Abstract
The aim was to describe inpatients with COVID-19 empirically prescribed heparinoid anticoagulants and compare resource utilization between prophylactic/low-dose and therapeutic/high-dose groups.
Introduction
Coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus resulted in a global pandemic, which affected hospitals worldwide. Coagulopathies are common among patients hospitalized with COVID-19 and are associated with more severe disease and mortality.1–4 In one study, estimated prevalence of thrombosis was 22% overall and 43% among patients admitted to intensive care units (ICUs). 5
The results of randomized, placebo-controlled trials indicate that anticoagulation decreases coagulopathy risk in the general population of acutely ill hospitalized patients.6–9 Among inpatients with COVID-19, anticoagulation has been a common management strategy, used in nearly 69% of US hospitalized patients in one early cohort study. 10 Studies indicate that timely thromboprophylaxis improves survival and other key clinical outcomes among inpatients with COVID-19, and therefore thromboprophylaxis is advised clinically and in medical societies’ guidelines, unless patients have contraindications.6,7,9,11–19
However, observational studies and clinical trials have produced mixed evidence as to the best anticoagulant dose level for patients with COVID-19. Some studies support higher-dose treatment (ie, a therapeutic dose or an intermediate-prophylactic dose, rather than a standard prophylactic dose) to help prevent microvascular thrombosis.20–23 Other studies suggest high-dose thromboprophylaxis is associated with lower rates of thromboembolic events and mortality compared with low-dose treatment for critically ill patients with COVID-19.24,25 In contrast, two robust randomized trials of patients hospitalized with COVID-19 indicated therapeutic-dose anticoagulation only improved survival among patients who were not critically ill.26,27 Similar results were reported when comparing dosage levels among patients who were not admitted to ICUs. 28 These discrepant findings may reflect the biological complexity of coagulopathy in COVID-19 infection.29,30 While it seems clear that most patients hospitalized with COVID-19 will benefit from some level of anticoagulation, the optimal dose remains unclear and is the subject of ongoing clinical trials. 31
COVID-19 is associated with substantial healthcare resource utilization (HRU) and costs.32–37 In studies of hospitalized patients, ICU utilization ranged from 15% to 52%.33,35,37–39 Among inpatients with COVID-19, increased total cost and length of stay (LOS) are associated with greater comorbidity burden, poorer hematologic biomarkers, worse clinical severity, and certain demographic characteristics.32,35,37,39,40 Before the COVID-19 pandemic, studies in hospital and outpatient settings evaluated the cost-effectiveness and clinical efficacy of various thromboprophylaxis strategies.41–46 However, to our knowledge, no such studies of HRU and costs related to heparinoid anticoagulation dose level have been reported for patients hospitalized with COVID-19.
Using a large US database, this study compared HRU, costs, and mortality adjusting for confounders among inpatients with COVID-19 who received either prophylactic/low-dose or therapeutic/high-dose anticoagulation (heparin and/or enoxaparin). This study used real-world data for patients who sought hospital care, were diagnosed with COVID-19, and were empirically prescribed heparinoid anticoagulants early during the pandemic.
Methods
Data Source and Study Design
A retrospective cohort study was conducted using data from the Premier PINC AI™ Healthcare Database - Special Release (PHD-SR), which averages more than 8.5 million inpatient and 75 million outpatient encounters per year and captures 20–25% of all US inpatient hospital discharges. The PHD-SR is an extensive hospital-based database containing service and billing information. By means of a unique PHD-SR patient key, patient data can be tracked within a hospital system.
Between April 1, 2020, and November 30, 2020, 685,853 patients diagnosed at discharge with COVID-19 (ICD-10-CM U07.1) were identified in the PHD-SR. Hospitalized inpatients and hospital-based outpatients (eg, those seen in an emergency department or hospital-based clinic) were evaluated. The index visit was the COVID-19-related hospital inpatient admission or outpatient visit. After excluding patients aged <18 years (n = 33,234) and patients with cardiovascular diagnoses (ie, atrial fibrillation, stroke, pulmonary embolism/deep vein thrombosis, or pulmonary hypertension) or who received anticoagulants (ie, warfarin or the novel oral anticoagulants apixaban, rivaroxaban, dabigatran, or edoxaban) during the 6 months before the index visit (n = 121,662), the initial analytic sample included 530,957 adults. Patients with prior treatment and diagnoses were excluded because the aim was to assess empirical anticoagulant dosing in patients with COVID-19. Eligible adults diagnosed with COVID-19 included 174,422 inpatients and 356,535 outpatients from 880 hospitals. Only inpatients were evaluated for heparinoid anticoagulant dosage.
All data were compliant with the Health Insurance Portability and Accountability Act (HIPAA). Based on US Title 45 Code of Federal Regulations Part 46, institutional review board approval for this study was not required because the study used pre-existing de-identified hospital data that could not be linked to individuals.
