Abstract
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, acquired hematologic disorder commonly treated with complement inhibitors such as eculizumab, ravulizumab, and pegcetacoplan. This study aims to describe treatment patterns, healthcare resource utilization, and cost for newly diagnosed PNH patients in 2 large, health insurance claims databases: MarketScan and Optum. Among the 271 patients meeting the inclusion criteria in MarketScan, 57.9% were female, and the average age was 46.6 years. Among these newly diagnosed patients, 25.1% (n = 68) of patients received a PNH-specific pharmacologic treatment, and the average time from diagnosis to treatment was 4.7 months. The medication possession ratio was 97.0%, but discontinuation was common (58.8%). The average per-patient-per-month costs were $18,978, driven by pharmacy and infusion ($11,182), outpatient ($4086), and inpatient ($3318) costs. Despite the availability of multiple treatments, 39.9% of patients had an inpatient stay, and 50.9% had an emergency department visit. Better care management and the introduction of new treatment options are needed to address delays between diagnosis and treatment, and high rates of hospitalization and emergency department use among patients with PNH.
Keywords
Introduction
Paroxysmal nocturnal hemoglobinuria (PNH) is an acquired hematologic disorder that renders affected patients’ red blood cells susceptible to premature destruction (ie, hemolysis). 1 Specifically, PNH develops as a result of somatic mutation of the PIGA gene and proliferation of resulting defective stem cells. These defective stem cells do not form glycosyl phosphatidylinositol (GPI) anchors. In turn, GPI-anchored complement regulatory proteins cannot attach to cell membranes, and the complement system destroys these cells. Complement-induced hemolysis can cause a range of symptoms, including fatigue, headaches, dysphagia, chest and abdominal pain, and erectile dysfunction and impotence in men. Patients may also experience serious complications including thromboses and chronic kidney disease. The economic burden of PNH is high for patients, with transfusion-dependent patients experiencing annualized all-cause hospitalization costs often in excess of $100,000. 2 PNH is a rare disease that typically arises in relatively young adults. Among 4336 patients included in the International PNH Registry, the median age at disease onset was 35.5 years (interquartile range [IQR]: 23.9 53.4). 3 In a recent US analysis, the prevalence of PNH was 13 per 1,000,000 individuals in the US in 2017, and the incidence rate was 5.7 per 1,000,000 person years. 4
Several pharmaceutical treatment options exist to manage the symptoms and slow the progression of PNH. Eculizumab is an intravenous (IV) complement inhibitor that was approved by the US Food and Drug Administration (FDA) in 2007 to reduce hemolysis in patients with PNH. 5 Ravulizumab, another IV complement inhibitor, was approved in 2018 after demonstrating non-inferiority to eculizumab. 6 Pegcetacoplan is a subcutaneous complement inhibitor that was approved in 2021. 7 Patients may receive other treatments based on their symptoms, including anticoagulants, immunosuppressive agents, growth factors, or blood transfusions. 1 Bone marrow transplantation is the only curative option for PNH, though it is reserved for severe cases due to the high risk of morbidity and mortality. 1 Even among PNH patients treated with currently available therapy, suboptimal C5 inhibition or physiological stressors such as surgeries or infections may result in breakthrough hemolysis (BTH). 5 BTH is characterized by the reappearance of classical PNH symptoms and the return of intravascular hemolysis.8–11 A recent cost model determined that BTH caused significant costs for patients, driven by C5 inhibitor dosage adjustments, hospitalizations, and blood transfusions. 12
Currently, there is limited evidence in the literature on the treatment and drug adherence patterns for PNH patients that include ravulizumab. Furthermore, claims-based estimates of utilization among PNH patients have largely been limited by small sample sizes due to the low prevalence of the disease. To better understand the current treatment landscape and clinical burden, this study analyzed novel real-world data, including ravulizumab utilization and adherence, on treatment patterns, HCRU, and costs of newly diagnosed PNH patients.
Materials and Methods
Data Sources
This was a retrospective cohort study utilizing the MarketScan claims database using data from October 1, 2015 to June 30, 2021. MarketScan is a database of medical and pharmacy claims with approximately 43 million covered lives in the most recent full data year. Due to the low incidence of PNH, this analysis was repeated using the US Optum research administrative claims database to help validate whether the findings from the MarketScan data were robust to other data sources. The US Optum claims database contains medical and pharmacy claims and enrollment information for commercial and Medicare Advantage health plan members and is geographically diverse across the United States.
MarketScan has been used recently to study PNH and examine treatment patterns, HCRU, and cost for specific cohorts of patients.4,13 This study updates prior work and expands the scope to include new treatments (eg, ravulizumab, pegcetacoplan) using the same MarketScan data source. In addition, this study also uses the Optum data to explore whether the findings are similar in a different data source.
Study Population
This was a retrospective study of patients newly diagnosed with PNH. Patients had a valid ICD-10 diagnosis code of PNH (D59.9) in any position from April 1, 2016 to December 31, 2020, were aged ≥18 years on the date of diagnosis, and had ≥6 months of continuous enrollment after the diagnosis date. In addition, patients were required to have at least 6 months of continuous enrollment prior to the diagnosis date with no PNH diagnosis.
Outcomes
The study began by looking at treatment selection, medication adherence, and medication switching patterns. For the treatment selection analysis, the rate of treatment and time to treatment were assessed for eculizumab and ravulizumab (no pegcetacoplan utilization was observed given it was approved by the FDA in May 2021), blood transfusion, and bone marrow transplant. The time from diagnosis to treatment was also evaluated. Medication adherence was measured based on 2 metrics: medication possession ratio (MPR) and proportion of days covered (PDC). 14 The time to discontinuation and the share of patients who discontinued therapy were also evaluated. Finally, we measured the share of patients who switched treatments during the observation period as well.
