Abstract
Introduction
Immunosuppressive therapy (IST) for acquired hemophilia A (AHA) results in remission within days to months in 60% to 80% of patients. However, little is known regarding the predictors of response.
Aim
This study aimed to identify the factors that influence response to treatment.
Methods
The data of 42 patients with AHA from three hospitals were retrospectively analyzed.
Results
All 42 AHA patients received IST; complete treatment data were available for 34 patients. The response rate was 60% among the 5/34 (14.7%) patients who received steroids alone, 70.8% among the 24/34 (70.6%) patients who received steroids plus cyclophosphamide, and 80% among the 5/34 (14.7%) patients who received steroids plus cyclophosphamide and rituximab. Overall, 29/34 (85.3%) patients achieved CR; 4/34 (13.8%) of them relapsed after a median time of 410 (21–1279) days. Adverse events occurred in 14/34 (41.2%) patients: 13/34 (38.2%) had infections and 1/34 (2.9%) developed pancytopenia. In univariate and multivariate Cox regression analyses, FVIII inhibitor titer ≥20 BU/mL was the only significant prognostic factor affecting time to CR. No variable had significant effect on OS.
Conclusion
FVIII inhibitory antibody titer ≥20 BU/mL appears to be an important predictor of time to complete response in patients with acquired hemophilia A treated with immunosuppressive therapy.
Introduction
Acquired hemophilia A (AHA) is a rare autoimmune disease with an incidence of approximately 1.5 per million per year. 1 In AHA, bleeding occurs due to neutralization of the activity of FVIII by inhibitory antibodies.2–4 Without proper treatment, fatal bleeding may occur, with mortality rate ranging from 9% to 41%. 5 Treatment is with hemostatic therapy to control acute bleeding and immunosuppressive therapy (IST) to eradicate FVIII inhibitory antibodies. In most cases, acute bleeding can be controlled by bypassing agents, 6 while steroids—either alone or in combination with cyclophosphamide—are an effective first-line IST for eradication of FVIII inhibitory antibodies. 7 Although 60% to 80% of patients respond to IST, the response is sometimes delayed and so there remains a risk of fatal hemorrhage during treatment.5,8–10 The treatment goal is to achieve rapid response without increasing risk of immunosuppression-related mortality. It is therefore important to select the appropriate initial therapy for each patient and, for this, the clinical and laboratory parameters that could predict outcome and guide treatment decisions need to be clearly identified.10–16 In previous studies, the prognostic factors associated with response to IST and overall survival (OS) have included residual FVIII level, high FVIII inhibitor titer, age ≥65 years, and malignancy.10,11,17
The aim of this retrospective multicenter study was to identify the factors that influence outcome in AHA patients receiving hemostatic treatment and IST in China.
Patients and Methods
Patients
The data of patients with AHA diagnosed and treated between January 2015 and August 2021 at Henan Provincial People's Hospital and the First Affiliated Hospital of Zhengzhou University and Nanyang Central Hospital were retrospectively analyzed. The clinical history, underlying diseases, physical examination findings, and laboratory test results of the selected patients were extracted from the medical records. The patients were diagnosed based on an acute or recent onset of bleeding, a prolonged activated partial thromboplastin time (aPTT), and reduced FVIII activity. Inhibitor detection screening was performed with the mixing test conducted using the aPTT method, which was considered positive if the aPTT of the mixed normal and the patient's plasma (1:1) incubated for 2 h persisted as prolonged. Inhibitor titre was determined using the Bethesda method. The local laboratory evaluated the aPTT, FVIII: C activity, and inhibitor titre. 18 Patients were eligible for inclusion if they had FVIII: C <50% and FVIII inhibitory antibody level >0.6 BU/mL. 6 Patients diagnosed with congenital hemophilia were excluded. A total of 42 patients met the eligibility criteria and were included in this study. This observational study was approved by the ethic committees of the involved hospitals and was in accordance with the Helsinki Declaration.
Hemorrhage
Hemorrhage was classified as minor or major. Major hemorrhage was defined according to the criteria proposed by Schulman et al.
19
as:
Fatal bleeding Symptomatic bleeding in a critical area or organ (eg, intracranial, intraspinal, intraocular, retroperitoneal, intraarticular, or pericardial hemorrhage, or intramuscular hemorrhage with compartment syndrome Bleeding causing a fall in hemoglobin level of 20 g/L (1.24 mmol/L) or more, or leading to transfusion of two or more units of whole blood or red cells.
