Abstract
This article seeks to review the current status of treatment and prevention of venous thromboembolic disease (VTE) in cancer patients after the addition of direct oral anticoagulants (DOAC) to the therapeutic arsenal available. The suitability of DOAC use in complex clinical situations, poorly represented in clinical trials, is controversial and difficult for care activity, making the recommendations in clinical practice guidelines the focus of special attention in this area. Recently, several randomized trials have compared low molecular weight heparin (LMWH) to DOAC for the management of CAT. Potential drug interactions with DOACs or the increased risk of bleeding in intraluminal tumors require special precautions, as do metastatic or primary brain disease and comorbid conditions, such as renal or liver failure, which are not suitably represented in pivotal studies. Furthermore, few data are available for situations involving elevated bleeding risk, with thrombocytopenia levels below the inclusion criterion of clinical trials, or recurrence during active anticoagulant therapy. Similarly, it is less clear that patients and physicians accept the presumption that oral DOAC administration is more convenient than subcutaneous LMWH, particularly when drug absorption may be compromised. The non-inclusion or under-representation of patients at higher risk for complications with anticoagulation in randomized clinical trials, makes their use complex in certain situations in health care. This paper provides a practical review of current clinical guideline recommendations regarding LMWH and/ or DOAC to treat and prevent CAT, as well as the most controversial clinical conditions for their use.
Keywords
Introduction
Properly preventing and treating venous thromboembolic disease (VTE) is part and parcel of comprehensive care for cancer patients. The elevated incidence of VTE in the oncological population and potential increase in complications arising from morbidity and mortality, due to both cancer and its treatment, highlights the complexity we face in the daily care of cancer-associated VTE (CAT). Low molecular weight heparins (LMWH) are considered more suitable than vitamin K antagonists for specific CAT prophylaxis and treatment after demonstrating better safety and efficacy profiles. 1 The incorporation of direct oral anticoagulants (DOACs) in CAT prevention and treatment clinical practice guidelines, following the publication of randomized studies in cancer patients, expands the therapeutic arsenal available, while simultaneously begging the question of how to optimize the use of the various alternatives in each individual case.2–4
Specific clinical situations, such as renal or hepatic insufficiency, patients with extreme weight, or tumors associated with a higher risk of bleeding are often excluded or under-represented in randomized studies, hindering customized clinical care in these conditions based on scientific evidence.
Taking the CAT prevention and treatment recommendations of the leading scientific societies’ clinical guidelines5–8 as a reference, we have conducted a comprehensive bibliographic review of anticoagulant management following a CAT event in certain complex situations. Similarly, we have evaluated the estimated risks, establishing areas of specific precaution for the use of DOACs versus what has been the standard of care to date, LMWH.
Thromboprophylaxis in Cancer Patients on Active Chemotherapy: General Recommendations
Antithrombotic prophylaxis in oncology patients undergoing active chemotherapy poses a dilemma because, while it seeks to lower the risk of CAT without increasing the risk of bleeding, it has failed to prove a definitive effect on survival. Proper patient selection for thromboprophylaxis is essential. The Khorana model 9 is the most widely used predictive model. It has been validated both internally and externally, and rates risk according to characteristics such as tumor location, body mass index (BMI), and baseline platelet, hemoglobin, and leukocyte counts, but lacks the ability to assess bleeding risk. Recently, new specific predictive models, such as Vienna-CATS, 10 PROTECHT, 11 CONKO, 12 or COMPASS, 13 among others, and adding genomic risk profiles 14 are used in an attempt to stratify thrombotic risk and indication for thromboprophylaxis more selectively.
Numerous studies have examined LMWH use in outpatient prophylaxis, underscoring the decrease in thrombotic events among patients with risk factors, without evidence of a significant increase in major or non-major bleeding (MB), revealing a general trend towards the benefit of anticoagulation in individuals classified as at high risk for CAT. (Results are displayed in Table 1.)
Randomized Studies of Thromboprophylaxis in Cancer Patients With Outpatient Treatment.