In addition to pharmaceutical treatment patterns, we also examined patient healthcare resource utilization of medical good and services. HCRU was measured for inpatient admissions, emergency department (ED) visits, office visits, and other outpatient visits. All analyses examined both all-cause HCRU (ie, services provided for any reason) and PNH-related HCRU (ie, services likely to be related to care for the patient's PNH). PNH-related HCRU was defined as any claim with an ICD-10 code of D59.5 in any position. For PNH-related HCRU, blood transfusions, bone marrow transplant, iron chelation therapy, and prednisone utilization were also assessed. Thromboembolic events were not specifically captured in the data, but some outpatient, inpatient, and ED visits may include thromboembolic events.
Finally, patient costs were measured for all-causes and PNH-related care. For both categories, we measured the total costs and costs for specific services such as inpatient admissions, ED visits, office visits, other outpatient visits, and pharmacy.
Statistical Analysis
All patients who met the inclusion criteria were included in all analyses, and all claims data available were used. Patient characteristics, including age, gender, region, insurance type, and NCI Comorbidity Index were assessed using descriptive statistics. The number and percent of patients are reported for binary and categorical variables, and the mean, standard deviation, median, and IQR are reported for continuous variables.
For the treatment pattern analysis, the number and percent of patients initiating—and transitioning to other—treatments were reported for each treatment. Mean, standard deviation, median, and IQR were calculated for the time to initiation of treatment and duration of treatment prior to discontinuation. General adherence was measured using MPR and PDC, the 2 most commonly used metrics used when analyzing health insurance claims data. 15 MPR was calculated as the number of days on treatment in the time frame divided by the total number of days in the time frame, where the time frame is the number of days between treatment initiation and discontinuation. PDC was calculated as the number of days covered divided by the number of days in the follow-up period observed in the data. For the switching analysis, the number and percent of patients for each therapy and each line are reported, along with the mean, standard deviation, median, and IQR for the time to switch.