Treatment Efficacy
Response to treatment was graded as:
10
Complete response (CR): FVIII: C >50%, undetectable inhibitor level, cessation of bleeding, and discontinuation of IST Relapse: Relapse was defined as the reemergence of a FVIII inhibitor after being in complete remission No response (NR): FVIII: C <50%, persistent inhibitor level, and/or active bleeding
Definitions
The time point of complete hemostasis was determined by the treating physician through chart review and was based on hemoglobin level and clinical and imaging findings. Time to CR was defined as the number of days from the start of IST to the achievement of CR. 10 The primary endpoint was the time to achieve CR after IST, and the secondary endpoints were hemostasis efficacy, CR rate, and OS.
Statistical Analysis
Descriptive analysis was performed for patient demographics, clinical characteristics, and laboratory indicators. Continuous variables were expressed as medians and ranges or interquartile ranges (IQRs), and compared using Student's t-test or Mann–Whitney U test. Categorical variables were expressed as percentages and compared using the Pearson's chi-square test or Fisher's exact test, as appropriate. The Kaplan–Meier method and the log-rank test were used to analyze time to CR and OS rate. Univariate and multivariate Cox regression analyses were performed to identify the factors associated with time to CR and OS. SPSS 26.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. Statistical significance was at P < .05.
Results
Baseline Characteristics of Patients
Table 1 shows the baseline characteristics of the 42 patients included in this study. The median age at diagnosis was 52 (9–92) years. Age and gender distribution revealed a small proportion of children and adolescents, with 42.9% of the overall cohort above 60 years (Figure 1). 11 of 18 (61.1%) of the senior age groups were dominated by men. In contrast, there was a female preponderance (10/13, 76.9%) in the childbearing age range (20–40 years). There was no significant correlation between FVIII antibody titers and FVIII levels (Figure 2).
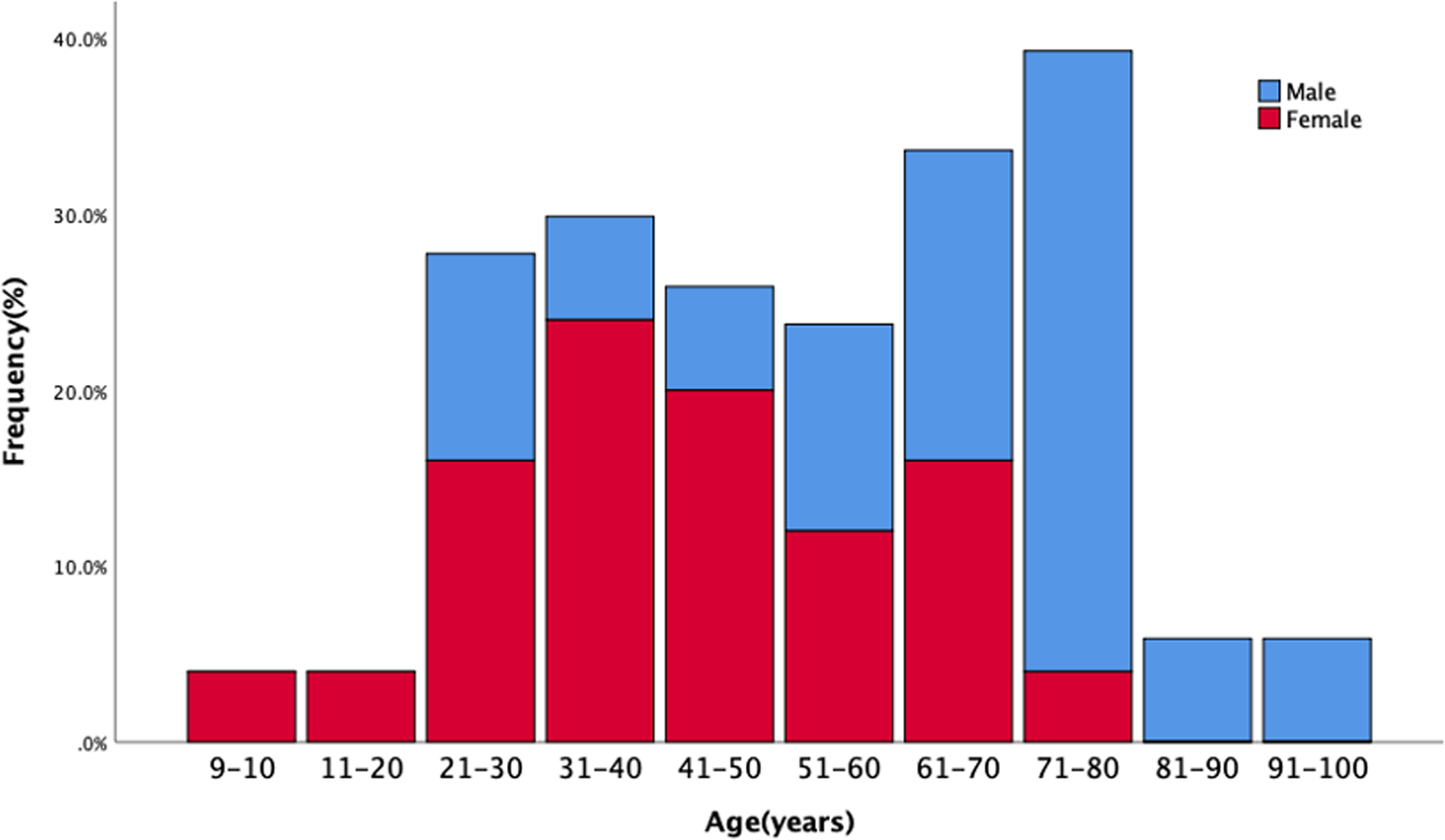
Distribution of patients diagnosed with acquired haemophilia A by age range and sex.

