Abstract
Sepsis-associated disseminated intravascular coagulation (DIC) is related to marked hemostatic changes such as transient thrombocytopenia secondary to the endogenous activation and consumption of platelets. This study measured markers of platelet function in 103 adult ICU patients with clinically established sepsis-associated DIC to determine the biomarker association with disease severity. Patients were categorized as having no DIC, nonovert DIC, or overt DIC using the International Society of Thrombosis and Hemostasis scoring system. Plasma levels of CD40L, platelet factor 4 (PF4), platelet-derived microparticles, and microparticle-associated tissue factor were quantified. Markers of platelet activation were significantly elevated in patients with DIC compared to healthy individuals. This increase was independent of platelet count. Levels of PF4 differed based on the severity of DIC and differentiated nonsurvivors and survivors. These findings suggest that the markers of platelet activation in DIC may not be regulated by the number of circulating platelets and may be independent of the factors leading to their consumption.
Introduction
Sepsis is a dysregulated host response to infection that can lead to life-threatening organ dysfunction and contributes significantly to mortality in the United States and worldwide.
1
Patients with sepsis can develop a systemic coagulation disorder known as sepsis-associated disseminated intravascular coagulation (DIC), which is characterized paradoxically by an increased risk of both bleeding and thrombosis.
2
The development of DIC doubles a septic patient’s risk of death.
3
Currently, there is no single biomarker or test that can provide a definitive diagnosis of DIC. The most widely accepted scoring system is provided by the International Society of Thrombosis and Hemostasis (ISTH).
4
The ISTH scoring system categorizes patient scores on a scale from 0 to 5 and is comprised of points awarded for reduced platelet count and fibrinogen level in addition to elevated
Although several biomarkers related to sepsis and DIC have been investigated over time, no single biomarker is capable of predicting the disease severity and prognosis. 5,6 A consequence of widespread coagulation activation includes exhaustion of platelet and clotting factors that can lead to severe bleeding and hemorrhage into tissues. 4 The role of platelets in the DIC inflammatory response is important to investigate as there may be platelet biomarkers that can better predict disease severity or DIC-associated mortality.
The development of thrombocytopenia secondary to platelet consumption is one of the most readily available clinical indicators of DIC development; however, the role of platelets in DIC is not merely one of passive consumption. The primary function of platelets is hemostatic in nature; platelets adhere to damaged endothelium, form a primary hemostatic plug to prevent blood loss, and ultimately contribute to the formation of a more stable fibrin clot. Platelets also bridge hemostasis, immunity, and inflammation. They are one of the first cell types to respond to compromised vasculature, invading pathogens, and sepsis. In addition to responding to damaged endothelium, platelet aggregation can be induced by contact with a pathogen or by high levels of circulating inflammatory factors. 7 Upon activation, platelets secrete their granular contents, releasing many inflammatory factors into circulation, to initiate the coagulation process. Accordingly, the analysis of platelet function, including factors linking platelet function with inflammation and endothelial function, is relevant to the understanding of sepsis and DIC.
In this investigation, several markers of platelet activation such as CD40L, platelet factor 4 (PF4), microparticles (MPs), and microparticle-associated tissue factor (MP-TF) have been measured to determine the extent of platelet activation in sepsis and DIC. Circulating levels of soluble CD40L may potentially serve as a reliable indicator of DIC severity and associated disease prognosis, as it is primarily derived from platelet activation. 8 Soluble CD40L has been detected in the plasma of patients with inflammatory conditions; however, specific studies related to sepsis-associated DIC are minimal.
Platelet factor 4 is a chemokine released from α granules of activated platelets. Bacterial presence activates platelets causing them to release their granulocytic contents. 9 Platelet factor 4 has been shown to charge-dependently bind to both Gram-positive and Gram-negative bacteria. Platelet factor 4 may play a role in DIC either through the enhancement of humoral immunity secondary to increased platelet activation and subsequent PF4 release or by some other mechanism.
Microparticles are small vesicles composed of cellular membrane and membrane proteins that are released into the circulation from numerous cell types, including platelets. Procoagulant MPs, defined by positive TF staining (MP-TF) via flow cytometry, have been detected in the plasma of patients with sepsis. 10,11 The production of MP-TF and procoagulant activity may be detrimental to the host in sepsis. The association between levels of factors generated or released by platelets and a patient’s clinical status or outcome may be confounded by the consumption of platelets during the coagulopathy characteristic of sepsis-associated DIC.