Among inpatients, anticoagulant use and dosing strategies were determined based on the maximum dose during days 0–3 of hospital admission. Total daily dose was categorized as prophylactic/low-dose (<160 mg enoxaparin or <15,000 units of heparin) or therapeutic/high-dose (≥160 mg enoxaparin or ≥15,000 units of heparin). Choice of enoxaparin versus heparin and dosage were at the discretion of individual clinicians’ assessment of the patient. To avoid confounding if these therapies were used for other conditions, doses greater than 320 mg enoxaparin or 25,000 units of heparin were excluded. Anticoagulation was not examined among outpatients because intensity of therapy in this population was expected to vary, which could potentially obscure associations with outcomes.
This cross-sectional study compared anticoagulant dose (ie, prophylactic/low-dose vs therapeutic/high-dose) in adult inpatients hospitalized with COVID-19. Primary study outcomes were ICU admission, LOS, total cost, and in-hospital mortality. Secondary outcomes assessed included clinical complications during hospitalization and readmissions within 60 days.
Patient Demographics and Visit and Clinical Characteristics
Data collected for all patients included age, primary insurance payer, and self-reported sex, race, and ethnicity. Individual comorbid conditions and overall risk were assessed using the Charlson Comorbidity Index (CCI)47,48 via primary and secondary ICD-10 diagnosis and procedure codes at discharge and 180 days before the index visit. Also ascertained were obesity, dyspnea, hypertension, dyslipidemia, respiratory support therapy, cardiac imaging tests, medications, and laboratory measures during the visit. Visit characteristics included admission point of origin, admission type, and discharge status, which hospitals submitted according to the uniform billing criteria of the Centers for Medicare and Medicaid Services. Hospital characteristics included geographic region (Midwest, Northeast, South, West), urban/rural location, teaching status, and number of beds.
Laboratory Measures and Medications
Laboratory measures and vital signs were assessed for all patients for whom these were available. (Not all hospitals in the PHD-SR contribute these data, and all tests also were not ordered for all patients.) Laboratory measures included D-dimer, C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), platelets, eosinophil count/percentage, partial pressure of oxygen, fibrinogen, white blood cell differential (WBC), and erythrocyte sedimentation rate. Additional details regarding determination of laboratory measures, such as testing manufacturer and kit used, were not available in the PHD-SR. Because laboratory measures could be assessed multiple times during hospitalization, both initial and final values were evaluated. If only a single measure was reported during the index visit, it was considered the initial exposure point value. Mean heart rate and diastolic and systolic blood pressure were calculated across the visit. Measurements were not corrected or filtered by minimum or maximum values. Medications assessed for use during the index visit consisted of ACE inhibitors, β-blockers, calcium channel blockers, corticosteroids, hydroxychloroquine, azithromycin, other antibiotics, remdesivir, tocilizumab, other antivirals, other anticoagulants (ie, not enoxaparin or heparin), and bronchodilators.
HRU, Costs, Complications, and Risk of Mortality
For inpatients, HRU was assessed by determining index hospital LOS (as submitted by hospitals), index ICU use (
Statistical Analyses
Results are reported as median (Q1, Q3) or n (%), and comparisons were performed using Wilcoxon Rank Sum and Chi-square tests, respectively. Power analysis was conducted prior to the study, and the sample sizes of the cohorts yielded powers of 0.8 or higher to detect differences in primary outcomes. Because of the large sample size, P < 0.01 was defined as significant. All analyses were conducted using SAS version 9.4.
Multivariable regression analyses were used to evaluate outcomes among recipients of anticoagulant heparinoids and address the selection bias due to differences in severity of patients between the two cohorts. For LOS and total cost, generalized linear models (GLM) with log link function and either negative binomial or gamma distribution, respectively, were used. GLM regression coefficients were exponentiated, and 95% confidence intervals for the exponentiated coefficients were reported. Logistic regression was used to assess in-hospital mortality and ICU utilization. Potential covariates for the regression models were evaluated for clinical relevance and statistical appropriateness (ie, overall frequency and differences between dose groups). Sensitivity analyses were completed to assess model fit and properties of variables in the model. Final covariates were retained in the models regardless of statistical significance with outcome measure.
Covariates in the final models included patient demographics (ie, age, sex, race, ethnicity, payer), visit characteristics (ie, admission type, point of origin, discharge status), hospital characteristics (ie, bed size, region, teaching status, urban/rural), clinical characteristics (ie, invasive mechanical ventilation, obesity, dyslipidemia, hypertension), individual comorbidities from the CCI (ie, myocardial infarction, congestive heart failure, cerebrovascular disease, dementia, chronic pulmonary disease, diabetes, cancer, renal disease), and concomitant medications. Discharge status (ie, a severity proxy) was not included in the model that assessed in-hospital mortality.