For HCRU, the number and percent of patients with each type of event are reported, and for patients with at least 1 event, the per-patient-per-month (PPPM) number of events is reported with the mean, standard deviation, median, and IQR. The number of hospital days PPPM is also reported for patients with at least 1 inpatient admission in the follow-up period.
Patient costs were measured on a PPPM basis. The mean, standard deviation, median, and IQR were reported for the entire follow-up period.
Sensitivity Analysis
Due to the rarity of PNH, we also evaluated outcomes using Optum claims data. In the Optum data, we identified that the age distribution of newly diagnosed patients was higher than the MarketScan population due to a higher portion of Medicare patients. To make the Optum results more comparable to MarketScan, we age-adjusted the Optum outcomes to be representative of the US population. After reweighting the sample, the comparison of Optum outcomes against MarketScan results was conducted qualitatively.
In addition to the complementary analysis using a second data source (ie, Optum), we also performed a sensitivity analysis with a baseline/washout period of 3 months and a 3-month follow-up period (compared to 6 months in the main analysis). The same outcomes were measured using this larger sample of patients for MarketScan and Optum to assess whether using a shorter washout period to identify newly diagnosed patients affected the findings.
Results
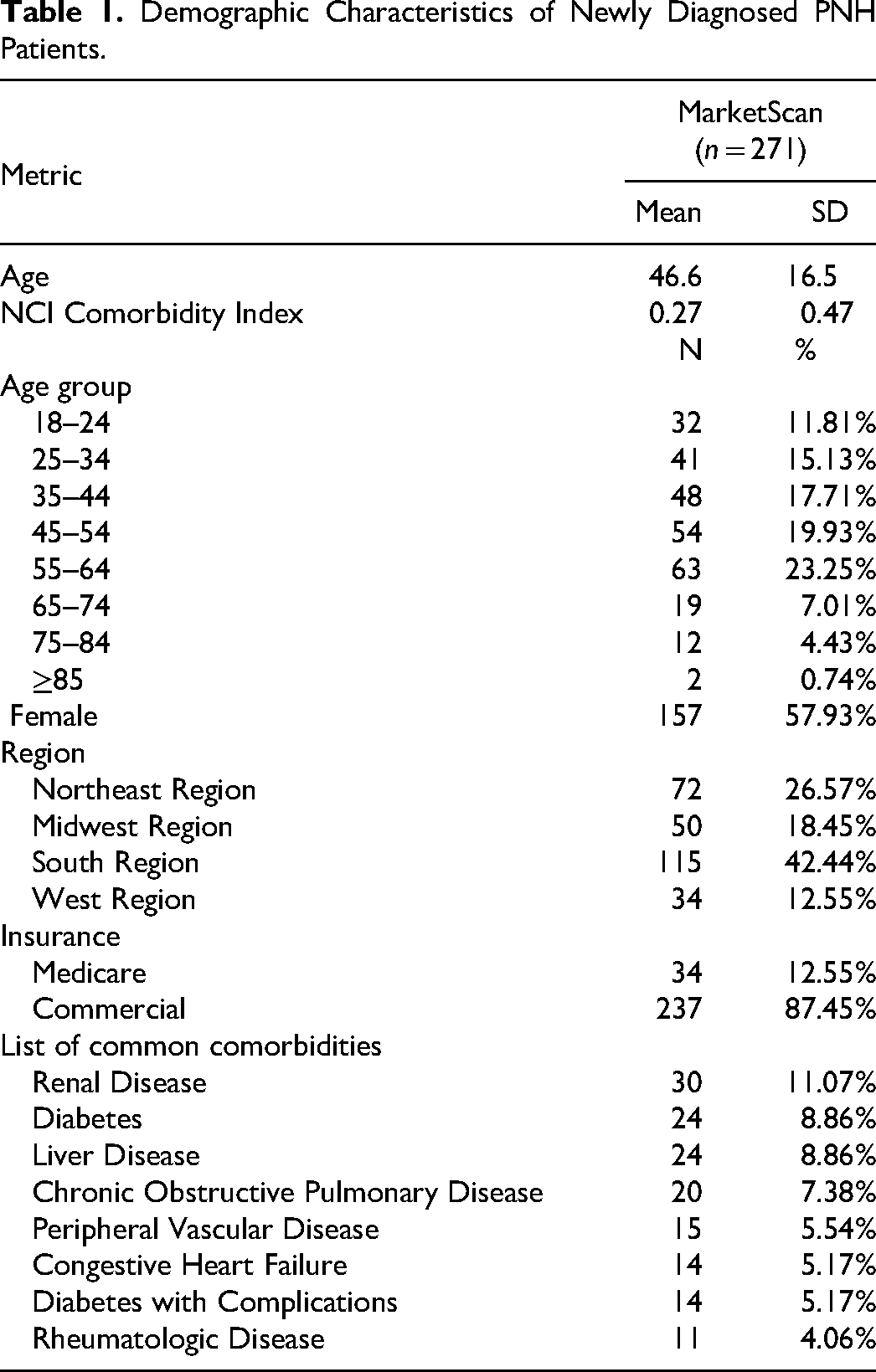
In the MarketScan data, 661 patients were identified as having an ICD-10 diagnosis of PNH (D59.5) between April 1, 2016 and December 31, 2020. After applying the exclusion criteria, 271 patients remained in the dataset for analysis. As shown in Table 1, 57.9% of newly diagnosed patients in the MarketScan data were female, the average age was 46.6, and the average NCI Comorbidity Index was 0.27.
Demographic Characteristics of Newly Diagnosed PNH Patients.
A large share of patients with PNH was not observed to receive any C5 inhibitor treatments. Specifically, 25.1% of patients with PNH (n = 68 out of 271) received any C5 inhibitor pharmacologic treatment (ie, eculizumab or ravulizumab). The average time from initial PNH diagnosis to treatment initiation among those treated was 140.9 days (SD = 307.9 days), with a median time of 48 days. (Table 2)
Treatment Rates and Time to Initiation of Treatment for Newly Diagnosed Patients.
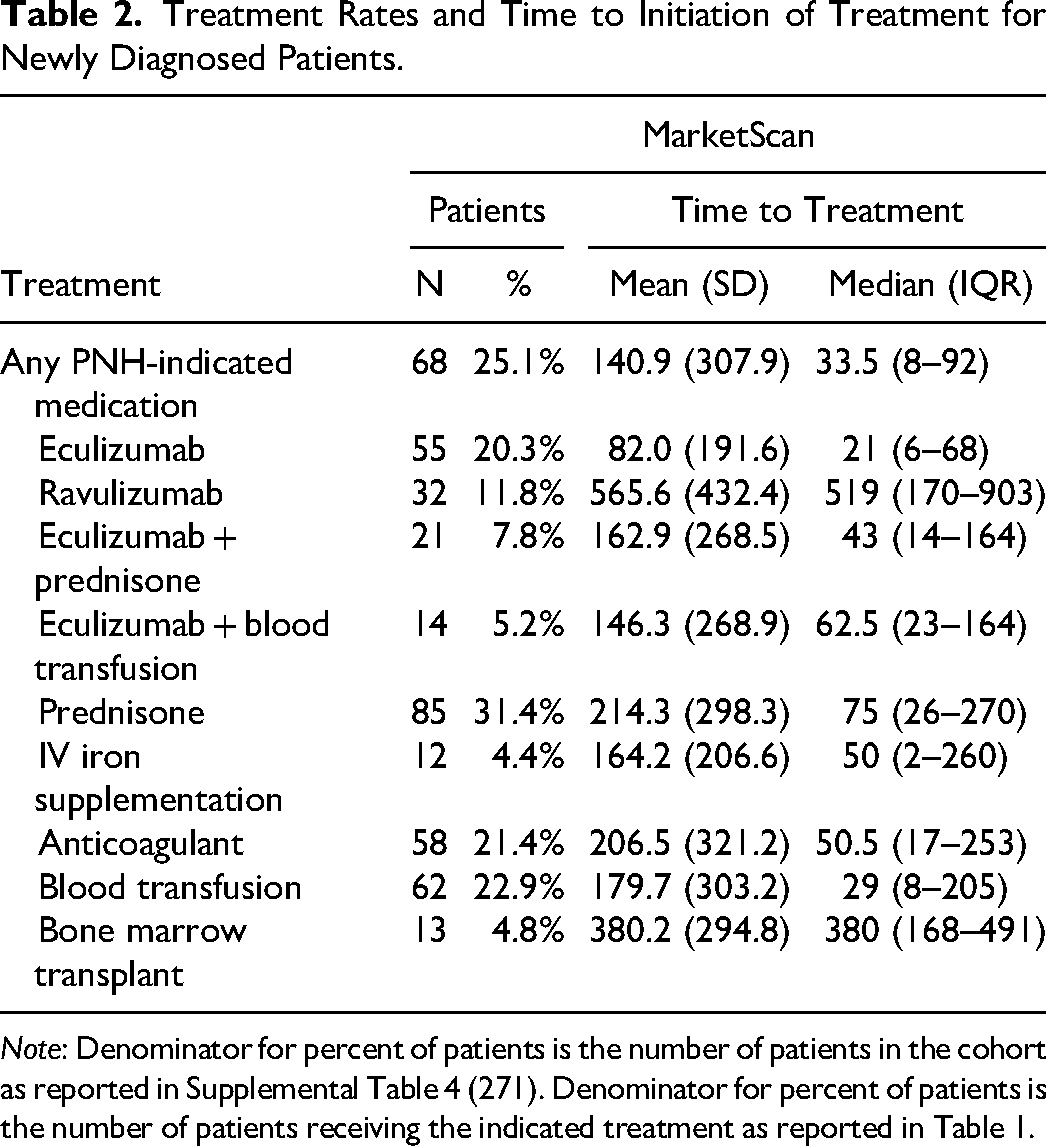
Note: Denominator for percent of patients is the number of patients in the cohort as reported in Supplemental Table 4 (271). Denominator for percent of patients is the number of patients receiving the indicated treatment as reported in Table 1.
