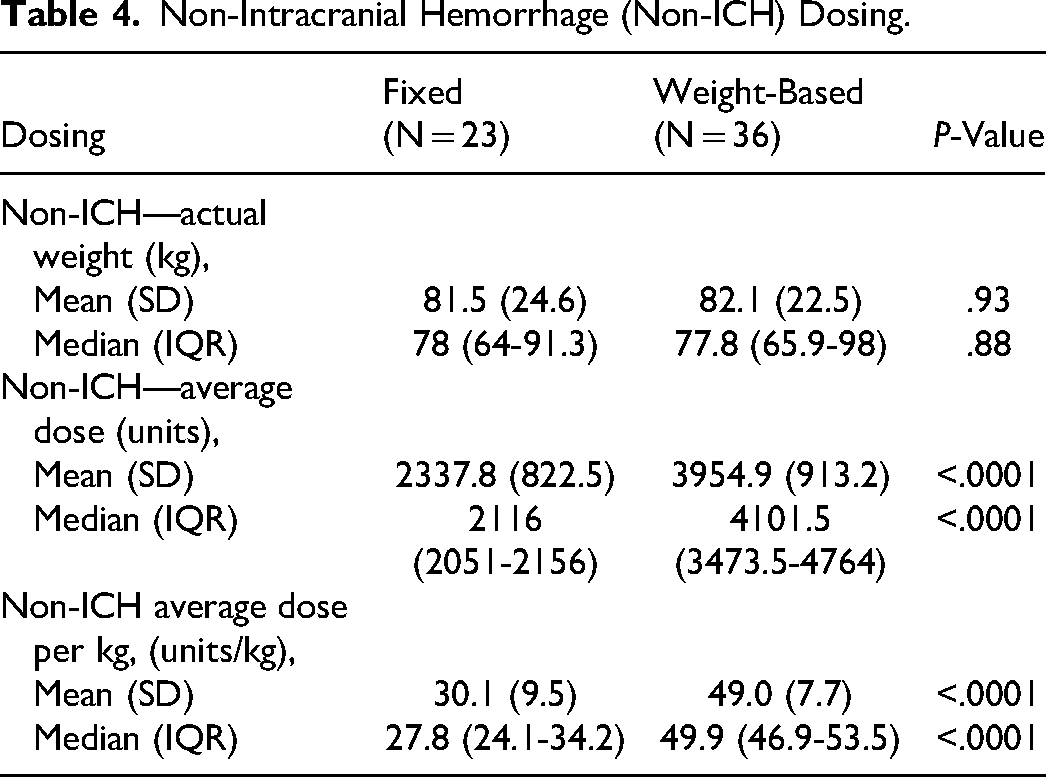
Demographics and dosing were separated by type of bleed (Tables 3 and 4). For ICH, the median dose was higher in the fixed-dose group (4818 units, IQR = 4796.5-4954.5 vs 3724 units, IQR = 3135-4429.5, P < .0001). For non-ICH, the median weight-based dose was higher (2116 units, IQR = 2051-2156 vs 4101.5 units, IQR = 3473.5-4764, P < .0001).
Intracranial Hemorrhage (ICH) Dosing.
Non-Intracranial Hemorrhage (Non-ICH) Dosing.
With respect to safety, nonthrombotic adverse events were absent for both dosing strategies. Three total cases of VTE were documented, 2 in the fixed-dose and 1 in the weight-based group (Table 5).
Disposition and Safety.
