Abstract
Iliac vein stenting for the treatment of iliac vein compression syndrome (IVCS) has been gradually developed. This article investigated the long-term patency and improvement of clinical symptoms after endovascular stenting for iliac vein obstruction patients. From 2020 to 2022, 83 patients at a single institution with IVCS underwent venous stent implantation and were divided into two groups: non-thrombotic IVCS (n = 55) and thrombotic IVCS (n = 28). The main stent-related outcomes include technical success, long-term patency, and thrombotic events. The technical success rate of all stent implantation was 100%. The mean length of hospital stay and cost were higher in the thrombotic IVCS group than in the non-thrombotic ICVS group, as well as the length of diseased vessel segment and the number of stents implanted were higher than in the control non-thrombotic group. The 1-, 2-, and 3-year patency rates were 85.4%, 80% and 66.7% in the thrombosis group, which were lower than 93.6%, 88.7%, and 87.5% in the control group (P = .0135, hazard ratio = 2.644). In addition, patients in both groups had a foreign body sensation after stent implantation, which resolved spontaneously within 1 year after surgery. Overall, there were statistically significant differences in long-term patency rate outcome between patients with thrombotic and non-thrombotic IVCS, the 1-, 2-, and 3-year patency rates in non-thrombotic IVCS patients were higher than those in thrombotic IVCS patients.
Keywords
Introduction
Iliac vein compression syndrome (IVCS), also known as May–Thurner syndrome (MTS), refers to a series of clinical symptoms caused by the out-flow obstruction of the left common iliac vein (CIV) compressed by the right common iliac artery. 1 The incidence of IVCS ranges from 20% to 30% in normal population, but clinically diagnosed IVCS only for 2% to 5%.2,3 Possible reasons are similarities to other chronic venous diseases (CVD), the need for invasive testing, and the concealment of symptoms.
Several retrospective studies and meta-analyses have presented the efficacy and safety about iliofemoral stent implantation, however the long-term patency and clinical improvements were shown little, especially patients with preoperative deep venous thrombosis (DVT).4–6 The outcomes may differ between patients with non-thrombotic IVCS and thrombotic IVCS. There is also a lack of standard methods for evaluating the degree of clinical symptom relief after stent implantation. Some studies have shown that CEAP (clinical-etiology-anatomy-pathophysiology) classification can be used as a quantitative evaluation of clinical severity of chronic deep venous disease of the lower extremities, but there is no clear evidence for the role of CEAP in evaluating the therapeutic effect after endovascular intervention in patients with chronic gold segment venous outflow obstruction. 7 So, there is a lack of reliable prognostic criteria for patients with thrombotic iliac vein occlusion. Whether CEAP can be applied to post-thrombotic syndrome (PTS) also needs further study.
The aim of this study was to report our experience with endovascular intervention to treat IVCS patients, elevating the effectiveness and long-term clinical improvements. Here, we presented the 3-year outcomes of stent implantation for IVCS patients with or without thrombosis obstruction. Furthermore, a multivariate regression model for long-term patency was established to facilitate the evaluation of patients with risk factors in clinical practice.
Methods
Patients
Patients diagnosed with IVCS at the Second Affiliated Hospital of Soochow University from January 2020 to July 2022 and received endovascular iliac vein stenting were recorded. Age, gender, length of stay, cost, clinical class of CEAP classification, previous thrombotic events, numbers of stent implantation, types of stents and other data were collected and analyzed.
Preoperative Assessments and Indications for Endovascular Intervention
Partial stenosis or complete occlusion caused by iliac or femoral vein thrombosis was considered to be thrombotic IVCS in patients who had previously undergone deep venous ultrasound, D-dimer and venous computerized tomography (CT) examination of lower limbs; otherwise, CT or angiography showed that iliac vein stenosis of more than 50% was considered to be non-thrombotic IVCS. During preoperative angiography, patients with obstruction or stenosis of the inferior vena cava were excluded, and no inferior vena cava filter or any thrombolysis catheter was present during surgical treatment. Endovascular iliofemoral intervention was performed mainly in patients with iliac vein stenosis ≥50% and collateral veins open at the stenosis lesion via computerized tomography venography (CTV). Patients with classical symptoms of IVCS including limb edema and pain, varicose, pigmentation, and ulcers, whereas CEAP score over 3 could also be considered requiring the treatment. Especially, the thrombotic IVCS group refers to patients whose thrombus is in the subacute (>30 days) or chronic (>90 days) stage and undergoing directly stenting without thrombolytic therapy during hospitalization.
