Abstract
The p.R147W mutation, the c.C6152T in exon 7, causing a change in amino acid from arginine to tryptophan of the PROC gene has been reported as a common mutation in Taiwanese populations with venous thromboembolism (VTE). The present study aimed to identify the prevalence of p.R147W in the Thai population and children with TE and the risk of developing TE. Patients aged ≤18 years diagnosed with TE were enrolled. The PROC gene was amplified by polymerase chain reaction using a specific primer in exon 7. The restriction fragment length polymorphism was designed using MwoI restriction enzyme. A total of 184 patients and 690 controls were enrolled. The most common diagnosis of TE was arterial ischemic stroke (AIS), at 100 (54.3%), followed by VTE, at 38 (20.6%), and cerebral venous sinus thrombosis (CVST), at 23 (12.5%). The prevalence of heterozygous and homozygous p.R147W in patients and controls was 9.5% versus 5.8% and 2.7% versus 0.1%, respectively. Heterozygous p.R147W had odds ratios (ORs) of 1.8 (95% confidence interval [CI]: 1.0-3.2, P = .04), 3.2 (95% CI: 1.2-8.2, P = .009), and 4.5 (95% CI: 1.6-12.8, P = .002) of developing overall TE, VTE, and CVST, respectively. Homozygous p.R147W had ORs of 20.2 (95% CI: 2.3-173.7, P < .001), 21.4 (95% CI: 2.2-207.9, P < .001), and 43.3 (95% CI: 3.8-490.6, P < .001) of developing overall TE, AIS, and CVST, respectively. This study suggested that p.R147W is a common mutation and increased risk of TE in Thai children.
Introduction
The thrombophilic risk factors reported as common in Thai and other Asian populations are anticoagulant deficiencies of protein C (PC) or protein S (PS), in contrast to factor V Leiden in Caucasians. 1 –4 Protein C, enhanced by its cofactor PS, functions as an inhibitor of activated factor V and factor VIII. Protein C deficiency is a risk factor in patients with thromboembolism (TE), mainly in venous TE (VTE) 5 such as deep vein thrombosis (DVT), pulmonary embolism, cerebral venous sinus thrombosis (CVST), and in infants with purpura fulminans. 6 Arterial TE (ATE) associated with PC deficiency is less common.
Protein C deficiency is inherited as an autosomal dominant trait. It is classified into 2 types: type 1—characterized by decreased levels of PC activity and antigen and type 2—characterized by decreased PC activity but normal PC antigen. The prevalence of PC deficiency is around 0.3% to 0.5% 7 in the Western population and 0.6% to 1.0% in patients with VTE. The prevalence of PC deficiency in the healthy Thai population and patients with VTE were 1.99% and 8.9%, respectively, which is higher than in Caucasian populations. 4,8,9 The homozygous or double heterozygous PC mutations cause severe PC deficiency and present with purpura fulminans and severe TE at a very young age. 10 Protein C deficiency is caused by mutations of the PC (PROC) gene, which is located in the chromosome 2q13-q14. The PROC gene consists of 9 exons. To date, over 300 mutations have been reported. 11 Studies of PC mutations found significant differences regarding the ethnic background. For example, the p.R230C, p.Q132stop, p.R306stop, and c.3363insC mutations were commonly found in Caucasian populations, 12,13 whereas p.R147W, the c.C6152T in exon 7, causing a change in amino acid from arginine to tryptophan, was a common mutation among Taiwanese Chinese. 14 In 6 Thai children with TE and had low levels of PC, p.R147W was identified in 3 patients. 10 P.R147W mutation results in 3 times lower affinity for binding to endothelial PC receptor. 15 The present study aimed to identify the prevalence of p.R147W in the Thai population and children with TE and to identify the risk of this mutation leading to the development of TE.
Methods
Inclusion Criteria
Patients aged ≤18 years diagnosed with TE by Doppler ultrasound or imaging studies were enrolled in this study after providing informed consent. Patients were diagnosed and treated at the Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand, or referred here for thrombophilic investigation. The controls were healthy participants of blood donors or healthy children without the history of TE.
Exclusion Criteria
Patients who were documented of PC mutations other than p.R147W included 3 patients with severe PC deficiency, 2 of whom had homozygous c.T7172G (p.C196G), whereas the other had double heterozygous c.T7172G and c.8871_8872TA (p.C196G and p.G385fs) causing severe PC deficiency, were excluded.