The purpose of this study was to determine the levels of the platelet activation biomarkers CD40L, PF4, MP, and MP-TF in the plasma of patients with DIC to determine markers that may predict disease severity and overall mortality. In addition, comparisons were made to determine the effect of platelet count on biomarker levels. These results will help to determine if platelet markers may be a better predictor for overall mortality associated with DIC.
Materials and Methods
Patient Samples
Plasma samples were collected, after obtaining consent, from 103 adult patients with sepsis and suspected DIC in the intensive care unit (ICU) within 48 hours of ICU admission. In this study, patients were required to meet the following criteria for systemic inflammatory response syndrome and have an identified or suspected focus of infection. Systemic inflammatory response syndrome was defined as the presence of 2 or more of the following: (1) temperature <36 °C or >38 °C, (2) heart rate >90 beats per minute, (3) respiratory rate >20 beats per minute or Pa
Whole blood was collected into 3.2% sodium citrate tube in a ratio of 9:1 and centrifuged to prepare platelet-poor plasma. Platelet-poor plasma was collected, aliquoted, and stored at −80 °C prior to sample analysis. In addition, plasma from 50 healthy volunteers (25 males and 25 females) was purchased from George King Biomedical. The ages of these volunteers ranged from 19 to 54 years, with a mean age of 32 years. These volunteers were nonsmokers, nonmedicated, and of geographically diverse origins.
Platelet Biomarkers
Commercially available enzyme-linked immunosorbent assays were used to perform these assays according to the manufacturer’s specifications. Assays for PF4, platelet-derived MP, and MP-TF were obtained from Hyphen BioMed. The assay for CD40L was obtained from R&D Systems.
The DIC Score
The ISTH 2001 scoring algorithm was used to assign a DIC score to each patient. This score is calculated on the basis of platelet count, international normalized ratio (INR),
ISTH scoring system for DIC.
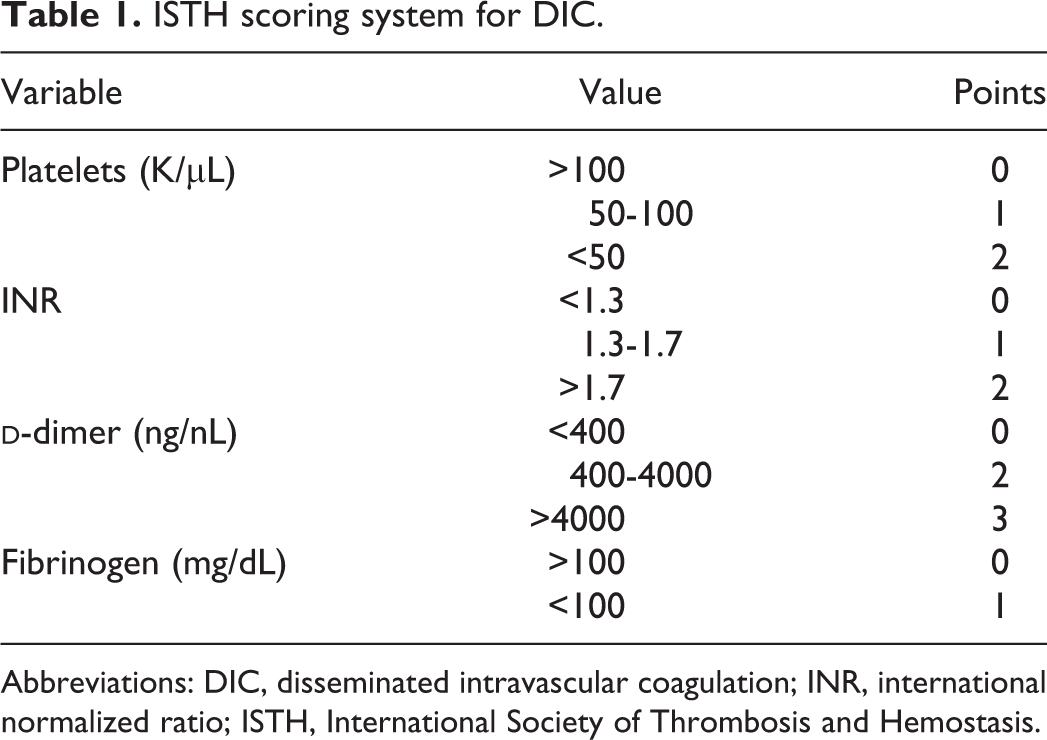
Abbreviations: DIC, disseminated intravascular coagulation; INR, international normalized ratio; ISTH, International Society of Thrombosis and Hemostasis.
