Abstract
The objective of this survey was to gain a real-world perspective on coagulation testing by evaluating the availability of various coagulation laboratory tests, assessing specific analytic and postanalytic steps in clinical laboratories in Korea.
Participants were surveyed using a 65-question questionnaire specifically focused on their coagulation testing practices related to prothrombin time (PT), activated partial thromboplastin time (aPTT), plasma-mixing studies, lupus anticoagulant (LA) tests, platelet function tests, coagulation factor assays, and the composition of hemostasis and thrombosis test panels. The survey was performed between July and September 2022.
The survey achieved a 77.9% (81 of 104) response rate. PT or aPTT tests were performed directly at all participating institutions, followed by D-dimer and fibrinogen tests, platelet function test, and plasma-mixing studies in order of frequency. Variations existed in the performance of mixing test and LA assessment. Patterns of coagulating testing differed depending on the size of the hospital. The survey revealed that most laboratories conducted coagulation tests following the international guidelines such as Clinical Laboratory Standards Institute guidelines and the Korean Laboratory Certification system. However, some coagulation tests, including mixing test and LA tests, are yet to be standardized in Korea.
Continuous education on coagulation test methods and internal and external quality control are required to encourage laboratories to enhance the performance of coagulation testing.
Keywords
Introduction
Coagulation, fibrinolytic, and anticoagulation protein systems, as well as the endothelium lining the walls of vessels, are in equilibrium to ensure adequate hemostasis; bleeding disorders occur when any of these variables are disrupted. A flaw or deficiency in a plasma protein, a defect in platelet quantity or function, or a defect in platelet-to-vessel wall adhesive contacts are usually the underlying causes of bleeding disorders.1,2 Thrombosis is a disease in which blood clots occur due to abnormal blood coagulation. Both genetic and acquired deficiencies, as well as abnormalities in regulatory factors, increase the risk of thrombosis. 3 These bleeding and thrombotic disorders are associated with significant morbidity and mortality and are major public health concerns.4,5
Laboratory testing of the coagulation process is utilized to diagnose most of the hemorrhagic and thrombotic disorders.6,7 Many coagulation tests exist, and due to the wide variations in methodologies and reagents, standardizations have been an issue for coagulation testing. Guidelines have been proposed for preanalytical, analytical, and postanalytical factors for coagulation tests by many institutes such as Clinical & Laboratory Standards Institute (CLSI), 8 International Society on Thrombosis and Haemostasis (ISTH), 9 International Council for Standardization in Haematology,10,11 and British Committee for Standards in Haematology. 7 Ensuring the accuracy of test results is of utmost importance. Despite the efforts, studies have shown variability in coagulation testing in different laboratories in many countries.6,12–15 There are no recent surveys assessing the overview of the real-world situation of coagulation testing on hemostasis and thrombosis. In Korea, clinical laboratories undergo the “Outstanding Laboratory Accreditation” by the Laboratory Medicine Foundation and participate in the proficiency testing program conducted by the Korean Association of External Quality Assessment Service, which is both mandatory for clinical testing. The Diagnostic Hematology Standardization Committee, organized by the Korean Society for Laboratory Hematology (KSLH), aimed to assess the current status of coagulation testing in Korea. This paper presents the outcomes of a comprehensive survey evaluating the availability of various coagulation laboratory tests, assessing specific analytic and postanalytic steps during the testing process. The survey aims to provide insights into regional variations, identify areas for improvement, and highlight gaps in knowledge or areas with conflicting practices within coagulating testing.
Materials and Methods
The initial phase involved identifying the survey subjects who were performing coagulation tests. This was followed by questionnaire design and execution of an extensive online survey.
To establish a list of survey subjects performing coagulation tests, we selected institutions where a laboratory medicine specialist, who is also a member of KSLH, is employed. If more than 1 specialist was employed at a single institution, duplicates were removed. Additionally, referral laboratories were excluded as they do not reflect the general situation of regular laboratories. Among these, 104 institutions, specifically secondary hospitals and above, were included. In Korea, secondary hospitals accommodate over 30 beds, whereas tertiary hospitals have more than 500 beds.