Analysis of FVIII level and FVIII antibody titre using linear regression.
Baseline Characteristics of Patients with Acquired Haemophilia A.

Drug: Aspirin (1 case).
Bleeding Symptoms and Hemostatic Therapy
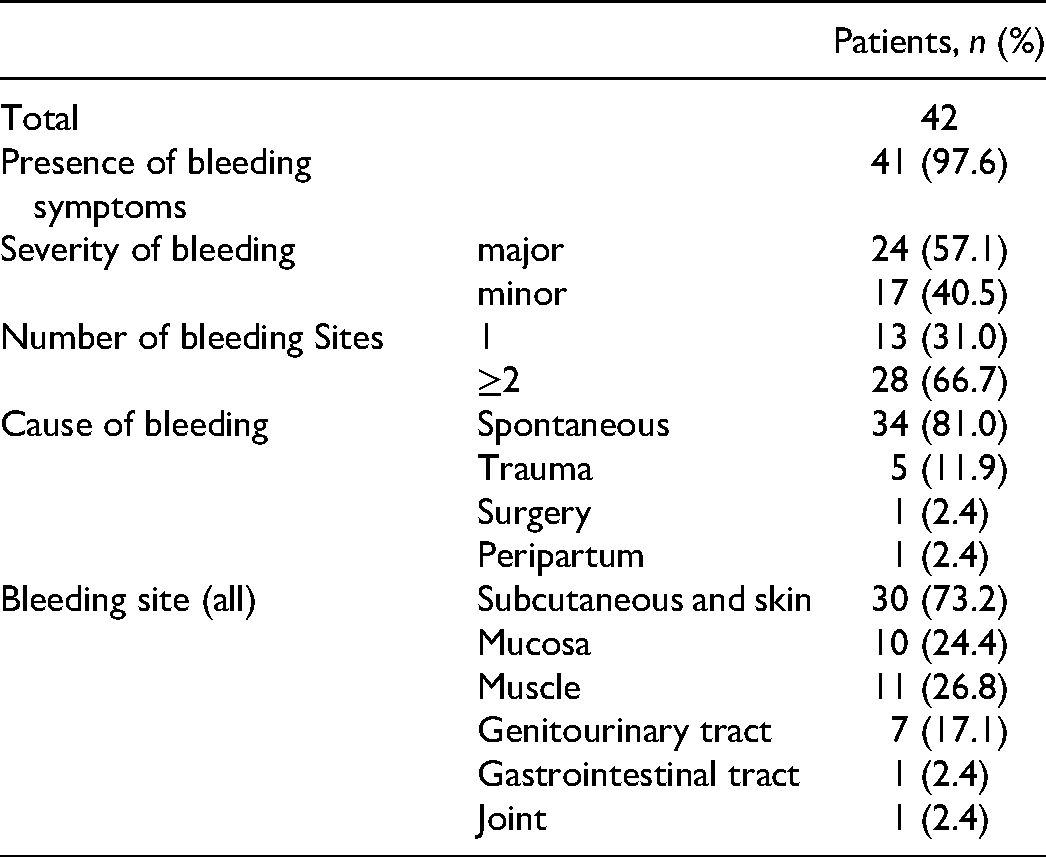
While 41/42 (97.6%) patients presented with bleeding, 1/42 (2.4%) patient only had prolonged activated partial thromboplastin time (aPTT) at admission and was diagnosed with AHA later. The median time from the first bleeding event to diagnosis (diagnostic delay) was 31.5 (range 0–684) days. The diagnostic delay was >30 days for 22/41 (53.7%) patients, 7–30 days for 17/41 (41.5%) patients, and <7 days for 2/41 (4.9%) patients. Table 2 summarizes the characteristics of the bleeding.
Summary of Characteristics Associated with Bleeding Symptoms.
