Abstract
Objective
Gynecologic cancer, including cervical, endometrial, and ovarian cancer, comprises the fifth leading type of cancer and is an important malignant disease in women. Previous studies in Western countries have reported respective prevalence rates for venous thromboembolism (VTE) of 3.3%–18.7%, 0.8%–8.1%, and 7.2%–20.9%. In this study, we aimed to identify the characteristics associated with VTE in Japanese patients.
Methods
We carried out a retrospective cohort study to compare the clinicopathological characteristics of patients with each gynecologic cancer with and without concomitant VTE. Patients: Patients with cervical, endometrial, or ovarian cancer treated at Fukui Prefectural Hospital, Japan, from April 2010 to March 2020.
Results
Among 699 patients with gynecologic cancer, 50 developed VTE within 5 years after their cancer diagnosis, including 16/357 patients with cervical cancer (5.6%), 12/185 with endometrial cancer (6.8%), and 22/157 with ovarian cancer (14.6%). The 1-year mortality rate after symptomatic VTE onset was 47.8%. The VTE group included significantly more older patients and more patients with advanced cancer or poor performance status compared with the non-VTE group. There was no significant difference in the rate of surgical treatment. Symptomatic, but not asymptomatic VTE, was associated with shorter survival.
Conclusion
Several baseline characteristics differed between patients with and without VTE. The incidences of VTE and some risk factors were similar in Japanese patients with gynecologic cancers compared with patients in other countries. Patients with VTE had some factors that worsened their prognosis, with patients with gynecologic cancer and symptomatic VTE having an especially poor prognosis.
Introduction
The Japanese Circulation Society published guidelines for the diagnosis, treatment, and prevention of pulmonary thromboembolism and deep vein thrombosis (DVT) in 2004. Venous thromboembolism (VTE) has since been diagnosed more frequently in Japan, partly because of improvements in the recognition and diagnosis of the disease. 1 Increasing attention is being paid to the importance of thrombosis in patients with cancer. Khorana et al reported that gynecologic cancer was associated with a high risk of VTE. 2 Gynecologic cancer, including cervical, endometrial, and ovarian cancer, comprises the fifth leading cancer in Japanese women. 3 Their respective prognoses are poor, with 5-year survival rates of only 22.5%, 20.1%, and 23.9%, in patients with stage IV cancers. 4 The morbidity and mortality of these types of cancers have been increasing in Japan during the 2000s. Notably, most patients diagnosed with cervical cancer are in their 20s to 40s, 5 while most patients diagnosed with endometrial or ovarian cancer are between their 40s and 50s,6,7 Japanese women can receive cervical cancer screening from the age of 20 years, but only 35.8% of women received this important screening in 2019, 8 which was fewer than in many other countries. 9 In contrast, no effective screening tests for endometrial and ovarian cancer have yet been established and their diagnoses may thus be delayed. Patients with gynecologic cancers tend to be relatively young and the cancer and/or its treatment may thus cause problems related to fertility preservation among patients of child-bearing age. These problems may be further exacerbated by the occurrence of thromboembolism, which is the second leading cause of death in patients with cancer. 10
Previous studies in Western countries have reported VTE prevalence rates of 3.3%–18.7% in patients with cervical cancer, 0.8%–8.1% in patients with endometrial cancer, and 7.2%–20.9% in patients with ovarian cancer.11–18 However, few studies have investigated this topic in Japan. In the present study, we aimed to identify the characteristics associated with the occurrence of VTE in Japanese patients with gynecologic cancer.