Patients who started C5 inhibitor therapy adhered to their treatment longer (average of 346.1 days and 369.4 days) compared to other forms of treatment (Supplemental Table 1). Nevertheless, discontinuation rates remained high, with 63.6% of patients receiving eculizumab discontinuing treatment and 50.0% of patients receiving ravulizumab discontinuing treatment. Of MarketScan patients receiving any treatment (eculizumab or ravulizumab), the MPR was 97% (92.7% had an MPR ≥ 80%) The MPR for eculizumab was 94%, and the MPR for ravulizumab was 99%. Due to high rates of discontinuation, however, PDC-based measures of adherence were much lower. PDC was 60% for patients receiving any treatment (eculizumab or ravulizumab), and only 38.2% of patients had a PDC ≥ 80%. The PDC for eculizumab was 48%, and the PDC for ravulizumab was 45%, due to switching and discontinuation (Supplemental Table 2).
More than 1 in 4 patients treated for PNH changed their C5 inhibitor medication. In the MarketScan sample, for patients who started on eculizumab, 33.3% later switched to ravulizumab, with an average time to switch of 654.9 days (SD = 304 days). Of patients who started on ravulizumab as their first line of treatment, 1 (7.1%) switched to eculizumab. (Figure 1)
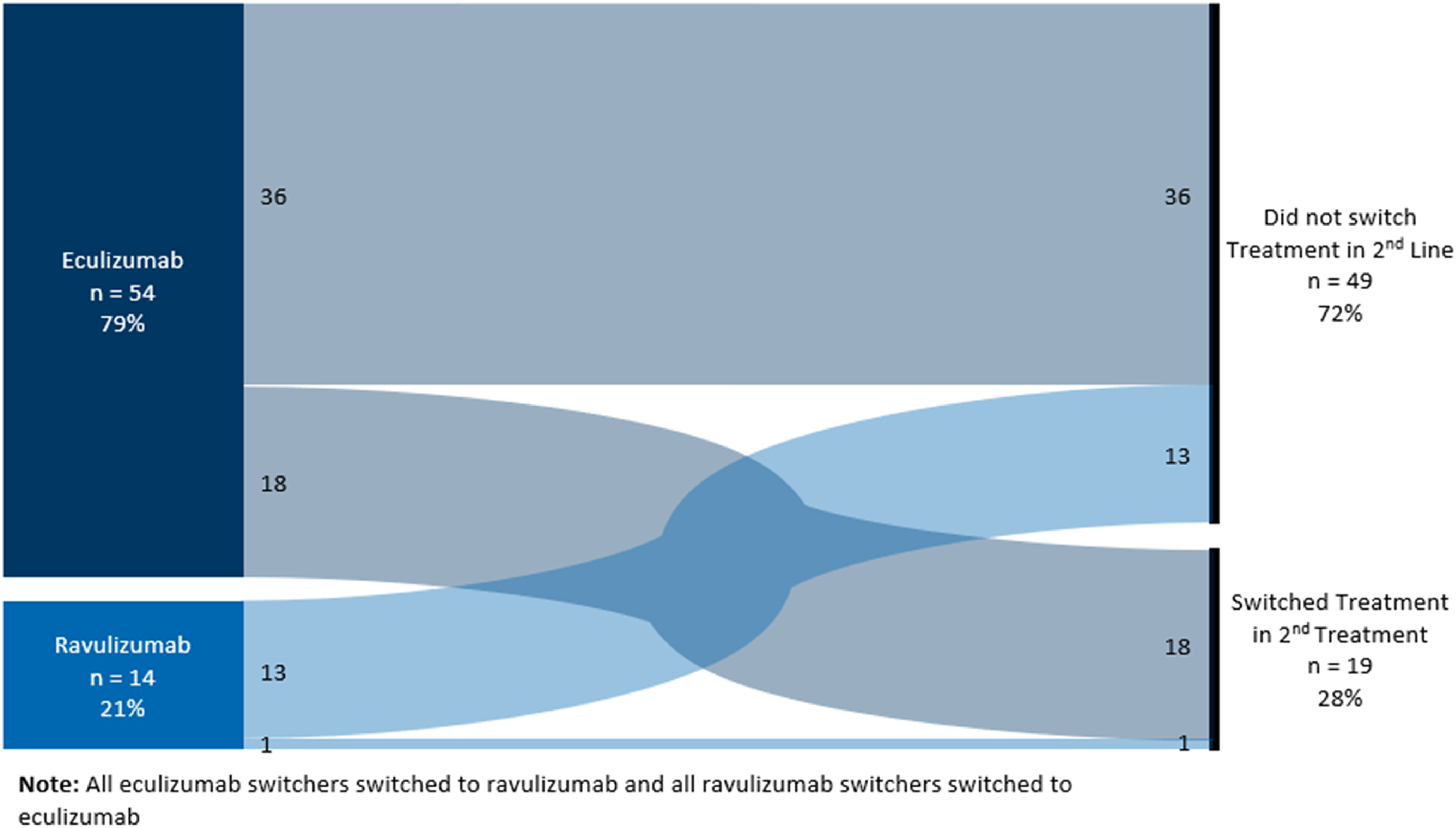
Lines of treatment and treatment switching for newly diagnosed patients, MarketScan.