39/41 patients (95.1%) received hemostatic treatment (Table 3). In general, bypass medications were employed regardless of FVIII: C in individuals with high inhibitor titers. 28/39 (71.8%) patients underwent hemostatic therapy combined first-line therapy with adjuvant therapy, while 11/39 (28.2%) patients used adjuvant therapy alone. Adjuvant therapy included: red blood cell transfusion (n = 10), antifibrinolytics (n = 10), artificial thrombin (n = 11), and other: fresh frozen plasma/cold precipitation (n = 26) (Table 3).
Summary of Haemostatic Treatment.

Abbreviations: PCC, prothrombin complex concentrates; NA, not-available; rFVIIa, an activated recombinant factor VII; Other: Fresh frozen plasma/Cryoprecipitate.
In China, activated prothrombin complex (aPCC; FEIBA®) was unavailable; therefore, prothrombin complex (PCC) was employed as a substitute. Among 28 patients treated for initial bleeding episodes, 15/28 (53.6%) patients received FVIII replacement therapy, 11/28 (39.3%) patients received PCC, and 2/28 (7.1%) patients received rFVIIa. There were no significant differences in age, sex, FVIII: C, inhibitor titer, and bleeding severity between the two groups of patients treated with FVIII replacement therapy and PCC. Bleeding was controlled in 8/11 (72.7%) patients treated with PCC and 7/15 (46.7%) patients treated with FVIII replacement therapy. There was no statistically significant difference in efficacy between the two groups treated with PCC or FVIII replacement treatment (OR = 3.048, 95% CI: 0.574–6.194, P > .05).
In 3/11 (27.3%), patients who did not respond to initial treatment with PCC, bleeding was controlled after recombinant activating factor VII (rFVIIa) treatment. 8/15 (53.3%) patients who failed initial FVIII replacement therapy, six patients received PCC as salvage therapy, and two patients received a combination of rFVIIa and PCC. Bleeding was controlled in 6/6 (100%) patients who received PCC as salvage therapy and in 1/2 (50%) patients who received a combination of rFVIIa and PCC. Only one patient failed bypass therapy; This patient was a pregnant woman with multiple systemic hemorrhages after cesarean delivery. She had a maximum inhibitor titer of 308.6 BU/mL, FVIII: C <1 IU/dL, and IST refractoriness. Eventually, she died of uncontrollable bleeding on day 14 after diagnosis.
Immunosuppressive Therapy and Adverse Events
All 42 patients received IST to eradicate antibodies, but details of treatment were available for only 34 patients. 34/34 (100%) patients received prednisolone (1–1.5 mg/kg/d) or an equivalent steroid as part of inhibitor eradication therapy; 32/34 (94.1%) patients received cyclophosphamide (1.5–2 mg/kg/d Oral or intravenous) combination therapy; 11/34 (32.4%) patients received rituximab (375 mg/m2/w for 4 weeks) as part of either first-line, second-line, or salvage therapy; and 2/34 (5.9%) patients received other immunosuppressive agents such as cyclosporine or sirolimus. The median follow-up time after the first IST was 116 (13–1372) days. Overall, 29/34 (85.3%) patients achieved CR, with median time to CR of 56 (13–309) days. Figure 3 shows the IST used and the response to treatment.