In adjunct analysis, data were examined separately for the full sample of adults with COVID-19 (ie, all inpatients and outpatients) stratified by patients age (ie, 18–49 years vs
Results
Dataset, Visit, and Hospital Characteristics
A total of 122,508 inpatients (70.2% of all inpatients) from 776 hospitals met criteria for heparinoid anticoagulant use and were included in analyses, of which 93,283 (76.1%) received prophylactic/low-dose and 29,225 (23.9%) received therapeutic/high-dose anticoagulation (Table 1). The high-dose group was a median of 3 years older and had a 6.3% lower proportion of female patients than the low-dose group. Distribution of Hispanic/Latino ethnicity was similar, but the high-dose group had more Black and fewer White patients than the low-dose group.
Patient Characteristics by Anticoagulation Dose Group. a
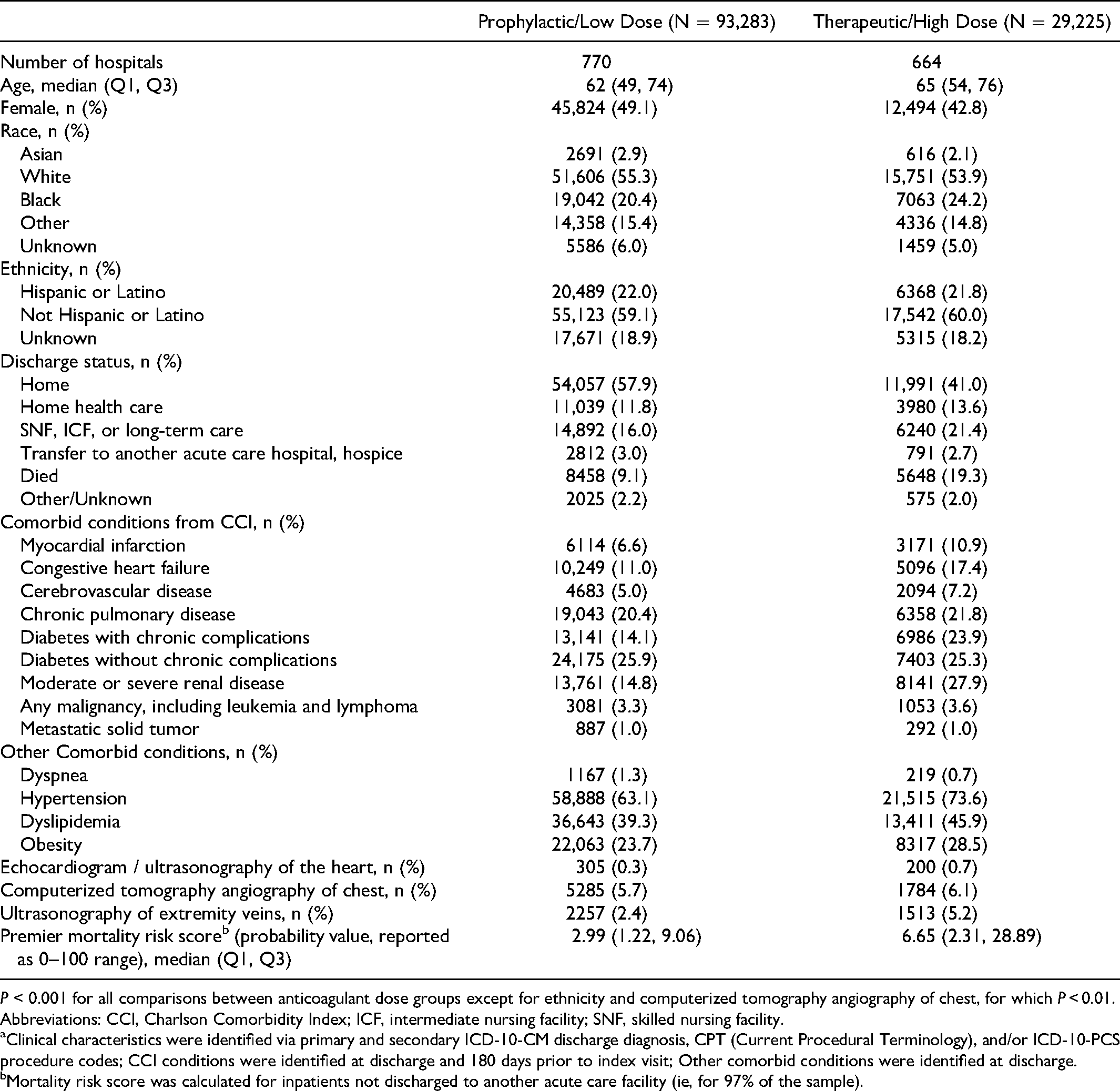
P < 0.001 for all comparisons between anticoagulant dose groups except for ethnicity and computerized tomography angiography of chest, for which P < 0.01.
Abbreviations: CCI, Charlson Comorbidity Index; ICF, intermediate nursing facility; SNF, skilled nursing facility.
Clinical characteristics were identified via primary and secondary ICD-10-CM discharge diagnosis, CPT (Current Procedural Terminology), and/or ICD-10-PCS procedure codes; CCI conditions were identified at discharge and 180 days prior to index visit; Other comorbid conditions were identified at discharge.