Patients and Methods
All procedures were conducted in accordance with the Declaration of Helsinki. All patients provided written informed consent for use of their data. This was a retrospective cohort study of patients with gynecologic cancer who were diagnosed and treated at Fukui Prefectural Hospital from April 2010 to March 2020. Patients who dropped out because they transferred to another hospital for treatment or who suspended hospital visits without receiving treatment at their own discretion after a cancer diagnosis at our hospital were excluded. In addition, because patients often develop VTE when their cancer is aggressive and during cancer treatment, we set the follow-up period as at least 1 year to assess the risk of VTE in patients receiving treatments. Cases treated at our hospital after a cancer diagnosis were included, even if they died within 1 year. Data for patients with a recorded diagnosis of VTE were extracted using the following criteria. The VTE group was defined as patients with proven VTE on imaging (eg, contrast-enhanced computed tomography [CT] 19 or echography of the lower limb veins), in patients with a high pretest clinical probability of VTE, according to the physician's opinion (eg, based on medical history, symptoms, physical findings, high D-dimer), or proven VTE detected coincidentally on contrast-enhanced CT performed for cancer diagnosis or follow-up, or investigation of other diseases. In the non-VTE group, contrast-enhanced CT was performed in 168 patients with cervical cancer after diagnosis (173 patient did not receive CT) and in all patients with endometrial or ovarian cancer. We checked all the radiology reports. Clinical characteristics (age, body mass index, smoking status, performance status, comorbidities, thrombotic history, disease stage, biomarkers before primary treatment, types of chemotherapy, and cause of death) and long-term outcomes were compared between the VTE and non-VTE groups. The sample size for endometrial and ovarian cancer was lower than that for cervical cancer, and we therefore compared each VTE group with the entire pooled population without VTE, in order to identify more risk factors. Performance status was assessed by Eastern Cooperative Oncology Group (ECOG) Performance Status Scale. 