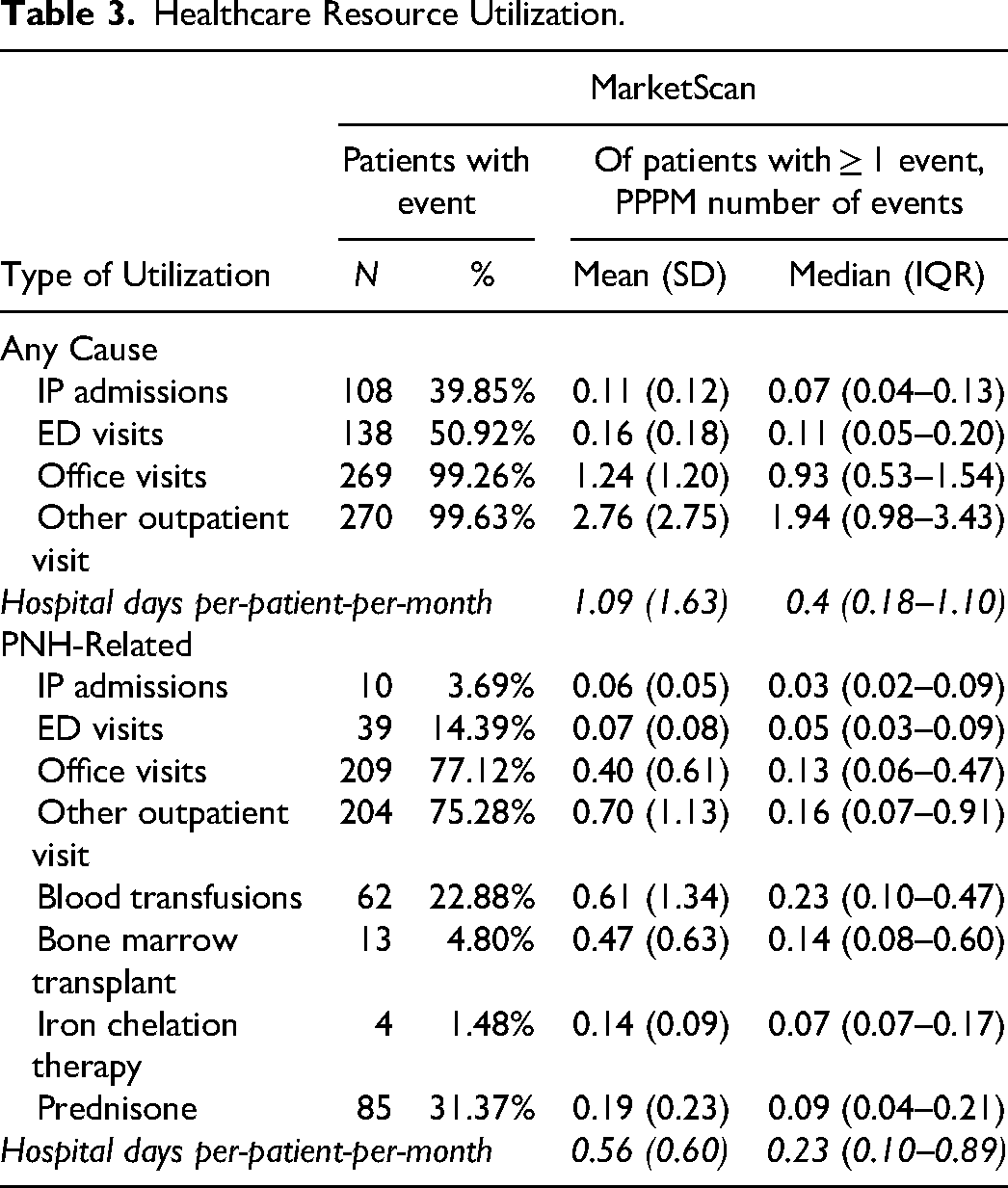
Inpatient and ED use was common among patients with PNH. Of the 271 patients in the MarketScan sample, 108 (39.9%) had an inpatient admission, 138 (50.9%) had an ED visit, 269 (99.3%) had an office visit, and 270 (99.6%) had a non-office/other outpatient visit for any cause. For PNH-related events, there were 10 patients (3.7%) with an inpatient admission, 39 (14.4%) with an ED visit, 209 (77.1%) with an office visit, and 204 (75.3%) with a non-office/other outpatient visit. Blood transfusions occurred in 62 patients (22.9%), while bone marrow transplant occurred in 13 patients (4.8%). (Table 3)
Healthcare Resource Utilization.
Costs incurred by patients with PNH were driven by pharmaceutical and inpatient costs. The MarketScan PPPM total cost for newly diagnosed patients was $18,978 or $227,736 per-patient-per-year (PPPY) across all follow-up months. Of these costs, 58.9% ($11,182 PPPM) were for pharmacy costs (including infusion costs), 21.5% ($4086 PPPM) were for other outpatient visits, 17.5% ($3318 PPPM) were for inpatient costs, 1.3% ($245 PPPM) were for office visits, and 0.8% ($146 PPPM) were for ED visits (Figure 2, Supplemental Table 3). The total PNH-related cost was $11,038 PPPM, accounting for 58.2% of the total cost. PNH-related pharmacy costs for eculizumab and ravulizumab, including administration, accounted for 81.4% ($8988) of PNH-related costs (no pegcetacoplan use was observed since it was approved in 2021).