Mortality risk score was calculated for inpatients not discharged to another acute care facility (ie, for 97% of the sample).
Recipients of heparinoid anticoagulants were primarily emergency/urgent admissions (96.8%) from home or a non-healthcare facility (80.7%); a small proportion (18.7%) were transferred from another healthcare facility. (See Appendix Table 1 in Supplemental Materials.) In the low-dose group, a higher proportion of patients was discharged home (with or without home healthcare; 69.7% total), and a lower proportion was transferred or discharged to another healthcare facility (19%), than in the high-dose group (54.6% discharged home; 24.1% discharged to other healthcare facility; P < 0.001 for the overall Chi-square).
Clinical Characteristics
Comorbidities and respiratory supportive therapies were more frequent and mortality risk was higher in the therapeutic/high- versus the prophylactic/low-dose group. The proportion of patients in the high-dose group with a CCI score
The therapeutic/high-dose group received more treatments and procedures to manage respiratory failure (Figure 1) and more median days of non-invasive mechanical ventilation and supplemental oxygen use (5 and 3 days, respectively, vs 4 and 2 days in the prophylactic/low-dose group; both P < 0.001). Although use was low (<6% overall), echocardiogram, extremity ultrasound, and computerized tomography angiography procedures were more frequent in the high-dose group.
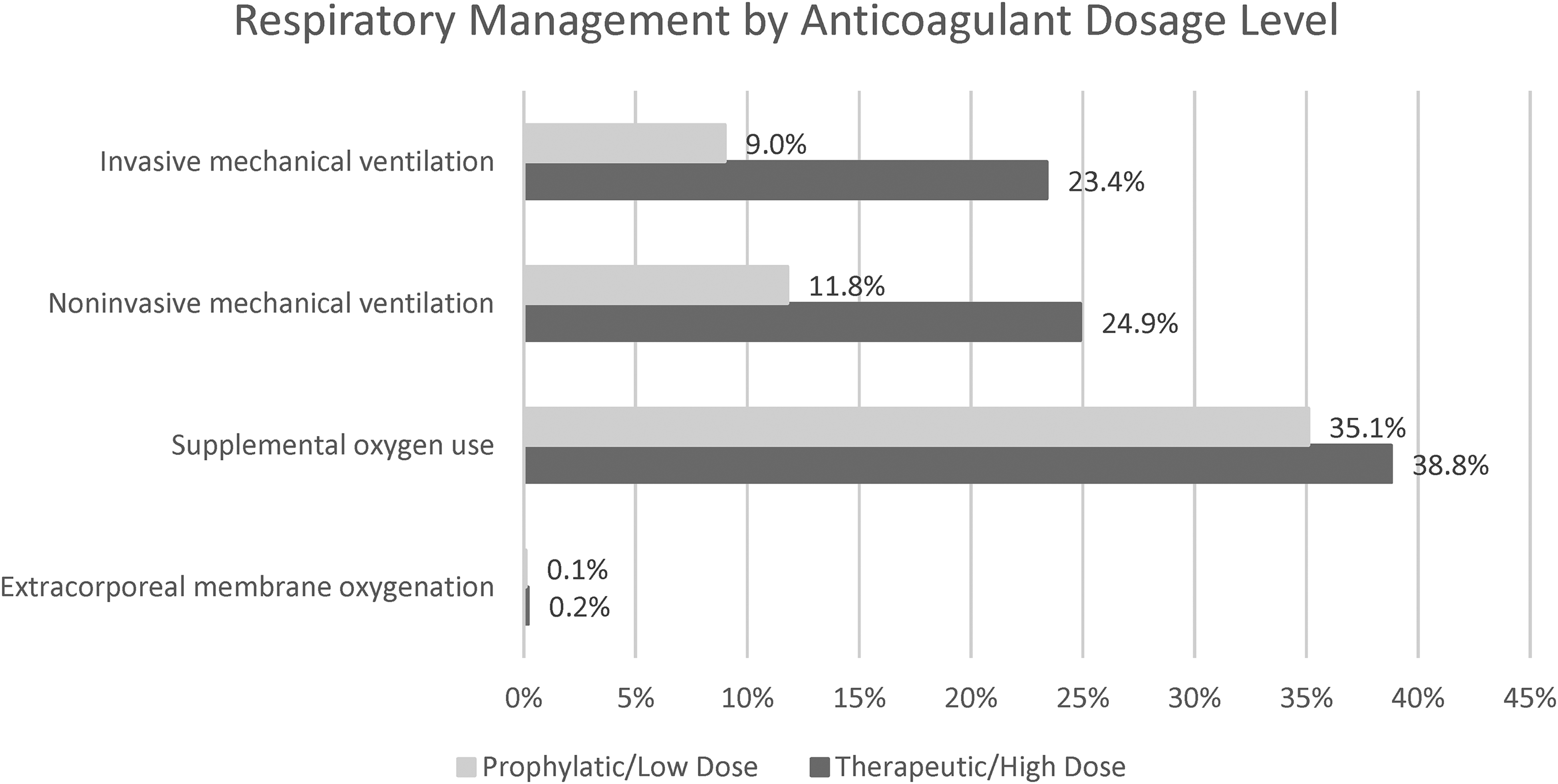
Respiratory Management by Anticoagulation Dose Group. P < 0.001 for all comparisons.