Blood and Data Collection
All patients and controls provided written informed consent, after which blood was collected in 2 syringes: an ethylenediamine tetra acetic acid (EDTA) tube and a sodium citrate tube with the ratio of blood: sodium citrate of 9:1. Then, blood in the EDTA tube and sodium citrate tube was centrifuged at 3000 rpm for 10 minutes to collect plasma and buffy coat. Plasma was kept at −80°C before the measurement of PC activity by automated functional clotting PC assay (Instrument laboratory [ACL 8000], Milan, Italy). The quality control of normal and abnormal PC levels was performed simultaneously during PC assay of the enrolled participants. In patients aged less than 5 years with low PC activities, PC activity were repeated until patients were more than 5 years old. 16 DNA was extracted from buffy coat using conventional phenol–chloroform method. Basic information including age, gender, and thromboembolic presentation was collected. The underlying diseases or acquired risk factors including the conditions reported to be a risk of TE, such as cyanotic congenital heart diseases, polyarteritis nodosa, and cerebral palsy with limited activity were also recorded. The study was approved by the Ramathibodi Hospital Review Board (ID 11-57-69).
Mutation Analysis
The polymerase chain reaction (PCR) amplification of exon 7 was performed using specific primers, as described in a previous study. 17 The restriction fragment length polymorphism (RFLP) was designed using MwoI restriction enzyme (New England Biolabs, England). After digestion, the product was identified in 10% polyacrylamide gel. Patient with wild type showed a band of 139 base pairs, heterozygous p.R147W showed 2 bands of 194 and 139 base pairs, whereas homozygous p.R147W showed a band of 194 base pairs. In order to confirm the RFLP results, 10 of the PCR products were also randomly submitted for DNA sequencing.
Statistical Analysis
Age and PC activities were expressed as mean (standard deviation [SD]). The Mann-Whitney U test was used to determine the differences in PC activities between the 2 groups. The χ2 test was used to determine the difference of p.R147W between the 2 groups. The risk of p.R147W to the occurrence of TE was expressed as an odds ratio with 95% confidence intervals (CI). Kaplan-Meier analysis was performed to determine the age onset of developing TE in p.R147W mutation. The receiver operating curve (ROC) analysis was performed to determine the use of PC activity for the diagnosis of PC deficiency. The sensitivity and specificity of PC activity were also calculated. A P value <.05 was considered statistically significant.
Results
A total of 184 patients and 690 controls, with mean (SD) ages of 8.8 (5.2 years; range 0-18.0) and 26.9 (11.2 years; range 8.0-56.0), respectively, were enrolled in the study. The number of participants according to gender (female:male) were 80:104 and 276:414 in patients and controls, respectively, P = .08. Diagnoses of TE consisted of arterial ischemic stroke (AIS) 100 (54.3%), VTE 38 (20.6%), systemic ATE 20 (10.9%), CVST 23 (12.5%), and multiple sites TE 3 (1.7%). Underlying diseases, identified in 52.2% of patients, were vasculitis 28 (15.3%), followed by malignancies 14 (7.4%), thalassemia diseases 12 (6.3%), heart diseases 11 (5.8%), hematologic diseases other than thalassemia disease 8 (4.2%), neurologic and genetic diseases 6 (3.2%) each, nephrologic and infectious diseases 4 (2.1%) each, and central venous catheter 3 (1.6%).
The incidence of heterozygous and homozygous p.R147W in patients and controls was 9.8% versus 5.8% and 2.7% versus 0.1%, respectively. Heterozygous p.R147W had odds ratios of 1.8 (95% CI: 1.0-3.2, P = .04), 3.2 (95% CI: 1.2-8.2, P = .009), and 4.5 (95% CI: 1.6-12.8, P = .002) of developing overall TE, VTE, and CVST, respectively (Table 1). In addition, heterozygous p.R147W in patients without underlying diseases had OR of 2.2 (95% CI: 1.1-4.6, P = .032) of developing TE (Table 1). In homozygous p.R147W, the ORs of developing overall TE, AIS, and VTE were 20.2 (95% CI: 2.3-173.7, P < .001), 21.4 (95% CI: 2.2-207.9, P < .001), and 43.3 (95% CI: 3.8-490.6, P < .001), respectively. The homozygous p.R147W also increased risk of TE in patients with or without underlying diseases (Table 1). The Kaplan-Meier analysis showed the median times to develop TE in wild type, heterozygote, and homozygote of p.R147W of 17.0, 14.0, and 5.1 years, respectively, P = 0.001 (Figure 1).
Odds Ratios for Developing TE and Allele Frequency of c.C6152T (p.R147W) of PROC Gene.