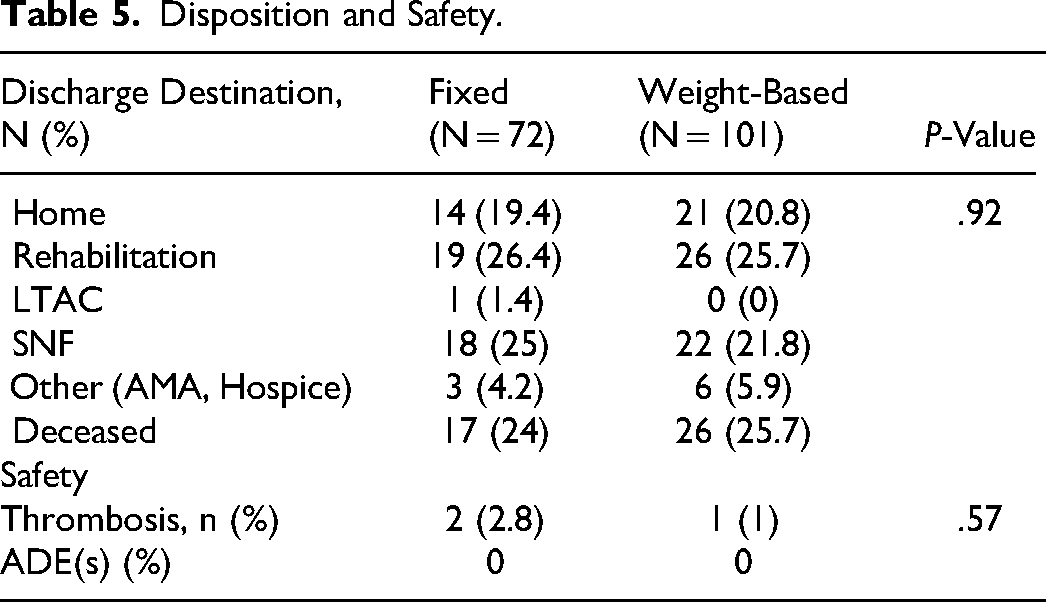
Main dispositions assessed included home healthcare, rehabilitation, assisted living, and hospice. The most common disposition after receiving PCC was rehabilitation, followed by skilled nursing care.
Discussion
To date, this is one of the first studies to compare weight-based versus fixed-dose methods of PCC for the reversal of FXaI effects. A related meta-analysis reviewed 25 studies that assessed fixed and variable PCC dosing strategies. 8 The meta-analysis found no significant differences in hemostatic effectiveness, hromboembolic events, or mortality rates between the dosing strategies. Upon further review of the individual studies, there was less delineation between true fixed-dose and weight-based dosing strategies. Fixed doses were set at 1500-2000 units or between 25 and 50 units/kg for ICH and major bleeds. Therefore, our study maintains novelty through the evaluation of the 5000 unit dose for ICH, which is the maximum dose allowed by the package insert.
In our study, the median time from PCC order entry to administration was not statistically different between fixed-dose and weight-based dosing strategies. This finding is in contrast to the 22-min reduction in time to administration found after implementation of a fixed-dose strategy for the reversal of VKAs. 5 One possible explanation is the nearly 24-h pharmacist coverage in the emergency department of our study site. This is a more complete coverage model than other local sites. More complete coverage enables pharmacists to assist with dose calculations, order entry, and communication with the central pharmacy. Collectively, these efforts expedite medication delivery to the bedside irrespective of the dosing strategy. Another explanation is that the weight-based dosing for FXaI-related hemorrhage is not lab-dependent as is dosing for VKAs. At the study site, the average wait time for routine labs is 1 to 2 hours. Therefore, it is possible that the larger difference previously observed for VKA reversal was due to delays in finalizing international normalized ratio. An additional explanation is that patients with ICH received a higher median dose of PCC (4818 vs 3724 units), which may have increased the time required to dissolve and mix the medication.
The fixed-dose mortality rate in our evaluation was 26.4%, which is consistent with the previous meta-analysis of weight-based PCC for ICH reversal. 9 While we did not find statistically significant differences between the 2 dosing strategies in relation to outcomes such as hemostasis and mortality, that does not rule out the possibility that a difference may exist between the 2 groups which were underpowered to detect.
We identified that the average dose varied based on type of bleed with the average fixed dose for ICH significantly higher than the weight-based dose while the reverse is true for non-ICH. Recalling that there were no significant differences in outcomes for ICH, there is a potential for major cost savings in this group. The weight-based method used approximately 1100 units less PCC for ICH and produced similar outcomes. One could argue that reducing the recommended fixed dose for ICH from 5000 units to 3500 or 4000 units could yield significant annual cost savings. For similar reasons, using the fixed dose method in non-ICH method could represent further cost savings.
Further research is needed to identify the optimal dose to balance safety, efficacy, and cost.