Laboratory Measures and Medications
Results of individual laboratory tests were available for 3.2% to 19.4% of patients, depending on the test and the hospital submitting the data. Data completeness was similar between the dose groups. Initial laboratory values were significantly higher in the high- versus low-dose group for D-dimer (difference: 0.5 mcg/ml), CRP (0.3 mg/L), NLR (1.1 units), and WBC (0.9 1000/L), and these differences persisted at final measurement (Table 2). Fibrinogen values exceeded normal in both dose groups and were statistically similar between groups. Despite some statistical differences, the two groups showed no clinically significant differences in blood pressure, heart rate, prothrombin time, or eosinophil percentage.
Laboratory Measures by Anticoagulation Dose Group. a

‡ P < 0.001, † P < 0.01 for comparisons between anticoagulant dose groups. Intra-group differences between initial and final laboratory measurements were not statistically tested.
When laboratory measures were assessed multiple times during hospitalization, initial and final values were evaluated for comparison between dosing groups. If only a single laboratory measure was reported, it was considered the initial exposure point value. Because a limited number of hospitals submit laboratory data, initial measures were available for 3–20% of all patients, depending on the test, and availability was similar across dosing groups. Sample sizes for evaluations of initial and final laboratory measurements ranged from n = 2949 to 19,042 for initial values and n = 1214 to 16,536 for final values in the prophylactic/low-dose group, and from n = 957 to 4958 for initial values and n = 477 to 4453 for final values in the therapeutic/high-dose group. Measures were not corrected or filtered by minimum or maximum values. Additional details regarding determination of laboratory measures, such as testing manufacturer and kit used, were not available in the database.
Concomitant medication use was more frequent in the high- versus low-dose group. Bronchodilators, β-blockers, calcium channel blockers, corticosteroids, and other antibiotic medications were prescribed 9% to 14% more frequently in the high-dose group, while azithromycin, remdesivir, other antiviral medications, and tocilizumab were prescribed 4% to 5% more frequently.
HRU, Costs, Clinical Complications, and Mortality
HRU was higher and complications were more frequently identified in the therapeutic/high- versus prophylactic/low-dose group. Median hospital LOS and median ICU LOS each were 3 days longer in the high-dose group (Figure 2), and more than one-third of patients in the high-dose group spent time in the ICU, compared with less than 20% of patients in the low-dose group (Figure 3). Total and ICU costs were nearly two times higher in the high-dose group compared with the low-dose group.
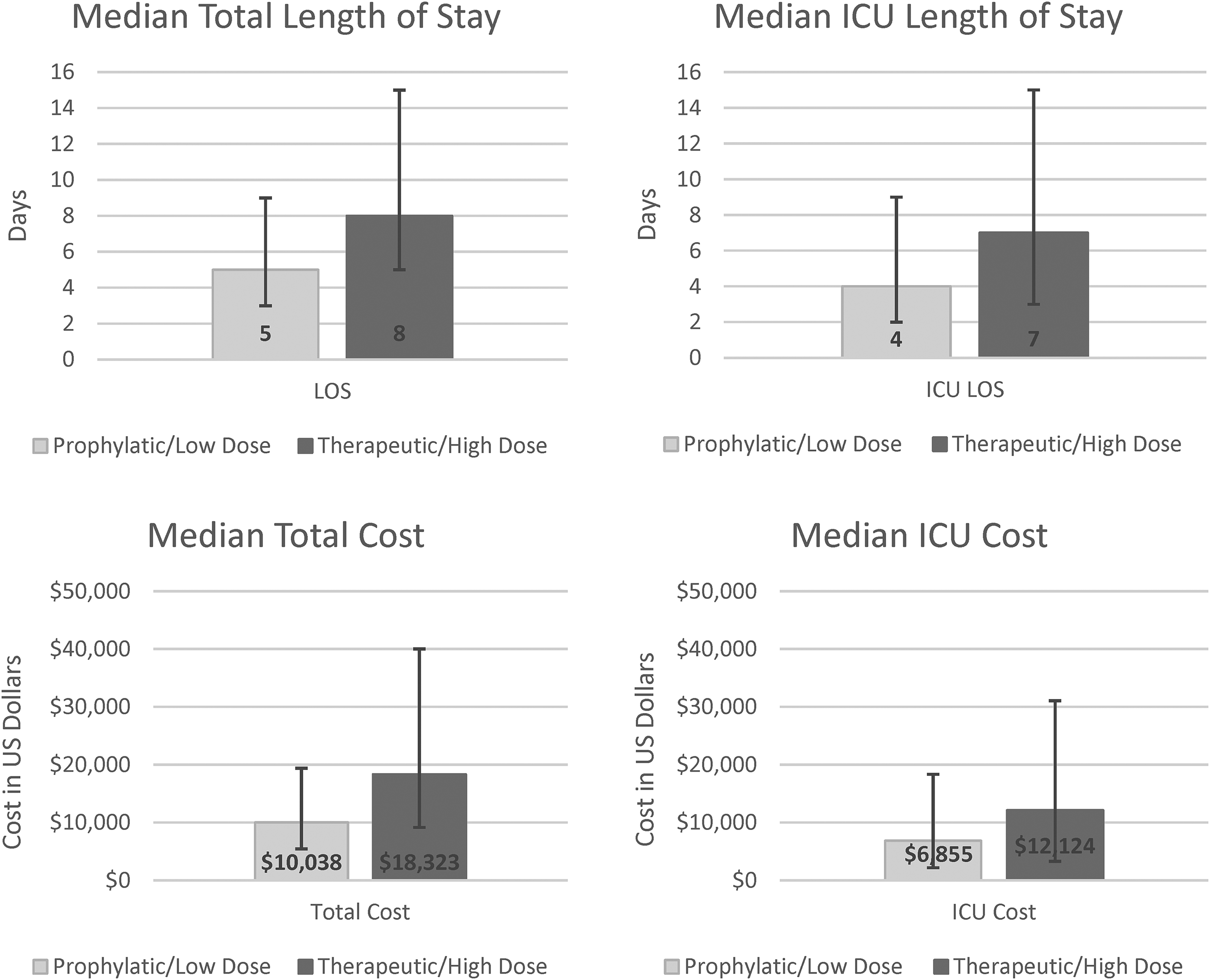
Median Length of Stay and Cost by Anticoagulation Dose Group.a P < 0.001 for all comparisons. Abbreviations: ICU, intensive care unit; LOS, length of stay. a Error bars show 25th to 75th percentile.

Outcomes by Anticoagulation Dose Group. P < 0.001 for all comparisons. Abbreviation: ICU, intensive care unit.