The recent publication of the AVERT 19 and CASSINI 20 trials with apixaban and rivaroxaban, respectively, have explored a new therapeutic alternative in intermediate-high risk subjects as defined by a Khorana score of ≥ 2. The study populations differ. Whereas the CASSINI study confirmed the absence of VTE at inclusion, had a more heterogeneous, higher-risk population, and yielded risk reduction outcomes that failed to achieve statistical significance, the AVERT study did produce statistically significant results.
The main international clinical guidelines include the studies described in their recommendations, as indicated below:
The
The
The guidelines of the
The recent
The NCCN guideline does not support primary prophylaxis in all cases. Instead, it highlights the importance of a good predictive model to calculate the individual's personal and tumor- and treatment-related risk factors. It recommends prophylaxis with rivaroxaban or apixaban for situations of high risk for thrombosis, as per the predictive model chosen, preferring DOACs over LMWHs, for 6 months. 7
All clinical guidelines stress how imperative it is to individualize benefit and risk. They call for a duration of 3-6 months of prophylaxis, and take into account contraindications to prophylactic anticoagulation (active bleeding, known coagulopathy, spinal interventions, thrombopenia…), as well as the need to adapt DOACs on the basis of each individual's hepatic and renal function.
Clinical Guideline Recommendations for Treating CAT
Classically, CAT has been treated with LMWH given the results reported. The CLOT study displayed a 52% reduction in the risk of VTE recurrence with LMWH versus VKA (HR 0.48; P = 0.002) without any increment in bleeding risk. 22 In the CATCH study, tinzaparin absolutely and significantly reduced the risk of recurrence of symptomatic DVT with no increase in MB and a significant decrease in clinically relevant, non-major bleeding. 23
The current approach to CAT has recently been modified based on the results of the Hokusai-VTE, Select-D, Adam-VTE, and Caravaggio studies,24–27 published in the last three years. The most salient results are summarized in Table 2.
Randomized Studies of DOACS Versus LMWHs for the Treatment of CAT.

When considering therapy for CAT, the following factors must be assessed 28 : bleeding risk, tumor type, tumor burden, thromboembolic burden, gastrointestinal surgery or intestinal absorption disorders, renal insufficiency (RI), drug-drug interaction, patient preference, body weight, comorbidities, and polypharmacy.
The ITAC-ISTH recommendations are: LMWH for initial anticoagulation (the first 5-10 days) or, in the absence of high risk for gastrointestinal or genitourinary bleeding, rivaroxaban or edoxaban (after 5 days of parenteral anticoagulation); DOACs or LMWHs for early maintenance anticoagulation (up to 6 months) (with the aforenamed precautions) and, for extended anticoagulation, beyond 6 months, the risk/benefit ratio should serve to individualize the decision in each case. 6
The ASCO guidelines recommend initial anticoagulation with LMWH or rivaroxaban, as long as there is no renal function impairment with creatinine clearance <30 ml/min. LMWH, edoxaban, or rivaroxaban is contemplated for the first 6 months and the decision to prolong anticoagulation beyond 6 months must be individualized. Subjects with active cancer, metastatic disease, or those receiving chemotherapy might be candidates.
Increased gastrointestinal, genitourinary, and mucosal bleeding has been reported with DOACs and drug-drug interactions must be scrutinized prior to administration. 5
Only the last three published guidelines include data from the Caravaggio study. The
In its November 18, 2020 update, it states that the prolonged anticoagulation therapies of choice for CAT are LMWHs and DOACs for 6 months. It emphasizes that apixaban is the only drug with a proven safety profile similar to LMWH. Edoxaban and rivaroxaban increase gastrointestinal and, probably, genitourinary bleeding risk.8,29
In the latest
In the current
As for choosing the agent to be used, the following factors must be taken into account: renal impairment and liver disease, individualized bleeding risk, possibility of reversing anticoagulation, outpatient versus inpatient, complete blood count (CBC) monitoring, convenience of administration, patient preference, and governmental approval. DOACs are preferable for cases without gastric and/or esophageal tumors (apixaban and rivaroxaban for initiation and edoxaban after prior LMWH) and LMWH for patients with these malignancies. 7 All guidelines recommend great caution when using DOACs in patients with gastrointestinal tumors, inasmuch as elevated bleeding risk is one of their most notorious complications.
