Abstract
Mortality rate in patients with COVID-19 increases in those admitted to the ICU. Activation of the coagulation system is associated with the worse disease outcomes. The aim of this study was to evaluate platelet activation and thrombotic biomarkers in hospitalized patients with COVID-19 during the second and third infection waves of the pandemic during 2021, following a previous report that included patients from the first wave. Sixty five patients were recruited and classified according to disease outcome; 10 healthy donors were included as a control group. Among prothrombotic biomarkers, t-PA concentrations (p < .0001), PAI-1 (0.0032) and D dimer (p = .0011) were higher in patients who developed critical COVID-19. We also found platelet activation via αIIbβIII expression (p < .0001) and higher presence of vWF-HMWM in severe COVID-19 (p < .0001). Several prothrombotic biomarkers are found to be increased since hospital admission in patients which lately present a worse disease outcome (ICU admission/death), among these, platelet activation, vWF increased plasma concentration and presence of HMWM seem to be of special interest. New studies regarding the predictive value of thrombotic biomarkers are needed as SARS-CoV-2 variants continue to emerge.
Introduction
Due to its rapid spread around the world, coronavirus disease 2019 (COVID-19) was declared as a pandemic in March 2020 by the World Health Organization (WHO). 1 It is an emerging sistemic disease caused by the SARS-CoV-2 virus, the newest member of the Coronaviridae family.2,3
In the last year it has been proven that the integrity of the endothelium is severely affected during the COVID 19 disease, therefore it has now been recognized as a vascular disease. While most of the infected patients present only mild respiratory symptoms, about 19% of COVID-19 patients develop pneumonic symptoms with rapid evolution to Acute Respiratory Distress Syndrome (ARDS) 4 and present a coagulopathy that frequently results in thrombotic events that lead to multiorgan failure, septic shock and death. 5 In most cases, the evolution of this infection towards severity is closely linked to a cytokine-mediated inflammatory storm that results in the activation of coagulation mechanisms leading to the formation of microthrombi in the pulmonary vasculature, frequently found in autopsies of deceased COVID-19 patients. 6
The hypercoagulable state in COVID-19 appears to be a consequence of platelet activation and endothelial cell damage caused by this cytokine burst, thereby producing a strong alteration of the hemostatic system. 7 Since the onset of the pandemic in 2020, thrombotic alterations have been described, ranging from thrombotic microangiopathy to pulmonary thromboembolism. 8 Although some prothrombotic biomarkers were recognized early in the pandemic, many new biomarkers of thrombotic risk have emerged.
Several studies on prothrombotic biomarkers were performed in the beginning of the pandemic, during 2020 and early 2021. As new variants of the SARS-CoV-2 virus emerge and the pandemic evolves, the question arises whether the new variants continue to behave in the same way from a prothrombotic point of view. The aim of this study was to evaluate platelet activation and thrombotic biomarkers in hospitalized patients with COVID-19 during the second and third infection waves during 2021 and correlate the results with the severity of the disease.
Patients and Methods
Patients
A total of 65 patients with diagnosis of COVID-19, admitted to the General Hospital “Dr Miguel Silva” in Morelia Mexico in the months of March to August 2021, during the second to third waves of infection, were included in this prospective observational study. SARS-CoV-2 infection was confirmed by reverse transcription polymerase chain reaction (RT-PCR) by the State Public Health Laboratory; all patients were ≥18 years of age. Samples from ten healthy volunteers were included as a Control Group, samples for this control group were obtained from August to December 2019 before the COVID-19 pandemic as control group for another study and were stored in our biobank, so no SARS-COV-2 infection is possible in them.
Informed consent was obtained from each patient, and the study was performed according to the ethical principles of medical research and local guidelines and approved by the local Ethics and Research Committee of the “Miguel Silva” General Hospital with the registration number 530/01/20.
Samples
For the purpose of this study, all venous blood samples were obtained at the time of admission or within the first 48 h after admission to the hospital by clean venipuncture, only those samples from confirmed SARS-CoV-2 infection were included in the study. Patients’ blood samples were further classified into two groups according to the evolution or final outcome of patients: the non-severe group included 37 patients that eventually developed severe COVID-19 but did not require admission to the ICU; the severe group included 28 patients who presented a severe disease with torpid evolution, required invasive mechanical ventilation (intubation) and therefore admission to the ICU and/or patients that eventually died. The following criteria were used by the intensive care staff to consider patients for invasive ventilation (intubation): clinical respiratory failure, PaO2, PaO2/FiO2 ratio (Kirby index) <200, blood gas analysis results, severe acidosis, failure of response to pronation and failure of response to high-flow ventilation.