A questionnaire comprising 65 questions that were subdivided into 7 sections was created. The sections were as follows: (1) hospital size, number and items of hemostasis, and thrombosis tests performed; (2) prothrombin time (PT) and activated partial thromboplastin time (aPTT); (3) plasma-mixing studies; (4) lupus coagulation test; (5) platelet function test; (6) coagulation factor assay; and (7) the composition of hemostasis and thrombosis test panels. Detailed instructions for answering the questionnaire were provided by each section. We also marked the items where multiple responses were allowed with the notation “multiple choice allowed.”
A list containing 104 laboratories located in general hospitals was collated, and the questionnaire was sent to the email addresses of the facilities. The questionnaire was distributed, and the survey data were collected between July and September 2022. First reminder was sent to each participant 18 days after mailing the survey. Second and third reminders were sent to all non-responders after 10 days each. Following up, we contacted individual participants who had not yet responded, using email or phone, to encourage their participation in the survey.
All collected data were processed in strict adherence to data confidentiality protocols. This study was approved by the Institutional Review Board (IRB) of Soonchunhyang University Seoul Hospital (SCHUH 2022-07-023).
Statistical Analysis
Data were collected through enumeration for the analysis. The findings were displayed in terms of relative frequencies (percentages) alongside the numbers of the total participant count. The analysis of the data was conducted with the Microsoft Excel 2016 software (Redmond, Washington, USA: Microsoft, 2016 Computer Software).
Results
Institution Size and Performance of Coagulation Laboratory Tests
Out of 104 laboratories, 81 completed the survey, and the response rate was 77.9%. Approximately 61.7% (50 of 81) of all the respondents were institutions with ≥ 501 beds and ≤ 1000 beds (Supplementary Table 1), while 21% (17of 81) of the institutions had ≤ 500 beds, and 14.8% (12 of 81) had > 1000 beds. A total of 39.5% (32 of 81) of the survey respondents performed 5001–10 000 PT tests monthly. Regarding aPTT, 38.3% (31 of 81) of the institutions that participated in the survey conducted 5001–10 000 tests monthly.