Special Situations:
Cerebral Tumors and Metastases
The most common central nervous system (CNS) tumors are metastases (24%), followed by high-grade gliomas (21%). These pathologies present a high risk of thrombotic events and bleeding. In some, such as renal and melanoma metastases, the risk of bleeding is even higher. 30 All these peculiarities force us to evaluate risk-benefit meticulously prior to initiating anticoagulation. In clinic practice, some tools are available to us, such as the bleeding risk index (PANWARDS), initially developed for non-oncologic patients, but which has also been evaluated in individuals with tumors. 31 The risk of bleeding can be up to 23% when scores exceed 25 (cutoff point).
Published data in this context focus primarily on prophylaxis in neurosurgical settings. The PRODIGE study (dalteparin vs placebo) exhibits a reduction in thrombotic risk over the first 6 months, albeit with more bleeding. 32 Several meta-analyses with heparins have confirmed this benefit, but not increased bleeding. 33
The standard of care in the treatment setting are LMWHs. At least 5 retrospective studies have focused on CNS tumors, concluding that LMWHs are both safe and effective.30,34 There is virtually no representation of CNS tumors in the bibliography on DOACs. The evidence we have is limited to a few retrospective studies, yielding encouraging results, as there appears to be no difference in efficacy and even less bleeding in the case of primary tumors. 35
The leading guidelines espouse heparins as standard for postoperative prophylaxis. 6 Both LMWHs and DOACs are included as treatment options. 5 The Spanish guideline (SEOM) is the only one that, according to the data described in the literature, refers to the option of reducing the dose of heparins by 25-50% when treating VTE in patients with brain metastases from renal tumors or melanomas, or in brain stem gliomas given the increased risk of hemorrhagic complications. 8
In conclusion, based on further scientific evidence, heparins are the treatment of choice (dalteparin, enoxaparin, tinzaparin) in this population. We should take into account the possibility of dose reduction in metastatic disease with a high risk of bleeding (melanoma, renal tumors, and gliomas). Treatment duration will depend on the risk of recurrence. At present we have little data to endorse treatment with DOACs, inasmuch as we only have retrospective studies, although they do appear to be safe and their use is already included in some guidelines.
Gastrointestinal and Genitouriniary Tumors
The clinical profile of VTE in oncologic patients differs depending on tumor location. Data extracted from RIETE (International Registry of Patients with VTE), which includes patients with breast (938), prostate (629), colorectal (1189), and lung (1191) cancers, indicate that 55% of patients had metastatic disease (42%, 36%, 53%, and 72%, respectively). During the course of anticoagulation therapy, the rate of thromboembolic recurrences (rVTE) was similar to that of MB in subjects with breast or colorectal cancer. In contrast, in individuals with prostate cancer, the rVTE was half that of the MB, and in those with lung cancer, it was double that of the MB. Thus, since the pattern of MB differs according to tumor type, different anticoagulation strategies should be designed according to tumor location. 36
In gastrointestinal (GI) and genitourinary (GU) tumors, there is a higher risk of bleeding at the tumor location and several studies report a higher incidence of bleeding at these levels in people receiving DOACs, as seen in the published analysis from the HOKUSAI study, in which the authors analyzed data obtained regarding VTE recurrence and bleeding according to tumor location. They found that with dalteparin, MB rates were lower in GI (3.6%) and GU (1.4%) tumors, than with edoxaban (12.7 and 4.6% in GI and GU respectively). 37 Several studies have likewise confirmed that the percentage of MB in GI tumors is clearly higher with rivaroxaban than with LMWHs.38–40
To conclude, in cases of GI or GU tumors and VTE, DOACs should be used with caution. Other available anticoagulation options should be evaluated until specific studies in this patient population to aid in identifying those that are not at increased risk of bleeding. 38
Gastrointestinal Absorption Alterations
Oncology patients undergoing surgy for tumors in the gastrointestinal tract (GIT) can have altered absorption of orally-administered drugs. When prescribing anticoagulation with DOACs, the following factors should be taken into account: administration with food or while fasting, changes in gastric pH due to antacids, site of absorption of DOACs and its possible alteration due to surgery of the GIT, use of a nasogastric tube (NGT), bioavailability of each DOAC, and the formulation of each drug. 41
Rivaroxaban and edoxaban are absorbed in the stomach, duodenum, and proximal ileum, with high bioavailability (80%-90%). Assimilation is pH-dependent and requires and acid environment for absorption. Therefore, in patients with gastrectomy or gastric bypass, their bioavailability may be diminished. In the case of rivaroxaban and in fasting conditions, alterations in dose-dependent absorption also occur, such that with a dosage of 2.5-10 mg there are no variations, but when the dose is 15-20 mg, the bioavailability is reduced by 66%; hence, it is recommended that this drug be taken with food.