Endovascular Technique and Postoperative Treatment
All procedures were performed in the digital subtraction angiography (DSA) hybrid operating room under local anesthesia with 5 mL of 1% lidocaine. Ultrasound-guided vascular access was obtained with 6F sheath through the ipsilateral deep vein proximal to the stenosis lesion, commonly through the femoral or popliteal vein. After finding the stenotic segment, occlusions were recanalized using a stiff 0.035″ hydrophilic guidewire in a vertebral catheter. Once the guidewire had passed through the narrow segment, it was first predilated with a small balloon, and then formed with progressive large balloons. The pressure pump was set at 15 atmospheres for 5 minutes. After angiography showed that there was still partial stenosis after balloon angiography, the iliac vein stent was implanted. The upper landing zone was set at most 1 cm above the confluence of the CIV with the inferior vena cava. The lower landing zone was beyond the distal stenosis segment treated. After that, venous angiography was performed from multiple angles to ensure patency of the lumen.
Direct smooth compression was applied after sheath removed. Patients were required to lie flat in bed for at least 12 hours with ankle pump exercises, but without bending the groin puncture site. All patients were discharged on 15 mg of rivaroxaban administered orally once a day to prevent in stent thrombosis. Moreover, patients can improve their clinical symptoms with the help of and oral vasoactive drugs (aescuven forte and diosmin).
Follow-Up
After intervention, patients were followed up as outpatients and patients who failed to attend the visit were contacted by phone. The time was scheduled at 1, 3, and 6 months, and then yearly. Patients were informed of the need for CTV and VCSS elevations, and only a few patients underwent venography.
Statistical Analysis
Continuous variables were presented as mean ± standard deviation (SD) and compared using the t test. Categorical variables were compared using the Chi-squared test. The Kaplan–Meier curve was used to estimate the postoperative stent patency rate of the two groups at different time points during the follow-up period, and the difference was compared by Mantel–Cox test. P values of <.05 were considered a statistically significant difference.
Results
According to the discharge diagnosis, patients were divided into two groups: thrombotic IVCS (n = 28) and non-thrombotic IVCS (n = 55). The mean age of the patients in the thrombotic and non-thrombotic group was 62.9 and 60.9 years, respectively (Table 1). There was also no specific difference in the ratio of male to female and preoperative CEAP classification in both groups (Table 1). Patients in two groups were at C3 and above, mainly concentrated on the C4 classification. We found that the degree of stenosis and the cumulative range of diseased vessels in the thrombotic patients were much greater than those in the non-thrombotic patients. In particular, thrombotic IVCS lesions were characterized by complete occlusion of the outflow tract, and also involve the external iliac vein and common femoral vein (CFV) segments. Due to the long lesion vessels, two to three stents were generally implanted in the thrombotic patients (Table 1). A total of 57.1% of patients underwent stent placement with double stents, while 25% underwent with triple stents in thrombotic groups (Table 1). The length of hospital stay for patients in the thrombotic group was 8.5 ± 3.9 days, which was more than that in the control group. The overall cost of hospitalization was also high in patients with thrombotic IVCS.
Characteristics and Clinical Classification of Patients.

Abbreviations: SD, standard deviation; N, number; IVCS, iliac vein compression syndrome; M, male; yrs, years; CEAP, clinical-etiology-anatomy-pathophysiology; CIV, common iliac vein; EIV, external iliac vein; CFV, common femoral vein.
*P < .05 (Thrombotic vs non-Thrombotic Groups).