20 Disease stage was grouped into non-advanced stage, including patients treated mainly with surgery, and advanced stage, including patients treated mainly with chemotherapy (cervical cancer: IIIA-IVB; endometrial cancer: IVA-IVB; ovarian cancer: III-IV). Data collected for the VTE group included symptoms, severity, classification of VTE, treatment for VTE, and outcomes. Differences between the VTE and non-VTE groups were compared using Fisher's exact test, unpaired t-test, or log-rank test. Cumulative incidence was assessed using Gray's test. Cox proportional-hazards model was used to estimate the hazard ratio (HR) and 95% confidence interval (CI) for factors associated with mortality in whole cohort. All p-values were two sided, and p-values ≤0.05 were considered statistically significant. All statistical analyses were performed using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). 21
Results
A total of 807 patients with gynecological cancer were registered at Fukui Prefectural Hospital during the 10-year study period, of whom 699 patients (cervical cancer: 357 cases; endometrial cancer: 185 cases; ovarian cancer: 157 cases) were followed for at least 1 year after their diagnosis (Figure 1). The histological classifications are shown in Figure 2. Overall, 134 patients (19.2%) had advanced stage cancer, including 63 patients with cervical cancer (17.6%), 14 patients with endometrial cancer (7.6%), and 57 patients with ovarian cancer (36.3%). The respective 5-year survival rates for patients with advanced-stage cancers were 58.3%, 26.4%, and 39.4%. Overall, 50 (7.2%) patients developed VTE, with cumulative incidences at 5 years of 5.6%, 6.8%, and 14.6% for cervical, endometrial, and ovarian cancer, respectively. Ten patients were diagnosed simultaneously with VTE and gynecological cancer. Sensitivity analysis including data for 108 patients excluded from this study resulted in similar findings (5.3%, 6.6%, and 13.8%, respectively). The baseline characteristics of the patients included in and excluded from the analysis are compared in Table 1. There were no significant differences between the two groups except for the numbers of patients with a history of chronic lung disease. However, it should be noted that some data were lacking for patients excluded from the analysis.
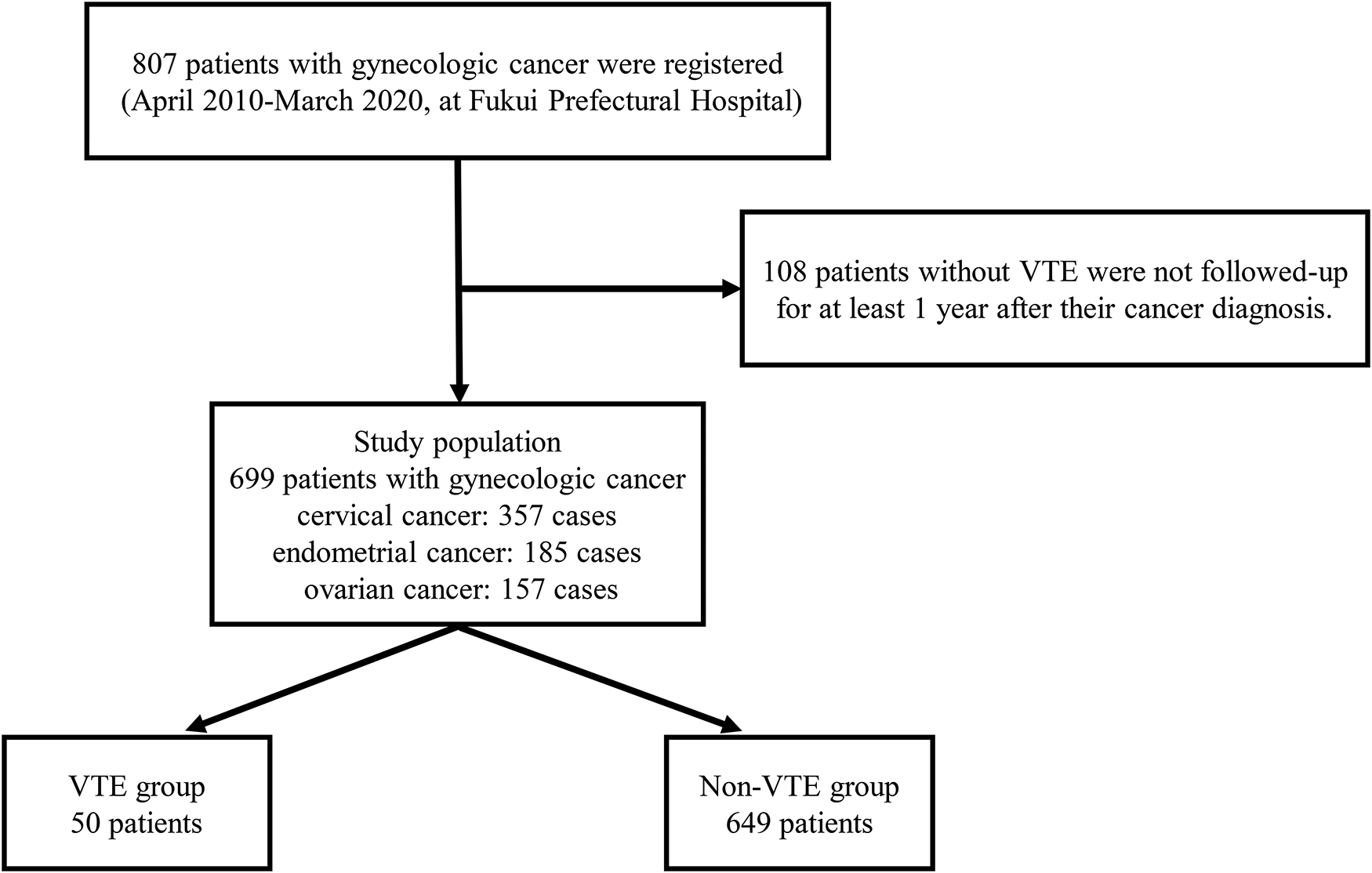
Study flow chart. VTE: venous thromboembolism.

The histological classifications
Comparison of Baseline Characteristics of the 108 Patients Excluded from the Analysis with the Included Participants.