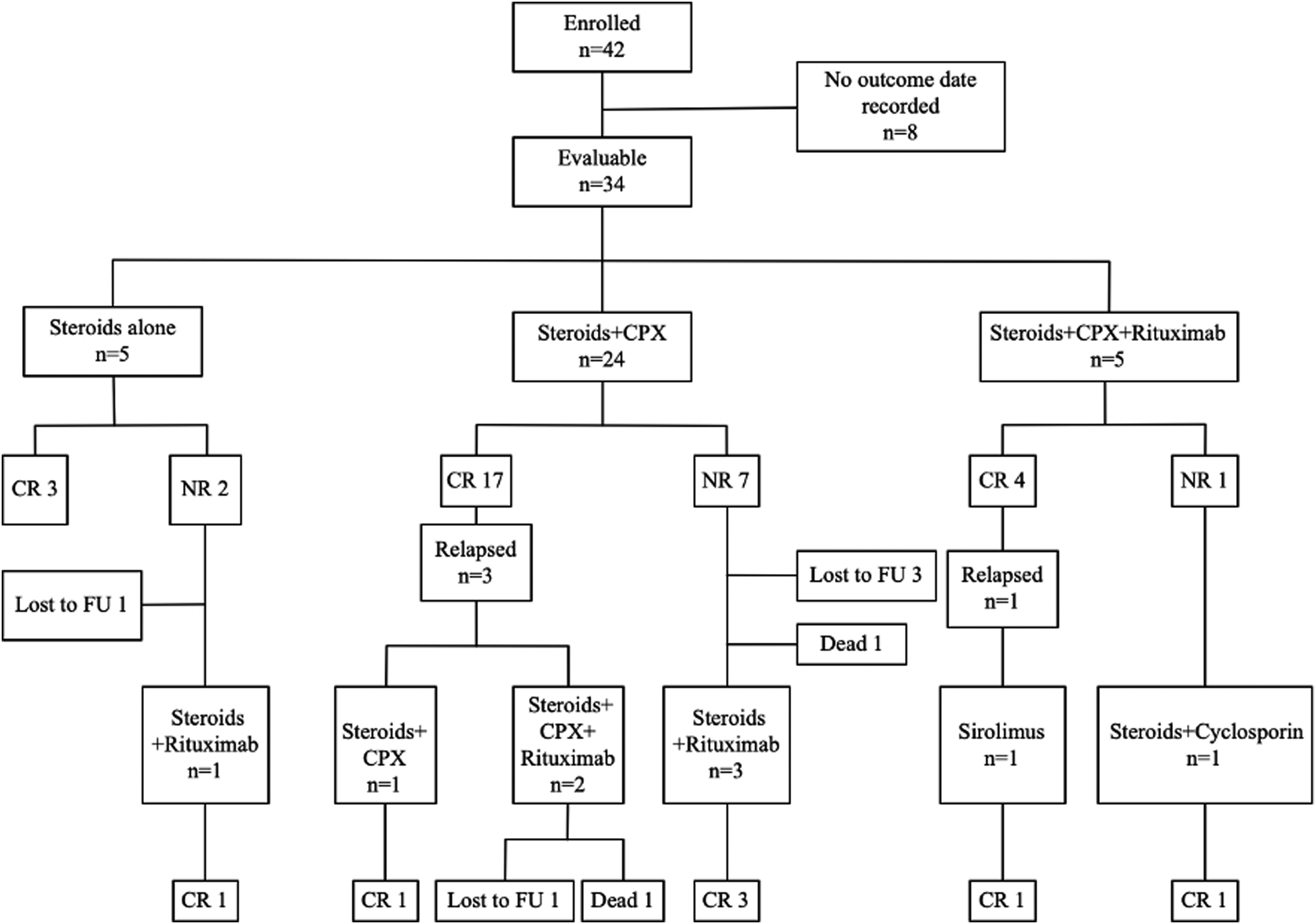
Flow and outcome of immunosuppressive therapy. Abbreviations: IST, immunosuppressive therapy; CPX, cyclophosphamide; CR, complete remission; PR, partial remission; NR, no response; Ref, refractory; FU, follow-up.
Among 5/34 (14.7%) patients treated with steroids alone, the response rate was 60.0%; one patient who did not respond to steroids monotherapy responded to second-line IST with steroids plus rituximab.
Steroids plus cyclophosphamide was used as IST in 24/34 (70.6%) patients. CR was achieved in 17 patients (70.8% response rate). However, three patients relapsed. Of these three patients, one achieved lasting CR after 13 days with continuation of steroids plus cyclophosphamide as salvage regimen. The other two patients received steroids plus cyclophosphamide and rituximab as salvage regimen, but both patients failed salvage IST. Of these two patients, one relapsed 1279 days after achieving CR on first-line therapy and was later diagnosed with adenocarcinoma of the pulmonary mucosa; the patient was eventually lost to follow-up. The other patient relapsed 777 days after achieving CR on first-line therapy and was later diagnosed with prostate cancer; the patient eventually died of refractory hemorrhage. Of the seven patients who failed first-line therapy with steroids and cyclophosphamide, three received steroids plus rituximab as second-line therapy, and all three achieved CR (100% response rate). One of the patients died due to refractory bleeding despite hemostatic therapy and IST. The other three patients showed no response even after 4 to 6 weeks of treatment and were eventually lost to follow-up; two of the three patients did not achieve PR, but had an increase in VIII activity.
Of the five (14.7%) patients who received steroids plus cyclophosphamide and rituximab as first-line IST, four achieved CR (response rate of 80.0%; Table 4). One of the four patients relapsed 21 days after achieving CR; the patient then received sirolimus as salvage regimen and re-achieved CR at 14 days after start of sirolimus. The one patient who failed first-line therapy with steroids plus cyclophosphamide achieved CR after treatment with steroids plus cyclosporine as second-line therapy.
Complete Remission Rates of First-Line Immunosuppressive Therapies Based on the Treatment Regimens of 34 Patients who Received IST.

Data are presented as number of patients with complete remission/number of treated patients (%).
*P value = .023 when compared to patients with FVIII Ab<20 BU/mL.