Compared with the prophylactic/low-dose group, the therapeutic/high-dose group had significantly (P < 0.001) greater frequencies of clinically relevant complications, including acute respiratory failure (60.2% vs 54.9%), sepsis (37.6% vs 26.3%), acute respiratory distress syndrome (11.7% vs 4.7%), venous thromboembolism (9.4% vs 2.5%), and pulmonary embolism (5.5% vs 1.3%). (See Appendix Table 2 in Supplemental Materials.) Inpatient mortality in the high-dose group was double that of the low-dose group. Moreover, among discharged patients, those who had received high-dose anticoagulation were more likely to be readmitted within 60 days.
Adjusted Analyses
Multivariable models (Table 3) substantiated our findings of poorer outcomes and greater HRU in the therapeutic/high- versus prophylactic/low-dose group. After adjusting for demographic, visit-related, and clinical factors, high-dose anticoagulation was associated with a 45% increase in odds of ICU utilization and a 26% increase in odds of in-hospital mortality compared to low-dose. After adjusting for covariates, high-dose anticoagulation was associated with 28% higher average total cost and 21% longer average LOS compared to low-dose. (For associations between covariates and outcomes, see Appendix Tables 3–6 in Supplemental Materials.)
Multivariable Regression Analyses a Comparing Therapeutic/High-Dose Versus Prophylactic/Low-Dose Anticoagulation Groups.

P < 0.001 for all associations.
Abbreviation: ICU, intensive care unit.
The following covariates were also included in each model: demographics (ie, age, sex, race, ethnicity, primary insurance payer), visit characteristics (ie, admission type, point of origin, discharge status), hospital characteristics (ie, bed size, geographic region, teaching status, and urban/rural population served), clinical characteristics (ie, invasive mechanical ventilation, obesity, dyslipidemia, hypertension), individual comorbidities from the Charlson Comorbidity Index (ie, myocardial infarction, congestive heart failure, cerebrovascular disease, dementia, chronic pulmonary disease, any diabetes [diabetes with chronic complication / diabetes without chronic complication], any cancer [any malignancy, including leukemia and lymphoma / metastatic solid tumor], moderate or severe renal disease), and concomitant medications (ie, ACE inhibitor, β-blocker, bronchodilator, calcium channel blocker, corticosteroid, hydroxychloroquine, azithromycin, other antibiotic medication, other anticoagulants [ie, not enoxaparin or heparin], remdesivir, other antiviral medication, tocilizumab). Discharge status was not included in the model examining in-hospital mortality. N = 122,508 inpatients for all models except total cost, where N = 104,569.
Estimates are the odds ratio for logistic regression models and the exponentiated coefficient for GLM. Thus, after adjusting for covariates in separate models, patients receiving therapeutic/high-dose anticoagulation had a 26% increase in odds of in-hospital mortality and a 45% increase in odds of ICU admission compared with patients receiving prophylactic/low-dose anticoagulation. After adjusting for covariates in separate models, average total cost per day was 28% greater and average LOS was 21% longer among inpatients receiving therapeutic/high-dose anticoagulation compared with inpatients receiving prophylactic/low-dose anticoagulation.