PT or aPTT tests were performed directly at all the institutions (100%), followed in order of frequency by D-dimer (97.5%), fibrinogen (95.1%), platelet function test including platelet drug response assay (95.1%), and plasma-mixing study (87.7%). On the other hand, diagnostic tests for thrombotic diseases, such as protein S and C activity and factor assays, were usually outsourced by most laboratories (86.4%, 84.0%, and 81.5%, respectively). Except for essential tests required for the outstanding laboratory accreditation program in Korea, such as PT, aPTT, D-dimer, platelet function test, and fibrinogen, special coagulation tests were performed at large institutions with > 500 beds (Figure 1).
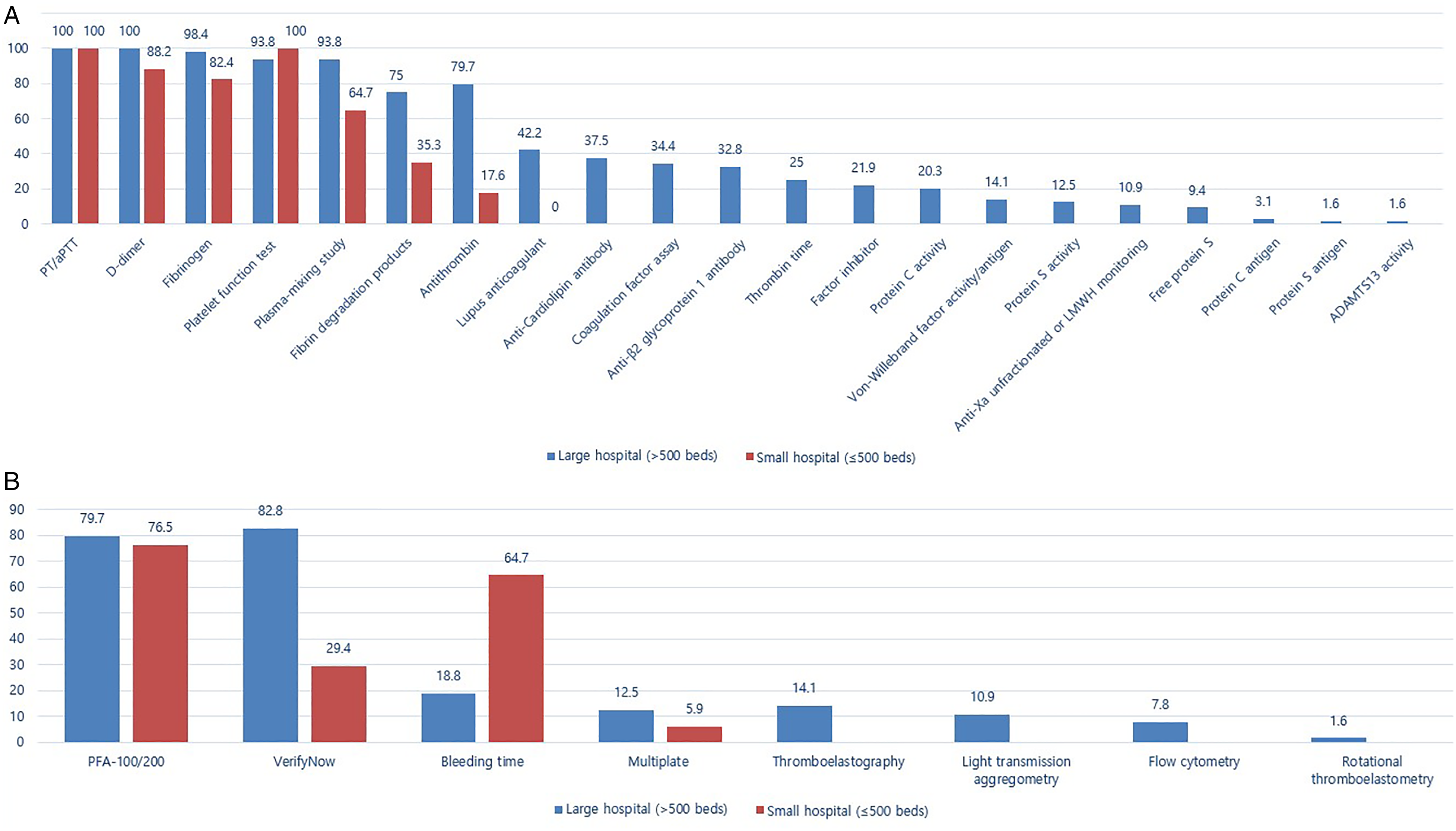
Comparison of the frequency of coagulation tests in large (<500 beds) versus small hospitals (≤500 beds). The numbers on the upper side of the bars indicate the percentage of the institutions performing those tests. (A) All coagulation tests, (B) Platelet function tests according to the test methods.
Testing Practices for PT and aPTT
In total, 96.3% (78 of 81) of the participating institutions reported PT results in seconds, percent, and international normalized ratio (INR) (Supplementary Table 2). Approximately 88.9% (72 of 81) of the institutions in this survey reset the mean normal PT (MNPT) when changing the reagent type, lot number, or instrument. Approximately 88.9% (64 of 72) of the institutions reported using 20 samples for the MNPT calculation. In total, 95.9% (69 of 72) of the used samples were obtained from individuals with normal PT results who had undergone a health checkup; however, 2 institutions used commercialized reference materials, and 1 used the manufacturer's MNPT results.
To check the correctness of INR calculations, 82.7% (67 of 81) of the institutions used excel formulas. Additionally, 77.8% (63 of 81) of the institutions verified the INR reference values by ensuring that at least 90% of samples from 20 healthy individuals fell within the INR reference range.
Approximately 72.8% (59 of 81) of the institutions responded that the aPTT-based heparin therapeutic range was established; however, 54.2% (32 of 59) of these laboratories used the ex vivo method recommended by the CLSI guideline. 16 Anti-factor Xa assays for heparin or low molecular weight heparin (LMWH) monitoring were performed in 16% (13 of 81) of the institutions. Furthermore, 96.3% (78 of 81) of the institutions complied with the requirement of regularly evaluating platelet counts in platelet-poor plasma used for coagulation testing at least once a year as part of the Excellent laboratory certification.