The characteristics of the VTE and non-VTE groups are summarized in Table 2. Among all patients with gynecologic cancer, the VTE group included significantly older patients, significantly more patients with PS 3-4, thrombotic history, advanced-stage disease, anemia, leukocytosis and more patients receiving chemotherapy than the non-VTE group. There were no significant associations with body mass index, smoking, comorbidities (except for stroke history), pelvic surgery, and laparoscopic surgery. One patient with VTE and two patients without VTE had thrombotic histories. Eleven patients developed cerebral infarction after their cancer diagnosis. There were no cases of central venous catheter-related thrombosis. Total of 281 patients with gynecologic cancer received primary chemotherapy (cisplatin (CDDP), 74 cases; paclitaxel and carboplatin combination therapy (TC), 193 cases; docetaxel and carboplatin combination therapy (DC), 7 cases; others, 7 cases). Thirty-six patients with VTE received chemotherapy (CDDP, 9 cases; TC, 26 cases; DC, 1 case) and 23 cases developed VTE after receiving chemotherapy (CDDP, 8 cases; TC, 15 cases).
Baseline Characteristics of Patients With Gynecologic Cancer.
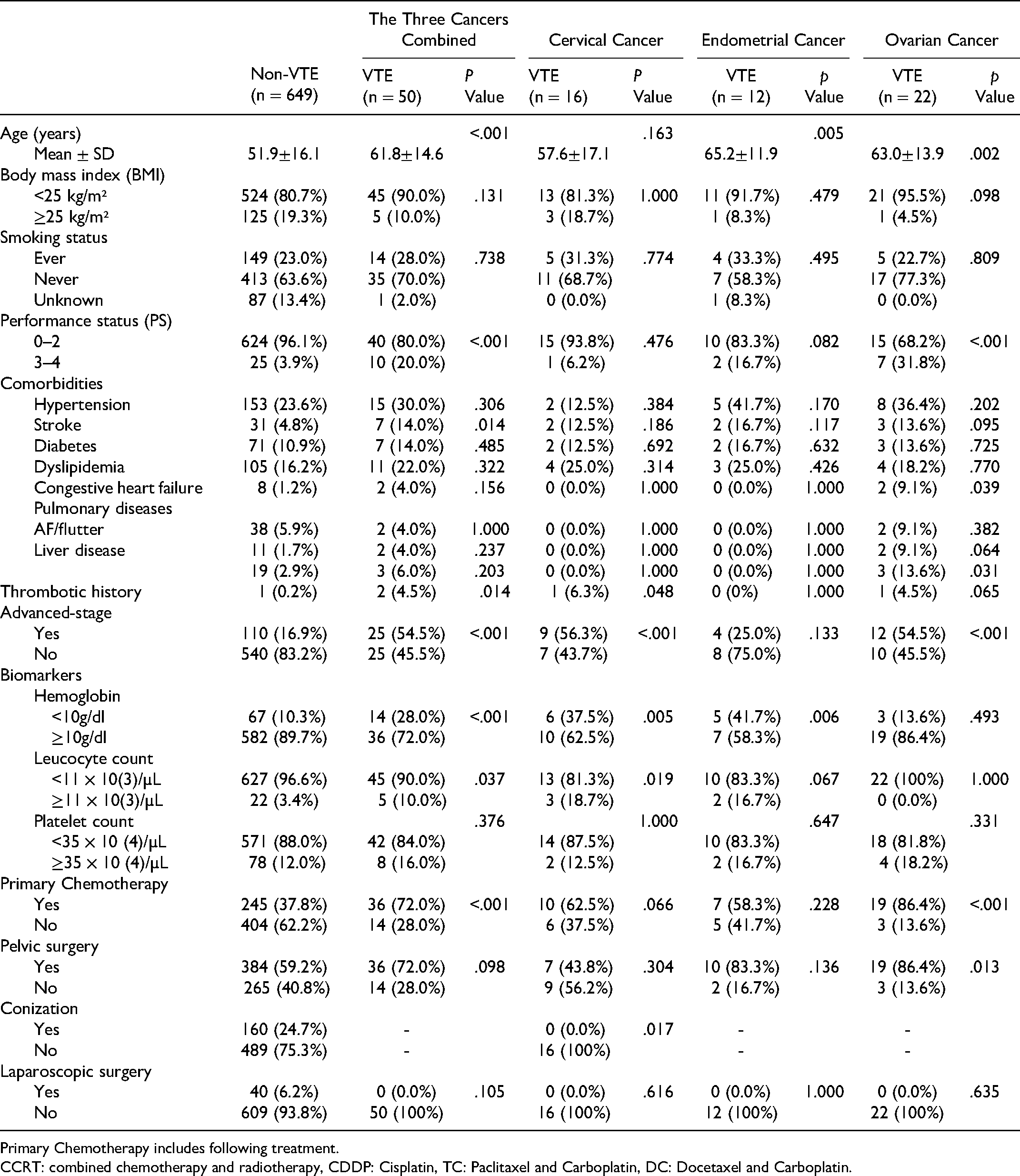
Primary Chemotherapy includes following treatment.
CCRT: combined chemotherapy and radiotherapy, CDDP: Cisplatin, TC: Paclitaxel and Carboplatin, DC: Docetaxel and Carboplatin.
Forty-four (88%) of the 50 patients in the VTE group developed pulmonary thromboembolism or DVT and six (12%) developed other thromboembolisms (ovarian vein: 4; internal jugular vein: 1; coronary vein: 1). Survival curves for the three gynecologic cancers alone and combined are shown in Figures 3 and 4. The median survival time from cancer diagnosis tended to be shorter in the VTE group than in the non-VTE group for all three cancers, with significant differences for the entire cohort (5.43 years vs Not Available: NA, P < .001), cervical cancer (5.92 years vs NA, P < .001) and endometrial cancer (3.14 years vs NA, P < .001), but not ovarian cancer (5.39 years vs NA, P = .079). Among patients with ovarian cancer, the median survival time was significantly shorter in patients with symptomatic VTE than in patients without VTE (4.82 years vs NA, P = .001); however, the median survival times were similar in patients without VTE and those with asymptomatic VTE. A total of 101 (14.4%) patients died, mostly from the gynecological cancer itself (78.2%), followed by infections (5%), and heart disease (5%). Two patients died from thromboembolism (VTE: 1; cerebral infarction: 1).