Blood samples from all patients were obtained within the first 48 h of admission to the hospital and there was no significant difference in clinical severity between the two groups at the time of admission/sampling. Patients admitted to the hospital had been home treated with standard medication such as paracetamol but were not anticoagulated at the time of sampling. Blood samples were collected in vacutainer tubes with sodium citrate solution at a final concentration of 3.2% (Becton Dickinson, Franklin Lakes, NJ, USA). The first tube (2 mL) of blood were discarded to avoid platelet activation due to venipuncture. For platelet activation assays all samples were processed within 4 h of collection. Platelet-rich plasma (PRP) was obtained by slow centrifugation at 157.81 × g for 10 min at room temperature (RT), subsequently PRP was incubated for 30 min under dark conditions. Samples were processed with minimal handling and Tyrodes buffer (5 mM HEPES, 137 mM NaCl, 2.7 mM NaHCO3, 0.36 mM NaH2PO4, 2 mM CaCl2, 5 mM glucose, 0.2% BSA, pH 7.4) was used to obtain a final concentration of 1 × 107 cells per milliliter. For prothrombotic biomarkers studies, plasma was obtained at 1933.20 × g for 15 min and preserved at −70 °C until studied.
Prothrombotic Biomarkers
Plasma concentrations of prothrombotic biomarkers: D-Dimer, PAI-1, tPA, FT and F-IX were assessed by flow cytometry using a LEGENDplex™ Human Thrombosis Panel (10-plex) from BioLegend® kit. This is a multiplex immunoassay based on antibody-sensitized beads. Briefly, plasma was incubated for 2 h with analyte-specific antibody-conjugated beads, which are differentiated by size and internal fluorescence intensity. This allows them to act as capture beads, so that each analyte will bind to its corresponding bead. After a wash, a cocktail of biotinylated detection antibodies is added, which will bind to their specific analyte that is bound to the capture beads, forming a capture bead-analyte-detection antibody sandwich. Subsequently, streptavidin-phycoerythrin (SA-PE) is added and after 30 min of incubation, samples were read using a CytoFLEX BECKMAN COULTER® flow cytometer for analysis.
Von Willebrand Factor
IMUBIND® vWF REF ELISA Kit from BioMEDICA Diagnostics was used to assess vWF plasma concentration, following the instructions provided by the manufacturer. The ELISA plate was read at 450 nm in a Thermo Scientific Multiskan FC reader®.
VWF Multimeric Analysis
Von Willebrand Factor multimeric structure analysis was performed by western blot using a discontinuous vertical electrophoresis sodium dodecyl sulfate 1% to 2% agarose mini-gels, at 29V for 10–12 h at a constant temperature of 4 °C, in a vertical MiniProtean Cel (BioRad, USA). A constant concentration of vWF protein was loaded in gels for each sample, based on previous quantification of vWF plasma concentrations for each patient. Subsequently, proteins were transferred to a polyvinylidene fluoride nylon membrane at 18 V for 1 h using semidry transfer conditions (Tans-Blot SD SemiDry Transfer Cell; BioRad). Membranes were blocked with albumin and then incubated using a polyclonal rabbit antihuman vWF antibody (Dako, Glostrup, Denmark) as a primary antibody and polyclonal swine antirabbit horseradish peroxidase–labeled immunoglobulins (Dako) as secondary antibody, both from DAKO® Cat A0082 and DAKO® P0217.
WB luminol reagent (SCBT® sc-2048) was used for development on the ChemiDoc MP Imaging System Biorad® gel documenter. The obtained patterns were scanned and subjected to densitometric analysis using ImagenJ and ImagenLab. Multimers were classified as low molecular weight (LMW-vWFM; corresponding to bands 1–5 in vWFM analysis), intermediate molecular weight (IMW-vWFM; bands 6–10) and high molecular weight (HMW-vWFM; bands ≥11). 9 For quantitative analyses, we calculated the densitometric area only of HMW-vWFM from each study group.