Adjunct Analyses on the Full Sample
Results of adjunct analyses on the initial sample of more than half a million adults diagnosed with COVID-19 in the PHD were also assessed and stratified by age groups. Among all patients, mean age was 51.4 years (SD: 19.5 years), 47.1% were age 18–49 years, and 52.9% were >50 years. More than half (53.6%) were female, 20.5% were Black, and 23.9% were Hispanic or Latino. Most patients had private insurance (42.6%) and were urgent/emergency admissions (74.3%) from home or a non-healthcare facility (77.3%). (See Appendix Table 7 in Supplemental Materials). Regional distribution of hospitals generally reflected the PHD-SR (53.5% South, 21.1% Midwest, 14.1% Northeast, and 11.3% West), whereas proportions of patients treated in urban facilities (84.5%) and teaching hospitals (42.9%) reflected early COVID-19 spread in the US.
HRU and costs were evaluated in the full sample. Among all inpatients with COVID-19 (ie, including those not prescribed anticoagulants), mean and median LOS were 8.3 (SD 9.2) days and 5 days, respectively. Median LOS was 2 days longer among inpatients aged
Nearly 5% of the approximately half million inpatients and outpatients in this study died at the hospital, and mortality was significantly higher among individuals aged
Discussion
To our knowledge, this is the first real-world data study to examine associations between anticoagulant dose, HRU, and costs among patients hospitalized with COVID-19. Among 122,508 patients hospitalized with COVID-19 and prescribed heparinoids, HRU, hospital costs, morbidity, and mortality all were significantly lower with prophylactic/low-dose versus therapeutic/high-dose anticoagulation. After adjusting for multiple demographics, visit-related, and clinical factors, the low-dose anticoagulant group was associated with 21% to 45% lower risk of ICU utilization, inpatient mortality, LOS, and total cost compared with the high-dose group. Thus, despite likely intention of clinicians to treat more rigorously patients with greater comorbidities and disease severity, no benefit was observed when evaluating associations between higher anticoagulant dose and outcomes among patients hospitalized with COVID-19.
In this study, overall findings on hospital LOS, ICU utilization, and cost were in line with results from studies of patients with COVID-19 in which assessment was not based on anticoagulant dose.32–35,37–40 Among all inpatients, median LOS was approximately two days shorter than that reported by others,32,34,39,50 but after examining anticoagulant use, we found that LOS was about 3 days longer among patients receiving high- versus low-dose heparinoids. Total hospitalization costs in this study were similar to others 35 but were significantly higher among recipients of high- versus low-dose anticoagulation. Similarly, overall ICU utilization was in line with that reported in other studies of patients with COVID-19,32,33,39 but was twice as high among patients who received high- versus low-dose anticoagulation, and consistent with another retrospective study reporting a three-fold increase in ICU admission. 30 Variations in HRU and costs across studies probably reflect differences in study time periods, geographic locations, and severity of illness indicators.32,36,40,51
Anticoagulant therapy and dosage are some of the many factors associated with mortality among patients hospitalized with COVID-19.14,20–22,52–54 Mortality among inpatients with COVID-19 has varied across time and studies, with rates ranging from 7% to 37%.10,24–27,32,33,35,36,38,39,55,56 In general, a reduction of in-hospital mortality is found with any anticoagulant regimen compared with no anticoagulation.11,25,30,52,55,57–59 Additionally, studies have demonstrated reduced mortality for patients receiving anticoagulants prior to hospitalization for COVID-19 or post-discharge.60–62 In the current study, mortality was similar overall to that in other general studies of COVID-19,32,38 but was noticeably higher among recipients of high-dose anticoagulation. These findings align with results from previous studies of patients with COVID-19 in which standard prophylactic-dose anticoagulation was associated with lower in-hospital mortality, and/or intermediate-to-therapeutic dose treatment was not associated with improved survival.11,30,52,55,57–59
However, other studies of patients with COVID-19 have reported discrepant associations between anticoagulant dose and mortality among patients receiving high-dose thromboprophylaxis.24,57 Some studies have reported higher mortality among non-critically ill patients receiving prophylactic- versus therapeutic-dose anticoagulation, but no differential effect of dosage in critically ill patients.26,27,30 Our results contrast with studies supporting the use of higher-dose anticoagulation in patients with COVID-19.20–23
In addition to higher HRU during hospitalization, patients with COVID-19 can develop persistent symptoms requiring additional treatment, emergency visits, and readmissions that continue to burden the healthcare system post-discharge.38,51,63,64 Further, patients hospitalized with COVID-19 have higher rates of subsequent mortality.63,64
Guidelines from the American Society of Hematology assert that the incremental cost of thromboprophylaxis "is negligible relative to the total costs" of COVID-19 hospitalization. 53 Experts recommend that hospital clinicians assess venous thromboembolism risk in patients hospitalized with COVID-19 just as they would for other acutely ill patients; as such, high-risk patients should receive anticoagulants, unless contraindicated.16,20,22,65,66