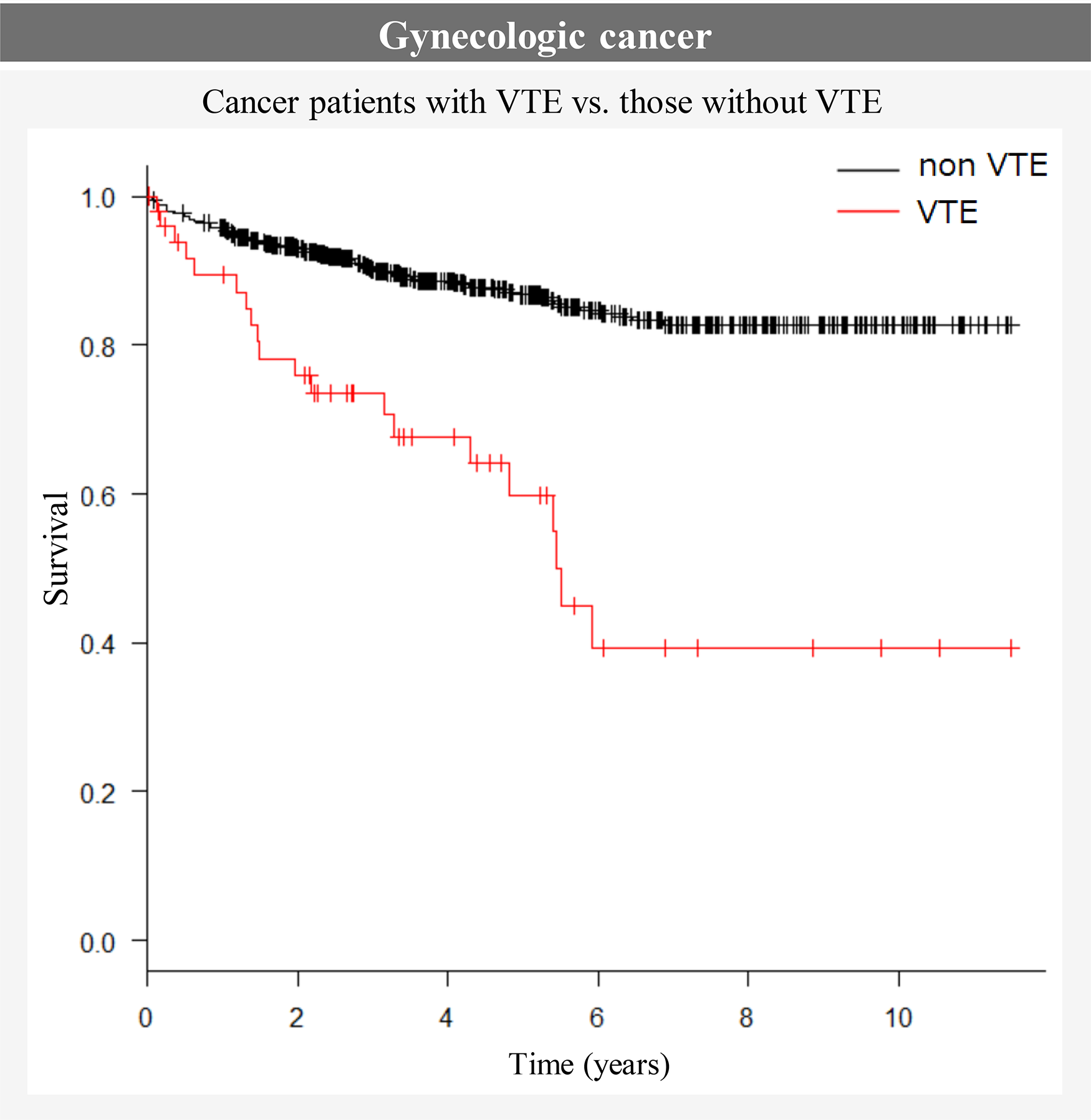
Survival curves for the three gynecologic cancers combined.
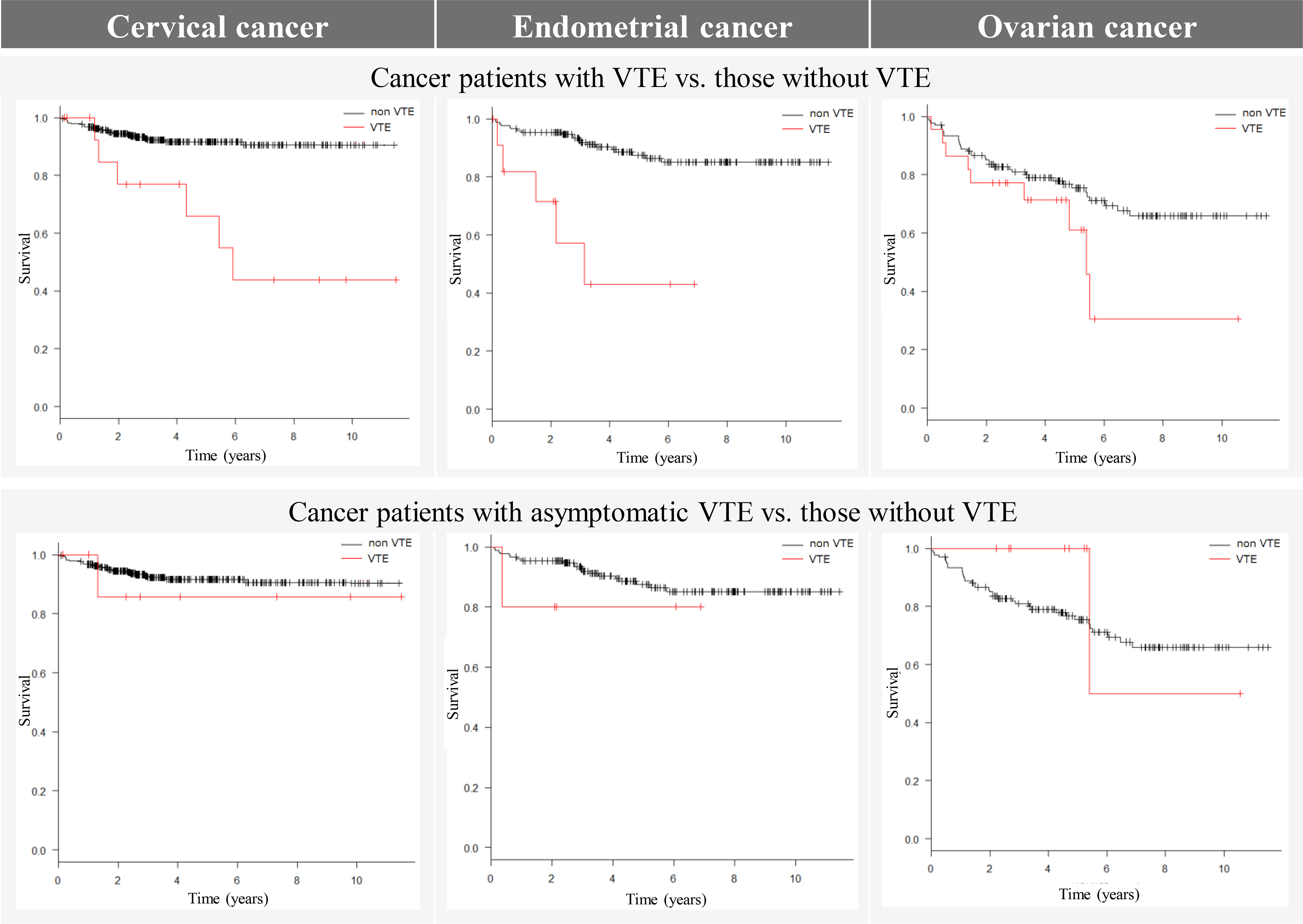
Comparison of survival of gynecologic cancer patients with and without VTE.

The period from cancer diagnosis to VTE onset. *1: Cases diagnosed simultaneously with VTE and gynecological cancer.
The incidence of VTE in patients with gynecological cancer was highest within the first year after cancer diagnosis and decreased with time, but VTE also occurred in the chronic phase (Figure 5). The median times from cancer diagnosis to VTE onset were 218 days for patients with cervical cancer, 161 days for endometrial cancer, and 21 days for ovarian cancer. Twenty-seven (54%) patients had symptomatic VTE. The VTE was not severe in most patients, but one patient had a cardiac arrest and one patient had a massive pulmonary thromboembolism. Among the 36 patients who developed DVT, 11 had central-type thrombosis and 25 had peripheral-type lower limb vein thrombosis. Twenty-three (46%) patients had asymptomatic VTE, including 16 patients in whom VTE was detected by contrast-enhanced CT or echography because of high D-dimer score, and the other seven were diagnosed by CT performed for diagnosis of cancer or assessment of the therapeutic effect. Twenty of the 50 patients with VTE died. The overall 1-year mortality after VTE onset was 29.5%, but the mortality in patients with symptomatic VTE was 47.8%. About the treatments for VTE, 38 patients received anticoagulant therapy. Twenty-six patients received mainly direct oral anticoagulants (25 edoxaban 30 or 60 mg once daily [60 mg in patients ≥60 kg and 30 mg in patients <60 kg]; 1 rivaroxaban 15 mg twice daily). All patients received the recommended dose of oral anticoagulants, considering their body weight and creatinine clearance. Nine patients received warfarin, two received only unfractionated heparin, and seven received an inferior vena cava filter. Twelve patients did not receive anticoagulant therapy because they were considered non-serious (eight cases, all asymptomatic and few thrombi), were in the terminal phase of life (two cases), or because they were about to undergo surgery (two cases). Anticoagulation therapy was discontinued in four patients due to bleeding (gastrointestinal bleeding: two; genitourinary bleeding: two). No symptomatic recurrence of VTE was observed in the study period.
