Abstract
Background
The D-dimer (DD) assay is an essential biological test for the diagnosis and monitoring of thrombotic conditions. DD testing is usually not performed as part of the routine laboratory management of patients with hemophilia (PWH). There is an increasing concern about the risk of thrombotic complications in PWH, which is likely related to age, cardiovascular risk factors, invasive thrombogenic procedures, over-correction of Factor VIII (FVIII) or FIX, or administration of new therapeutic agents mimicking FVIII or rebalancing coagulation.
Objective
This retrospective study sought to assess the basal DD levels in PWH treated prophylactically with FVIII, and to evaluate potential changes after switching to emicizumab.
Method
Patients over 18 years of age treated with emicizumab within a single center over the period 2017-2022 were included in the study.
Result
DD levels were measured in 40 adult PWH (37 severe/ three moderate / two with FVIII inhibitor) with a median age of 46 years (range: 19-82; Q1-Q3: 30,25-56,5)
Conclusion
Most adult PWH on prophylaxis using FVIII display DD levels within the normal range, which remain unchanged after switching to emicizumab. In view of these reassuring results, monitoring of DDs at the start of emicizumab treatment does not appear necessary but could be considered when combined with other bypassing agents or high dose FVIII.
Keywords
What is the NEW aspect of your work? Evaluation of D-dimers before and after initiation of emicizumab in haemophilia patients. What is the CENTRAL finding of your work: Initiation of emicizumab treatment does not influence d-dimer kinetics What is (or could be) the SPECIFIC clinical relevance of your work? These data support that there is no increased thrombogenicity under emicizumab therapy
Introduction
Hemophilia A (HA) is a congenital X-linked bleeding disorder caused by Factor VIII (FVIII) deficiency, with a prevalence of 17.1/100.000 males. 1 It is further classified into mild, moderate, and severe according to residual FVIII levels. 2 For patients with hemophilia (PWH) exhibiting a severe bleeding phenotype, the only available treatment has long been intravenous prophylaxis using FVIII concentrates in order to reduce and prevent joint, muscle, and soft tissue bleeding complications, as well as life-threatening bleeds like intracranial hemorrhage. 3 However, the burden of intravenous injections administered up to several times a week, residual risk of bleeding, and development of neutralizing antibodies in 30% of cases represent major limitations to replacement based on FVIII concentrates. 4 Given this context, non-substitutive therapies have been developed, validated, and introduced, including emicizumab. 5 This agent is a humanized bispecific monoclonal antibody with activated FIX and FX binding sites, mimicking FVIII in its cofactor activity. Emicizumab has been largely adopted and is now being used in around 10,000 patients worldwide, with or without neutralizing antibodies.6,7
There is a growing concern about the risk of thrombotic complications in PWH, whether related to age, concomitant cardiovascular risk factors, thrombogenic invasive procedures, over-correction of FVIII, or administration of new agents like emicizumab. Arterial and venous thrombotic events have indeed been reported in PWH, who were treated with emicizumab combined with activated prothrombin complex concentrates, 8 as well as in patients treated with rebalancing agents, namely a tissue factor pathway (TFPI) monoclonal antibody 9 and antithrombin-lowering agents. 10 It is therefore paramount to be able to rely on simple and readily available biological tests allowing for detecting and monitoring the development of any prothrombotic state in PWH who are treated with these agents, thus possibly at increased risk of thrombosis.
The D-dimer (DD) assay represents an important assay for the diagnosis and monitoring of venous thrombotic conditions. 11 The DD assay is not typically part of routine laboratory management of PWHs. This retrospective study sought to assess the basal DD levels in PWH treated with FVIII concentrates, and to evaluate their possible alterations following emicizumab initiation.