There were 3 total VTE events within 30 days of PCC administration. The singular case of thrombosis in the weight-based group was diagnosed in a patient with a past medical history of chronic VTE. Anticoagulation was held from the time of PCC to the diagnosis date of the unilateral VTE, totaling 14 days. Although this timeline suggests PCC was a contributing factor to the thrombosis, the patient was already taking rivaroxaban for a chronic deep vein thrombosis prior to PCC. To further support the time of onset, the ultrasound read suggested that the appearance was consistent with a nonocclusive and potentially chronic post-thrombotic change. In the fixed-dose group, 1 case of VTE was diagnosed as age-indeterminate prior to PCC and persisted after PCC. The second case of VTE in the fixed-dose group was reported as a chronic occlusion 6 days prior to PCC. Factoring in these pre-existing diagnoses, there was seemingly 1 true case of VTE associated with weight-based PCC and this was weak correlation. The researchers do not postulate that either dosing strategy increases VTE risk because both were far below the 5% estimated average risk associated with PCC. 10 Ultimately, the consensus of the researchers is that both PCC dosing methods are safe.
Limitations
Our study is also limited by design: this was a retrospective cohort conducted at a single site, which may limit the external validity. The retrospective design also limited the ability to consistently assess hemostatic efficacy. While every effort was made to review all radiologic documentation and physician assessment of bleeding, repeat imaging and timing of that imaging were at the discretion of the providing team.
The possibility of a type II error cannot be excluded, as we did not perform a power calculation.
The conclusions drawn from our study may be affected by a limited ability to control confounding. We did not control or adjust for any variables, including medical resident involvement with order placement. Previous studies have found that physicians with lower levels of experience are more likely to make a prescribing error.11,12 If a prescribing error is made, it may take pharmacists longer to verify the order. If the distribution of orders from medical residents differed between the weight-based and fixed dosing groups then that could have impacted our primary outcome. Similarly, there may have been other confounders that could have affected our secondary outcomes of death and hemostasis such as ICH subtype, subjectivity associated with imaging interpretations, and additional blood products administered which we did not control for and which could affect our findings. An additional source of error could arise as physicians were able to choose if patients received the fixed or weight-based regimen after April 25, 2022. This could have led to a selection bias where patients were more likely to receive one treatment or another based on various characteristics. Weight-based doses were used in a total of 17 patients during the fixed-dose era. This amount is not insignificant, making up 16.8% of the weight-based group and taking away 19.0% of the subjects in the fixed-dose group. If those characteristics were also related to the outcomes, this could have affected our findings.
Strengths
Despite the limitations, our study had several strengths, including assessing safety outcomes similarly to prior studies. 13 Additionally, conducting the study at a single site with a consistent group of experienced pharmacists verifying orders, minimized the potential variability in the primary outcome of verification time. Further, the data collection was guided by a protocol and carried out by a single pharmacist which minimized the risk of variability in how data were collected and coded. The timeframes used before and after the implementation of the fixed dosing protocol were consistent which could ameliorate any potential seasonal effects. While there were missing data, the amount was generally low with only 1.2% of subjects having partially missing baseline characteristics and 0.6% of subjects missing from the outcome data. Given the low numbers, it is unlikely that missing data could change the conclusions.
Conclusions
We were unable to detect a significant difference in hemostasis, mortality, or time to administration using either method of PCC dosing. Both methods were safe with a low risk of PCC-associated VTE. Both the fixed and weight-based PCC dosing methods may be used for the reversal of FXaI effects, but the comparative cost, safety, and efficacy should be further evaluated.
Footnotes
Authors's note
Micheal Gionfriddo is also affiliated with Mayo Clinic, Knowledge and Evaluation Research Unit, Rochester, NY, USA.
Declaration of Conflicting Interests
One of the authors, Taylor J Miller, is on the Speakers Bureau for rivaroxaban. All other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
IRB Approval
The study was approved by the University of Pittsburgh Institutional Review Board.
Presentation History
The study was presented at the University of Pittsburgh's Resident and Fellow Research Symposium on June 1, 2023.
Accessing Research Materials
Underlying research materials related to our paper can be accessed by contacting the corresponding author, Brett R. Snyder. The corresponding author has access to these records through the primary study site.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