The technical success rate was 100% in both groups. No patient experienced pulmonary embolism or wound infection and hematoma due to puncture during the interventional procedure and peri-interventional period. Postoperative complications during this study were presented in Table 2. Our focus during follow-up was on complications related to the procedure, stent, and re-thrombosis. Overall, patients in the thrombosis group experienced a prolonged duration of foreign body sensation compared to those without thrombosis (30 days: 53.6% vs 14.5%; 6 months: 42.3% vs 8%; 1 year: 37.5% vs 6.9%; 2 year: 30% vs 3.3%; 3 year: 41.7% vs 0). This can be attributed to the involvement of CIV, external iliac vein, and femoral vein segments in the diseased vessels of the thrombosis group. The implanted stent spanning across the inguinal region may cause discomfort when bending down or squatting up, which raises concerns about its potential impact on long-term quality of life for patients. There was no significant correlation between back pain and stent foreign body sensation observed. Both groups exhibited more than a 50% incidence of back pain at both postoperative intervals of 30 days and 3 months; however, this gradually subsided by 1 year after surgery (Table 2). The rate of in-stent restenosis was higher in the thrombosis group with early days (<3 months) significantly associated with deep vein thrombosis. In fact, previous studies have indicated that low back pain is a typical symptom following iliac vein stenting possibly due to factors such as oversize stents and intraoperative balloon dilation.5,8
Main Complications During Follow-Up Period.
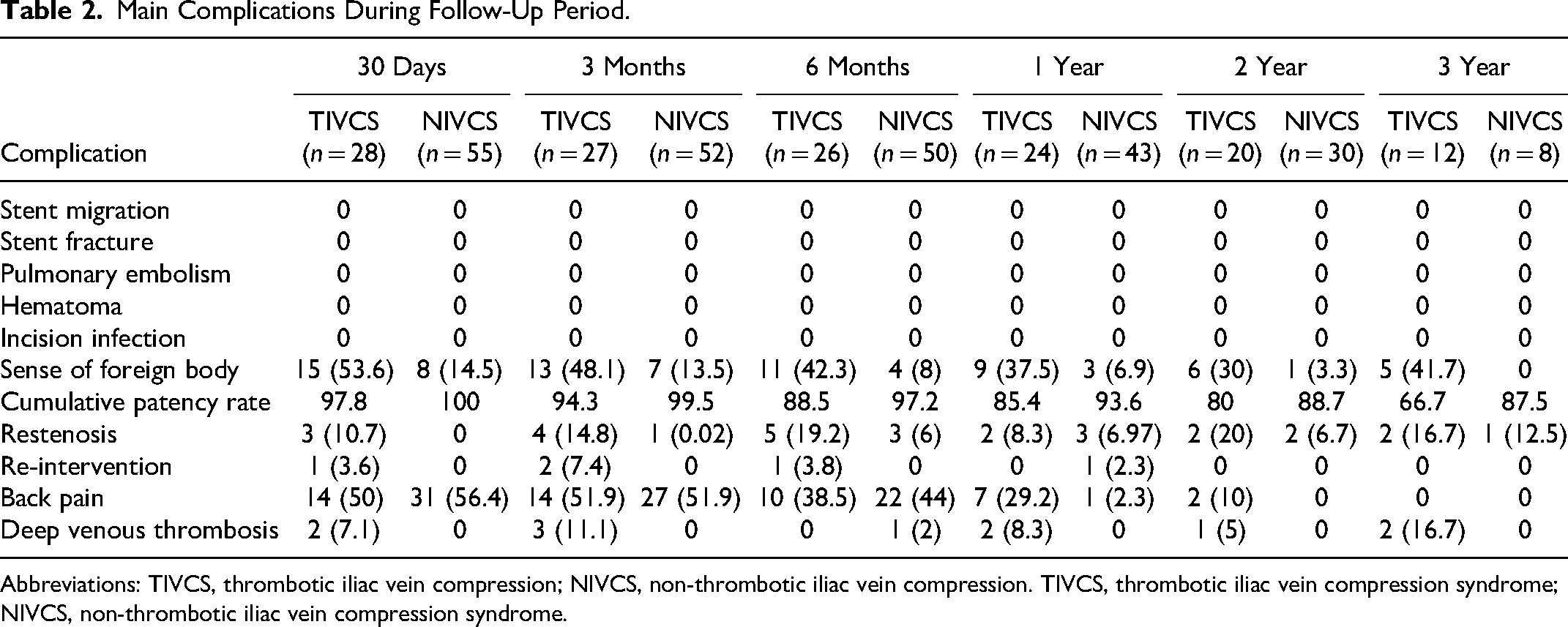
Abbreviations: TIVCS, thrombotic iliac vein compression; NIVCS, non-thrombotic iliac vein compression. TIVCS, thrombotic iliac vein compression syndrome; NIVCS, non-thrombotic iliac vein compression syndrome.