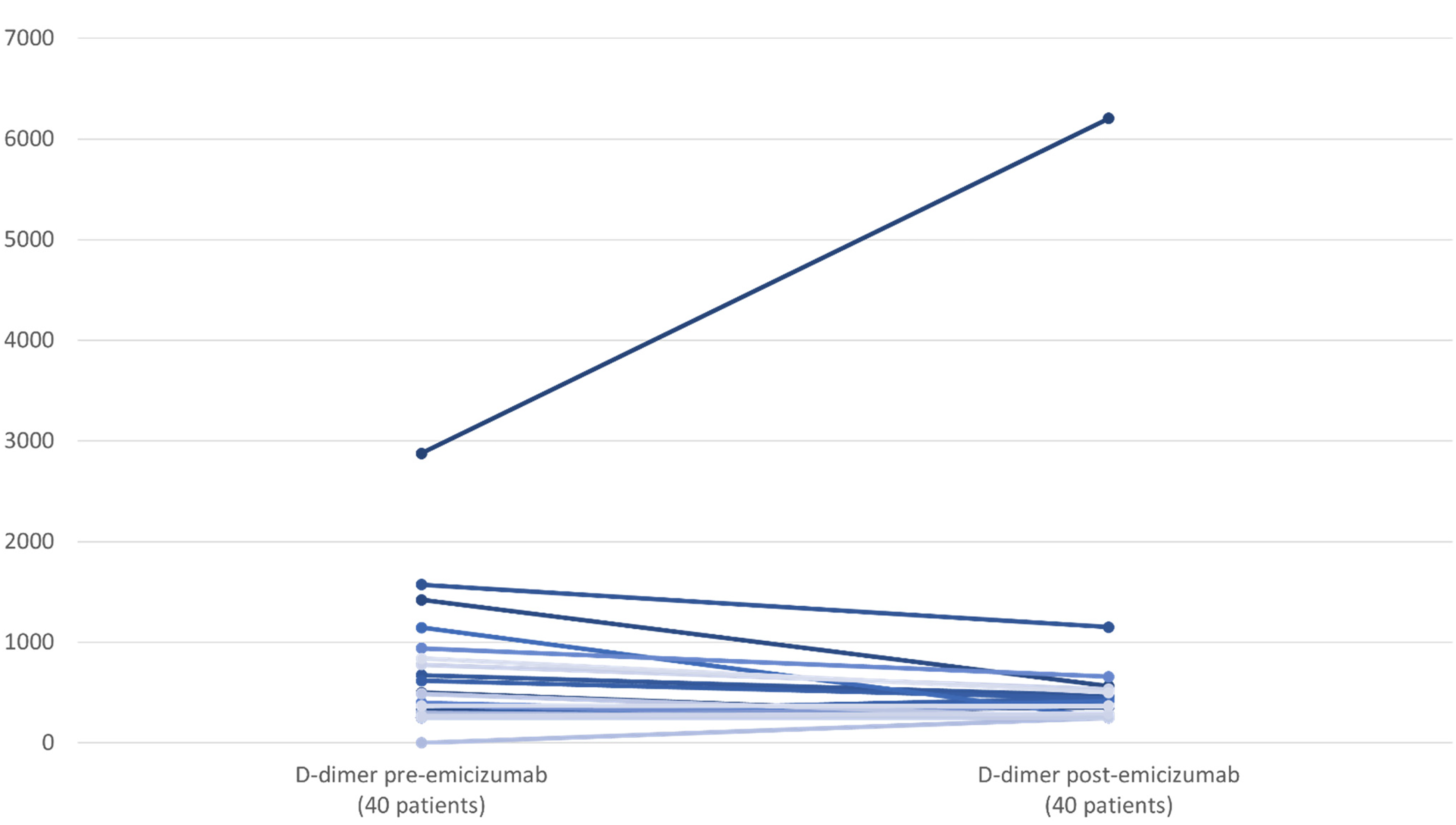
Evolution of D-dimer after emicizumab initiation.