Apixaban, in turn, is principally absorbed in the distal small intestine and ascending colon; consequently, it is independent of gastric pH for assimilation. In situations of small bowel or right colon resection, the use of this drug should be avoided given the potential reduction in its absorption.
We know that dabigatran is absorbed in the upper GIT. Its formulation in capsule form is therefore not recommended for use in NGT. Its absorption is pH-dependent and requires an acidic environment; hence, its bioavailability could decrease subjects with gastrectomy or gastric bypass. 42
Renal Insufficiency
RI is a common and underestimated complication if we rely solely on serum creatinine level; the glomerular filtration rate (GFR) should be calculated. 43
RI alters plasma protein binding, volume of distribution, and non-renal elimination of many drugs, which increases toxicity or determines treatment inefficiency. This clinical situation increases the risk of thrombotic events and MB secondary to anticoagulant therapy.
Patients with severe RI (glomerular filtration rate < 30 ml/ min) included in randomized studies with LMWHs do not exceed 2% of the study sample. Pharmacodynamic studies follow different methodologies, are not comparable, and the recommendation for dose adjustments should not be extrapolated and must be made according to the technical data sheet. 44
LMWHs are excreted mainly by the renal route, with potential dose accumulation due to decreased clearance and increased elimination time. 45
Bemiparin, enoxaparin and nadroparin have increased renal elimination, whereas tinzaparin and dalteparin are less dependent on elimination by GFR. 46
The CLOT and CATCH studies, that compared the use of LMWHs versus antivitamin K oral anticoagulants, included a limited number with GFR < 60 ml/min (24% and 15%, respectively). Post-hoc analyses of patients with RI exhibited similar safety profiles with no evidence of excess bleeding due to LMWH accumulation in RI.47,48
As for the use of DOACs, randomized trials conducted in cancer patients excluded patients with severe RI (GFR < 30 ml/ min).24–27 The FDA and EMA have approved the use of DOACs in patients with RI and GFR > 15 ml/ min, recommending dose adjustment according to pharmacokinetics.
Currently, the existing data are insufficient to recommend routine use of DOACs to treat/ prevent thrombosis in these patients.
Clinical guidelines and expert consensus give preference to the use of LMWHs, particularly in the presence of additional factors that increase the risk of bleeding.
Liver Failure
The association between liver damage and the coagulation cascade is multifactorial and complex. On the one hand, we find a state of coagulopathy due to alterations in the synthesis of coagulation factors, with prolongation of PT and APTT, but which correlates poorly with the risk of hemorrhage. On the other hand, partial deficiency of anticoagulant proteins is accompanied by improvements in thrombin synthesis, which determines a procoagulant state. This fact would explain why the belief that patients with chronic liver failure are self-anticoagulated has been proven to be false. 49
Subjects with chronic liver failure receive less prophylactic anticoagulant therapy than those without. Randomized trials have failed to validate the safety of LMWH prophylaxis in this setting. Retrospective studies reveal that death and gastrointestinal bleeding rates are not increased in this cohort. Prescription of anticoagulation therapy in these individuals is affected by other factors, such as personal experience, routine in clinical practice, and whether the patient refuses treatment. 50
Clinical guidelines and expert consensus contraindicate anticoagulation with severe or uncompensated coagulopathy, given the high risk of bleeding. NCCN guidelines consider the presence of liver failure to be an absolute contraindication for DOACs. Given that DOACs entail a higher risk of bleeding than LMWHs and that there are no adequate safety data in this scenario, if anticoagulation treatment is required, priority should be given to treatment with LMWHs.