The overall stent patency rate was 72.5% at 3 years follow-up (Figure 1A). The 1-year, 2-year, and 3-year patency rates were 85.4%, 80%, and 66.7%, respectively, in the thrombosis group, and 93.6%, 88.7%, and 87.5%, respectively, in the non-thrombosis group (P = .0135, hazard ratio = 2.644, Figure 1B).
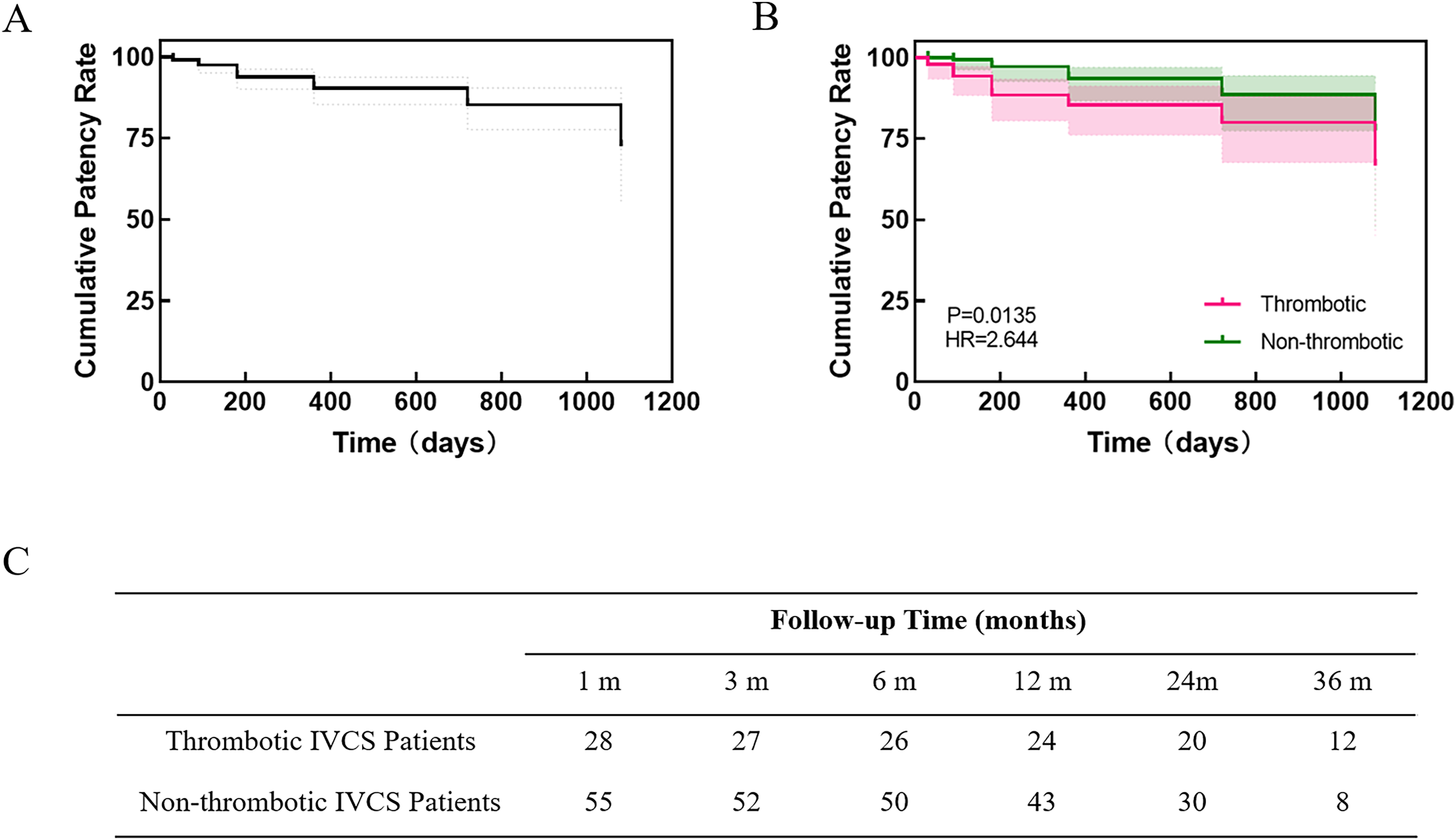
The Kaplan–Meier curve showing the cumulative patency rate. (A) Among all of patients. (B) A comparison between the thrombotic iliac vein compression syndrome and non-thrombotic iliac vein compression syndrome. (C) The follow-up conditions.
Discussion
IVCS was first anatomically defined in 1957 and considered a low incidence. 9 Based on angiography, multidetector computer tomography (CT), and magnetic resonance venography (MRV), the actual incidence of clinically diagnosed IVCS is higher than expected. 1 In patients with DVT, this proportion will reach 18% to 49% and be more common on the left side. 10 With the development of endovascular therapy technology, many endovascular intervention techniques have been tried, especially stent implantation, which can be used to selectively reduce the clinical symptoms of IVCS patients and to restore and maintain venous patency in IVCS patients. 11 A number of studies have confirmed the high clinical success rate and safety of endovascular treatment in IVCS patients by selectively reducing the clinical symptoms and restoring and maintaining venous patency, highlighting the important impact of stent implantation on improving the quality of life of patients with chronic iliac and femoral venous obstructive lesions.
Studies have shown that stent implantation for the treatment of non-thrombotic IVCS has a high stent patency rate, low mortality and low in-stent restenosis rate.12–14 The postoperative patency rate of iliofemoral vein stent in patients with PTS is not satisfactory. A number of clinical studies have shown that the recurrence rate of stent thrombosis in PTS patients within 30 days after stent implantation is 3.4%. 