Patients and Methods
We retrospectively included all PWH treated with emicizumab at the Saint Luc University Hospital (UCLouvain, Brussels, Belgium) during the period 2017-2022. Among the 76 patients identified, 23 were excluded because of their young age (< 18), and 13 patients were excluded due to lack of biological data three months after initiation of emicizumab. Blood parameters (FVIII, C-reactive protein [CRP], fibrinogen [Fg] and DD) measured before and after emicizumab treatment initiation were available for 40 patients. The median time between the two blood tests was 791 days, with a single blood test (including DD) performed 445 days (median value) after initiation of emicizumab. FVIII concentrates were measured using chromogenic assay with human reagents before and after emicizumab initiation (Biophen FVIII:C; Hyphen biomed). DD determination was performed using a latex enhanced turbidimetric immunoassay (HemosIL D-Dimer HS 500). Data collection was approved by the ethics committee at Saint-Luc University Hospital (UCLouvain, Brussels, Belgium).
Statistical Analysis
The data were analyzed using Excel software and expressed as median. Shapiro test confirmed that the collected data were not normally distributed, which could be expected given the relatively small sample size. Mean differences between pre- and post- emicizumab initiation were therefore evaluated using the non-parametric Wilcoxon signed-rank test, with each test performed at a 5% alpha level. In addition, a non-parametric Spearman's rank correlation test was applied to evaluate correlation between data (FVIII and DD levels). P value <0.05 was considered statistically significant.
Results
The 40 (39 males and 1 female) participants displayed a median age of 46 years (range: 19-82; Q1-Q3: 30,25-56,5), and suffered from severe (n = 37) to moderate (n = 3) hemophilia A, either with (n = 2) or without inhibitor (n = 38). Prior to emicizumab initiation, all participants were treated prophylactically with FEIBA® (n = 2), Advate® (n = 12), Elocta® (n = 8), Factane® (n = 2), Adenovy® (n = 2), Refacto® (n = 6), Octanate® (n = 1) or Kovaltry® (n = 7). The biological data were collected during routine consultation visits prior to emicizumab introduction and at least 3 months thereafter. None of the 53 patients on emicizumab discontinued the treatment. None of the 53 patients exhibited thromboembolic complications, nor were any on them receiving preventive or curative antithrombotic treatment.
Our cohort displayed a high prevalence of cardiovascular risk factors, despite a fairly young average age, as reflected by the numerous patients with diabetes (n = 6), arterial hypertension (n = 11), coronary artery disease (n = 1), dyslipidemia (n = 4), obesity (n = 5), smoking (n = 11), alcohol consumption (n = 4), or sedentary lifestyle favored by joint morbidity (n = 12). In our cohort, there was additionally one case of hepato-carcinoma, three cases of human immunodeficiency virus (HIV) infection with undetectable viral loads, as well as 20 patients with HCV infection (hepatitis C virus), yet all eradicated (Table 1).
Baseline Demographics and Disease Characteristics.

†: Hepatocarinoma, hypothyroidism.
Prior to initiating emicizumab treatment (Table 2), the median FVIII concentration on prophylactic treatment was 3 IU/mL (range: 1-47; Q1-Q3: 1-8,5), median CRP concentration was 1 mg/dL (range: 1-114; Q1-Q3: 1-3,8), median Fg concentration was 375,5 mg/dL (range: 267-578; Q1-Q3: 330-417), and median DD concentration was 257 ng/mL (range: 250-2876; Q1-Q3: 250-493,5). Nine patients had an age-adjusted DD level above normal (cut-off of age-adjusted DD obtained by age*10 in ng/mL, for patients over 50 years old). This was later explained by conditions as follows (Table 3): mild unexplained inflammatory syndrome characterized by a C-reactive protein under 20 mg/L without identified infection, autoimmune disease, or cancer (n = 3), an active neoplasia (n = 1), recent muscle tear (n = 1), Covid-19 infection (n = 1), recently suspected hemarthrosis (n = 1), as well as skin ulcer (n = 1).12, 13
FVIII, Fibrinogen, CRP, and D-dimer, before and after Initiating Emicizumab Prophylaxis.