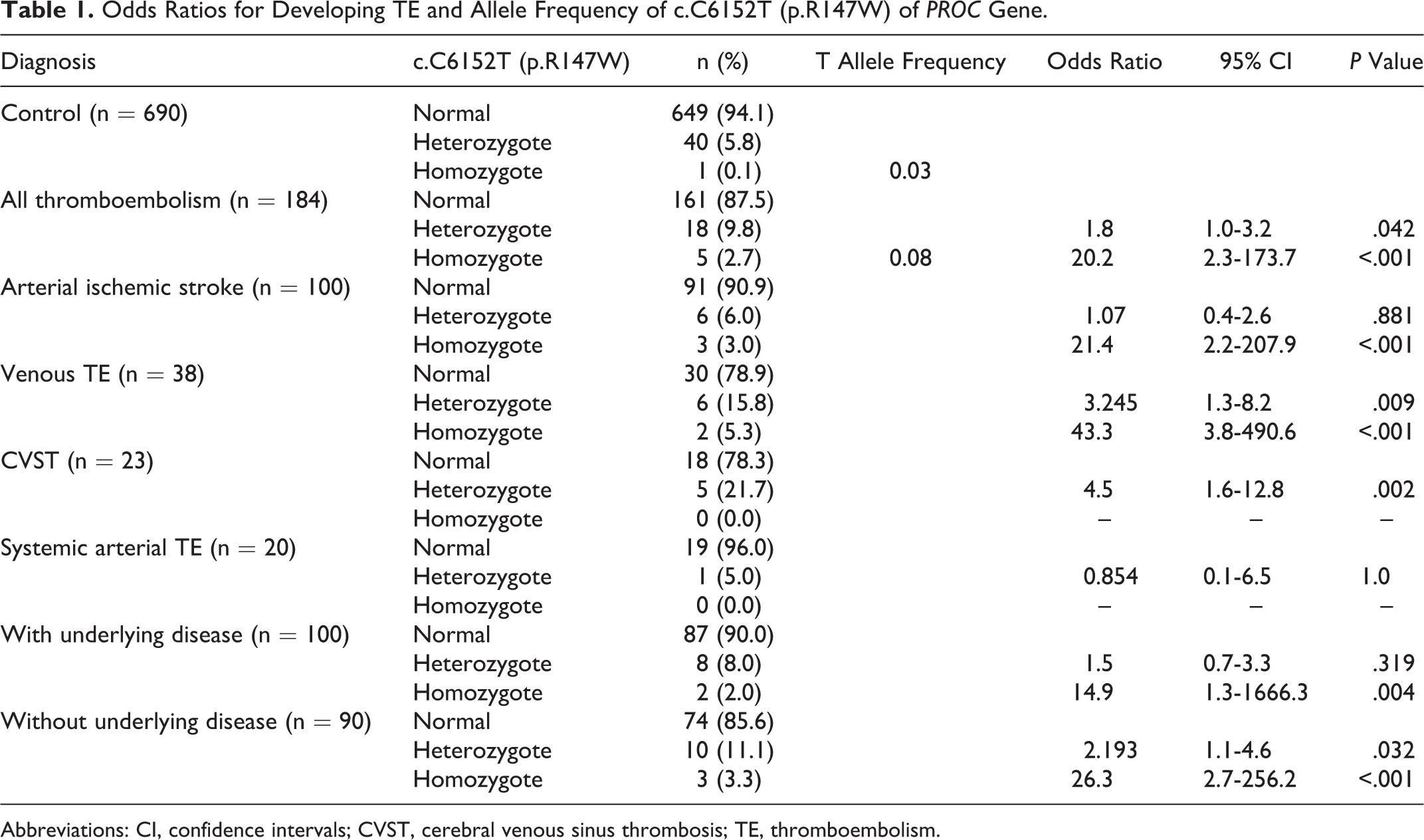
Abbreviations: CI, confidence intervals; CVST, cerebral venous sinus thrombosis; TE, thromboembolism.