Platelet Activity Evaluation
PRP was obtained by centrifugation, diluted in Tyrodes buffer and incubated for 20 min with CD41/PECy7 (BioLegend Cat. No. 303718) as identity marker, PAC-1/FITC (BioLegend Cat. No. 362804) and CD62/PE (BioLegend Cat. No. 304906), were both used as activation markers (glycoprotein αIIbβIII and P-selectin, respectively). IgG1 k (BioLegend Cat. No. 400125), FITC Mouse IgM k Isotype (BioLegend Cat. No. 401605) and, Mouse IgG1 k Isotype (BioLegend Cat. No. 400111) were used as isotype control respectively. Subsequently, platelets were fixed with paraformaldehyde at 4%. Dark conditions and minimal handling were used during the assay to avoid external activation of platelets. As positive controls of platelet activation we used known activation agonists ADP, collagen and epinephrine and the acquisition was performed by flow cytometry as reported before by our group. 10 Results were analyzed using FlowJo v 10.8.0.
Statistical Analysis
Categorical variables were expressed as frequencies and percentages. Continuous variables that followed a normal distribution were reported as mean and standard deviation; those that did not meet the assumption of normality were expressed as median and interquartile range. Normality analysis was performed using the Kolmogorov-Smirnov test and the Anderson-Darling test as appropriate. Homogeneity of variances was evaluated with the Levene test. Comparison between groups in variables with normal distribution and homogeneity of variances was performed by analysis of variance (ANOVA) with Tukey's post hoc test and those that did not meet the homogeneity assumption were evaluated with Welch's ANOVA and Games-Howell's post hoc test. Some variables were transformed using the Log function in order to obtain a normal distribution; after the analysis, antilogarithm was applied to interpret the results. Non-parametric variables were analyzed with Kruskal-Wallis ANOVA and Dunn's post hoc test. For clinical data, Mann–Whitney U test was used to compare non-parametric data. For parametric data, an unpaired Student's t test was performed, and data were reported as mean and standard deviation.
Ethics Aspects
The research studies were conducted in accordance with the Declaration of Helsinki under protocols approved by the ethics committees of the respective institutions. Informed consent was obtained from all donors or their legal representatives.
Results
Study Population
Between March and August 2021, a total of 65 COVID-19 adult patients were included in this study. Patients were classified into two groups according to evolution or outcome: the non-severe group consisted of 37 (56.9%) patients who subsequently developed severe COVID-19 but did not required admission to the intensive care unit; the severe disease group included 28 (43.1%) patients who presented a torpid evolution and required mechanical ventilation and admission to the ICU and/or fatality. Demographic and clinical characteristics of both study groups are presented in Table 1. The mean age was similar between study groups (p = .0580). However, there was a