Abstract
Introduction
Calf muscular vein thrombosis (CMVT) is defined as thrombosis in the soleal and gastrocnemius calf muscle veins. The soleal and gastrocnemius veins, distinct from the posterior tibial or peroneal veins, are intramuscular veins forming a sinus or plexus of veins. 1 Post-stroke paralysis is a common factor contributing to calf deep vein thrombosis (calf DVT), evidence shows that the calf muscle veins are the most common thrombosis locations after stroke (>90%). 2 Although CMVT has been known entity for 40 years, 3 thrombosis in the calf muscle has not yet been fully elucidated, and conclusive research on this disease entity is limited. But this does not imply that CMVT is of no clinical importance. The calf muscular vein has been suggested as being the origin of thrombosis in lower limbs, 4 and a proportion of the CMVT propagate rapidly into DVT and pulmonary embolism (PE), 1 which may lead to devastating consequences among hospitalized patients. Therefore, early and timely CMVT risk assessment after stroke should be highlighted.
More often asymptomatic than proximal DVT, CMVT may escape diagnosis if not ruled out in detail during patient evaluation, especially for stroke patients during rehabilitation who usually have impaired consciousness, cognitive impairment, or sensory deficits.5,6 Clinical studies rarely focused on CMVT until the shift from venography to venous ultrasonography.7-9 Duplex venous scanning (DVS) has been recommended as the standard diagnostic modality for CMVT because of its comparable accuracy, reduced invasiveness, and cost savings compared to venography. 10
To our knowledge, the literature on the prevention of CMVT in stroke patients during rehabilitation is sparse. Only a small amount of evidence suggested that the risk factors associated with CMVT in patients with cerebral hemorrhage included higher age, higher level of D-dimer, lower Glasgow Coma Score (GCS), and lower affected side muscle strength. 6 However, other factors, such as the patient's degree of paralysis and daily mobility skills, 11 which may affect the mobility of hemiplegic patients during rehabilitation, have not been considered.
Individualized estimation of stroke patients during rehabilitation with CMVT could be useful for obtaining a critical time window for clinical rehabilitation intervention and treatment. The nomograms, in which certain risk factors act together to allow precise predictions, are often applied to a variety of diseases.12-14 Nevertheless, to the best of our knowledge, nomograms for CMVT in stroke patients based on their degree of hemiplegic limb dysfunction and daily mobility skills during rehabilitation have not yet been developed.
This research aimed to establish an accurate and individualized prediction nomogram for CMVT after stroke during rehabilitation to facilitate clinical prediction of CMVT.
Methods
Study Design and Patients
This study was a retrospective observational study and data collection occurred from December 2015 to February 2019, all patients were from the Rehabilitation Medicine Center of the First Affiliated Hospital of Nanjing Medical University, China. All patients who were diagnosed with stroke (ischemic or hemorrhagic) by brain computed tomography (CT) or magnetic resonance imaging (MRI) scans were enrolled. Patients were excluded if they had (1) lower limb fractures; (2) malignant tumors; (3) inherited coagulopathy; (4) with a history of lower extremity venous thrombosis; (5) incomplete medical records.
Date Collection
Baseline demographic characteristics, including age and gender, were collected in detail from the electronic medical records of these patients. The clinical parameters included the subtype of stroke, hypertension, diabetes mellitus, coronary heart disease, chronic obstructive pulmonary disease(COPD), history of smoking, history of alcohol, and history of stroke were also assessed. Meanwhile, limb functional parameters were evaluated by professional rehabilitation physicians within 24 h after admission to the rehabilitation center, including the daily mobility skills, Brunnstrom stage (lower extremity), and calf triceps surae tone. The daily mobility skills, consisting of transfers, movements, and stairs, are part of the activities of daily living (ADL), which are evaluated by the Barthel index. Among them, transfers and mobility are 0–15 points, stairs are 0–10 points, and the total score of the Barthel index is 0–100 points.11,15 The Brunnstrom stage is a short and easily administered measure for assessing the motor function that is usually used individually to describe a patient's motor function. 16 This test describes the movement recovery based on the degree of spasticity and voluntary movement after a stroke. Stages 1–6 of the Brunnstrom stage represent different stages of motor recovery. 17 The calf triceps surae tone is assessed by the Ashworth Scale which is a simple 5-point scale that enables clinicians to quantify the amount of muscle resistance encountered during passive exercise. 18 Muscle spasticity can decrease the range of movement of the joints, which can disturb the function of the calf muscle venous pump. 19 Relevant assessment scale details were explained in earlier literature.11,15,18,20,21 Additionally, baseline coagulation parameters, blood lipid indexes, and hemoglobin levels were measured within 24 h of admission. Furthermore, the treatment measures of anticoagulant and antiplatelet were recorded. A color duplex ultrasound (DUS) examination of both lower extremities was routinely performed 3 days (3 ± 2 days) after admission by two experienced ultrasound physicians who were blind to the baseline rehabilitation status of each patient.