Thrombocytopenia
Thrombocytopenia is defined as a platelet count <100,000/µL.
Its pathogenesis is multifactorial
51
:
In solid tumors, it is often the consequence of chemotherapy treatment. It is common at diagnosis in individuals with hematologic tumors. Other associated conditions (infections, drugs, coagulopathy due to consumption, etc) can affect the degree of thrombocytopenia
VTE and thrombocytopenia are more usual in cancer patients. Treatment of both concomitant processes is highly heterogeneous
52
and specific studies are needed in this population to determine an optimal management strategy, given that cases of VTE, cancer, and thrombopenia are excluded from studies with both LMWHs and DOACs. Thrombopenia notwithstanding, the risk of recurrent VTE remains high.
There is a paucity of data on the use of DOACs in patients with VTE and severe thrombocytopenia (< 50,000/µL). 53 Clinical practice and recommendations in the leading guidelines indicate that LMWHs can be contemplated in this subgroup of patients if the dosage is titrated according to bleeding risk and the patient's individual risk factors. For example, in case of acute VTE with low risk of progression or established VTE, LMWHs could be prescribed at 50% of the previous dose or at a prophylactic dose. Its use below 20 000 platelets/µL is not contemplated.5,7
High Risk of Bleeding
Cancer patients have a higher risk of bleeding associated with anticoagulation therapy than non-cancer patients.
54
The factors associated with fatal bleeding during the first 3 months of VTE treatment remain unclear and, while there are several predictive models of MB due to anticoagulant therapy, none are specific to the oncology population; for example:
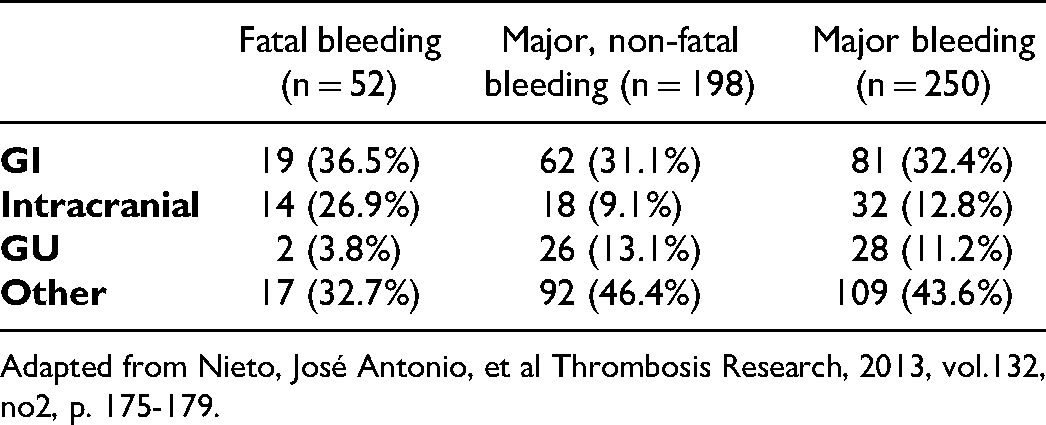
In the RIETE study, of the 24 395 patients evaluated, 546 (2.24%) had MB and 135 (0.55%) suffered a fatal hemorrhage. The GIT was the most common site (40%), followed by intracranial hemorrhage (25%). Fatal bleeding was independently associated with the factors reflected in the table (Table 3) present at the time of VTE diagnosis.
55
Another tool that evaluates the risk of bleeding in subjects about to undergo anticoagulant treatment is the IMPROVE scale, which establishes a high bleeding risk when the score is Bleeding Rates According to Tumor Location Based on Subanalysis of the Riete Study. Adapted from Nieto, José Antonio, et al Thrombosis Research, 2013, vol.132, no2, p. 175-179.