significant prevalence of male patients (p = .0340). The most frequent comorbidities found were diabetes, hypertension and obesity. Diabetes was significantly more prevalent in the severe disease group (p = .0170). In clinical laboratory tests, the presence of leukocytosis (p = .0002), neutrophilia (p < .0001) and lymphopenia (p < .0001) in severely ill patients were significant, as well as a higher erythrocyte sedimentation rate (p = .0244) and fibrinogen levels (p = .0124). In addition, mean platelet volume was found to be elevated in both groups of patients (p = .0006).
Clinical and Demographic Characteristics of the Study Population.
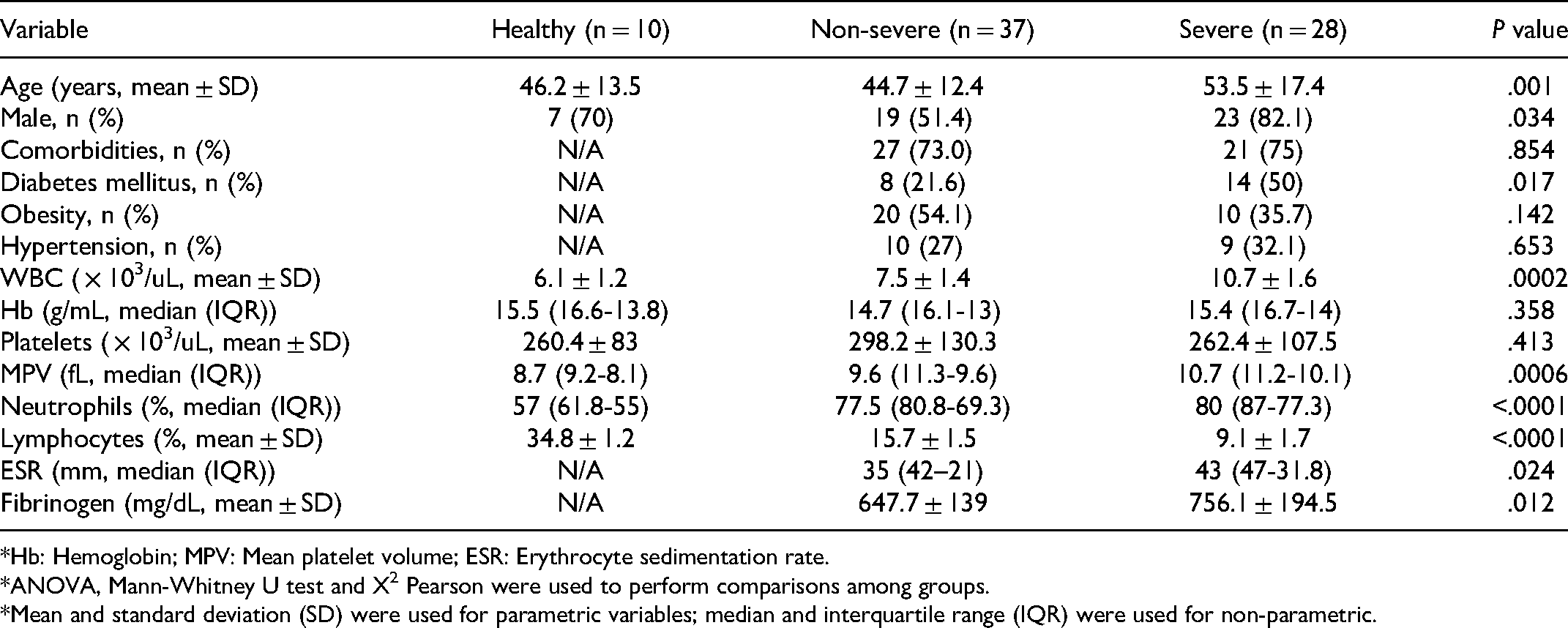
*Hb: Hemoglobin; MPV: Mean platelet volume; ESR: Erythrocyte sedimentation rate.
*ANOVA, Mann-Whitney U test and X2 Pearson were used to perform comparisons among groups.
*Mean and standard deviation (SD) were used for parametric variables; median and interquartile range (IQR) were used for non-parametric.
tPA and PAI-1
Significant differences were found in the concentrations of tPA in both study groups as can be seen in Figure 1. Concentration levels for the non-severe disease group were 5.44503 ± 1.72 ng/ml (p = .0009) and 6.93426 ± 1.56 ng/ml for the severe illness group (p < .0001) respectively; concentrations in the control group were 2.77332 ± 1.49 ng/ml. PAI-1 concentration levels were significantly higher only in the severe illness group (48.86523 ± 1.85 ng/ml) compared to healthy subjects (23.65919 ± 1.60 ng/ml) (p = .0032) These results are shown in Figure 2.

Comparison of t-PA plasma concentrations in patients with COVID-19 according to severity of the disease. tPA was significantly higher in patients of the severe COVID group compared to the control group. (ANOVA, Post Hoc Tukey **** p < .0001).

Comparison of PAI-1 plasma concentrations in patients with COVID-19 according to severity of the disease. PAI-1 was significantly higher in severe COVID-19 compared to the control group. (ANOVA, Post Hoc Tukey **p < .01).
D Dimer, F-IX and TF
The D dimer concentrations were also higher in both study groups (p = .0011) as shown in Figure 3, the severe group (17,179.08 ± 4.21 pg/ml) showed higher concentrations than the non-severe group (8356.03 ± 2.76 pg/ml) (p = .0492) and control group (3443.45 ± 1.78 pg/ml) (p = .0010). No differences were found in F-IX and TF concentrations for any study group. These results are shown in Figures 4 and 5.

Comparison of plasma concentrations of D-dimer in patients with COVID-19 according to severity of the disease. The figure shows how D-dimer levels were significantly higher in patients with worse outcomes (ANOVA Welch, Post Hoc Games-Howell **p < .01, *** p < .001).

Comparison of plasma concentrations of F-IX in patients with COVID-19 according to the evolution of the disease. No statistical difference was found among groups (ANOVA Kruskal-Wallis p = .5108).

Comparison of plasma concentrations of TF in patients with COVID-19 according to the evolution of the disease. No statistical difference was found among groups (ANOVA Kruskal-Wallis p = .8028).