Testing Practices for Plasma-Mixing Studies
Approximately 87.7% (71 of 81) of the institutions performed mixing studies (Supplementary Table 3): 55% (39 of 71) performed 1–30 tests annually, excluding quality control; 18.3% (13 of 71) conducted > 100 tests annually; and 70.4% (50 of 71) conducted incubation tests in addition to the mixing studies to confirm that a factor inhibitor was present. The incubation temperature was 36.5°C to 37°C at 98% (49 of 50) of the institutions, and the incubation time was 1 h at 60% (30 of 50) of the institutions and 2 h at 30% (15 of 50). Approximately 53.5% (38 of 71) of the institutions used in-house normal plasma for mixing tests, whereas 46.5% (33 of 71) used commercialized plasma. To generate a normal pooled plasma (NPP), 60.5% (23 of 38) of the institutions used 20 normal specimens in accordance with the Outstanding Laboratory Accreditation standards/checklists. The selection criteria for normal specimens were diverse; most of the institutions chose normal samples that fell within the PT and aPTT reference ranges, while some verified the results of the coagulation factor assay. In addition, some institutions reviewed the patient's history of receiving anticoagulants and the presence of coagulation disorders.
Mixing studies were performed at 71.8% (51 of 71) of the institutions using the 1:1 ratio of patient specimen to normal plasma, while 25.3% (18 of 71) used mixing ratios of 1:4 and 4:1. When reporting the results of mixing studies, 78.9% (56 of 71) of the institutions included a text-format report with mixing results and expert opinion on PT or aPTT normalization or correction, while 12.7% (9 of 71) reported only the results of mixing studies. Regarding the criteria used for determining whether the patient's PT or aPTT results were corrected or not after mixing NPP, 45.1% (32 of 71) of the institutions confirmed that PT or aPTT results fell within the reference range after mixing with normal plasma; 19.7% (14 of 71) used the percentage correction method (Chang method); and 12.7% (9 of 71) used the index of circulation anticoagulant (Rosner index) calculation formulas. A total of 8.5% (6 of 71) of the institutions responded that they used all 3 methods mentioned in the survey. Regarding the specimens with non-corrected mixing results, implying the presence of anticoagulants in plasma, 88.7% (63 of 71) of the institutions responded that they added a comment indicating that “the presence of anticoagulant drugs such as heparin or direct thrombin inhibitors, cannot be excluded.” Only 1 institution (1.4%) performed thrombin time measurement to detect the presence of a thrombin inhibitor, while 9.8% (7 of 71) neither performed additional tests nor added comments.
Testing Practices for Lupus Anticoagulant
Approximately 33.3% (27 of 81) of the institutions performed lupus anticoagulant (LA) tests (Supplementary Table 4); 59.3% (16 of 27) of which had 501–1000 beds, while the others had > 1000 beds. In order of frequency, the most commonly used methods for LA testing were dilute Russell's viper venom time (DRVVT), followed by both DRVVT and aPTT, and aPTT only. Approximately 51.9% (14 of 27) of the institutions indicated that they would perform a confirmatory test for LA if the screening test results were positive, while 37% (10 of 27) answered that they would perform a confirmatory test in all cases. Regarding the LA test, 74.1% of the institutions indicated that the mixing test was important; however, although the order of screening, mixing, and confirmatory tests varied among the institutions, the screen-confirm-mix was the most used at 25.9% (7 of 27). Regarding whether tests were performed after checking anticoagulants, 81.5% (22 of 27) of the institutions indicated that they would conduct tests regardless of drug administration, while 11.1% (3 of 27) mentioned that they would recommend testing after discontinuing anticoagulants for a while. None of the institutions checked the concentration of unfractionated heparin (UFH) or LMWH before performing the LA test.
