Abstract
Protein C (PC) deficiency, caused by mutations of the PROC gene, is a common inherited risk factor of thromboembolism (TE) among Thai people. This study aimed to investigate the association of 3 single nucleotide polymorphisms (SNPs; −1654 C/T, −1641 A/G, −1461A/T) at the PROC promoter region with PC activity and the risk of developing TE. A total of 216 patient s with TE, diagnosed at aged 0 to 20 years, and 102 healthy adults were enrolled. The SNPs were identified by Sanger sequencing. Protein C activity was measured using an automated functional clotting assay. Linear and logistic regression analyses were used to determine the association of SNPs with PC activity and the risk of TE. Patients and controls with homozygous TAA (119.6% ± 26.1%) and CGT haplotypes (102.7% ± 22.6%) had significantly lower PC activity than those with a homozygous CAA haplotype (140.4% ± 44.9%); P = .027 and .016, respectively. However, none of these haplotypes increased the risk of TE. This study suggested that the 3 PROC promoter SNPs were shown to be associated with lower PC activity but did not increase the risk of TE.
Introduction
Protein C (PC), a vitamin K-dependent plasma anticoagulant, is activated by the thrombin–thrombomodulin complex at the surface of endothelial cells. Activated PC, augmented by protein S (PS), inhibits factor V and factor VIII in the coagulation cascade. Thus, a deficiency in PC leads to an increased risk of developing thromboembolism (TE), mainly venous TE (VTE).
Protein C deficiency is the most common inherited thrombophilia in Asian populations, 1,2 while the factor V Leiden and prothrombin 20210 mutations are the most common in Caucasians. 3,4 Protein C deficiency is classified into 2 types: type 1 displays decreases in both PC activity and antigen levels, and type 2 displays decreased PC activity with normal PC antigen levels. Protein C deficiency is caused by mutations of the PROC gene.
PROC, located on chromosome 2q13-q14, is 11 kilobases in length 5 and comprises nine exons and 8 introns. 6 Transcription starts at exon 1 while translation starts at exon 2. 6 The most common mutation of PROC gene among Thai and Taiwanese Chinese populations is R147W 7 or R189W, according to Human Genome Variation Society. In Caucasians, there are 4 common mutations of PROC: R230C, Q132stop, R306stop, and 3363insC. 8,9 While most of the known mutations occur within the coding region of PROC, there have been reports that single nucleotide polymorphisms (SNPs) at the promoter region are associated with PC activity and increased thrombotic risk. Single nucleotide polymorphisms at the PROC promoter region significantly impact the regulation of transcription via disruption of the binding sites for transcription factors, namely hepatocyte nuclear factor (HNF)-1, HNF-3, 10 and the nuclear factor I/CCAAT transcription factor family. 11 Moreover, the 2 promoter SNPs (−1654C/T and −1641A/G) are in very high linkage disequilibrium with 2 SNPs in intron 2 (857 and 894) which have significant nuclear factors binding. 12
Three SNPs (−1654C/T, −1641A/G, and −1476A/T) have been identified in the promoter region of PROC. 13 –16 Associations of these SNPs with PC levels and the risk of developing TE were found in many studies of adult populations. In Dutch patients, 14 homozygous CGT was found to increase the risk of developing TE (odds ratio [OR]: 1.6; 95% CI: 1.0-2.5) with a significantly lower mean PC level of 94% compared to homozygous TAA at 116% (P < .001). In accordance with a study in France, 15 CG haplotypes (−1654C/T and −1641A/G SNP combination) also had significantly lower PC antigen levels in both homozygous (103.1%) and heterozygous (106.0%) patients compared to non-carriers (111.5%; P = .010), and an increased risk of TE (OR: 1.4; 95% CI: 1.0-1.9). In Chinese patients, the TT genotype at −1654C/T of PROC was found to be associated with pulmonary TE (OR: 2.2; 95% CI: 1.2-4.0). 16 However, no similar studies have been conducted in children.