**P value = .002 when compared to patients with FVIII Ab<20 BU/mL.
IST-related adverse events occurred in 14/34 (41.2%) patients; the adverse events included infections (13/34, 38.2% patients) and pancytopenia (1/34, 2.9% patients).
Prognostic Factors for Remission
The impact of the following factors on time to CR and 12-month OS were evaluated: sex (male vs female), age (<65 vs ≥65 years), WHO-PS >2, underlying disorders (autoimmune disease, malignancy), FVIII: C level (<1% vs 1%–50%), and inhibitor titer (<20 BU/mL vs ≥20 BU/mL). To reduce bias, we exclude the rituximab-based regimen group from the analysis.
In univariate Cox regression analysis, only FVIII inhibitor titer ≥20 BU/mL was a significant prognostic factor for time to reach CR (HR, 0.31; 95% CI, 0.12–0.80; P = .01; Table 5). Sex, age ≥65 years, WHO-PS >2, malignancy, autoimmune disease, and FVII: C <1%, did not affect the time to CR. Of note, among patients treated with steroids plus cyclophosphamide as first-line IST, the CR rates were lower in patients with inhibitor titers ≥20 BU/mL than in patients with inhibitor titers <20 BU/mL (45.5% vs 92.3%, P = .023, Table 4). Time to reach CR was significantly longer in patients with FVIII inhibitor titers ≥20 BU/mL than in patients with inhibitor titers <20 BU/mL: 77 days (95% CI, 0.0-226.8) versus 44 days (95% CI, 23.0–65.0), P = .01 (Figure 4).

Time to achieve complete remission. (A) Total patients, (B) The patients were divided into two groups according to the antibody titre: patients with FVIII antibody titres ≥20 BU/mL (red line n = 16) and patients with FVIII antibody titres <20 BU/mL (blue line n = 21).
CR According to Baseline Characteristics: Univariate Analysis.
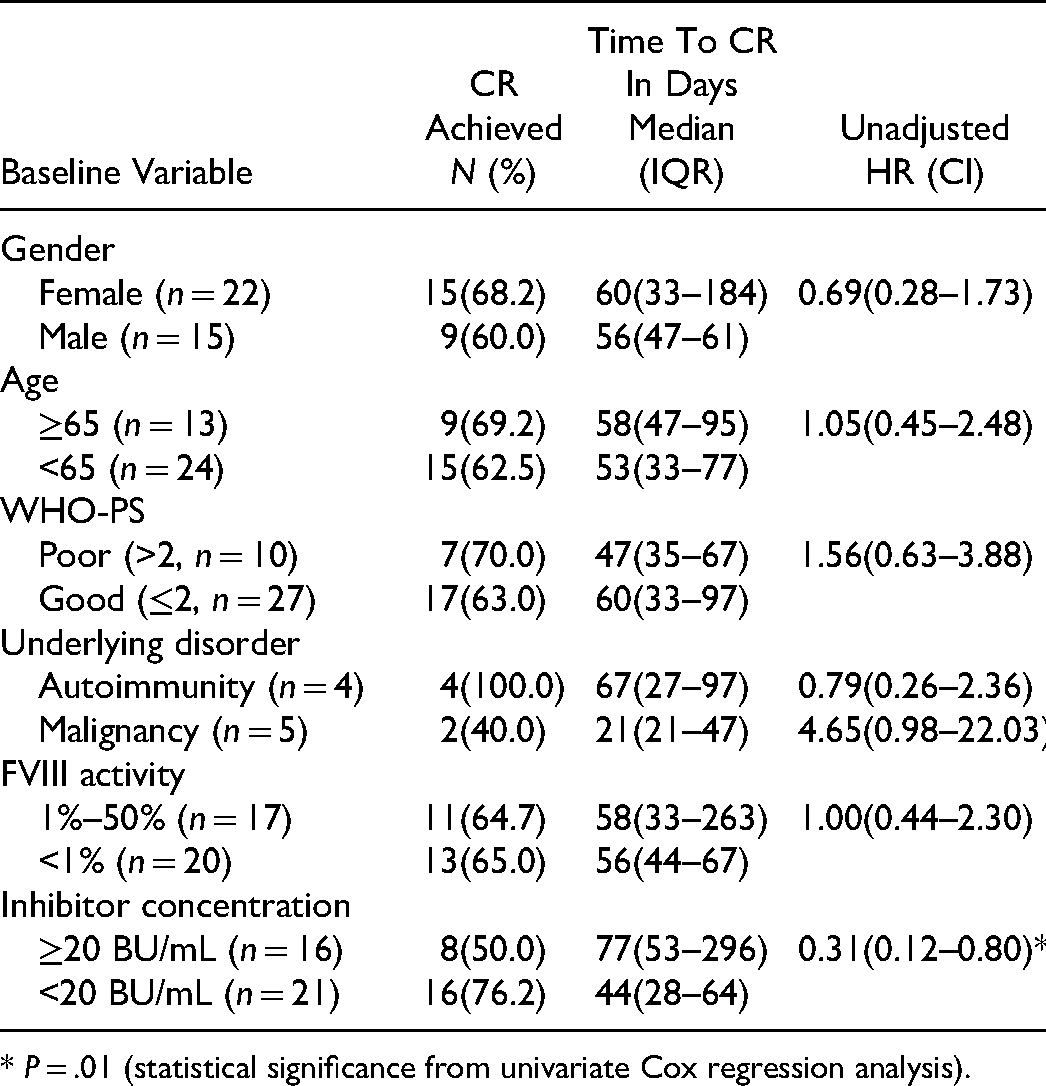
* P = .01 (statistical significance from univariate Cox regression analysis).