Approximately 66.7% (18 of 27) of the institutions used commercial NPP for normalization, while the rest manufactured it themselves; only 1 institution used both. A total of 12 institutions answered the question, “how many donors are needed for normalization?” and most of them (66.7%, 8 of 12) answered that at least 20 donors were needed. Regarding the cutoff value, 51.9% (14 of 27) of the institutions responded that they used the cutoff value provided by the manufacturer; however, some of the institutions (33.3%, 9 of 27) replied that they used the 97.5th percentile. When calculating the cutoff value, 29.6% (8.27) of the institutions required plasma from 20 to 60 donors, followed by requiring > 120 and 60 to 120 donors, in order of frequency. Approximately 51.9% (14 of 27) of the institutions answered that they consider the mixing test to be normalized or corrected by only confirming the normalization of clotting time. Interpretive reporting was reported by 63% (17 of 27) of the institutions. Internal quality control with commercial controls was performed in 85.2% (23 of 27) of the institutions, and most of them performed it every test day, followed by once a week and twice a week in order of frequency. For external quality control, 55.6% (15 of 27) of the institutions conducted comparison tests between laboratories, followed by conducting the College of American Pathologists surveys and Sysmex proficiency test programs in order of frequency. One institution answered that it had not yet implemented external quality control.
Testing Practices for Platelet Function Tests
Platelet function tests were directly performed at 95.1% (77 of 81) of the institutions, most of which used the INNOVANCE PFA-100/200® system (Siemens Healthcare, Germany) and VerifyNow (Accumetrics, San Diego, CA) (Supplementary Table 5). PFA-100/200 was the most commonly used platelet function test regardless of the number of hospital beds; furthermore, except for the VerifyNow, bleeding time, and Multiplate (Roche Diagnostics, Mannheim, Germany), other tests were usually performed at large institutions with > 500 beds (Figure. 1).
In 62.3% (48 of 77) of the institutions, platelet function tests were performed continuously throughout the day, although platelet function tests using light transmission aggregometry (LTA) and flow cytometry were not available at night. Approximately 49.4% (38 of 77) of the institutions offered interpretative reports, for the PFA-100/200, VerifyNow, and LTA tests, in order of frequency. In their interpretation reports of PFA-100/200, approximately 56.3% (36 of 64) of the institutions reported hematocrit value as a factor that could affect PFA closure time, 54.7% (35 of 64) reported medication history, and 51.6% (33 of 64) reported platelet count, while 9.4% (6 of 64) reported ABO blood type. Two institutions reviewed the patient's underlying disease and symptoms related to platelet function when reporting the PFA-100/200 results.
Approximately 80.6% (54 of 67) of the institutions provided a therapeutic range or cutoff values on the VerifyNow and Multiplate analyzers. While performing the VerifyNow P2Y12 test, 55.2% (32 of 58) of the institutions reported both the P2Y12 reaction unit (PRU) level and inhibition rates; 39.6% (23 of 58) of the institutions reported only the PRU level; and 5.2% (3 of 58) of the institutions reported only the inhibition rate. Nine institutions that conducted thromboelastography (Haemonetics Corporation, Braintree, MA) and rotational thromboelastometry (Pentapharm GmbH, Munich, Germany) used various reporting items that differed from an institution to another.
Regarding platelet function tests, 81.8% (63 of 77) of the institutions implemented internal quality control, and 27.3% (21 of 77) implemented external quality control. Quality control was mainly performed for PFA-100/200 and VerifyNow tests.
Testing Practices for Coagulation Factor Assays
Approximately 27.2% (22 of 81) of the institutions performed coagulation factor assays (Supplementary Table 6); factor VIII was performed in-house in all (100%) of the institutions, followed by factor IX, factor V, factor XII, and factor VII, in order of factors directly assayed in the laboratory. Various factor assays were sent to other laboratories. Among them, factor XIII was being referred from the highest number of laboratories to other laboratories (77.3%, 17 of 22). Among the institutions that did not perform all factor assay items within the laboratory, 83.1% (9 of 59) sent all factor assays to another laboratory. Majority (68.2%, 15 of 22) of the institutions who performed coagulation factor assays in-house, verified the manufacturer's reference range, which involved factors VIII, IX, V, VII, and XII, in order of frequency. Regarding internal quality control, all the laboratories (22 of 22) used commercial quality controls; additionally, 1 of the laboratories used quality controls that they had manufactured themselves. Internal quality controls were performed every test day in 90.9% (20 of 22) of the laboratories. Regarding external quality control, 81.8% (18 of 22) of the institutions participated in the Korean Association of External Quality Assessment Service.