This study aimed to investigate the association of the SNPs −1654 C/T, −1641 A/G and −1461A/T at the promoter region of PROC gene with the risk of developing TE in pediatric population and their associations to the PC activity.
Methods
Study Design
This study is a case–control study that enrolled 206 patients, diagnosed with either arterial TE (ATE) or VTE at the Department of Pediatrics, Ramathibodi Hospital, Mahidol University, Bangkok, Thailand between November 1998 and October 2018. The diagnosis of TE was confirmed by imaging study. The investigation of thrombophilia in patients included PC, PS and antithrombin activity, homocysteine level, and antiphospholipid antibody. The 102 control participants were healthy adults to ensure that the controls will not later develop TE because subjects with heterozygous PC deficiency usually present with TE during childhood until adulthood. 17 This study, for which we used leftover DNA samples from patients and control participants that were obtained for other, unrelated research purposes, was approved by the Ethics Committee Involving Human Subjects of Ramathibodi Hospital Faculty of Medicine (ID 03-61-66).
Patients
Inclusion criteria
Patients were children and adolescents aged ≤20 years at the time of diagnosis with either ATE or VTE. The diagnosis of TE was confirmed by imaging study. The control group was healthy adults aged 20 to 56 years without a history of TE.
Exclusion criteria
To study the association of the SNPs and the development of TE, patients who did not have DNA samples were excluded. To study the association of the SNPs and PC activities, patients with the following factors were excluded: not having PC activities, known mutations of the PROC gene, age <6 years, 18 receiving warfarin therapy, nephrotic syndrome, liver diseases, and cyanotic heart disease. 19
Laboratory Procedures
DNA extraction, PCR amplification, and purification
DNA samples were extracted from venous blood using a salting-out method. We used the numbering system of Foster et al, in which position 1 marks the beginning of translation. 5 A 574-bp fragment of the PROC promoter region (−1920 to −1347) was amplified by polymerase chain reaction (PCR) using specific primers as described previously: 16 5′-ATTGGGATGGCATGTTCATTG-3′ and 5′-CCCTGGCTGGA GGATTCAG-3′. The PCR conditions were as follows: 95 oC for 5 minutes; 35 cycles at 95 oC for 45 seconds, 58 oC for 45 seconds and 72 oC for 60 seconds; and 72 oC for 7 minutes. After the PCR products were confirmed by electrophoresis through a 1.2% agarose gel and ethidium bromide staining, the products were purified using a Geneaid purification kit (Geneaid Biotech).
DNA sequencing and mutation analysis
Sanger DNA sequencing on samples from all patients and controls was conducted by Humanizing Genomics Macrogen, from which mutation analysis of the −1738 A/G, −1654 C/T, −1641 A/G, and −1461A/T SNPs at the promoter region of PROC was performed.
Protein C activity
Protein C activity was measured using an automated functional clotting assay (Instrument Laboratory [ACL 8000]) of all of the control participant samples. For patients, PC activity was tested after the diagnosis of TE. In patients with low PC activity levels, the activity assays were repeated to confirm the results. Protein C investigation was included as a part of the thrombophilia investigation in our institute.
Statistical Analysis
Descriptive statistics for age, gender, and PC activity were calculated using STATA version 14.2 (StataCorp). Genotype data were checked for genotyping error based on significant deviation from Hardy-Weinberg equilibrium in the control group using PLINK version 1.07. 20 Calculations involving individual SNPs including their allele frequencies and testing for association between each SNP and the risks of developing TE were also conducted using genotypic logistic regression analysis with heterozygous/homozygous coding (--hethom option) as well as global association tests consisting of both genotypic (“2 degrees of freedom” ie, testing the effect of each genotype separately) Fisher exact test and Cochran-Armitage trend test in PLINK.