VWF and Structural Analysis
Circulating vWF plasma concentration was quantified and then its multimeric structure was analyzed. Similarly, higher concentrations of vWF were found in both groups of patients, non-severe (147.6 ± 28.77 IU/ml) and severe (146.7 ± 29.16 IU/ml), when compared with healthy donors (90.17 ± 31.43 IU/ml) as shown in Figure 6A. Structural and densitometric analysis identified HMW-vWF and their densitometric area was quantified; results from western blot and densitometric analysis are shown in Figure 6B. Significant differences were found between the diseased patient groups and the healthy control group (4.28 ± 1.25%) (p < .0001). There was also a significant difference between the severe (13.58 ± 1.48%) and non-severe (7.72 ± 1.49%) groups (p = .0008), with higher HMW-vFW in patients with a worse disease course as shown in Figure 6C. A ROC curve analysis showed that the best cut-off point for the densitometric area was 9.51%, with a sensitivity of 80.0%% and a specificity of 77.8% as depicted in Figure 7.

Vwf plasma concentrations and analysis of its multimeric structure in patients with COVID-19 according to the evolution of the disease. (A) Comparison of plasma concentration of vWF. (B) Structural and densitometric analysis of vWF and quantification of HMW-vWF. (C) Densitometric area percentage of HMW-vWF. Densitometric area were transformed with the Log function to achieve a normal distribution. The figure shows high presence of HMW-vWF in patients who progressed to severity. (ANOVA, Post Hoc Tukey **p < .01, ***p < .001, ****p < .0001.).
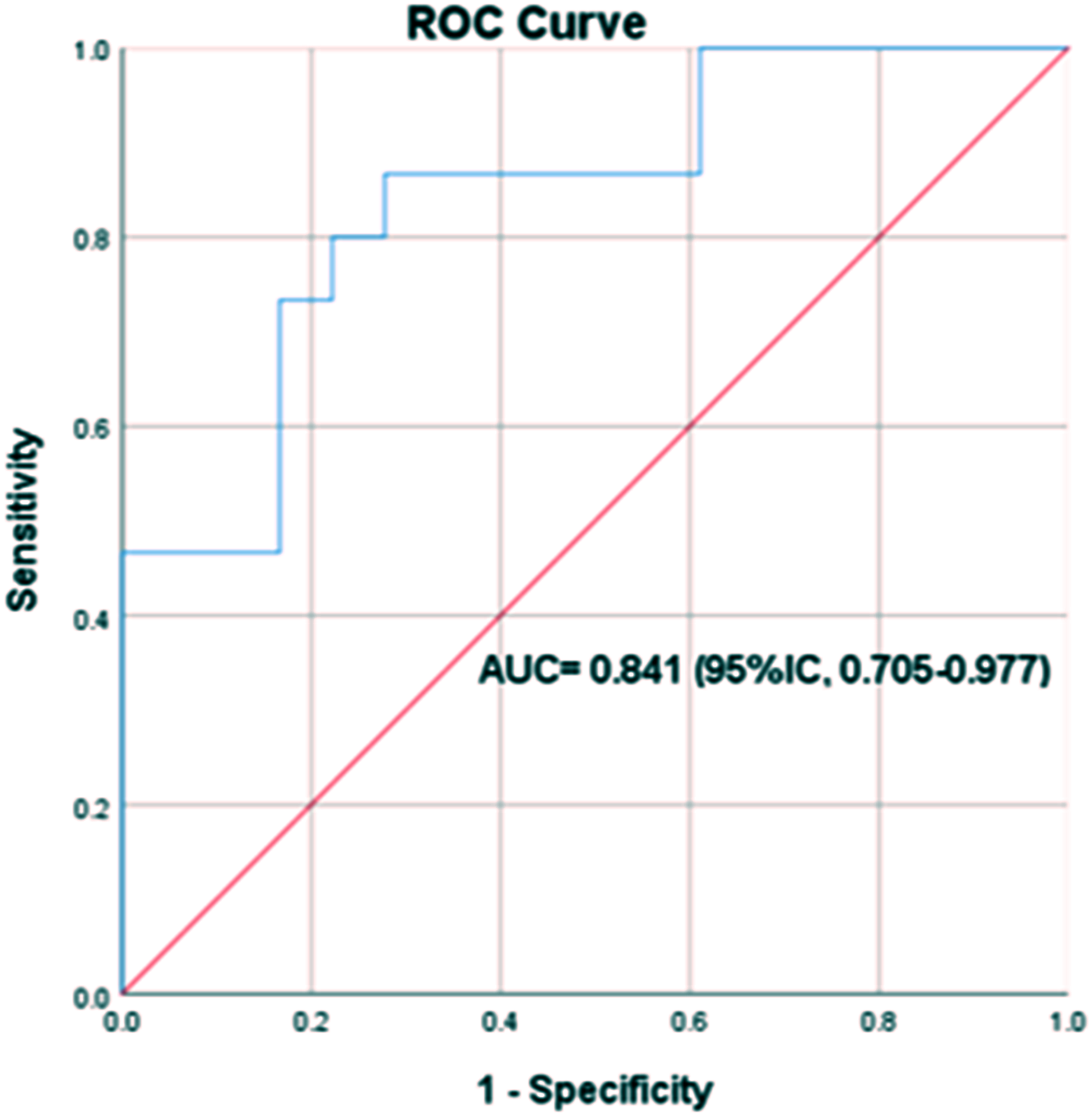
Receiver operating characteristic (ROC) curve of percent multimeric density of HMW-vWFs in patients with COVID-19. AUC (ROC of HMW-vWF %): 0.841 (p < .0001).