In multivariate Cox regression analysis, inhibitor titer ≥20 BU/mL was the only significant factor affecting time to CR (HR, 0.07; 95% CI, 0.02–0.31; P < .001; Table 6). The 12-month OS rate was 97.3%. In Cox regression analysis, none of the evaluated factors had a significant effect on 12-month OS.
Predictors of Remission: Multivariate Analysis.

Data are presented as adjusted HR (CI).
*P < .01.
Discussion
This study is a multicenter retrospective analysis of AHA in central China to investigate the current status of AHA and to identify factors associated with response to IST. Only FVIII inhibitor antibody titer was a predictor of early complete response after IST. Patients with FVIII inhibitor titers ≥20 BU/mL took more time to achieve CR and had lower CR rates. This is the fourth study reporting FVIII inhibitor antibody titer ≥20 BU/mL as a predictor of response to IST.10,15,17
AHA is a rare bleeding disorder that occurs predominantly in adults. Our data show a female predominance, which is consistent with larger-scale studies in the UKHCDO cohort and the CARE cohort, but contrary to the male predominance observed in the AHASR cohort.9,20,21 Incidence increases with age, with a median age of approximately 78 years in some studies.9,21,22 The age distribution was biphasic, with the primary peak occurring between 68 and 80 years and the second between 20 and 30 years.5,10,23 In our cohort, the two peaks were consistent with previous studies. However, the median age (52 years) and primary peak age were much younger than those reported in other studies. Interestingly, our cohort's median age was the same as that of the CARE cohort. 20 We hypothesize that referral bias may have had some effect on this cohort. Younger people with AHA are more likely to be referred for further consultation than older patients.
A high number of idiopathic AHA cases were described, and the most common underlying illnesses, autoimmune disorders and malignant, were consistent with those previously reported.5,8,9,24–30
The bleeding pattern was not different from previously reported, with a high incidence of subcutaneous and cutaneous mucosal bleeding. Most patients are seen for spontaneous bleeding, and the lack of awareness of the disease has led to delayed and missed diagnoses. In the EACH2 study, 5 the median time from presenting bleeding symptoms to diagnosis was only 3 days. In the study of AHASR, 21 the median time was 17 days. Our data shows the median time from the onset of bleeding to diagnosis was 31.5 (0–684) days, which is similar to the CARE data (30 days), indicating that patients are at high risk of severe bleeding for a more extended period. Delayed diagnosis may due to two factors: lack of awareness of AHA or facilities to measure FVIII: C and inhibitor titers, indicating the unmet need in the diagnosis of AHA, especially in countries with limited resources.
Although bypass agents such as recombinant activated FVII (rFVIIa) and activated PCC (aPCC) are recommended as the best hemostatic treatment,6,12 they may not be available or affordable to Chinese patients. Lusher et al. 31 compared PCC with aPCC from the same manufacturer and found no difference in efficacy; response rates were 50% for PCC and 51.7% for aPCC. Our results also suggest that PCC is effective and safe for controlling acute bleeding when compared with previous studies using aPCC.5,32 The main drawback of PCC is the risk of thrombotic complications. However, in the present study, no thrombotic events were observed. So PCC is a reasonable option for hemostatic treatment when rFVIIa and aPCC are unavailable or unaffordable. In our study, rFVIIa was mainly used as second or third-line salvage therapy for patients with poor response to PCC or FVIII.