Haplotypes of 3 SNPs (−1654C/T, −1641A/G, and −1461A/T) were estimated using the expectation-maximization algorithm through the haplo.em function of haplo.stats package version 1.7.9 21 on R 3.6.2. 22 Mean and standard deviation of the PC activity in each haplotype and haplotype-pair group were calculated using R. Association between haplotypes and the TE outcomes were assessed using logistic additive effect model through haplo.stats’s haplo.cc function. This is a wrapper function that calculates the haplotype frequencies in the cases and controls, performs the logistic regression through the haplo.glm function 23 with logit link function, and performs the global score test for association. 24 Association between haplotypes and PC activity, adjusted for gender and mean-centered age, was assessed using a linear additive effect model through the haplo.glm function. The association between haplotype-pairs and PC activity, also adjusted for gender and mean-centered age, was modeled using R’s lm (linear model) function. The results of these tests were considered statistically significant when P < .05.
Results
A total of 318 participants were enrolled: 216 patients aged 0 to 20 years with a diagnosis of TE and 102 healthy adult control participants without a history of TE. The median (interquartile range) ages of the patients were 8.5 (3.7-12.6) and the controls 29.0 (24.0-33.8) years, P < 0.001. The control group comprised of adults whose level of PC activity had reached a mature level and were healthy without TE until adulthood. The gender distribution was 53.2% male and 46.8% female in the patient group, and 30.4% male and 69.6% female in the control group (P < .001).
A total of 216 patients and 102 controls were analyzed for the presence of the 3 promoter SNPs and associations with TE. Those with PC activity data, which met the inclusion and exclusion criteria (82 patients and 102 controls), were further analyzed for the associations of the SNPs with PC activity. The mean ± SD of PC activity in patients was 118.6% ± 40.7%, and 131.8% ± 26.1% in controls. The median (range) of PC activity in patients was 111.0 (64% to 250%) and 129.5 (69% to 211%) in control. None of the patients and controls had a PC activity below the respective age-appropriate normal level. The difference in mean PC activity levels between the patients and the controls adjusted by age and gender was significant (P = .0125).
Diagnoses of TE in the patient group were as follows: ATE in 136 (63%) with arterial ischemic stroke (112; 51.8%), systemic ATE (23; 10.6%) and purpura fulminans (2; 0.9%; VTE in 78 (36.1%) with deep vein thrombosis (DVT) and/or pulmonary embolism (29; 13.4%), cavernous sinus thrombosis (28; 13%), portal vein thrombosis (PVT; 12; 5.6%), and other venous TEs (9; 4.1%); and both ATE and VTE in 2 (0.9%) with catastrophic anti-phospholipid syndrome. Patients with both ATE and VTE were allocated into both the ATE and VTE groups for subgroup analysis.
While most (115; 53.2%) of the patients had no underlying disease, 101 (46.8%) patients had the following underlying diseases: malignancy in 18 (8.4%); thalassemia in 12 (5.5%); systemic lupus erythematous in 11 (5.1%); vasculitis in 9 (4.2%); heart disease in 9 (4.2%); liver disease in 8 (3.7%); infection in 7 (3.2%); catheter-related TE in 7 (3.2%); neurologic disease in 6 (2.8%); chromosomal anomaly in 6 (2.8%); nephrotic syndrome and end-stage renal disease in 5 (2.3%); and vascular anomaly in 3 (1.4%). The investigation of thrombophilia demonstrated hyperhomocysteinemia in 16 (7.4%) patients, phospholipid antibody in 11 (5.1%), PC deficiency in 4 (1.9%), PS deficiency in 2 (0.9%), and homocysteinuria in 2 (0.9%).
Genetic Analysis and Identification of a New PROC SNP (−1738G/A)
We determined the genotypes of the 3 known SNPs: −1654C/T (CC, CT, and TT), −1641A/G (AA, AG, and GG), and −1461A/T (AA, AT, and TT). The distribution of allele frequencies in patients and controls at −1654C/T was C: T = 0.48: 0.52 and 0.47: 0.53, respectively (P = .799), at −1641A/G was A: G = 0.80: 0.20 and 0.82: 0.18 (P = .524), and at −1641A/G was A: T = 0.80: 0.20 and 0.82: 0.18 (P = .524).