Statistical Analysis
According to whether the data were normally distributed, continuous variables were presented as mean ± SD or median (quartiles), categorical variables as frequencies, and percentages (%).
Group comparisons between CMVT and no CMVT were performed using one-way ANOVA, Kruskal-Wallis test, or Pearson chi-square test, as appropriate.
To choose the potential predictive features from stroke patients, we utilized the least absolute shrinkage and selection operator (LASSO) regression, which is an effective high-dimensional prediction method. Meanwhile, the optimal value of λ was determined via fivefold cross-validation. 22 Then, we performed multivariable logistic regression analysis by the features selected in the LASSO regression model to identify statistically significant predictors, which were then utilized to construct a nomogram. The features were presented as odds ratio (OR), 95% confidence interval (95% CI), and p-value.
To assess the overall discriminatory ability and calibration of the nomogram the calibration curve was plotted, meanwhile, the Hosmer-Lemeshow test was calculated (a significant test statistic indicated that the model is not well calibrated). In internal validation, Harrell's concordance index (C-index) and bootstrapping (1000 bootstrap replicates) were computed. 23 Finally, to evaluate the clinical benefits of the CMVT nomogram, we performed decision curve analysis (DCA) to measure the net benefits of different threshold probabilities in the stroke cohort. 24 Statistics R software (Version 4.1.2; https://www.R-project.org) was used for statistical analyzes, and P < .05 was considered statistically significant.
Results
Patients' Characteristics
A total of 360 eligible patients completed the investigation. According to ultrasound evaluation, all patients were separated into CMVT and no CMVT groups (mean age 60.07 ± 14.15 years).
Of these patients, 34.2% (123/360) were CMVT patients. All patients’ baseline demographic information, clinical and limb functional parameters, laboratory and treatment features in the two groups are given in Table 1. Of the 28 features collected from patients, four features were chosen based on non-zero coefficients calculated by LASSO logistic regression analysis (Figure 1(A) and (B)). These selected features included age, Brunnstrom stage (lower extremity), D-dimer, and antiplatelet therapy. These features were subsequently included in multivariate logistic regression analysis.

Features selection using the LASSO binary logistic regression model. Notes: (A) Log (Lambda) value of the 28 features in the LASSO model. A coefficient profile plot was produced against the log (lambda) sequence. (B) Parameter selection in the LASSO model used fivefold cross-validation via minimum criterion. Partial likelihood deviation (binomial deviation) curves and logarithmic (lambda) curves were plotted. Use the minimum standard and 1se (1-SE standard) of the minimum standard to draw a vertical dashed line at the optimal value. The optimal lambda produced four nonzero coefficients. Abbreviations: LASSO, least absolute shrinkage and selection operator; SE, standard error.
Differences between Demographic and Clinical Characteristics of CMVT and No CMVT Groups.

Notes: 0, 5, 10, 15 in each item of the Barthel Index represent different scores, the overall score on the Barthel Index ranges from 0–100; 1–6 in the Brunnstrom stage represent different stages of motor function stages; 0–4 on the Ashworth Scale (triceps surae) represent different spastic stages.
Abbreviations: CMVT, calf muscular vein thromboses; COPD, chronic obstructive pulmonary disease; PT, prothrombin time; APTT, activated partial thromboplastin time; FIB, fibrinogen; TT, thrombin time; TC, total cholesterol; TG, triglycerides; HDL, high-density lipoprotein; LDL, low density lipoprotein; HGB, hemoglobin.
Development of an Individualized Prediction Model
A multivariate logistic regression analysis identified that age, Brunnstrom stage (lower extremity), D-dimer, and antiplatelet therapy were independent predictive factors affecting CMVT (Table 2). The above independent predictors were then incorporated to develop a predictive nomogram (Figure 2).