Testing Practices for Hemorrhagic and Thrombotic Disease Panels
Overall, 39.5% (32 of 81) of the institutions used hemorrhagic and thrombotic disease panels to validate the convenience of test prescriptions and operated the disseminated intravascular coagulation (DIC), thrombosis, bleeding disorder, and antiphospholipid antibody panels. Approximately 18.8% (6 of 32) of the institutions provided interpretive reports for panels. The DIC panel included D-dimer, PT and aPTT, quantitative fibrinogen degradation product (FDP), antithrombin III, and fibrinogen tests, in order of frequency. The thrombosis panel included D-dimer, PT and aPTT, antithrombin III, protein C activity (functional), protein S (free), and LA (DRVVT), in order of frequency. The bleeding disorder panel included PT, aPTT, PFA (epinephrine), PFA (adenosine diphosphate [ADP]), D-dimer, and fibrinogen tests, in order of frequency. The antiphospholipid antibody panel included LA (DRVVT), cardiolipin antibodies, IgG and IgM, anti-beta 2 glycoprotein I antibody tests, and others. Apart from the panels mentioned above, 6.3% (2 of 32) of the institutions used a von Willebrand Disease panel. This panel included PT, aPTT, factor VIII activity, von Willebrand factor antigen, and von Willebrand factor ristocetin cofactor, or a combination of PT, aPTT, and von Willebrand factor antigen.
Discussion
Through the survey, we have identified variations in coagulation testing aspects performed in clinical laboratories in Korea. This underscores the need for implementation, as well as further harmonization and standardization for coagulation testing.
In this study, PT and aPTT were the most frequently performed coagulation tests, followed by fibrinogen and D-dimer, which was similar to other surveys.6,13,14 Due to its limited sensitivity and low negative predictive value, bleeding time is no longer advised for use in routine preoperative evaluation, 17 but was still being performed in Korea, and the usage was comparable to other international studies other than those in the United States.6,13,14
Most of the institutions that participated in this survey study performed the MNPT calculation and INR verification in accordance with the CLSI guidelines.18–20 Therapeutic aPTT ranges for UFH therapy should be determined at each institution, 16 and in our survey, 72.8% of the institutions offered the heparin therapeutic ranges, and half of the institutions determined the therapeutic range using ex vivo plasma samples. Many institutions used in vitro heparin spiking, contrary to the current recommendations, due to difficulties in obtaining sufficient numbers of heparinized patient specimens, especially in small laboratories. 21 Comparable results were found in surveys conducted in the United States and Canada regarding the establishment of heparin therapeutic ranges12,13; however, fewer institutions in Croatia and Thailand have set up their own ranges.6,14 This demonstrates the importance of laboratory accreditation such as those developed by the College of American Pathologists and Laboratory Medicine Foundation of Korea, which recommends setting therapeutic aPTT range for UFH therapy. On the other hand, only 16% of the institutions in our survey responded to the use of the anti-factor Xa assay for LMWH monitoring, which is very low compared to the surveys from Canada and the United States, possibly since the assay was recently introduced in Korea.12,13
More than 80% of the institutions performed mixing studies, which was a higher score compared to previous surveys. 6 In the current study, most laboratories have chosen either in-house or commercialized plasma as NPP for mixing studies. According to the CLSI guidelines, in-house normal plasma to be used as NPP should include at least 20 normal individuals with > 80 U/dl of all coagulation factors, a platelet count of < 10 × 109/L, and the absence of LA.22–24 The majority of the institutions in our survey examined PT and aPTT results in samples selected for NPP generation. It is recommended to follow the CLSI guidelines or use commercialized plasma for obtaining reliable mixing study results. To detect the presence of factor inhibitors, samples were incubated at 37°C for 1 or 2 h,25,26 and the detection of factor inhibitors was conducted in most laboratories. Several methods can be used to interpret mixing studies; 1 recommendation stated in the CLSI guidelines is to ensure that the coagulation results after mixing are within the normal reference range. 22 Generally, this method can be easily applied and does not require setting cutoff values that need further verification; however, it is effective only for samples with a single factor deficiency. Moreover, samples with slightly prolonged clotting time caused by a weak inhibitor can be falsely corrected owing to inhibitor dilution. 27 Two other commonly used methods for interpreting mixing studies are the percent correction method and the index of circulating anticoagulant (ICA) or Rosner index, which were developed for LA investigations.25,28 The discriminatory power of the percent correction method was improved by testing samples with an additional mixing study of the 4:1 ratio. 29 Since no single interpretation approach has shown 100% sensitivity or specificity for identifying factor deficiencies or anticoagulants, it is crucial for laboratories to establish local cutoff values for the analyzer, normal plasma, and reagent types in use for the applicable formula. 25 Moreover, laboratories should carry out procedures for detecting heparin or other antithrombotic medications that inhibit coagulation if a sample's mixing study results suggest the presence of an inhibitor. Alternatively, non-corrected mixing results can be reported, considering that the effect of inhibitor medications cannot be ruled out.30,31 In this investigation, most institutions added comments regarding the presence of anticoagulant or direct thrombin inhibitor in accordance with the Korean Laboratory Accreditation guidelines.