The distribution of genotypes of each SNP is shown in Table 1. Combinations of all 3 SNPs resulted in the identification of 3 haplotypes (TAA, CAA, and CGT). The distributions of each haplotype in patients and controls are also shown in Table 1. The most common haplotype was CAA.
Genotype Frequencies of the Single Nucleotide Polymorphisms (SNPs; −1654C/T, −1641A/G, −1461A/T) at Promoter Region of PROC Gene and Odds Ratios for Developing TE in All Patients, Arterial Thromboembolism (ATE) Group, and Venous Thromboembolism (VTE) Group.a
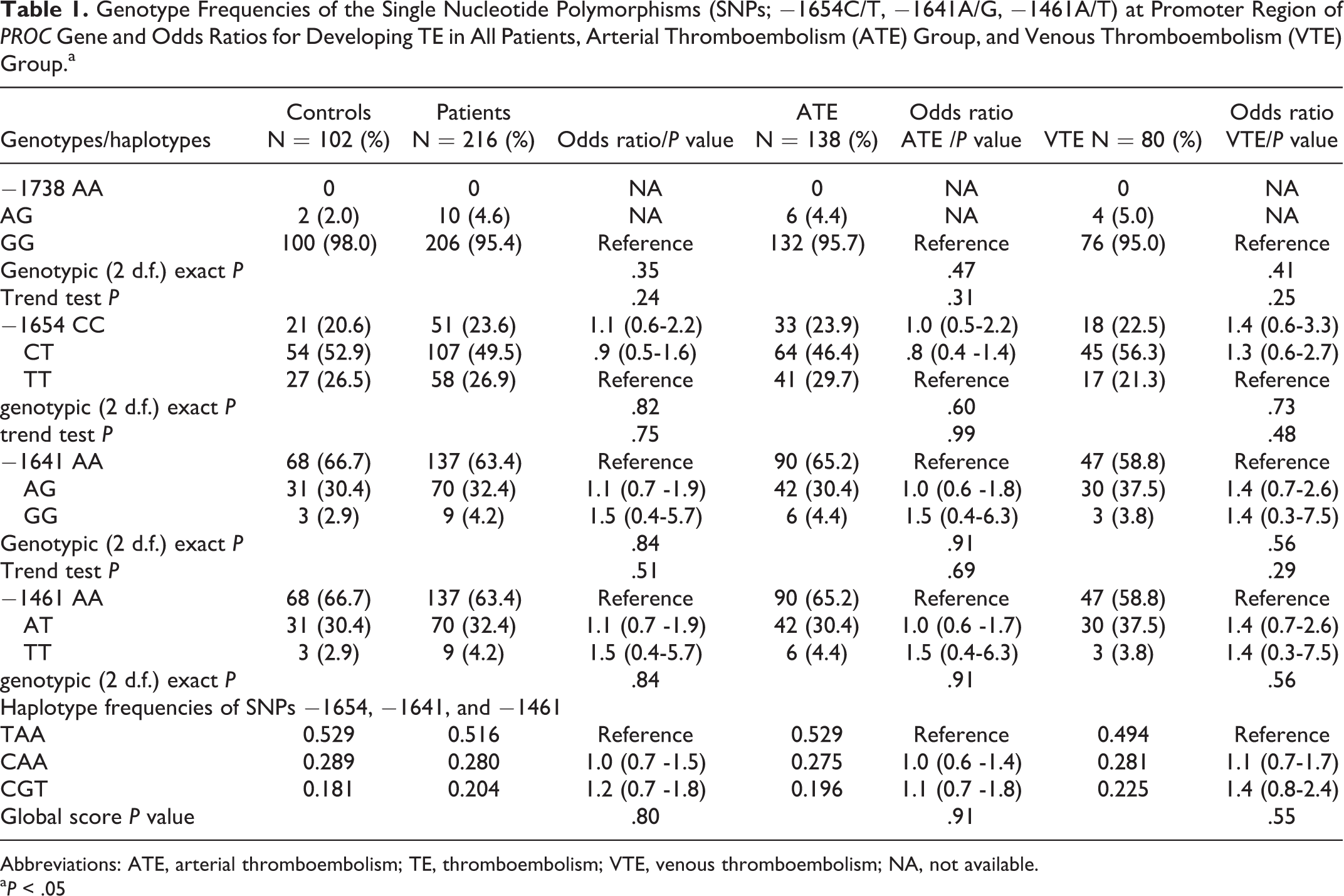
Abbreviations: ATE, arterial thromboembolism; TE, thromboembolism; VTE, venous thromboembolism; NA, not available.