Statistical Analysis
Data are presented as mean ± SD or mean ± SEM as specified. P < .05 was used as the cutoff for statistical significance. Results were tabulated and stored using Microsoft Excel (Microsoft Corporation). Statistical analysis was performed and graphs were generated using GraphPad Prism (GraphPad Inc).
Biomarker levels in patient populations are presented as mean ± SEM. Differences in platelet biomarker levels between the 3 groups and the healthy control cohort were assessed using the Kruskal-Wallis analysis of variance with Dunn multiple comparison test and P < .05 as the cutoff for significance. Differences in biomarker levels between the 2 patient groups (ie, survivors and nonsurvivors) were analyzed using the Mann-Whitney test. Predictive values were analyzed using receiver–operator curve analysis, with the main output for this analysis being the area under the curve (AUC).
Results
Patient Cohort Baseline Characteristics
Plasma samples were collected from 103 adult ICU patients with sepsis. Patient cohort baseline characteristics are shown in Table 2, and disease severity and outcome information are shown in Table 3. The demographics of this cohort are within the range typical for sepsis patients in the literature. This includes the age distribution (57 ± 18.5 years, mean ± SD) and the gender balance (46.6% male). The healthy control group consisted of 50% male, 50% female, and had a mean age of 32 years.
Baseline Characteristics of the Utah Patient Cohort.

Abbreviation: BMI, body mass index.
Outcome and Disease Severity Information.

Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; DVT, deep vein thrombosis; MI, myocardial infarction; PE, pulmonary embolism; SOFA, Sequential Organ Failure Assessment.
The primary outcome measure in this patient cohort was 28-day mortality. This cohort consisted of 88 survivors and 15 nonsurvivors, resulting in an overall 28-day mortality rate of 14.6%. Since this study included both patients with only sepsis and nonovert DIC as well as those with severe DIC, our mortality rate was slightly lower than the estimated 20% to 30% seen in other studies. 1,4
Association of Baseline Platelet Biomarkers With DIC Score
A DIC score was assigned using the ISTH scoring algorithm for overt DIC as previously described. Of the 103 patients, 20 had sepsis without DIC, 59 had sepsis and nonovert DIC, and 24 had sepsis and overt DIC.
Significant variation in levels of platelet biomarkers (MP-TF, CD40L, PF4, and MP) based on DIC score was observed. Although Figure 1 demonstrates a trend toward changes in platelet markers between patients in the different DIC score categories compared to healthy controls, no significant differences in CD40L, MP, MP-TF, or PF4 were observed based on DIC severity. Significant elevation in all platelet markers was observed in all groups compared to the healthy controls.

Baseline platelet biomarker levels stratified by disseminated intravascular coagulation score. Significance calculated between groups using the Kruskal-Wallis analysis of variance with Dunn multiple comparison test and P < .05 as the cutoff for significance (indicated by *). Data are shown as mean ± SEM. (A) Levels of Microparticles-tissue factor (MP-TF); (B) Levels of CD 40 ligand (CD40L); (C) Levels of Platelet factor 4 (PF4); (D) Levels of Microparticles (MP).
Association of Biomarkers With Platelet Count
Recently, significant associations have been demonstrated between levels of platelets and hemostatic, inflammatory, and endothelial markers in patients with sepsis-associated coagulopathy. 12 In line with this analysis, patients were divided into groups based on platelet count on ICU day 0 of <100 000/µL (n = 21), 100 000 to 149 000/µL (n = 20), or ≥150 000/µL (normal range; n = 61). The normal range for platelet count is 150 000 to 400 000/μL. Patients with a platelet count of 100 000 to 149 000/µL have thrombocytopenia; however, a platelet count within this range does not pose a significant bleeding risk. For patients with a typical platelet count of close to 150 000/μL, a platelet count within the 100 000 to 149 000/µL range may not represent a significant drop in platelet count. A platelet count of <100 000/µL indicates a marked drop in platelets from the normal range and may lead to an increased risk of bleeding. A platelet count of <50 000/µL indicates a markedly increased bleeding risk; however, only 3 patients fell within this range in this cohort, and thus, these patients could not be analyzed separately.