15 Peng et al retrospectively analyzed 489 patients with PTS treated with stent implantation, including stent implantation in the CIV, CFV, and external iliac vein. 5 The average follow-up time was 1.5 to 36.2 months, and the 1-year and 3-year patency rates were 83.36% and 67.98%, respectively. Especially for severe PTS, the incidence of in-stent stenosis is higher.16,17
Studies on the mid- and long-term unfavorable outcomes of stent implantation in PTS are mainly clinical retrospective cohort studies and case-control studies. The literature reports that the factors affecting stent thrombosis after stent implantation in PTS include: iliac vein compression, poor stent position, distal iliofemoral vein or below inguinal ligament, obstructed distal femoral vein inflow, residual thrombus in the stent, and inadequate anticoagulation.18,19 However, there is still no progress in the research of in-stent stenosis after PTS, especially there is no scientific and reasonable experimental study on the pathological changes and biological process of early in-stent thrombosis and mid and long-term in stent restenosis after PTS.
In our study, we found thrombotic IVCS patients showed a worse patency rate, which may be closely related to the length of the lesion, the number of stents implanted, and the difference in clinical manifestations reflected in stent foreign body sensation and long-term back pain.
The analysis result was limited by the small number of patients. Large study groups of patients with or without thrombosis events should be constructed to elevate the long-term outcome of stents.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Anhui Engineering Technology Research Center of Biochemical Pharmaceutical, Bengbu Medical College (grant number 2022SYKFD03).