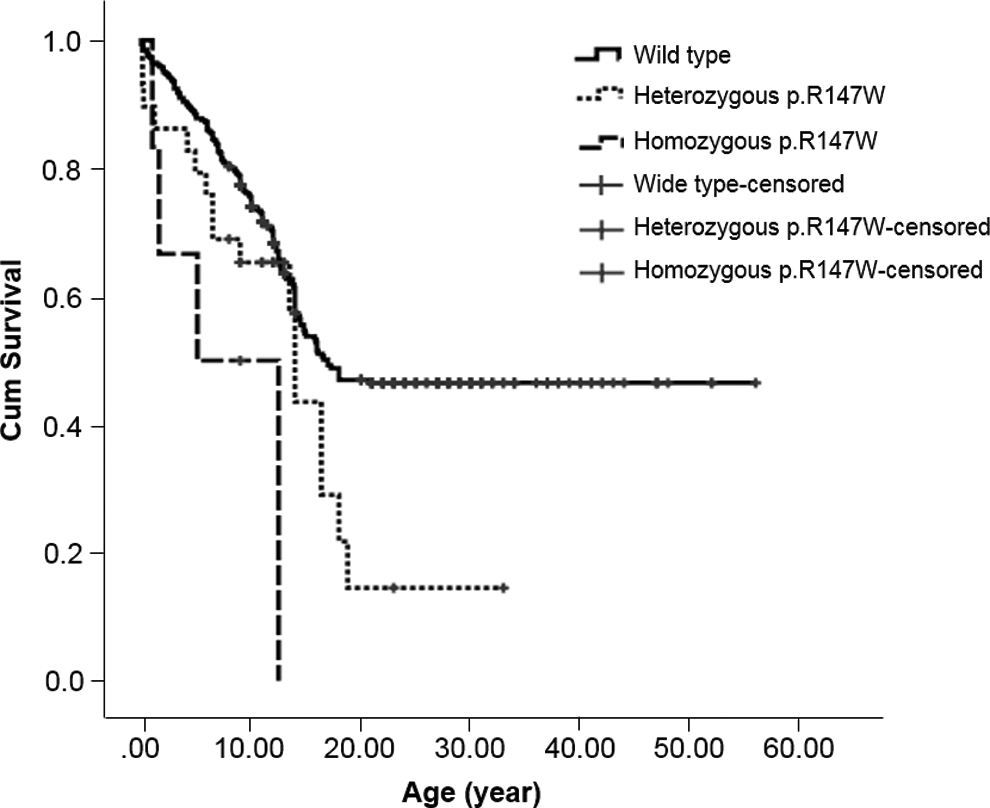
Kaplan-Meier analysis of age at diagnosis of thromboembolism in patients with wild type, heterozygote, and homozygote of p.R147W.
Protein C activities in 291 wild type, 22 heterozygote, and 5 homozygote of p.R147W had mean (SD; range) levels of 120.4% (33.5%; 63%-319%), 80.0% (30.5%; 37%-168%), and 44.3% (20.0%; 14.4%-73.1%), respectively, P < .001. The ROC was performed to determine the use of PC activity for the diagnosis of PC deficiency according to the p.R147W mutation. The area under the curve was 0.854, P < .001 and demonstrated that PC activity <94% had sensitivity and specificity of 75.0% and 80.1%, respectively (Figure 2).
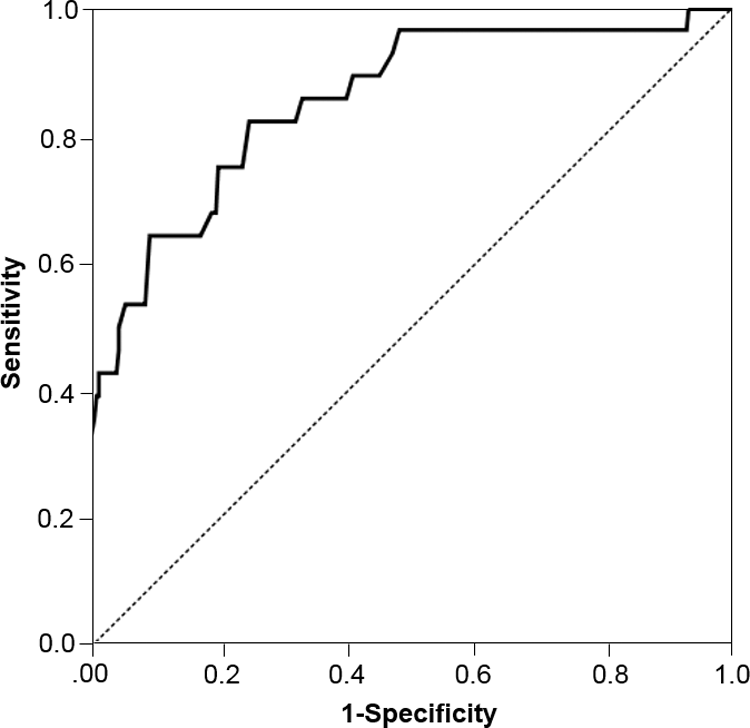
Receiver operating curve of plasma protein C activity for the diagnosis protein C deficiency according to p.R147W mutation.