Adjustments of Doac Doses According to Summary of Product Characteristics.

No*: not recommended if >120 kgs or BMI >40.
Catheter-Associated Thrombosis
There are 2 types of catheters, centrally implanted and peripherally inserted central catheters (PICC), the latter being the ones that entail the highest risk of thrombosis. The incidence of asymptomatic and symptomatic thrombosis related to PICC ranges from 2.2-58% and 2%-15%, respectively 57 and the diagnostic test of choice is echo-Doppler, reserving CT angiography for specific locations. There are explicit guidelines for managing catheter- associated thrombosis. Secondary, not primary prophylaxis is recommended, after 3-6 months of treatment if the catheter has not been removed and, in case of replacement, treatment should be maintained for at least the first 30 days post-insertion of the new device. 58
None of the pivotal DOAC studies stratify as per the presence or absence of a central catheter. Two studies with rivaroxaban have been published that specifically address this population. The first one was retrospective, with 83 patients, and found that only 3.6% had the catheter removed and severe bleeding presented in 2.4%. 59 The other was a prospective study of 70 patients all of whom retained their line; thrombosis reoccurred in 1.43% and bleeding developed in 12.9%. 60
The pre-eminent clinical guidelines endorse LMWHs as the treatment of choice for at least 3 months and even indefinitely, if the catheter is maintained (which is advisable unless it fails to function or associated complications arise).5–8
In conclusion, LMWHs should be used for both treatment and secondary prophylaxis, given the current dearth of evidence with respect to DOACs in this context.
CAT Recurrence
Recurrences of thrombotic events in cancer patients are relatively common place up to 11%. This phenomenon tends to happen in the first months following the initial event; is more prevalent in advanced disease, increases as survival worsens. 61 At a practical level, though risk tables do exist, such as the modified Ottawa Score (MOS), it has only been validated in cases of cancer managed on an outpatient basis. 62
In recurrences, the drug of choice is LMWH and the general recommendation is to maintain them for at least 6 months. The initial maneuver should be to ensure that patients have had no prior reduction in medication; if they have, anticoagulant treatment should be reestablished at optimal doses. If the heparin regimen is perfectly adjusted for the patient, there are retrospective studies that support increasing it up to 120-125% at the time of recurrence. 63 This recommendation is included in the different clinical guidelines in which this dose increase or switching to DOACs are the therapeutic options put forth for individuals on LMWH therapy, despite the absence of evidence that advocates DOACs in this context. In those cases, in which recurrence occurs while on DOAC treatment, the action of choice is to begin LMWHs. Over the long term, the possibility of switching to DOACs after the first 6 months of treatment is contemplated.5–8
Therefore, in light of the data currently available, our recommendation is that the drug of choice is LMWH for thrombotic recurrence. Another important issue is treatment duration following a second episode; it appears that indefinite therapy is the most appropriate course of action, particularly in cases of active tumor, unless contraindicated.
Drug-Drug Interactions
Drug-drug interactions are one of the areas of greatest concern when prescribing DOACs for oncology patients undergoing treatment with antineoplastic drugs. Clinical practice guidelines endorse evaluating potential interactions when initiating or modifying each new line of oncologic therapy in patients receiving concomitant anticoagulation.5–8
LMWHs are compatible with almost all antitumor drugs. However, all DOACs known to date are P-glycoprotein substrates for their metabolism. Furthermore, apixaban and rivaroxaban are substrates of cytochrome P450 (CYP3A4), while neither edoxaban nor dabigatran are substrates of this metabolic pathway. 2 This is one of the metabolic pathways followed by both antitumor and supportive drugs administered to these patients.
Consequently, introducing drugs metabolized via this pathway can potentially increase the risk of recurrence by decreasing plasma concentrations of DOACs, while inhibitory drugs can increase plasma concentrations of DOACs. As a result, there would be a risk of increasing potential bleeding. At present, there are scarcely any studies that yield data on clinically relevant interactions between anticancer therapy and anticoagulants and their pharmacokinetic and pharmacodynamic effects in oncology patients. 64
Extreme Weights
There is no solid scientific evidence on the potential drawbacks of using anticoagulant drugs at fixed doses, but given that typically, they have been dosed by weight, there is concern about possible recurrences and/ or bleeding when DOACs are administered at unadjusted dosages to high- or low- weight individuals. 65 Generally, LMWHs are dosed in terms of IU/kg, with few restrictions on use in extreme weight situations.