Figure 2 shows the level of individual platelet markers stratified by platelet count. CD40L and MP increased with increasing platelet count, with statistically significant differences between patients with normal platelet counts of ≥150 000/µL compared to those of <100 000/µL for both parameters and for patients with a platelet count of ≥150 000/µL compared to those with a platelet count of 100 000 to 149 000/µL for MP. The trend observed for PF4 was more complex, with the highest level of PF4 measured in patients with platelets of 100 000 to 149 000/µL and significant differences between 100 000 to 149 000/µL and <100 000/µL and between ≤150 000/µL and <100 000/μL. Levels of MP-TF showed no difference between varying platelet counts.

Baseline platelet biomarker levels in patients stratified by platelet count. Significance calculated between groups using the Kruskal-Wallis analysis of variance with Dunn multiple comparison test and P < .05 as the cutoff for significance (indicated by *). Data are shown as mean ± SEM. (A) Levels of Microparticles-tissue factor (MP-TF); (B) Levels of CD 40 ligand (CD40L); (C) Levels of Platelet factor 4 (PF4); (D) Levels of Microparticles (MP).
Association of Baseline Biomarkers With Mortality
The 28-day mortality in this patient cohort was 14.6% (88 survivors and 15 nonsurvivors). Information on time to mortality was not available. As shown in Figure 3, minimal associations were observed between markers of platelet function and patient outcome. Only PF4 showed a significant association with mortality (P = .016, AUC = 0.70), with significantly lower levels observed in nonsurvivors compared to survivors. All other platelet biomarkers had weak predictive values for mortality (AUC: CD40L, 0.55; MP, 0.53; MP/TF, 0.62).
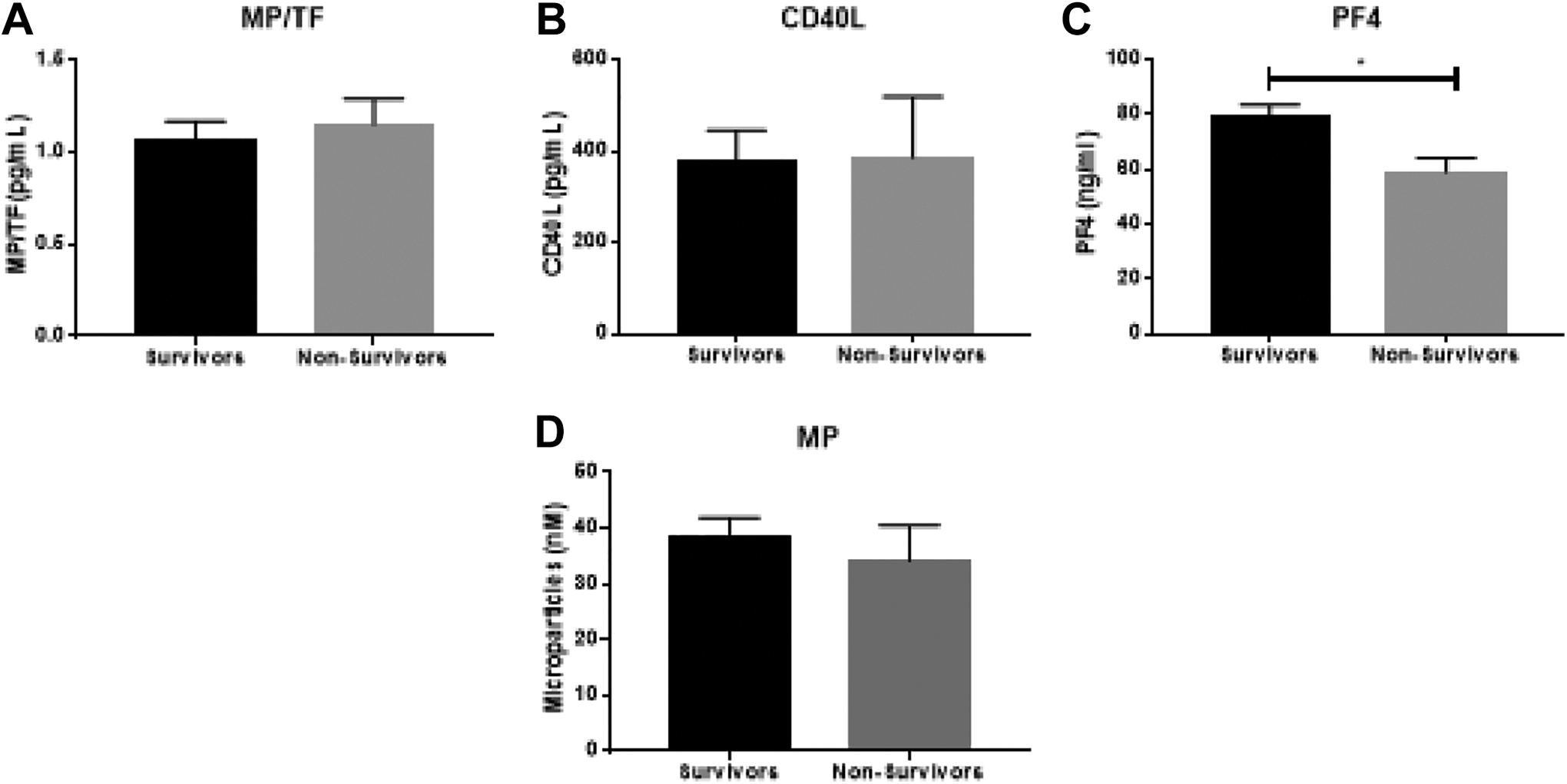
Association of baseline platelet biomarker levels with survival. Significance calculated between groups using the Mann-Whitney test with P < .05 as the cutoff for significance (indicated by *). Data are shown as mean ± SEM. (A) Levels of Microparticles-tissue factor (MP-TF); (B) Levels of CD 40 ligand (CD40L); (C) Levels of Platelet factor 4 (PF4); (D) Levels of Microparticles (MP).
Discussion
The incidence of sepsis, on a global scale, remains high. In 2017, a reported 48.9 million cases of sepsis were recorded worldwide and 11.0 million sepsis-related deaths occurred. Overall, sepsis-related deaths comprised 19.7% of all global deaths. 13 –15 The cost of sepsis in the United States is also staggeringly high as it costs health care more than US$24 billion each year to treat. 14 Sepsis complicated by DIC accounts for approximately 20% to 40% of septic cases. 14,15 Since DIC can frequently lead to fatal respiratory failure, its mortality rate is noticeable higher than that observed in patients without DIC. Therefore, the early diagnosis and treatment of sepsis-induced DIC is critical for improving prognosis. 16,17