a P < .05
We also identified a new SNP at −1738G/A that had 2 genotypes (GG and AG) in both patients and controls with allele frequencies A: G = 0.02: 0.98 and 0.01: 0.99, respectively (P = .355). However, the frequency of the A allele at −1738G/A in both groups was very low compared with the other SNPs (−1654, −1641, and −1461). Thus, we did not include −1738G/A in combination with the other SNPs in the determination of genotypes.
Association of SNPs With PC Activity
Protein C activity of patients whose age ≥6 years without factors affecting the level, and PC activity of controls according to each haplotype and haplotype pairs of the SNPs −1654C/T, −1641A/G, and −1461A/T are shown in Table 2. The results of multiple linear regression analysis of the SNPs and PC activity (adjusted for age and gender) are shown in Table 3. The data of PC activity from both patient and control groups were pooled together in this analysis based on haplotypes and haplotype pairs. Patients with homozygous CGT haplotype had the lowest PC activity (102.3% ± 22.6%), followed by homozygous TAA haplotype (119.6% ± 26.1%). Both homozygous CGT and TAA had significantly lower PC activity than those with a homozygous CAA haplotype (140.4% ± 44.9%; P = .027 and .016, respectively). The haplotype pairs and PC activity result remained the same for the subgroup analysis in the patient and control groups. There was a trend of lower PC activity in the homozygous TAA (119.6% ± 26.1%) and CGT haplotypes (102.7% ± 22.6%) compared with the non-carriers of TAA (126.8% ± 38.4%) and the non-carriers of CGT (126.2% ± 35.5%) respectively, but the differences were not statistically significant. The homozygous CAA haplotype group (140.4% ± 44.9%) seemed to have higher PC activity compared with the non-carriers (122.9% ± 28.7%), but the differences were not statistically significant either.
Protein C Activity in Individuals With 0, 1, or 2 Copies of Each Haplotype of the SNPs −1654C/T, −1641A/ G, and −1461A/T and Each Genotypes.

aThe numbers are based on the case–control samples from this study and therefore not representing the haplotype distributions in the population.
bMean ± SD.
Multiple Linear Regression Analysis of Protein C Activity According to Haplotypes and Haplotype Pairs of SNPs; −1654C/T, −1641A/ G, and −1461A/T, Adjusted for Age and Gender.