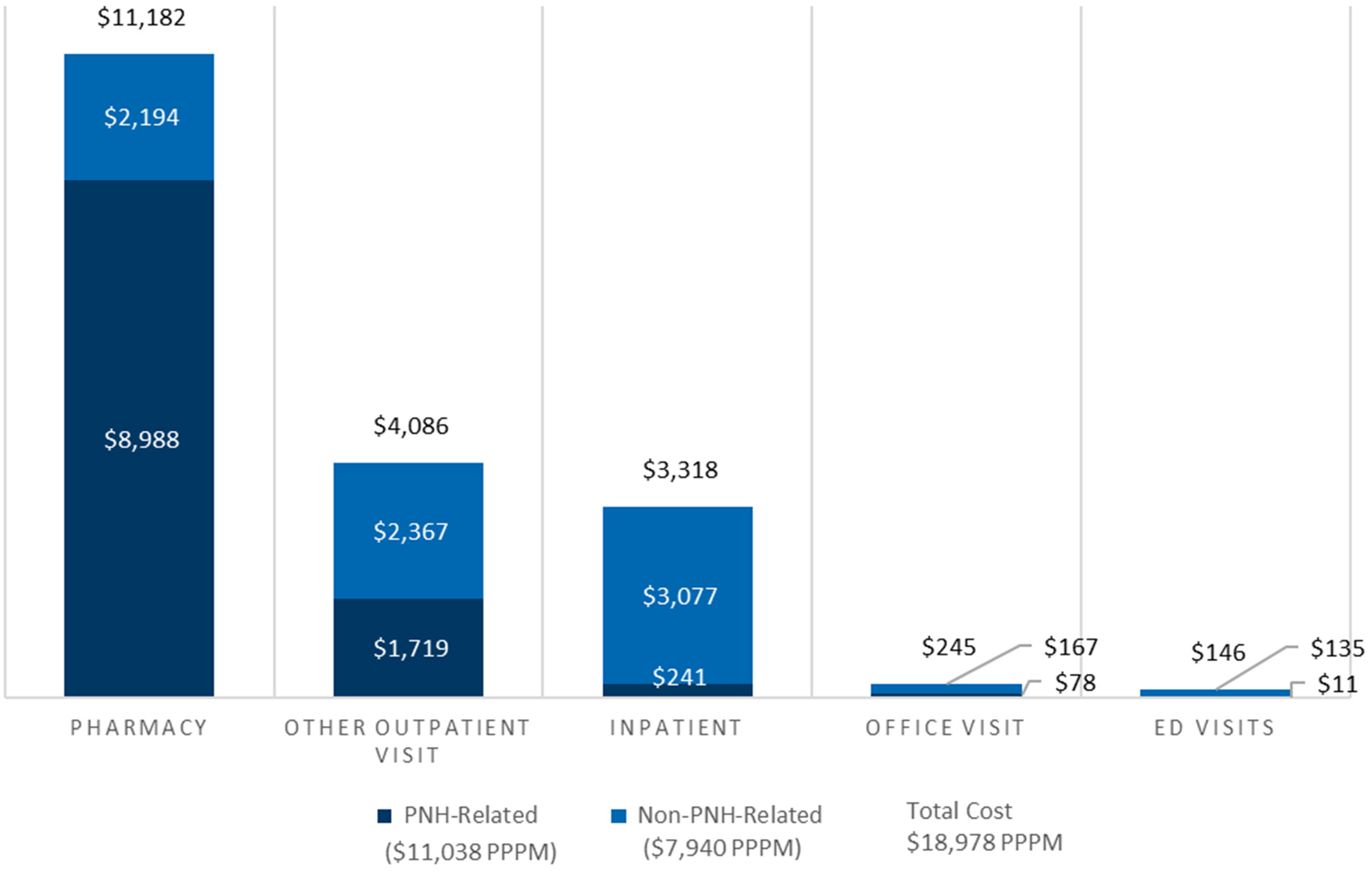
PPPM cost for newly diagnosed patients.
Sensitivity Analyses
The results using the Optum data were similar qualitatively, although fewer patients received C5 inhibitor therapy, and among those treated, medication switching rates were higher. In the Optum sample of 381 patients, 50.1% were female, and the average age was 61.3 years. After reweighting, 52.8% were female, and the average age was 56.2 (Supplemental Table 4). A lower share of patients in the Optum data received any PNH-specific pharmacologic treatment (11.8% compared to 25.1% in MarketScan) with a shorter average time from diagnosis to treatment initiation among those treated (47.9 days compared to 140.9 in MarketScan) (Supplemental Table 5). Of the 39 patients receiving eculizumab, 29 (74.4%) discontinued treatment for 60 days or over, whereas 6 (24%) of those receiving ravulizumab discontinued treatment. Compared to MarketScan, the eculizumab discontinuation rate was higher in Optum (74.4% compared to 63.6%), but the ravulizumab discontinuation rate was lower in Optum (24% vs 50%). The mean duration of treatment prior to discontinuation for either of the medications was 219.7 days, shorter than the MarketScan duration of 346.1 days (Supplemental Table 5). Of Optum patients receiving any treatment (eculizumab or ravulizumab), the MPR was lower at 88% (compared to 97% for MarketScan), and fewer patients (73.3% vs 92.7% in MarketScan) had an MPR ≥ 80%. The PDC was 60% for patients receiving any treatment (eculizumab or ravulizumab), and 37.8% had a PDC ≥ 80%, similar to the results of MarketScan (Supplemental Table 7). In Optum, for patients who started on eculizumab, 48.7% later switched to ravulizumab, with an average time to switch of 450.4 days (SD = 382.7 days) and no patients switched from ravulizumab to eculizumab. The rate of switching observed in Optum was higher than that of MarketScan (48.7% vs 33.3%), and the average time to switch was lower in Optum (450.4 days vs 654.9 days) (Supplemental Table 8).