Discussion
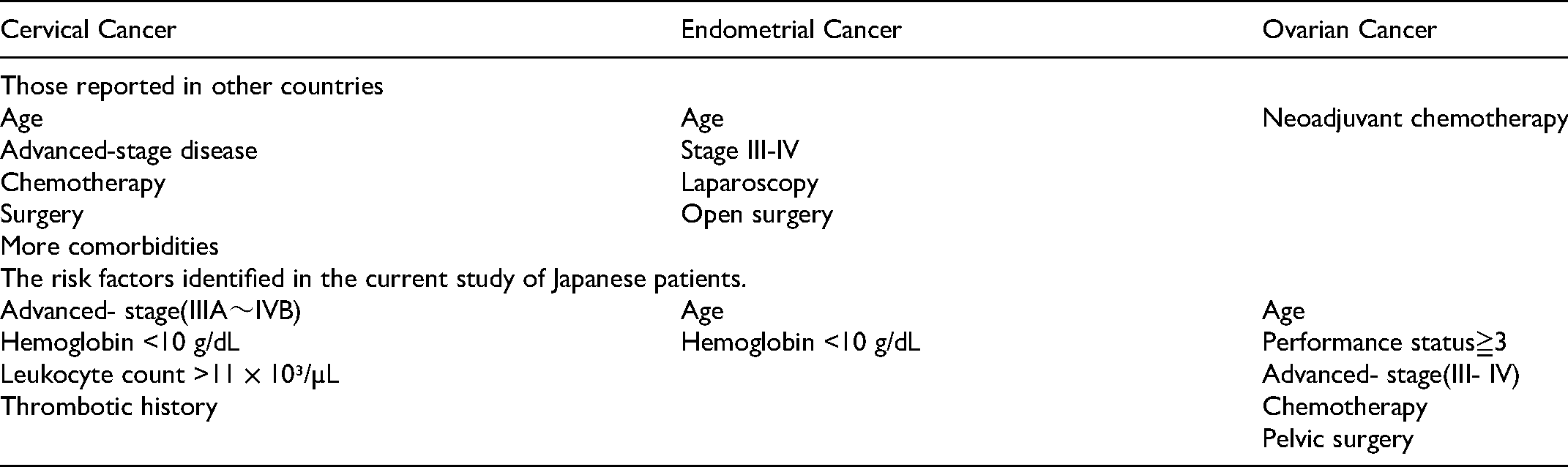
The overall incidence of gynecological cancer-associated VTE in our study of Japanese patients was similar to that reported in studies from other countries. In particular, the incidence of VTE in patients with ovarian cancer was relatively high. Ovarian cancer has been reported to have among the highest incidences of VTE among all solid tumors, 22 possibly because of a combination of widespread disease at diagnosis, venous stasis from pelvic masses, and chemotherapeutic agents, all of which are known to promote coagulation. 23 One report suggested that thromboprophylaxis during neoadjuvant chemotherapy should be assessed prospectively. 18 The fact that the incidence of VTE was highest within the first year after cancer diagnosis and then decreased with time may be related not only to cancer aggression, but also to cancer treatments. Table 3 compares the risk factors reported in other countries with those identified in the current study of Japanese patients.11–13,18,24,25 There was no significant difference in the rates of surgical treatment for cervical cancer and endometrial cancer. Additionally, regarding blood parameters, using the Khorana risk score, hemoglobin level <10g/dl, leukocyte count >11 × 10³ /µL or a platelet count ≥ 35 × 10⁴ /µL were predictors of thrombosis. In this study, some biomarkers before primary treatment (anemia and leukocytosis in patients with cervical cancer, anemia in patients with endometrial cancer) were significantly associated with VTE. This finding may support the predictive ability of the Khorana risk score. 2 Among patients with cervical cancer or endometrial cancer, survival rates were significantly lower in patients with VTE compared with patients without VTE. Although survival also tended to be shorter in patients with ovarian cancer, the difference was not significant. This may have been influenced by the relatively poor prognosis of patients with ovarian cancer, who tend to present with advanced disease. An adjusted analysis considering the risk factors for the whole cohort according to a Cox proportional hazards model showed that age, PS 3–4, advanced stage, anemia, and leukocytosis were significant predictors of poor survival (Table 4). However, the development of VTE was not an independent factor. The poor prognosis of the VTE group may be because patients with VTE had some factors that worsened their prognosis, rather than because VTE directly shortened their survival. Symptomatic VTE shortened survival, with a 1- year mortality from VTE diagnosis in patients with symptomatic VTE of 47.8%. The COMMAND VTE Registry is a multicenter registry of Japanese patients with cancer and symptomatic VTE. Yamashita et al reported that the 1-year mortality rate for all patients in the registry after a diagnosis of VTE was 49.6%, 26 which was similar to the 1-year mortality rate in our study, confirming the similarly poor prognoses of patients with symptomatic VTE with gynecological and other cancers. In contrast, there was no significant difference in survival between the 23 patients with asymptomatic VTE and those with VTE. However, most patients with asymptomatic VTE had high D-dimer scores in regular blood tests and underwent CT or echography, and other patients with asymptomatic VTE may have been missed. This suggests that the early detection of thrombosis, when the patient has few symptoms, may reduce the risk of shortened survival due to VTE. However, given the level of medical care in Japan, only one patient in this study died directly from VTE. Considering that age, advanced cancer, and poor performance status were listed as risk factors for VTE, the shorter survival period in patients with symptomatic VTE may be due to the poor cancer-related prognosis of patients with gynecologic cancer in Japan who develop VTE, rather than to the VTE itself. However, several studies have found an association between VTE onset and poorer prognosis,11–13 and diagnosing asymptomatic VTE may improve the prognosis in patients with gynecologic cancer. Eight patients with VTE received no anticoagulation therapy because of slight thrombi in side branches of the anterior/posterior tibial veins or peroneal veins, or because of a high bleeding risk, such as elderly patients and patients with a history of bleeding event, stroke, or chronic renal or liver dysfunction. Thirteen patients discontinued anticoagulation therapy after disappearance of the thrombus. There were no subsequent confirmed cases of exacerbation or recurrence of VTE during the research period. However, close attention should be paid to the recurrence of VTE, given that a history of VTE is known to be an important risk factor for VTE, and current guidelines recommend that cancer patients with VTE should receive long-term anticoagulant therapy until their cancer is cured. 