The 2020 LA/antiphospholipid antibodies guideline proposed by ISTH provide a guidance for performing LA testing, recommending both DRVVT and aPTT-based methods with DRVVT being the initial test. 9 However, our survey results showed that 96.3% (26 of 27) of the institutions perform DRVVT, and only 44.4% (12 of 27) conducted aPTT-based method while the 2019 ISTH Scientific and Standardization Committee survey reported a higher percentage of DRVVT and aPTT using reagent in other countries, 98.9% and 79.7%, respectively. 15 The lower proportion of the institutions performing aPTT-based method may be possibly due to the reimbursement issues for multiple LA tests in Korea. When performing the 3-step process of screening, mixing, and confirming with both tests, if 1 of the tests yields a positive result, it should be interpreted as positive. Regarding the timing of the confirmatory test, the majority of the institutions reported conducting it when the screening test was prolonged, followed by those who perform it on all samples. In the 2019 ISTH survey, while the majority also conducted the test when the screening test was prolonged, there were more institutions that performed it in cases where there was an increase in both screening and mixing tests, compared to those that conducted it on all samples. 15 The ISTH recommends conducting the mixing step (using the screen reagent) and confirmatory test simultaneously after the screening test and suggests that if the confirmatory clotting time is prolonged, the mixing test using the confirm reagent can be performed in the confirmatory step. This approach is useful in cases where factor deficiency is suspected as well as in cases where the use of VKA prolongs the clotting time or when there is excessive LA that exceeds the reagent's phospholipid capacity.9,22,32 According to our survey results, 81.5% (22 of 27) of the institutions proceed with testing in patients undergoing anticoagulant therapy regardless of the treatment. However, the 2019 ISTH survey results show that a lesser percentage, 52.8%, perform LA testing despite on VKA, 33.5% for those on LMWH or UFH, and 70.3% for DOACs. Even among those who deemed the test appropriately, many institutions emphasized the need to check INR or anti-Xa assay results. Notably, about half of the institutions were unsure about DOACs, suggesting a need for providing education on the guidelines on LA testing in patients receiving anticoagulant therapy. 15 For the mixing step in LA test, in-house prepared NPP is preferred; however, commercial lyophilized or frozen NPP can also be used. 9 According to the literature, 9 NPP should involve at least 40 donors; however, only 11.1% (3 of 27) of the institutions participated in this study stated that they prepared NPP using the plasma of at least 40 donors. When setting the cutoff value, it is recommended to calculate the 99th percentile of at least 120 normal samples.9,33 Alternatively, if the manufacturer provides a cutoff value based on a sufficiently large donor population and appropriate statistical modeling, the value can be used after verification. According to the results of this survey, only 51.9% (14 of 27) institutions in Korea routinely verified the manufacturer's cutoff value, emphasizing further education on the use after verification. In the interpretation of normalization/correction in the mixing test, “normalized clotting time only” was the most common at 51.9% (14 of 27), followed by the Rosner index. This was similar to the findings of the 2019 ISTH survey. 15 According to the guideline of 2020 ISTH, it is recommended to include explanations regarding the results and provide information regarding the patient’s history of receiving anticoagulant therapy in the report. 9 Approximately 63.0% (17 of PRU27) of the institutions responded that they provide interpretative reporting.