Platelet Activation Analysis
Platelet activation was assessed via glycoprotein αIIbβIII and P-selectin surface expression. Thirty-five patients were included for this assay, 21 form the non-severe group and 14 from the severe group. In both groups platelet activation was found to be higher than normal, geometric mean of αIIbβIII expression was higher in patients with worse clinical course (3696 ± 1197) than in non-severe patients (2670 ± 609) (p = .0029) and healthy volunteers (2276 ± 616) (p = .0009). Moreover, P-selectin was significantly higher in both patient groups, non-severe (8239 ± 3332) (p = .0005) and severe (10,453 ± 5063) (p < .0001), compared to the control group (1801 ± 545 GM). These results are shown in Figure 8.
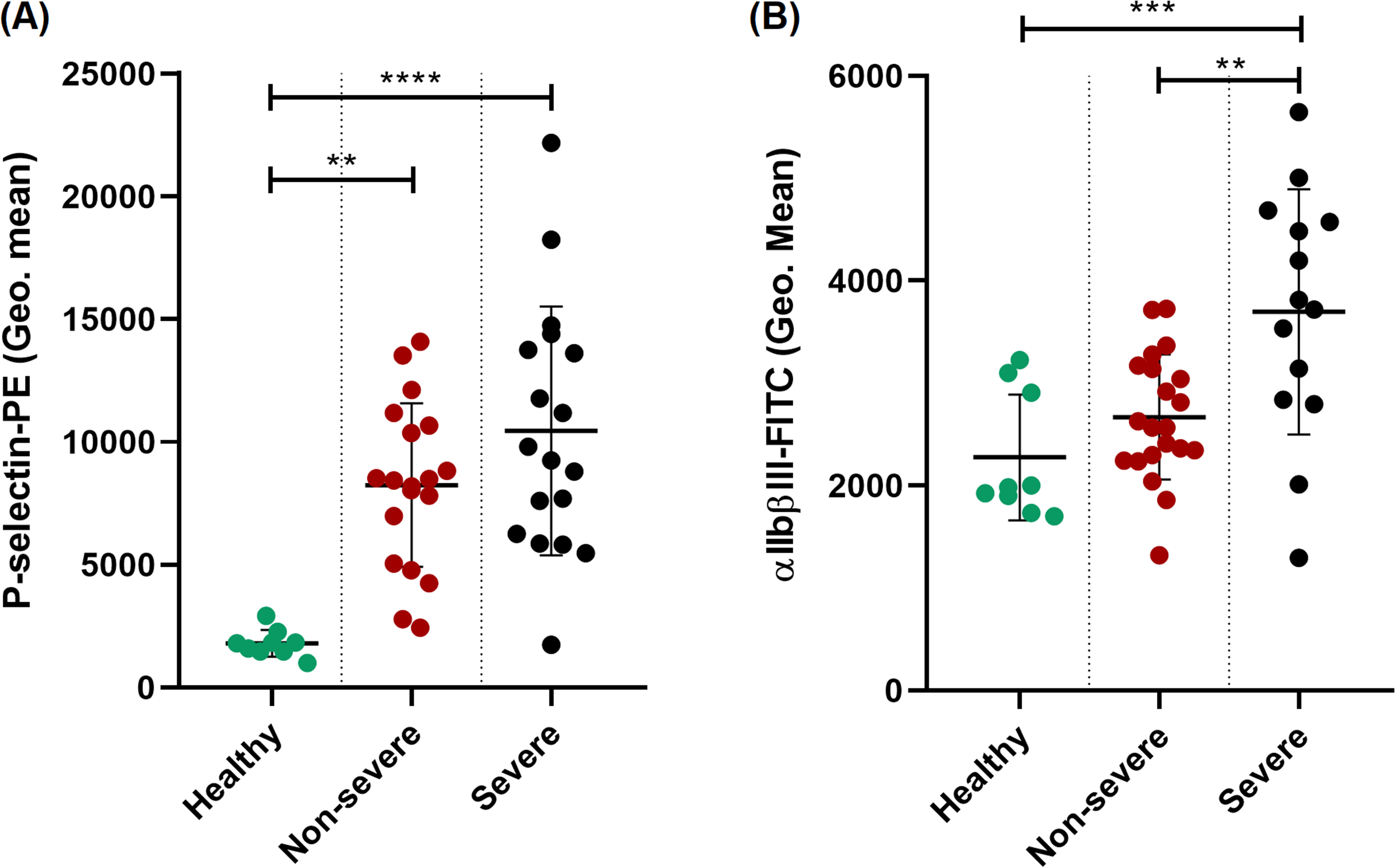
Platelet activation in patients affected by COVID-19 according to the evolution of the disease. (A) Comparison of geometric mean expression of CD41 (P-selectin-PE). (B) Comparison of geometric mean expression of PAC-1 (αIIbβIII-FITC). Platelets from patients in the severe group showed higher expression of activation markers. (ANOVA, post Hoc Tukey **p < .01, *** p < .001, **** p < .0001).
Discussion
COVID-19 is a life-threatening disease in some individuals. Since the beginning of the pandemic, it has been described that patients with severe disease develop a pathological inflammatory response that results in severe tissue damage, endothelial dysfunction and coagulopathy that favors the development of thrombotic complications and increased mortality. 11 The aim of this study was to evaluate whether certain biomarkers of an hypercoagulable state were altered in patients before the development of a critical condition and therefore if this was associated with a worse prognosis of the disease. In the present study we provide evidence that several prothrombotic biomarkers such as tPA, PAI-1, D-dimer, vWF, its multimeric structure and platelet activation could predict a higher risk of developing severe disease independently of pre-existing risk factors.