This study examined the relationship between biomarkers of platelet function and the severity of DIC in a cohort of patients with sepsis and well-defined coagulopathy. Although several different criteria systems currently exist to determine the presence of a sepsis-induced coagulopathy, the ISTH score has been utilized as the main source for identifying patients with overt versus nonovert cases of DIC. Each of the criterion have their own set of advantages and disadvantages. The ISTH score, for example, has been reported to have poor sensitivity, particularly in patients with infectious diseases. On the other hand, Ding et al found that the ISTH score was best able to predict nonsurvivors in the ICU and was also proven to be an independent risk factor for mortality. 18 Umemura et al similarly reported that ISTH overt DIC screening on the day of ICU admission was associated with a lower mortality, and the association became stronger if the screening was repeated 2 days later, suggesting that DIC screening alone might be sufficient for reducing mortality. 18,19 It is important to emphasize, however, that the ISTH score only incorporates platelet count and not platelet function as one of its criterion.
It may be important to look beyond platelet count into platelet function to help identify patients at high risk for developing DIC and its associated complications such as thrombosis. This theory is consistent with other studies that have focused on the contribution of platelets to the pathophysiology of sepsis beyond a reduced platelet count since platelets are well-known players in coagulation and likely mediators of DIC development. In addition to their role in thrombosis and hemostasis, platelets are also acknowledged as essential activators of the immune response, reacting to infection and disturbed tissue integrity and contributing to inflammation, pathogen killing, and tissue repair. 20 A deeper understanding of the contribution of platelet biomarkers to the development of sepsis-associated DIC is necessary to determine their ultimate clinical significance. This is a complex matter to consider as platelets are not only inflammatory mediators that contribute to vascular and tissue injury but also play important roles in resolution of inflammation and tissue repair. 21 This study supports the contributory role of platelet biomarkers despite overall reduced platelet counts, as significant elevations in platelet biomarkers were observed despite any correlation with DIC severity.
In this study, levels of biomarkers of platelet activation (CD40L, MP, MP-TF, and PF4) increased compared to normal healthy controls. When stratified according to ISTH severity, there was no difference observed between patients with sepsis alone and patients with sepsis and either nonovert or overt DIC. This suggests that although platelet activation plays a role in sepsis-associated DIC pathophysiology, the severity of the DIC may not be predicted by platelet function.