Patients with D-dimer Elevation before or after Initiation of Emicizumab.

Blood control was performed minimum three months after initiating emicizumab with a median of 445 days, no significant changes in tested parameters were revealed (Table 2), with median values of 373,5 mg/dL (range: 171-678; Q1-Q3: 340,25-398) for Fg (p = 0.8), 1 mg/dL (range: 1-10,4; Q1-Q3: 1-3,15) for CRP (p = 0.6), and 250 ng/mL (range: 250-6205; Q1-Q3: 250-380,25) for DD (p = 0.9). These assays were performed as part of a follow-up visit in the absence of active bleeding, hospitalization or adjunctive therapy with FVIII or FEIBA®. As expected, the mean basal FVIII level measured by chromogenic assay with human reagents was 29% (range: 5-48; Q1-Q3: 17,25-33,5) (p = 0.01), clearly reflecting the emicizumab effect. DD levels remained unchanged in 17 patients. The four patients with increased DD on emicizumab were older and had significantly higher CRP and Fg (Table 4). No correlation was found between measured FVIII and DD levels. Clinically, no thromboembolic events were reported during the study period.
Differences between Patients with Normal DD Vs Positive DD on Emicizumab.

Discussion
The DD assay is a very important diagnostic tool for managing suspected venous thromboembolic disease (VTE) within the general population, with a high negative predictive value. 11 Nevertheless, the DD assay has not yet been validated in PWH, given that thromboembolic disease is rather unusual in this population, even in the event of major orthopedic procedures.12,14 The estimated prevalence of VTE among patients with inherited bleeding diseases has been estimated at 1/27,000 according to Girolami. 15 Depending on the EUHASS registry, 331 cases of thrombosis were reported between 2008 and 2022. 16 Due to VTE's low prevalence in PWH, DD has not yet become a routine monitoring parameter, as evidenced by the recent publication pertaining to coagulation tests in PWH under emicizumab treatment. 17 Indeed, this article described the biological disturbances related to using emicizumab, and proposed recommendations on activated partial thromboplastin time (aPTT), FVIII/FIX/FXI/and FXII, protein S assays, in addition to tests for lack of response to activated protein C (APC-R) assays, yet without reporting its impact on DD kinetics. However, this paradigm is likely to be disrupted owing to a higher thromboembolic risk (arterial and venous), due to an increased average age within this population, concomitant presence of cardiovascular risk factors (as seen in our population), and the performance of thrombogenic invasive procedures and potential overcorrection of FVIII. 18 This is likely further exacerbated by the increasing use of emicizumab, as this compound has been associated with improved bleeding control in PWH with and without inhibitors. 19 Yet, of note is that this agent also increases thrombotic risk when used in conjunction with bypassing agents like activated prothrombin complex concentrates (FEIBA®). Moreover, thrombotic events have been reported in patients receiving a monoclonal antibody and antithrombin lowering agent.20,21
The HAVEN 1 study evaluated the emicizumab efficacy in terms of bleeding control in hemophilia A patients with an inhibitor, in addition to its safety, as assessed by measuring Fg, DD, FIX, FX, and prothrombin. 5 In this study, no significant DD elevation was recorded compared to the control group. A DD elevation was found in two patients, which was, however, explained by their aPCC (FEIBA®) use. This was confirmed in vitro by demonstrating excessive thrombin generation upon aPCC addition in emicizumab-treated patients. 22 In clinical terms, excessive thrombin generation was shown correlated with development of thrombosis and thrombotic microangiopathy, when emicizumab and FEIBA® (cumulative dose >100 U/kg/24 h) were concomitantly given. 23 Based on this finding, FEIBA® should be avoided not only during emicizumab's 30-day half-life, but also for at least 6 months thereafter. Indeed, during this period, residual emicizumab levels can persist in plasma, as suggested by recent guidelines. In case of absolute necessity, lower aPCC doses (not exceeding either 50 IU/Kg or 100 IU/Kg total per day) should be employed, together with close patient monitoring as regards the development of thrombosis or thrombotic microangiopathy. 24 The use of emicizumab was also evaluated in elderly patients (>50 years) with a higher cardiovascular risk profile than previously studied populations. During an observation period of 400 days, hemostatic efficacy was confirmed with no increase in arterial thrombotic events. 25