27 In the present study, four patients discontinued anticoagulant therapy due to bleeding. In particular, genitourinary bleeding should be considered in patients with gynecological cancer undergoing direct oral anticoagulant therapy, because the genitourinary system acts as a metabolic and excretion pathway for these agents. Although some reports have suggested the use of perioperative thromboprophylaxis,28,29 there is currently no consensus on the efficacy of thromboprophylaxis and an early diagnosis of VTE is considered to be more important. Previous reports about cervical cancer and endometrial cancer from other countries have revealed that surgical treatment was a risk factor for VTE;24,25,30 however, the current study with both of cancers found no significant difference between the VTE and non-VTE groups in terms of the proportions of patients who received pelvic or laparoscopic surgery. This result may show the efficacy of standard perioperative VTE preventive measures in Japan, such as compression stockings, circulating plasma volume management, and early bed-leaving. Both the early diagnosis of VTE by regular screening of D-dimer and the lack of an increase in VTE during the perioperative period may be related to improvements in the recognition and diagnosis of VTE among gynecologists. Gynecologic cancers tend to be diagnosed in relatively younger women than other types of cancer; however, the inclusion of some older patients may increase the risk of gynecologic cancer-associated VTE. Advanced stage was identified as a risk factor for VTE in patients with cervical cancer, and the development of VTE may shorten the survival period, indicating the importance of improving the rate of cervical cancer screening in young women in Japan. However, screening has not been shown to improve overall survival related to endometrial or ovarian cancer. Compared with cervical cancer, more patients with endometrial and ovarian cancers have advanced-stage disease or have received chemotherapy, which may increase the incidence of VTE and shorten the survival period. In this study, four patients developed ovarian vein thrombosis (three right and one left ovarian vein). Ovarian vein thrombosis typically tends to occur in patients after parturition, especially after Caesarean section. Fever and lower abdominal pain are the primary symptoms, and expansion of the ovarian vein is sometimes detected by imaging examinations. There is currently no consensus regarding the need for anticoagulant therapy for ovarian vein thrombosis, however, 13% of patients with ovarian thrombosis developed pulmonary thromboembolism due to thrombosis progression to the inferior vena cava. 31 No patients with ovarian vein thrombosis had severe thromboembolism in the present study.
Comparing the Risk Factors Reported in Other Countries with Those Identified in the Current Study of Japanese Patients.
An Adjusted Analysis with Cox Proportional-Hazards Model Considering the Risk Factors.

Inf: Infinity.
The current study had a few limitations. This was a retrospective study using data from our electronic medical records system and was specific to patients diagnosed with and treated for both gynecological cancer and VTE at our hospital. Our findings are therefore not necessarily applicable to other patient cohorts. Regarding the survival periods in patients with and without VTE, the median was not reached in patients without VTE during the study period, and patients may therefore need to be observed for longer in order to make an exact assessment. Additionally, this study included patients with asymptomatic VTE who were diagnosed coincidentally by elevated D-dimer levels during regular blood examinations or CT for diagnosis of cancer or assessment of therapeutic effects. However, not all patients registered in this study, especially those only receiving palliative treatment, received regular screening for D-dimer or CT, and the incidence of VTE may thus have been underestimated. A prospective study of VTE in patients with gynecologic cancer is needed to verify our findings.
Conclusions
We carried out a retrospective study of patients with gynecologic cancer who developed VTE. Several baseline characteristics differed between patients with and without VTE. The incidence of VTE and some of the risk factors were similar in Japanese patients with gynecologic cancer compared with patients in other countries
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