The interpretation of levels of factors released by platelets in patients with sepsis and DIC is complicated by the dual processes of platelet activation and platelet consumption. Activated platelets may secrete factors at high levels. However, this effect may be canceled out by sufficient reduction in platelet number. However, it is important to understand platelet function in sepsis and DIC, as platelets are involved not only in hemostasis but also in the response to infection. Previous analyses have shown that sepsis patients with severe thrombocytopenia demonstrate a different molecular profile than those with platelet counts within the normal range. 12 Accordingly, patients were divided into groups of normal platelet count (≥150 000/μL), mild thrombocytopenia (100 000-149 000/μL), and moderate to severe thrombocytopenia (<100 000/μL) for subanalyses.
In this study, platelet-related biomarkers demonstrated an overall inverse relationship with decreased platelet counts, but not to DIC severity. It is important to keep in mind, however, that platelet depletion is a major characteristic of sepsis-associated DIC, as circulating platelets are reduced due to a consumptive coagulopathy. Reduced platelet count is included in the ISTH algorithm for the diagnosis of DIC and thrombocytopenia, in conjunction with elevated INR which is commonly used as a screening test for sepsis-associated coagulopathy. Analysis of the association of the level of platelet biomarkers with platelet count revealed that the circulating levels of these biomarkers are largely controlled by platelet depletion. Platelet depletion was also reflected in the association with hemostatic markers. 22,23 The increases in INR and decrease in fibrinogen observed in patients with reduced platelet counts are reflective of the same consumptive process. 22
The relationship of platelet biomarkers to platelet count was largely dictated by platelet depletion. CD40L, PF4, and MP were significantly reduced in patients with reduced platelet counts compared to those within the normal range. The reduction of these markers in patients with dramatically reduced platelet counts indicates that the degree of platelet consumption in these patients overcomes the increased secretion of these markers by activated platelets. The relatively limited association of platelet biomarkers with the severity of DIC and mortality is attributable to this same process rather than to a lack of involvement of platelet-related factors in disease pathophysiology. Measurement of PFs at an earlier time point may provide insight into the disease progression of patients who are likely to develop coagulopathy, as dramatic platelet activation precedes platelet depletion. Measurement of platelet count at both early and late time points in disease would also allow for analysis of not only platelet count but change in a patient’s platelet count over time. Due to the wide range of normal platelet counts (150 000-400 000/μL), some patients who have experienced a drop in platelet count of more than 50% may still be classified as having a normal platelet count, whereas some patients with only minor drops in platelet count may be classified as thrombocytopenic. Further study of this phenomenon should occur in patients with more detailed information available regarding the timing of the onset of illness.
Despite the increased incidence of mortality in DIC sepsis and the major role that platelets have in its disease pathology, in this study, only PF4 showed significant association with mortality. Platelet factor 4, in fact, may be protective since survivors of DIC sepsis exhibited higher levels of PF4 than nonsurvivors. Platelet activation during sepsis triggers the release of PF4 from the alpha granules of activated platelets. The release of PF4 can lead to downstream stimulation of alveolar macrophages in order to produce chemokines promoting neutrophil recruitment and edema formation. 24 Moreover, PF4 also accelerates generation of activated protein C, which can counteract the increasing procoagulant state during sepsis that may promote DIC. Similar to protective findings of PF4 in sepsis DIC patients in this study, PF4 has been found to increase survival in cases of endotoxic shock. 25
In this study, markers of platelet activation were significantly elevated in patients with sepsis and DIC compared to healthy individuals. This increase was largely independent of platelet count, underscoring the importance of activation processes to this disease process. Of the markers studied, only PF4 showed statistically significant difference based on DIC score status or mortality and differentiated the nonsurvivors compared to survivors. Levels of CD40L, MP-TF, PF4, and MP showed significant association with platelet count, increasing in a stepwise manner with increases in platelet count. The overall elevated levels of platelet activation markers in DIC help to underscore the importance of platelets to this disease process. These studies also suggest that the markers of platelet activation in sepsis-associated DIC are primarily regulated by the number of circulating platelets and may be independent of the factors leading to their consumption.
Footnotes
Acknowledgements
The authors have acknowledged the skillful assistance of the staff of the Hemostasis Research Laboratories of the Department of Pathology and the Loyola University Medical Center. The authors are thankful to Dr Eva Wojick, Chair of the Department of Pathology, for her support in facilitating this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