To the best of our knowledge, this is the first study to provide assessment of the level of DD in the real life, before and after initiation of emicizumab. Indeed, this assay is not typically carried out in routine practice, and only scarcely described in the literature. Based on our study data, most PWH on FVIII prophylactic replacement therapy displayed low or unmeasurable DD levels, as previously demonstrated by Tardy-Poncet in a PWH A/B cohort with minor to severe hemophilia. 26 The reasons for high DD levels detected in some PWH do not differ from those recorded in the general population. In our small cohort, concurrent infection, neoplasia, or bleeding states clearly explained the DD elevations that were observed in some patients. A significant DD (>2000 ng/ml) elevation was indeed recorded in one patient exhibiting a progressing neoplasia. The main finding of our study confirmed that switching to emicizumab did not impact DD levels (Figure 1).
Even if only few studies have investigated DD levels in PWH who were treated with FVIII concentrates or new agents, the DD assay is now being included as a monitoring tool in ongoing studies, such as the evaluation of fitusiran (https://www.clinicaltrials.gov/: NCT03417102), and anti-TFPI (https://www.clinicaltrials.gov/: NCT02531815). Initiation of Fitusiran or Concizumab in hemophiliac and healthy individuals has been shown to increase DD and prothrombin fragment 1 + 2 concentrations. This was not associated with clinically significant alterations in coagulation markers such as fibrinogen, prothrombin time, activated partial thromboplastin time and platelets.27,28 In addition, DD level assessment is now being considered an inclusion criterion for new clinical protocols (ClinicalTrials.gov Identifier: NCT04073498). It cannot be excluded and it is even likely that the DD assay could increasingly be applied in view of therapeutic PWH monitoring.
Limitations of our study include its retrospective nature and the limited number of participants included. Not all adult patients in our haemophilia treatment center could be included because DD test results before or after the change were not available for the entire study population. Nevertheless, there is no reason to believe that the non-inclusion of 13 patients influenced the validity of our results. None of the 53 patients on emicizumab discontinued the treatment.
The vast majority of our PWH were evaluated in the absence of infection, neoplasia, or concomitant treatment with FVIII/bypassing agents. Therefore, it cannot be excluded that in such situations, the physiological increase in DD would have been amplified by the underlying emicizumab treatment. Finally, although our study used the DD assay which is readily available, one cannot rule out that thrombin generation or prothrombin fragments F1 + 2 assays would have been more sensitive to detect pro-coagulable alterations.
Conclusion
This retrospective study demonstrated that nearly all adult PWH on FVIII prophylaxis exhibit normal DD concentrations, which remained unchanged after switching to emicizumab. These data support that there is no increased thrombogenicity under emicizumab therapy, at least in the absence of infection, neoplasia, or concomitant adjuvant hemostatic treatments. These results must, however, be confirmed in a larger PWH population under emicizumab and rebalancing therapies. In view of these reassuring results, monitoring of DDs at the start of emicizumab treatment does not appear necessary but could be considered when combined with other bypassing agents or high dose FVIII
Footnotes
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Funding
This study was made without any funding.
Author Contributions
Conceptualization: C. Hermans / Data collection : C. Hermans – C. Lambert / Data curation and analysis : M. Iarossi / Writing – original draft : M. Iarossi / Writing review and editing : C. Hermans and M. Iarossi
Statement of Ethics
The study protocol was approved by the ethical committee of the Saint-Luc University Hospital (UCLouvain, Brussels, Belgium).
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