In this study, the CR rate for first-line IST was 70.6%, which is consistent with previous studies.5,8–10 The EACH2 study showed a higher CR rate in patients receiving combination therapy (steroids plus cyclophosphamide) than in those receiving steroids monotherapy (85% vs 58%). However, the UKHCDO study found no difference between the two regimens.9,11 Our study also found no significant difference in response rates between patients receiving the two regimens, but this may be due to the small number of patients in the cohort receiving steroids as monotherapy. In this study, one patient who received steroids plus cyclophosphamide and rituximab as first-line therapy did not respond; this patient eventually achieved CR after receiving steroids plus cyclosporine as a second-line regimen. Another patient who relapsed after receiving steroids plus cyclophosphamide and rituximab eventually recovered after receiving sirolimus as a salvage regimen. These data suggest immunosuppressive agents other than those routinely used could be helpful as second-line and salvage therapy. Some of our patients received rituximab-based regimen as first-line therapy, with a CR rate of (80.0%). Studies of the CARE and the AHASR both found that rituximab-based regimens achieved good outcomes and did not take a long time to achieve CR.20,21 Also, in a recently published study, 22 a CyDRi regimen was used and achieved a high (96.9%) stable CR and low treatment-related toxicity.
In the present study, of a total of 29 patients who achieved CR, four (13.8%) relapsed after a median time of 410 (21-1279) days. Of the four patients, two achieved CR after salvage therapy. However, one of these patients relapsed again and was diagnosed with mucinous adenocarcinoma of the lung; the patient was lost to follow-up after surgical treatment. The other patient also suffered another relapse and was later diagnosed with prostate cancer; the patient subsequently died of refractory bleeding. Thus, two of the four patients (50%) who relapsed in our cohort were diagnosed with malignant tumors after recurrence, which suggests that evidence of tumor, autoimmune disease, and other etiologies should be sought in patients who relapse. 29 14/34 (41.2%) patients in our cohort experienced treatment-related side effects, possibly related to the higher proportion of patients receiving steroids combined with cyclophosphamide. However, there was no incidence of death due to infection, sepsis, or neutropenia, which may be related to the younger median age of our cohort. Two deaths (2/34, 5.9%) were recorded in our cohort: both deaths were due to refractory bleeding (one patient after cesarean section).
Several authors have attempted to identify the predictors of response to IST and to establish an effective treatment protocol. In the EACH2 registry study, inhibitor titer <16 BU/mL and high level of FVIII: C were reported as predictors of rapid response. 11 In the prospective GTH2010 study, inhibitor titer ≥20 BU/mL was reported as a predictor of poor CR; however, in patients with FVIII level ≥1 IU/dL and inhibitor titer <20 BU/mL, there was 11 times higher probability of achieving PR with steroids monotherapy within 21 days. 10 A French study also reported that patients with FVIII level ≥1 IU/dL and inhibitor titer <20 BU/mL had a shorter time to PR after steroids monotherapy (15 days vs 41 days). 8 Consistent with the GTH2010 study, our study showed that inhibitor titer ≥20 BU/mL was a predictor of IST response. Further, among patients receiving steroids plus cyclophosphamide as first-line IST, we found significant difference in CR rates after stratification according to inhibitor titers (<20 BU/mL vs ≥20 BU/mL): CR rates, 92.3% versus 45.5%, respectively (P = .023; Table 4; Figure 3).
This study has some limitations. Most importantly, this was a retrospective study with a small sample size, so bias is inevitable. Although this study may not have obtained accurate demographics of AHA patients, the results are still meaningful. A national prospective study using risk-stratified AHA treatment protocols would help accurately identify the epidemiology of AHA in China.
Conclusions
This retrospective multicenter study from China suggests that FVIII inhibitory antibody titer is a predictor of early complete response after IST in patients with acquired hemophilia A. Patients with FVIII inhibitory antibody level ≥20 BU/mL take longer to achieve complete response and have lower rates of complete response after IST. These patients should therefore be considered for steroids and another immunosuppressant (cyclophosphamide or rituximab) as first-line therapy, and steroids plus sirolimus or cyclosporine as second-line or salvage therapy. Future prospective studies or registry analyses should attempt to identify other clinical or laboratory variables affecting time to complete response and overall survival. Such information will help physicians achieve optimal outcomes.
Footnotes
Acknowledgements
This work was supported by Science and Technology Department of Henan Province (No. 222102310152) and Department of Human Resources and social Security of Henan Province (No. CKQ20220001).
Author Contributions
Concept and design: Yanhui Liu, Xiang Ruan; Acquisition of data: Yanhui Liu, Xiang Ruan, Pingchon Lei, Baojun Shang, Zunmin Zhu, Shengmei Chen, Dao Wang, Ruijuan Wang, Xiqing Li, Fei Xue; Analysis and interpretation of data: Yanhui Liu, Xiang Ruan; Writing the manuscript: Yanhui Liu, Xiang Ruan; Revising the manuscript: Yanhui Liu, Xiang Ruan.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Human Resources and Social Security of Henan Province, Science and Technology Department of Henan Province, (grant number CKQ20220001, 222102310152).