Clinical observations and autopsy studies suggest a complex set of events whereby thrombosis and anticoagulation precede COVID-19 related deaths.55,67 Several mechanisms for hypercoagulopathy and thrombosis in COVID-19 patients have been proposed, including endothelial cell injury, increases in circulating plasminogen, cytokine activation, formation of leukocyte-platelet aggregates, acute systemic inflammation, and progressive organ dysfunction.2,20,29,66–73 Whether a specific type of anticoagulant is preferable in the management of patients with COVID-19 remains unclear. 53
Some limitations of this study, such as reliance upon accurate and complete diagnoses and procedure coding, are inherent to any analysis of an administrative database. Additionally, since patients in the two anticoagulant dose groups were not randomly assigned, there was potential selection bias because patients with more severe cardiovascular symptoms might be more likely to receive therapeutic/high-dose versus prophylactic/low-dose anticoagulation. Although the assessment of the lab measures showed no clinically significant differences in blood pressure, heart rate, fibrinogen, prothrombin time, or eosinophil percentage between the two groups, and patient demographics, comorbidities and severity were controlled for in regression analyses, it is possible that unknown confounding factors or unobservable clinical differences could contribute to the differences in the outcomes between the two dosage groups. Low-high classification and intent of anticoagulant dosing could not be further delineated in this analysis. Comparisons between inpatients with no prescribed anticoagulants and prophylactic/low-dose anticoagulation, or proportion of enoxaparin versus heparin within anticoagulant groups were not explored in this study. Because anticoagulant dosage was examined retrospectively, regression models controlled for critical illness overall rather than dichotomizing patients. Nonetheless, this study captured empiric anticoagulant use and outcomes during a time when clinicians had little guidance on how to treat patients with COVID-19.
This study is notable for its use of real-world data to examine anticoagulant dosage and management approaches in more than 100,000 inpatients with COVID-19. Large administrative databases facilitate the larger-scale exploration of patient care and outcomes during outbreaks of established and emerging diseases. Instituting standards of care that maximize overall patient benefits is essential, not only during pandemic peaks – which lead to high levels of treatment fatigue and have a serious impact on healthcare professional resources – but also more generally, as COVID-19 is likely to become endemic. As the course of COVID-19 in hospitalized patients has become clearer and COVID-specific treatments continue to be developed, clinicians should feel comfortable using established methods of care that include prophylactic/low-dose anticoagulant heparinoids.
Conclusion
Hospitals and patients benefit from lower HRU and costs. This study provides real-world data that in patients hospitalized with COVID-19, use of prophylactic/low-dose anticoagulation is associated with shorter LOS, lower costs, lower proportions of ICU admissions and in-hospital deaths, and better clinical outcomes compared to therapeutic/high dose after controlling for clinical differences. Higher anticoagulant use as a therapeutic intervention did not result in observed improvements in outcomes. Therefore, prophylactic/low-dose anticoagulant therapy may be appropriate for hospitalized patients with COVID-19 and warrants further investigation.
Supplemental Material
sj-docx-1-cat-10.1177_10760296221137848 - Supplemental material for Assessment of Healthcare Resource Utilization by Anticoagulant Heparinoid Dosage Level in Patients Hospitalized with COVID-19
Supplemental material, sj-docx-1-cat-10.1177_10760296221137848 for Assessment of Healthcare Resource Utilization by Anticoagulant Heparinoid Dosage Level in Patients Hospitalized with COVID-19 by Keith Robinson, Laura L. Schott, Tom Matthews, Manu Tyagi, Vivek B. Ajmani, Nancy Sacco and Zhun Cao in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgements
Amy Karon, DVM, MPH, senior medical writer with PINC AI™ Applied Sciences, Premier Inc., provided literature review, manuscript editing, and publication support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Meitheal Pharmaceuticals, Inc. Publication of study results was not contingent on the sponsor's approval or censorship of the manuscript.
Financial Disclosures
Keith Robinson, an employee of Syneos Health, provided consulting services to Meitheal Pharmaceuticals, Inc. on this project. Tom Matthews is a former employee of Meitheal Pharmaceuticals, Inc. and a current employee of Xentria, Inc. Vivek B. Ajmani, Zhun Cao, Laura L. Schott, and Manu Tyagi are employees of Premier, Inc. which was contracted by Meitheal Pharmaceuticals, Inc. for this project. Nancy Sacco is a former employee of Meitheal Pharmaceuticals, Inc. and a current employee of SiteBridge Research, Inc., and Hexima Ltd.
Informed Consent
Informed consent for patient information published in this article was not obtained because the PHD has been certified as deidentified and is not considered human subjects research. Study data and recorded information could not be identified directly or through identifiers linked to individuals.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