For HCRU and cost outcomes in Optum, the results show a high level of resource utilization and cost for newly diagnosed PNH patients with inpatient admission rates higher in Optum. Of the 381 patients in the Optum age-adjusted sample, 212 (55.6%) had an inpatient admission, higher than the MarketScan percent of 39.9% (Supplemental Table 9). The percent of patients with an ED visit was similar at 48.0% (compared to 50.9% in MarketScan), as were the office and non-office/other outpatient visits at roughly 99%. For PNH-related events, more patients had an inpatient admission where PNH was coded on the claim in Optum at 25.7% (compared to 3.7% in MarketScan), but fewer had a PNH-related ED visit (4.2% in Optum vs 14.4% in MarketScan). Blood transfusions occurred in 94 patients (24.7%), while bone marrow transplant occurred in 14 patients (3.7%), both similar to the percent of patients in MarketScan. The Optum PPPM mean total cost for newly diagnosed patients was lower at $14,537 or $174,444 PPPY across all follow-up months (compared to MarketScan at $18,978 PPPM and $227,736 PPPY). This difference was driven by slightly lower costs across all categories except office visits, which were higher in Optum. Of the total costs in Optum, $9458 PPPM (65.1%) were for pharmacy costs (including infusion costs), $2649 PPPM (18.2%) were for inpatient costs, $1844 PPPM (12.7%) were for other outpatient visits, $516 PPPM (3.5%) were for office visits, and $69 PPPM (0.5%) were for ED visits (Supplemental Table 10), shares similar to those in MarketScan. The total PNH-related cost was $7725 PPPM, accounting for 53.1% of the total cost. PNH-related PPPM pharmacy costs for eculizumab and ravulizumab (no pegcetacoplan use was observed since it was approved in 2021), including administration, were $6526 (84.5% of total PNH-related costs). PPPM PNH-related inpatient costs were $833 (10.8% of total PNH-related costs), office visits were $92 (1.2%), and other outpatient visits were $273 (3.5%). Overall, despite Optum average costs being somewhat lower ($14,437 vs $18,978 in MarketScan), the median PPPM costs were actually higher in Optum as compared to MarketScan ($2954 in Optum vs $2606 in MarketScan).
The results of the sensitivity analyses that required a 3-month baseline/washout period, and 3 months did not materially change the findings in either data set (Supplemental Table 11 to 17). While the number of patients in both the MarketScan and Optum data increased under less restrictive washout period criteria, neither the treatment pattern statistics among newly diagnosed PNH patients, demographics, or utilization and cost figures changed by more than a standard deviation. The results still show low treatment rates, high MPR, and high discontinuation rates. HCRU and costs show similar levels as well, with pharmacy costs slightly higher in the sensitivity analysis.
Discussion
Despite the availability of multiple C5 inhibitor therapies, PNH patients continue to have unmet medical needs and high medical costs. Only 25.1% of patients with PNH initiated treatment with eculizumab or ravulizumab. However, 50.9% of patients had at least 1 ED visit, and 39.9% of patients had an inpatient admission in the follow-up period after the initial PNH diagnosis, contributing to a PPPM cost of $18,978 during the follow-up period ($227,736 PPPY). Patients were also still often reliant on other treatments, as 31.4% of patients were treated with prednisone, 21.4% received an anticoagulant, and 22.9% had at least 1 blood transfusion. Adherence, as measured by the MPR, is high for PNH treatment at 97.1%. However, discontinuation rates were 58.8% in this population. High discontinuation rates may be due to the development of anti-drug antibodies, which have been found in other studies 16 to lead to a loss of clinical efficacy.
While other studies have used claims data to measure HCRU and cost in PNH patients,13,17,18 our analysis not only uses more recent data, but also confirms the findings using 2 separate claims data sources. Despite the overall findings being similar in MarketScan and Optum, there were some differences to note. First, 11.8% of newly diagnosed PNH patients initiated treatment with eculizumab or ravulizumab, compared to 25.1% in MarketScan. The rates of ED visits were similar, at 48.0% (compared to 50.9% in MarketScan), while the rates of inpatient admissions were higher at 55.6% (compared to 39.85% in MarketScan). The total PPPM cost was also lower at $14,537 PPPM (compared to $18,978 in MarketScan), due in part to lower outpatient visit and pharmacy costs, but median PPPM costs in Optum were actually higher as compared to MarketScan. These differences could be driven by several factors, including differences in the underlying population, the small sample size, and variation in prices, especially for eculizumab and ravulizumab. While these differences suggest that caution should be taken in generalizing specific information about treatment patterns, HCRU, and cost from a PNH population in 1 data source, they still demonstrate high levels of unmet medical need and high medical costs.
This study also documents novel findings on treatment patterns that include the availability of ravulizumab. Despite the study period covering years both before and after the FDA approval of ravulizumab, 79.4% of newly diagnosed patients in the MarketScan sample who initiated a C5 inhibitor started on eculizumab. Of those, 33.3% of patients later switched to ravulizumab, with an average time to switch of 655 days. For Optum, 86.7% started on eculizumab, and 48.7% switched to ravulizumab with an average time to switch of 450 days. Though the sample of newly diagnosed patients who were diagnosed in 2019 and 2020 (after the availability of ravulizumab) is too small for a meaningful statistical analysis, the analysis of this subpopulation of patients suggests that consistent with the overall results, patients are often still starting on eculizumab, and roughly half of patients later switch to ravulizumab.