In phase III studies conducted with DOACs versus LMWHs in cancer patients, only the dose of edoxaban was decreased to 30 mg/ day for subjects weighing < 60 kg 24 with no change in cases of obesity. Clinical practice guidelines recommend care when using them in individuals weighing > 120 kg and indicate the need for more data in patients with extreme weights before indicating routine use in this setting. 5 DOAC dose modification as per their technical data sheets are shown in Table 4. (Table 4)
Patient Preference and Adherence
CT patients experience the diagnosis of thrombosis as life-threatening, but anecdotal in the overall oncologic process. Multiple factors impact treatment decision-making, such as clinical experience, professional training, routine workplace practice, etc Patients should be empowered to make decisions about what treatment is necessary and why it is prescribed, with full information regarding benefits and harm. While practitioners often consider LMWH treatment too aggressive because of its route of administration, patients view the injection as a necessary evil to prevent future symptomatology and insignificant compared to the rest of their oncologic treatment as long as it does not interfere with it. Their main concern is how the thromboembolic event may affect the treatment of their tumor.70,71 Furthermore, patients integrate LMWH treatment into their daily routine and normalize it.
As for DOAC use in cancer patients, an increased risk of clinically relevant bleeding must be taken into account, especially in gastrointestinal malignancies (Hokusai and SELECT-D).24,25 The increased possibility of drug-drug interactions must be compared and considered between DOACs versus LMWHs. 3 Given the absence of head-to-head comparative studies and the heterogeneity among clinical trials, it is not possible to recommend the use of one type of DOAC over another.
Conclusions
International guidelines on the management of CAT have recently incorporated DOACs into their recommendations, for both treatment and prevention in stratified high-risk patients. In contrast to the well-established use of LMWHs in this context, pre-treatment evaluation with DOACs should cautiously consider the presence of tumors or special clinical situations, which are not always well-represented in randomized trials (Table 5). The possibility of drug-drug interactions in patients receiving active oncological treatment, who