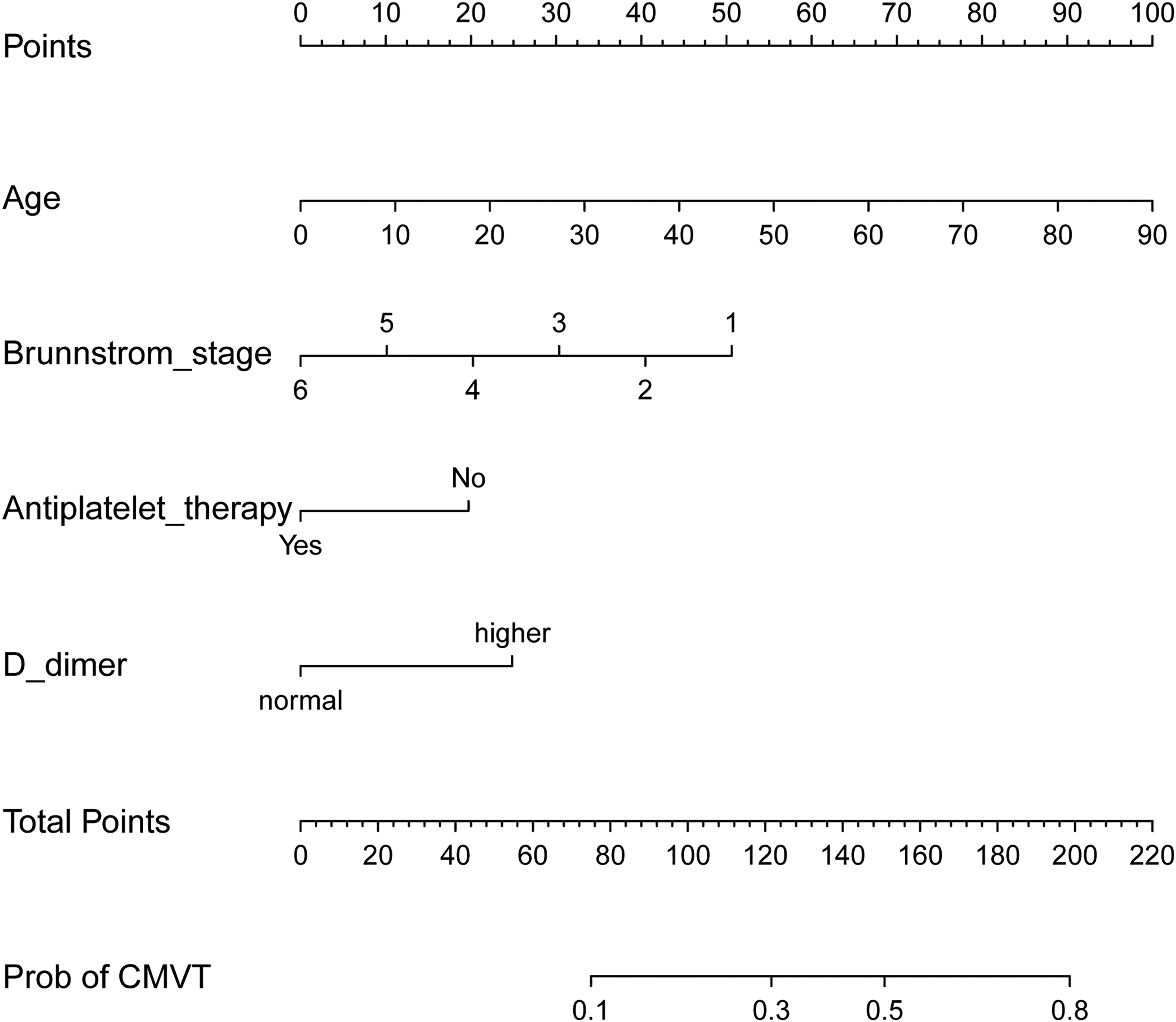
Developed CMVT nomogram. Notes: the CMVT nomogram was developed in the cohort, with the age, Brunnstrom stage (lower extremity), D-dimer, and antiplatelet therapy.
Prediction Factors for CMVT in Stroke Patients.

Note: β is the regression coefficient.
Abbreviation: CI, confidence interval.
Performance and Validation of the Nomogram
In this cohort, the calibration curve of the CMVT risk nomogram for stroke patients showed good agreement (Figure 3). The C-index for the CMVT prediction nomogram was 0.718 (95% CI: 0.663-0.773) and was confirmed to be 0.706 via bootstrapping validation indicating good discrimination against the model. In addition, the Hosmer–Lemeshow test, P = .217, suggested that the model was of goodness-of-fit.

Calibration curves of the CMVT nomogram prediction in the cohort. Notes: the x-axis represents the predicted CMVT risk. The y-axis represents the actual diagnosed CMVT. The diagonal dotted line depicts a perfect prediction by an ideal model. The solid line reflects the performance of the nomogram; a closer fit to the diagonal dotted line indicates a better prediction.
Clinical use
Finally, the CMVT DCA for the CMVT nomogram is presented in Figure 4. The decision curve revealed that if the threshold probability of a patient or doctor was >6% and <50%, using this nomogram to predict the calf muscular vein thromboses added more benefits than either the predict-all-patients as CMVT or the predict-none-patients as CMVT.

Decision curve analysis for the CMVT nomogram. Notes: The blue line represents the CMVT risk model, the fine line represents the predict-all-patients as CMVT, and the thick line represents the predict-none-patients as CMVT.