This study aligns with existing literature on PNH that demonstrates a high clinical burden and cost of disease and adds new insights with more recent data for newly diagnosed patients. An international PNH registry study found high rates of hemolysis (55.8%) and impaired renal function (42.8%) at baseline, along with history of major adverse vascular events. 3 These findings have been further supported by claims-based analyses, which show high rates of PNH-related symptoms and HCRU. Cheng et al (2021) found that 36.4% of PNH patients treated with eculizumab have a hospitalization, and 19.2% have an ED visit in the follow-up period after starting eculizumab treatment, and Jalbert et al (2019) found that 31.5% had been hospitalized in the year prior to the incident PNH diagnosis.4,13 More recent research has indicated continued need for blood transfusions, with 39.5% of patients receiving therapy receiving at least on transfusion after starting treatment, and 20.6% with 4 or more transfusions, with lower rates for patients treated with ravulizumab.17,18 Our study analyzes similar outcomes for all newly diagnosed patients, including those that do not start treatment with eculizumab or ravulizumab, and shows that for this cohort of patients, the rates of inpatient admissions are similar at 39.9%, but the rates of ED visits are higher at 50.9%. Our findings also include more patients due the inclusion of 2 data sources, as well as more recent data through June 2021.
There is limited evidence on treatment patterns for PNH patients, and this study provides an updated perspective on treatment patterns that includes ravulizumab, but also documents new findings on adherence. One recent claims-based study found that for newly diagnosed patients, 10.3% started on eculizumab within 1 year of PNH diagnosis with average time to treatment of 60.5 days. 4 Further, within 1 year of incident diagnosis, roughly one-third of patients discontinued eculizumab. The current study finds similar, though somewhat higher, rates of treatment initiation for a C5 inhibitor, either eculizumab or ravulizumab (20.3%), and provides new findings on rates of treatment and time to treatment initiation for ravulizumab. We found that 25% of newly diagnosed patients initiate treatment with a C5 inhibitor, with 79% starting on eculizumab and 21% starting on ravulizumab. These findings are roughly similar across our 2 data sources. We also examined adherence with MPR and PDC, which to our knowledge have not previously been quantified in the literature. We found an MPR of 97%, but due to high discontinuation rates (58.8%), PDC is lower at 60% for newly diagnosed patients from the MarketScan data. Findings were similar using Optum data, and for the prevalent, treated population, though for the latter, PDC was somewhat higher at 78%.
Our study has several limitations. First, the sample sizes from MarketScan and Optum were relatively small as PNH is a rare disease, limiting the ability to conduct formal statistical tests of differences in patient characteristics and outcomes across treatments. Second, as the patient population was limited to US patients insured by private health plans or Medicare Advantage, our results may not represent the broader US PNH population (eg, individuals covered by Medicaid, or those without insurance). Moreover, MarketScan's Medicare population is also limited to those who with employer-sponsored Medicare supplement insurance, 19 which may include more individuals from higher socioeconomic status. Third, the costs in this study focused on direct medical costs from paid claims and do not include any indirect or societal costs. Medical-related absenteeism costs have been estimated at $7756 PPPY for transfusion-dependent PNH patients, 13 but these costs are not included in our study. Fourth, the study did not evaluate differences in adverse event rates across treatments. To the extent that the adverse events resulted in any additional health care resource utilization, however, the impact would be captured in the total cost estimates. Lastly, current treatment patterns, HCRU, and cost may be changing given that ravulizumab was approved during the study period (December 2018) 20 and pegcetacoplan was approved for PNH in May 2021. Due to the timeframe of this study, no pegcetacoplan use was observed among patients meeting the follow-up window inclusion criteria.
Conclusion
Despite the availability of new treatment options, only 25.1% of newly diagnosed patients with PNH received any PNH-specific pharmacologic treatment. Even among those patients who were treated, the average patient initiated a PNH-indicated treatment 5 months after diagnosis. Once treatment was received, most patients were adherent to their medication while on therapy, but more than half of patients discontinued therapy. Healthcare resource utilization and costs remain high with 39.1% of PNH patients having an inpatient visit after diagnosis. Nearly 1 in 5 newly diagnosed PNH patients still require at least 1 blood transfusion, signaling some unmet medical need in PNH. Future research should re-examine treatment trends and HCRU with additional data sources, particularly as new treatments become available to PNH patients.
Supplemental Material
sj-docx-1-cat-10.1177_10760296231213073 - Supplemental material for Treatment Patterns and Healthcare Resource Utilization of Patients With Paroxysmal Nocturnal Hemoglobinuria: A Retrospective Claims Data Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296231213073 for Treatment Patterns and Healthcare Resource Utilization of Patients With Paroxysmal Nocturnal Hemoglobinuria: A Retrospective Claims Data Analysis by Denise Clayton, Jason Shafrin, Glorian Yen, Soyon Lee, Lincy Geevarghese, Yulin Shi, Luyang He, Ying Shen and Anem Waheed in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
The authors would like to thank Frank Li for support conducting the statistical analyses.
Author Contributions
DC, JS, GY, LG, and AW designed the study. YShi, LH, and YShen conducted the analyses. DC, JS, GY, SY, LG, and AW interpreted the data. DC and JS wrote the first draft of the manuscript. All authors critically commented on the manuscript and approved the final version.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DC and JS are employed by FTI Consulting, which receives funding from Novartis and other life sciences and healthcare organizations. GY, SL, LG, LH, and YS are employed by Novartis. AW consults for Novartis. YS is employed by KMK Consulting Inc. and receives funding from Novartis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Novartis Pharma.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