have alterations, or special situations, such as extreme weights, that involve secondary modifications in absorption and increased potential bleeding in people with gastrointestinal or urological endoluminal tumors, are some of the precautions pointed out when establishing therapeutic recommendations with DOACs in CAT. Baseline comorbidities, such as renal or hepatic insufficiency, and the increased risk of bleeding in situations like thrombocytopenia must be factored into the recommendations of expert guidelines based on management extracted from the experience, which is still scarce in the use of DOACs.
Treatment of CAT In Specific Situations.
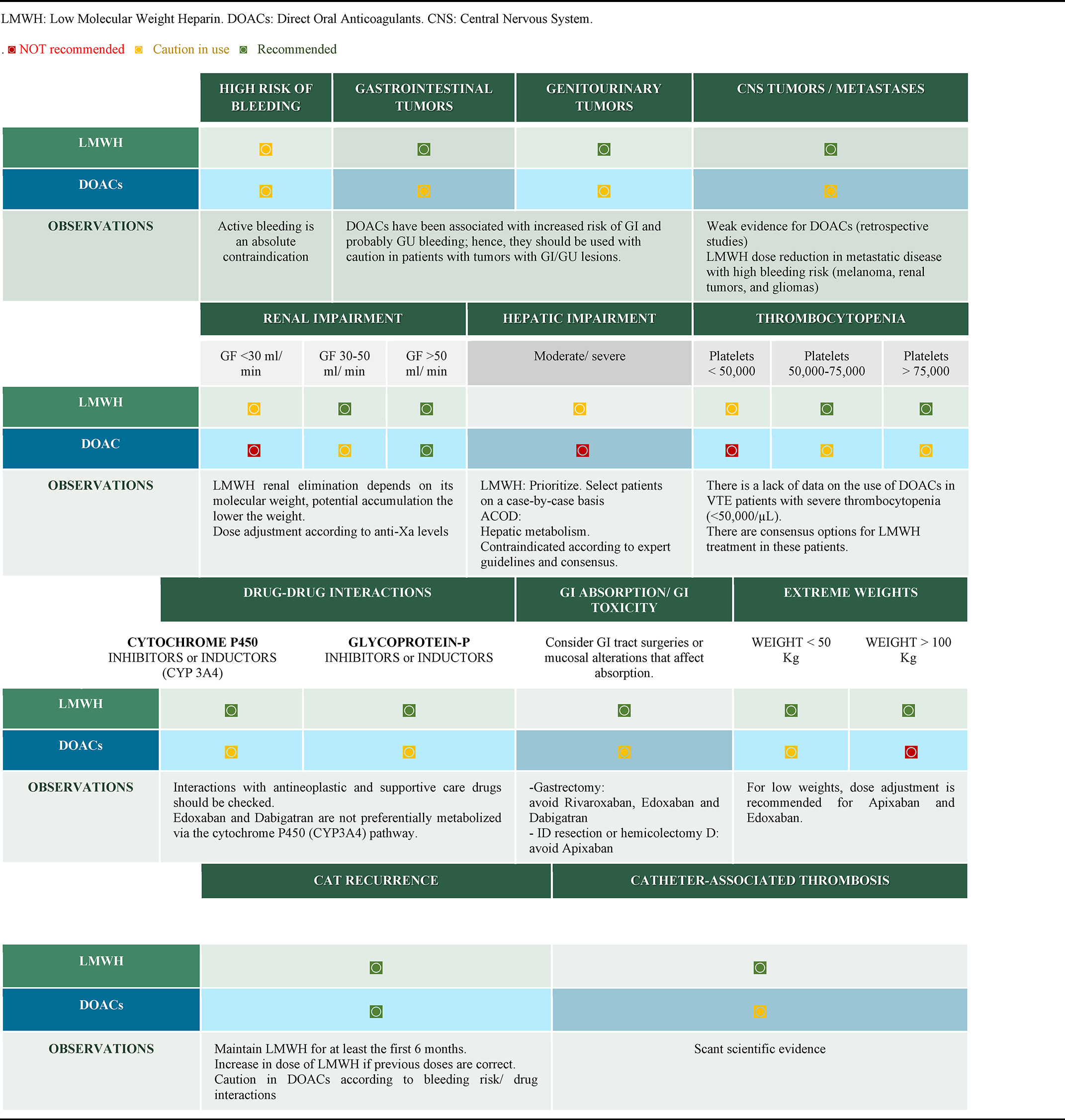
Individualization in the search of optimal strategies to treat CAT must undoubtedly be founded on the data derived from scientific evidence, while fully aware of the gaps that this may leave in situations that are poorly supported by research for more careful and safer patient care.
Footnotes
Acknowledgements
We thank Priscilla Chase Duran for editing the manuscript.
Author Contributions
M.S. coordinated the study. All authors developed the project, provided clinical information, comments, drafted, and improved the manuscript and participated in the critical review of the document.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
MSF has participated in advisory boards for Pfizer; has received Speaker fees from LEO Pharma, Rovi, Sanofi; has received Travel and accommodation support for attending symposia from Sanofi, LEO Pharma, Rovi. MEBV has participated in advisory boards for Bayer, Pfizer; has received Speaker fees from Rovi, LEO Pharma; has received Travel and accommodation support for attending symposia from LEO Pharma. BCB has participated in advisory boards for BMS, Sanofi; has received Speaker fees from BMS, Sanofi, Rovi, LEO Pharma; has received Travel and accommodation support for attending symposia from BMS, Rovi. PGV: has participated in advisory boards for Leo Pharma; has received Speaker fees from LEO Pharma; has received Travel and accommodation support for attending symposia from Sanofi. MLVT: has no conflict of interest to declare. MELP: has no conflict of interest to declare. DAR has no conflict of interest to declare.
Ethics Approval and Consent to Participate
The current study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. For this type of study, no formal consent is required.