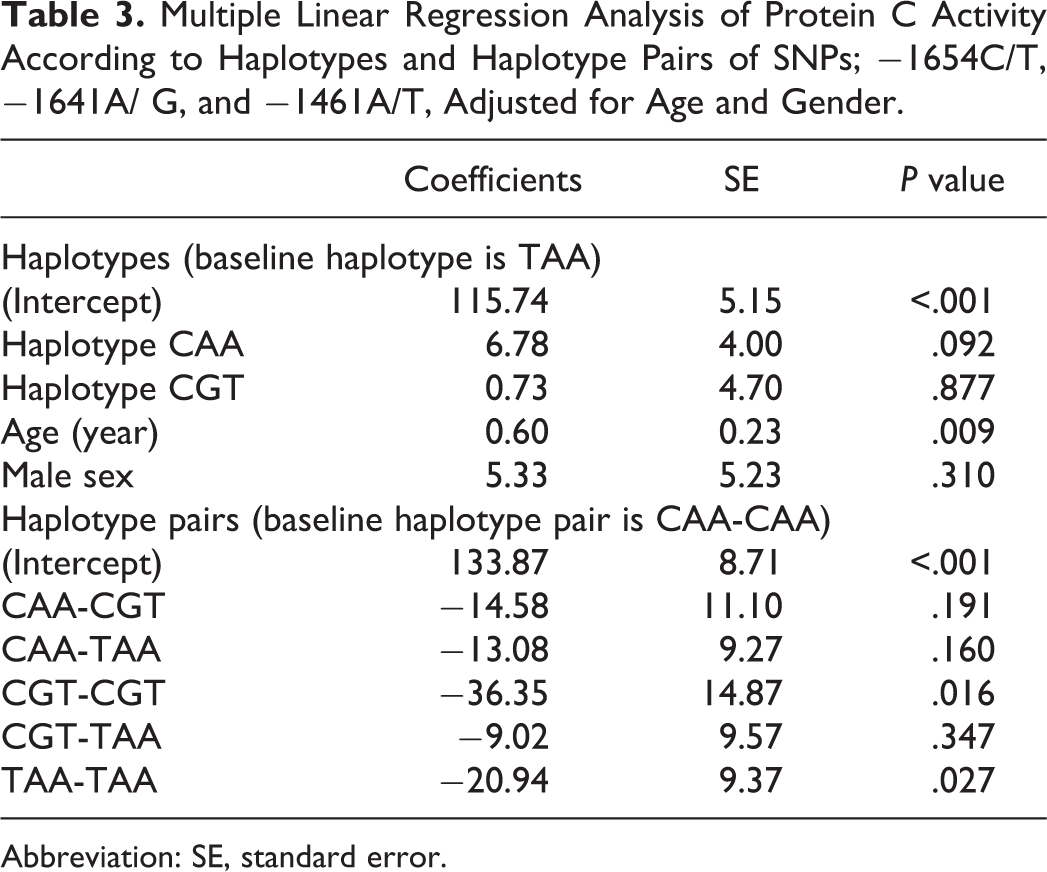
Abbreviation: SE, standard error.
Association of SNPs With the Risk of Developing TE
As shown in Table 1, none of the genotypes and haplotypes increased the risk of developing TE. Subgroup analyses of patients with ATE and VTE compared to controls revealed no differences in the genotype and haplotype distributions of both groups.
Discussion
The present study aimed to determine the associations of 3 known PROC promoter region SNPs with PC activity and the risk of developing TE in a Thai population. The study of the SNPs and risk of developing TE was performed in 216 children and adolescents who were diagnosed with either VTE or ATE and 102 healthy adult controls. In the patient group, VTE comprised DVT, pulmonary embolism, cerebral venous sinus thrombosis, and PVT, while ATE comprised arterial ischemic stroke and systemic arterial TE. However, to determine the association of the SNPs and PC activity, the study included PC activity of all controls and only patients whose age ≥6 years because the maturation of PC reaches an adult level at this age. 18
While previous studies demonstrated the association of the PROC promoter SNPs with the risk of developing TE and PC activity in patients in the Netherlands, 14 France, 15 and Brazil, 25 only 1 study in China was conducted in an Asian population. 16 Besides, most of the previous studies were conducted in adult patients with DVT. The strength of our study was the relatively large number of pediatric patients with both VTE and ATE, as PC deficiency can also cause ATE in pediatric populations. 7 Also, the ethnicity effects on the SNPs distribution in Thais had never been reported.