The hypercoagulable state observed in severe COVID-19 has been attributed to an exacerbated innate immune response driven by proinflammatory cytokines and extracellular traps from neutrophils. 12 In the present study elevated leukocyte concentrations with a high percentage of neutrophils were detected in patients who required admission to the ICU. This inflammatory environment, together with pathogen-associated patterns (PAMP's) and reactive oxygen species (ROS), stimulates the activation of endothelial cells and thus the production and release of procoagulant factors such as fibrinogen, tPA, PAI-1 and vWF.13,14 Our results show an increase in tPA in patients hospitalized for COVID-19, which may be attributed to the interaction between proinflammatory cytokines and the endothelium, the stimulation of the latter then releases tPA. 15 On the other hand, concentrations of PAI-1 were higher in the severe disease group. These findings were also reported by Henry et al implying that an abnormality in the fibrinolytic process is taking place, this can contribute to a greater hypercoagulability state that may lead to ARDS development and worsening of the patient status due to microthrombus formation and fibrin accumulation in the alveolar niche.15,16 Similar to t-PA, D dimer, which is a product of fibrin degradation during fibrinolysis, is considered an independent marker of impaired fibrinolysis and a strong predictor of death in COVID-19. 17 In the present study increased D dimer and fibrinogen concentrations were found in both study groups, indicating that the fibrinolytic system is active despite elevated PAI-1 concentrations and probably because tPA is also elevated; however, tPA probably decreases at some point in the course of the disease or its concentration is exceeded to a greater extent by PAI-1, resulting in hypofibrinolysis that contributes to patients worse outcome. 18 Han and Pandey have reported SARS-CoV-2-triggered endothelial dysfunction leads to robust release of PAI-1 19 in vitro. This finding in addition to the growing clinical evidence that older patients and those with preexisting cardiometabolic or chronic inflammatory diseases, who are more likely to have higher baseline PAI-1 levels, are at an increased risk for severe disease; this suggests the plausibility of targeted PAI-1 inhibition as a potential treatment for patients with COVID-19.