Platelet function tests are usually performed to screen for platelet aggregation disorders or to determine the effect of antiplatelet medications. Although LTA is considered as the gold standard, only a few large hospital laboratories (>500 beds) performed LTA since it is a complex, labor-intensive, and time-consuming method for sample manipulation; moreover, its results are affected by preanalytical variables, such as hematocrit value, age, platelet count, and operator error.34,35 Since commonly used PFA-100/200 and VerifyNow assays can also be affected by factors such as hematocrit value, platelet count, and specific medications, these limitations should be informed to the clinicians. 35 VerifyNow offers 2 quantitative measurements of clopidogrel resistance: the PRU and the inhibition rate. Many institutions in this survey (55.2%) reported both PRU and inhibition rates. The PRU level was impacted by hematocrit and hemoglobin and the correlation between PRU value and inhibition rate is non-linear; therefore, thromboembolic risk can be determined by a single PRU value, with the inhibition rate providing as an additional parameter. 36 Most studies that used the VerifyNow P2Y12 assay evaluated platelet function based on the PRU value, while only a few used the inhibition rate. 37 The standardization of platelet function tests is an important consideration for the quality of test performance, as the lack of standardization will lead to diagnostic discrepancies between different laboratories. 38 External and internal quality control are critical to ensure the quality of testing practices; however, both are challenging given the theoretical requirements for the production, storage, and shipment of large volumes of stabilized normal and pathologic blood samples. 39 In our study, many institutions reported using internal quality control processes, mainly in PFA-100/200 and VerifyNow. However, only external quality assessment (EQA) was performed in some institutions (27.3%), which was lower than the results in the United States, 40 since the Korean Association of External Quality Assessment program only recently started the EQA for platelet function tests from 2023 whereas CAP have been providing EQA for PFA-100 since 2006.
In this survey regarding factor assays, 68.2% (15 of 22) of the institutions validated the reference ranges provided by the manufacturers according to the CLSI guideline; each laboratory should verify or establish its own reference ranges for each factor assay. 41 Moreover, the established reference range should be reassessed when changes are carried out in instruments or reagents. Verification can be completed by testing 20 samples and investigating outliers, with no > 10% of values falling outside the reference range. When reference ranges are crucial for interpretation, a larger number of samples can be used for verification. According to the survey responses, internal quality control was mostly performed daily; however, according to the CLSI guidelines, it should be performed at least every 8 h and whenever reagents are changed.41 External quality control was performed through Korean Association of External Quality Assessment program and the CAP proficiency testing. Additionally, some institutions conducted comparisons with other laboratories’ results.
This study had some limitations. First, in comparison to small institutions (≤500 beds), large institutions (>500 beds) tended to participate in the survey since large institutions accounted for 80% of the survey respondents. Second, the response rate for subjective questions was lower than that for multiple-choice questions. Finally, the special conditions and expertise of the participating institutions were not taken into consideration, as this research is descriptive in nature regarding coagulation laboratory testing methods.
In our study, the participating institutions performed most of the coagulation tests in accordance with CLSI and ISTH guideline for analytical and postanalytical aspects. In the context of coagulation testing for patients undergoing anticoagulant therapy, certain practices were observed that were not in alignment with established guidelines. The establishment of a domestic consensus on this issue would also be necessary. Our results indicated the need for a standardized evidence-based laboratory testing guidelines for hemorrhagic and thrombotic disorders that would enhance the overall quality of coagulation testing, with the potential to inform the refinement of the guidelines in the field. Continuous education on coagulation test methods and internal and external quality control, including proficiency surveys, are required to encourage laboratories to enhance coagulation testing performance.
Supplemental Material
sj-doc-1-cat-10.1177_10760296241228239 - Supplemental material for Coagulation Testing in Real-World Setting: Insights From a Comprehensive Survey
Supplemental material, sj-doc-1-cat-10.1177_10760296241228239 for Coagulation Testing in Real-World Setting: Insights From a Comprehensive Survey by Hae In Bang, Ja Young Lee, Hyun-Young Kim, Saeam Shin, Myung Hyun Nam, In-Suk Kim, Ji Myung Kim, Jong-Hyun Yoon, Myung-Geun Shin, Sang Mee Hwang and Sun-Young Kong in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Korean Society for Laboratory Hematology.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