The haplotype and haplotype pair distributions in the control participants of this study were similar to those in the Chinese population study, but not to those in the Caucasian population studies. The comparison of haplotype pair frequencies in controls of the present study and the study in China 16 were CAA-TAA 31% versus 30.5%, TAA-TAA 26.5% versus 26.3%, CGT-TAA 21.6% versus 24.2%, CAA-CAA 8.8% versus 10.5%, CAA-CGT 8.8% versus 8.0%, and CGT-CGT 2.9% versus 0.5%. TAA was the most common haplotype among our study participants, followed by CAA and CGT, same as the Chinese study 16 (TAA 52.9% vs 53.7%, CAA 28.9% vs 29.7%, and CGT 18.1% vs 16.6%). The CGT haplotype (33.0%) was the most common in the Dutch study, 14 followed by TAA (30%) and CAA (25%). The CGA, CAT, TGT, TGA, TAT haplotypes were not identified among our 216 cases and 102 controls, whereas they were found in Dutch participants 14 at frequencies of 3%, 3%, 3%, 1%, and 1%, and in Brazilian Caucasians 25 at 10%, 7%, 11%, 4%, and 9%, respectively. The distributions of haplotype frequencies also varied when compared among 3 Brazilian ethnic groups (Brazilian Caucasians, Brazilian Blacks, and Amazonian Indians). 25 Taken together, it appears that ethnic background accounts for some of the differences in the SNPs in the promoter region of the PROC gene.
With regard to PC activity, patients with homozygous TAA and CGT haplotypes had significantly lower levels of PC activity compared with homozygous CAA patients, but not significantly lower than the heterozygous carriers or non-carriers of each haplotype. Homozygous CGT patients had the lowest PC activity in the present study, in accordance with the study in Dutch patients. 14 Also, in French study participants, the CG haplotype (−1654C/T and −1641A/G) had a significantly lower PC antigen level in both homozygous and heterozygous carriers compared with non-carriers. 15 However, in contrast to the Dutch study, 14 we did not find an association between homozygous CGT and the risk of developing TE in our study. Differences in the CGT haplotype frequencies between our controls (18.1%) and controls in the Dutch study (33.0%) might explain this discrepancy. 14
Protein C deficiency is the main cause of VTE. Although only 36.1% of our patients had VTE, the subgroup analysis of patients with VTE compared with controls also demonstrated no association of the SNPs with the risk of developing TE. Furthermore, because it has been known that PC deficiency can cause ATE in children, 17 we also analyzed the subgroup of ATE patients with healthy controls and did not find any association with TE.
There was no statistical differences in the haplotype and haplotype pair distributions of the PROC promoter SNPs between the patients and controls in this study. As all of our patient ages were less than 20 years, this study demonstrated that the SNPs were not associated with the risk of developing TE in children and adolescents. The possible explanation for the lack of association with the risk of developing TE was that we conducted the study in children and adolescents. Previous studies that demonstrated such associations were conducted in adult populations with a mean age of patients and a mean age at diagnosis that exceeded those of our patients. In the study in France, for example, the mean age of patients was 42.2 years, and the mean age at first VTE was 37.7 years. 15 Thus, to implicate our conclusion for the lifetime risk of developing TE or TE in adulthood, further longitudinal follow-up study, especially for those with PC level below the normal limit or in the borderline level range, is required. Also, in children with homozygous CGT and TAA, which have lower PC activity compared to those with homozygous CAA, the risk of developing TE may increase with the combination of other acquired precipitating factors causing low PC activity, such as infection and inflammation.
Although the present study excluded patients aged <6 years old for the PC activity study, the limitations of this study were PC level in our pediatric patients was lower than that in adult controls, which may be caused by age differences. Another limitation was the number of subjects in the individual haplotypes. The low number of patients in some haplotype categories could lower the power of the study.
In conclusion, 3 PROC promoter SNPs (−1654 C/T, −1641 A/G, and −1461A/T) were not demonstrated as a risk factor of TE in children and adolescents. There were associations of PC activity to those SNPs in the Thai population. Patients with homozygous TAA and CGT haplotypes had lower PC activity compared to homozygous CAA.
Footnotes
Author’ Note
N.U. performed and analyzed the study. J.E. was involved in statistical analysis. N.S. designed the study and reviewed the manuscript. P.K., A.C., and D.S. were involved in patient care. W.S. was involved in the DNA analysis and reviewed the manuscript. N.S. is a recipient of a Career Development Award from the Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand.
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Faculty of Medicine Ramathibodi Hospital.