No differences were observed in F-IX or TF concentrations. This is consistent with that reported by Martín-Rojas who found no consumption of coagulation factors in prothrombotic coagulopathy due to COVID-19. 20 On the other hand, another study also found no differences in plasma TF concentration between patients with COVID-19 and a control group, but did find elevated concentrations of TF in bronchoalveolar lavage fluid. 21
Another protein that is considered a marker of endothelial activity and therefore associated with prothrombotic activity is vWF. This multimeric glycoprotein plays a fundamental role in hemostatic processes. Under physiological conditions, this protein is stored in Weibel-Palade bodies (WPB) of endothelial cells and in alpha granules of platelets. When stored, vWF is found as HMW-vWF, as in circulation it is present predominantly as IMW-vWF and LMW-vWF because of its cleavage by the restriction protease ADAMTS-13.22,23 Our study shows increased levels of vWF in both study groups. These results are consistent with our previous report from 2021, when we reported significantly higher plasma concentrations of vWF in severe COVID-19 patients from the first wave when compared to non-severe patients and controls. These results also agree with those reported by Ward et. al., who compared the concentrations of vWF in COVID-19 patients against healthy controls. 24 Flora Peyvandi's group also reported increased vWF levels in severely ill patients, 25 with an elevated vWF to ADAMTS 13 activity ratio that was strongly associated with disease severity.
In the multimeric and densitometric analysis, HMW-vWF were higher in the group of patients that developed the more critical disease. This might be evidence that there is active degranulation of platelets and endothelial cell Weibel Palade bodies contributing to the pathophysiology of severe COVID-19. 26 HMWM have the capacity to activate and improve platelet adhesion and activation. vWF also carries coagulation factor VIII, which is activated upon detachment. Altogether the presence of vWF HMWM indicates that significant endothelial damage is occurring which favors a pathological deviation of hemostasis and with it, the formation of microthrombi in the pulmonary vasculature. 27
Within the immunothrombotic triad described in COVID-19, platelets are cells that play an important role since they are classically associated with the clotting process, furthermore, platelets have recently been described as immune cells mediating the inflammatory process. The numerous receptors present on their surface allow them to be activated by several stimuli. 28 Once activated, platelets express ligands that favor adhesion to other cells and aggregation, also, these cells release many factors involved in both thrombotic and inflammatory mechanisms. In this study, patients with COVID-19 that developed a worse illness course showed increased MPV and platelet hyperactivation determined by an elevated expression of P-selectin and integrin αIIbβIII. In addition, glycoprotein αIIbβIII showed a significant difference between both study groups being higher in patients with the worse outcome. Similar to our results, Leopold et al, reported increased expression of p-selectin and the αIIbβIII complex in COVID-19 patients. Moreover, they reported a high concentration of sCD40L and PF4 released from the alpha granules. 29 These results show that platelets play a key role in COVID-19 coagulopathy and in disease outcome.
With the continuation of this pandemic, different SARS-CoV-2 variants have emerged around the world. As the virus has evolved, clinical presentation of the disease has also changed. In a previous study published by our group 30 we reported higher concentrations of prothrombotic and inflammatory biomarkers in patients recruited from July to September 2020 during the first COVID-19 wave in Mexico. In this new study, samples were obtained from March through August 2021, during the second-third wave of COVID-19. Although in this study we did not have the possibility to sequence the specific variant of SARS-CoV-2 in our patients, different variants of SARS-CoV-2 were circulating in the world and in our country at the moment of the study. Our results show that the thrombophilic state of hospitalized COVID 19 patients persists even though new variants of the virus emerged during this period of time.
Conclusions
In conclusion, the prothrombotic state in patients hospitalized for COVID-19 seems to be caused by significant endothelial tissue damage, which is reflected by increased markers of coagulation and endothelial dysfunction. In addition, increased PAI-1, the presence of platelet hyperactivation with abundant αIIbβIII integrin expression phenotype, as well as the presence of large amounts of HMW-vWF promote a hypercoagulable and hypofibrinolytic microenvironment that leads to the formation of pulmonary and/or systemic microthrombosis, organ failure and death. Due to the important role of these biomarkers in the patient's evolution, they could be used as predictors of risk of critical condition and/or death and as potential targets for treatment development, furthermore, when measured on admission, before patients are critically ill, vWF parameters could be useful to identify patients that could potentially evolve to severity/death and who could benefit from early anticoagulation/oxygenation therapies.
Footnotes
Authorship Contributions
MEVS designed the research. MEVS, SLC, GDV, NGL, ACM conceived and designed the experiments; NGL and ACM trained fellows and supervised experiments, GDV performed experiments, data analysis and wrote the original manuscript; GDV, PGC and ATL obtained samples, performed laboratory studies and built the data base; SELC and ACM performed project management; MEVS and SELC reviewed and edited final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Consejo Nacional de Ciencia y Tecnología, (grant number 320085).