Discussion
Protein C deficiency is a common inherited thrombophilia in Asian countries, including Thailand. 1,3 Not only VTE, PC deficiency has been reported in children with ATE. 10 The p.R147W mutation in the PROC gene, a type 2 PC deficiency, is found to be common among Asian populations. 14,15
The present study demonstrated p.R147W as a common mutation in the Thai population. The prevalence rates of heterozygous and homozygous p.R147W, among 690 volunteers, were 5.8% and 0.1%, respectively. When compared to the previous study in Taiwanese, Chinese, the prevalence of heterozygote in this report was higher (5.8% vs 0.85%). 14 The calculated T allele frequency in the present study was 0.03 compared to that of Taiwanese population, 0.0043. 14 The higher incidence rate and allele frequency in the present study may be due to the different population or the theory of natural selection 18 . The difference in age-group in the present study did not affect the incidence of p.R147W, as it is equal when dividing the normal population into 2 groups of <20 and ≥20 years old. In patients with TE had higher prevalence rates of heterozygote, 9.8%, and homozygote, 2.7%, when compared to 5.8% and 0.1%, respectively, in the control participants. The heterozygous p.R147W had the ORs of developing VTE in children including CVST and VTE of 3.3 and 4.5, respectively. The increased OR to develop VTE also demonstrated in the previous report in adult populations of 5.1. 14 The homozygous p.R147W had much increased ORs of both AIS and VTE of 21.4 and 43.3, respectively. The Kaplan-Meier analysis demonstrated the median occurrence of TE to be the lowest in the homozygote (5.1 years old), followed by heterozygote (14 years old) and wild type (17 years old) of p.R147W, which supported the risk of this mutation for the development of TE in the pediatric age-group. The presentation ages of TE in patients with homozygous p.R147W in the present study were at the age of 2 years in 2 patients, 5 years in 1 patient, and 12.5 years in 2 patients. Three of them presented with AIS and the other 2 presented with DVT, while the report of homozygous or compound heterozygous of other PC mutations presented with severe or multiple sites TE such as purpura fulminans with or without AIS and systemic arterial TE since the infancy period, and most of the patients had the PC activities less than 5%. 10 The homozygous p.R147W in this report had PC activities of more than 10%, and this level can explain the TE symptoms at the older ages. However, the compound heterozygote of p.R147W with other mutations in the PROC gene may result in lower PC activities, meaning patients could present with severe TE.
As underlying diseases play an important role in the development of TE in children, the present study demonstrated that, in patients with significant underlying medical conditions which could explain TE development, heterozygous p.R147W was not a risk factor; however, homozygous p.R147W increased ORs in patients with and without underlying diseases.
Protein C activities were tested in participants with wild type, and heterozygous and homozygous mutations had the mean (SD; range) of 120.4% (33.5%; 63%-319%), 80.0% (30.5%; 37%-168%), and 44.3% (20.0%; 14%-73.1%), respectively. The reported levels of PC activities in wild type and heterozygous p.R147W mutation from the previous study were 104.8% (21.5%) and 59.8% (9.2%), respectively. 14 The levels from the previous report were lower than the present study which may be due to the different population and method of PC activity measurement. The ROC analysis in the present study demonstrated the moderate sensitivity and specificity of PC activities for the diagnosis of PC deficiency according to the genetic mutation. The results from the present study suggested that, in some patients with heterozygous mutation, the level of PC activity may overlap to the level of wild type, although there was significantly different of PC activities among wild type, heterozygote, and homozygote. Therefore, genetic identification is necessary to confirm the diagnosis. Despite, p.R147W increased the risk of TE in Thai children, no patients in the present study had recurrent TE events. The long-term follow-up is required to determine the recurrent risk.
Conclusion
All Thai pediatric patients with TE, the p.R147W of PROC gene should be included in the thrombophilic screening, since the heterozygote increases the risk of overall TE, VTE, and TE without underlying diseases. Furthermore, patients with homozygous mutation develop TE at the age younger than the patients with heterozygous mutation and had higher risk of TE. Although, no recurrent TE was observed in the present study, the prophylaxis in a high-risk situation may be required especially in homozygous p.R147W mutation.
Footnotes
Acknowledgments
The authors are grateful to physicians and paramedic personnel who were involved in taking care of the patients. Nongnuch Sirachainan is a recipient of Career Development Award from the Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Thailand Research Fund-Senior Research Scholar 2006 (A.C.) for their support.