Discussion
In this study, we developed and internally validated an individualized prediction nomogram for stroke patients during rehabilitation with CMVT. We found four easy-to-assess variables based on age, Brunnstrom stage(lower extremity), D-dimer, and antiplatelet therapy for healthcare professionals to predict the occurrence of CMVT after stroke. The model performs well in predicting the risk of CMVT in stroke patients. Internal validation in the model revealed good discrimination and calibration ability, especially, our high C-index in the interval validation demonstrated that this nomogram can be extensively and accurately used for a large sample size. In addition, the DCA showed that the model is of clinical significance for decision-making over a range of probability thresholds.
Consistent with previous findings, this study found that age and D-dimer were important risk factors for deep vein thrombosis.25,27 We showed that older age was more prone to develop CMVT. Age has been identified as a risk factor for calf vein thrombosis after stroke in many prediction models. Additionally, these studies indicated that the incidence of calf venous thrombosis increases with age in stroke patients.28,29 In agreement with previous studies, we have found that a high level of D-dimer is an independent risk factor for calf venous thrombosis.29,30 D-dimer is produced by fibrinolysis and is elevated in the presence of a thrombus, but the increase of the D-dimer level can also be elevated in other diseases and therefore cannot be used solely to confirm the presence of DVT. 31
In terms of antiplatelet therapy, studies have shown that antiplatelet therapy plays an important role in acute and long-term prevention of recurrent ischemic stroke,32-36 however, the effect of antiplatelet therapy on the prevention of venous thromboembolism after stroke has been controversial. Some previous studies have found that antiplatelet therapy provides modest protection against venous thromboembolism after acute stroke,32,36,37 while a large retrospective study showed that aspirin appeared ineffective in preventing DVT in patients undergoing stroke rehabilitation. 38 In this research, for CMVT, we found that the absence of antiplatelet therapy resulted in an increased incidence of thrombosis.
Brunnstrom stage(lower extremity) as a risk factor for CMVT was identified in our nomogram. The lower the Brunnstrom stage, the greater the likelihood of CMVT. Until now, only a few pieces of evidence observed a significant association between gait function and the Brunnstrom stage in post-stroke patients, which result revealed that the Brunnstrom stage influenced walking ability.39,40 We, therefore, infer that the Brunnstrom stage may affect thrombosis by affecting gait function. However, future research is needed to prove this hypothesis. In addition, although we have considered the patient's daily mobility skills in the screening of risk factors, unfortunately, this factor was not included in our prediction model. This may be attributed to the retrospective nature of our data, the small sample size, or other factors that require further research to validate in the future.
To our knowledge, two other predictive models exist for DVT in stroke patients. Pan et al 41 developed a nomogram to predict DVT in patients within 14 days after the onset of stroke in the neurology department (AUC = 0.756). Cheng et al 28 published a model to assess early distal deep vein thrombosis in acute ischemic stroke patients (AUC = 0.767). Compared with these two models, we aimed at patients who entered the rehabilitation center for later rehabilitation and focused on building a simple and easy-to-operate thrombosis model from the perspective of hemiplegic limb function. Therefore, for patients in the rehabilitation phase, our model has stronger clinical utility. More importantly, our model is aimed at CMVT, a disease that has not been fully developed. Therefore, our prediction model has significant clinical implications.
The most significant and final argument for the clinical use of the model is based on the need to explain the individual need for additional prediction. To justify the clinical usefulness, the DCA was applied in the present study. The decision curve revealed that if the threshold probability of a patient or doctor was >6% and <50%, using this nomogram to predict the calf muscular vein thromboses added more benefits than either the predict-all-patients as CMVT or the predict-none-patients as CMVT.
Limitations
However, this present study has several limitations. First, this research was based on a retrospective analysis of the single-centered study, Therefore, a multicentre and prospective trial is needed to validate the accuracy of the model. Second, not all risk factors are considered in this research, thus, the role of other potential predictors like infection may need to be considered in future models. Finally, external validation is still required to further evaluate the performance of our nomogram.
Conclusion
We developed and validated a tool for assessing the individualized prediction of CMVT in stroke patients during rehabilitation. This novel nomogram showed reasonable accuracy and discrimination. Clinicians and patients can take more effective steps to control exercise patterns and medical interventions based on individual risk assessments. Future prospective studies including external validation are warranted to assess the prediction performance of the model in different populations.
Footnotes
Acknowledgments
We acknowledge Mr Chen and his son for their unconditional support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethics approval was granted by the Ethics Committee of the First Affiliated Hospital of Nanjing Medical University (approved number [2021-SR-438]. Since all the data were de-identified, the informed consent was waived.
Funding
.This trial was funded by the Nanjing Municipal Science and Technology Bureau (Grant number of 2019060002). The funding bodies had no role in the study design, data collection, analysis, and interpretation of data.
