Abstract
Pulmonary embolism (PE) treatment depends on disease severity and risk of complications. Physician and institutional expertise may influence the use of reperfusion therapy (RT) such as systemic thrombolysis (SL) and catheter-directed interventions (CDI). We aimed to investigate the effects of a consensus-based treatment algorithm (TA) and subsequent implementation of PE response team (PERT) on RT modality choices and patient outcomes. A cohort of PE patients admitted to a tertiary care hospital between 2012 and 2017 was retrospectively evaluated. Demographics, clinical variables, RT selections, and patient outcomes during 3 consecutive 2-year periods (baseline, with TA, and with TA+PERT) were compared. Descriptive statistics were used for data analysis. A total of 1105 PE patients were admitted, and 112 received RT. Use of RT increased from 4.7% at baseline to 8.2% and 16.1% during the TA and TA+PERT periods. The primary RT modality transitioned from CDI to SL, and reduced-dose SL became most common. Treatment selection patterns remained unchanged after PERT introduction. Hospital length of stay decreased from 4.78 to 2.96 and 2.81 days (P < .001). Most of the hemorrhagic complications were minor, and their rates were similar across all 3 periods and between SL and CDI. No major hemorrhages occurred in patients treated with reduced-dose SL. In conclusion, TA and PERT represent components of a decision support system facilitating treatment modality selection, contributing to improved outcomes, and limiting complications. Treatment algorithm emerged as a factor providing consistency to PERT recommendations.
Introduction
Despite progress in research and care, management of patients with submassive and massive pulmonary embolism (PE) varies and depends on the physician’s and institution’s expertise. 1 Systemic thrombolysis (SL) has been shown to improve outcomes in hemodynamically unstable (massive, or high-risk) PE 2,3 and is generally accepted as the first-line intervention in this patient category. Submassive (intermediate risk) PE patients are heterogeneous in their clinical course and risk of treatment-related complications. 4 –6 Guidelines do not always encompass the complexity of clinical scenarios, and supporting data are not always robust. 7 Reperfusion therapy (RT) is recommended for patients who experience hemodynamic deterioration while treated with anticoagulation alone and selected normotensive patients at high risk for decompensation. 8 –10 A full-dose SL administered simultaneously with therapeutic anticoagulation significantly reduced the risk of hemodynamic decompensation but was associated with higher frequency of hemorrhagic complications, including intracranial hemorrhage (ICH). 6 Studies on reduced-dose SL suggest decreased bleeding risk and similar clinical outcomes but the data remain limited. 11 –14 Catheter-directed interventions (CDI) have been advocated as techniques associated with reduced bleeding risk but they require special expertise and have not been directly compared to SL. 15 –17 The concept of multidisciplinary PE response team (PERT) has emerged to address these challenges, 1,18 –21 and management recommendations that take into account experience and practice patterns of multiple treatment centers have been recently published. 22 The PERT implementation has been associated with increased use of RT and improved efficiency of care and outcomes, 23,24 but factors affecting treatment modality selections and their relation to patient outcomes have not been extensively studied. The purpose of this study was to examine the evolution of RT choices and patient outcomes during 3 consecutive 2-year periods: baseline, with the addition of a consensus-based treatment algorithm (TA), and then following PERT implementation (TA+PERT period) at a 631-bed tertiary care teaching hospital.
Methods
Study Design
This was a retrospective cohort study. The study was approved by Allina Health’s Institutional Review Board with a waiver of individual informed consent. The data were extracted from electronic health records (EHR). Patients were identified using International Classification of Diseases, Ninth Revision and Tenth Revision codes associated with the primary PE diagnosis. Two investigators performed manual chart abstraction for variables not readily available in the EHR extract.
Definitions
Massive PE: Hypotension (systolic blood pressure < 90 or a decrease by ≥ 40 mmHg from baseline for a period > 15 minutes), shock, or cardiac arrest. Submassive PE: Right ventricular (RV) dysfunction/strain on echocardiogram or computed tomography, and/or elevated biomarkers (troponin or brain natriuretic peptide) without hypotension.
Major bleeding 25 : Fatal bleeding and/or symptomatic bleeding in a critical area or organ, such as intracranial, intraspinal, intraocular, retroperitoneal, intra-articular or pericardial, or intramuscular with compartment syndrome, and/or bleeding causing a fall in hemoglobin level of 2 g/dL or more, or leading to transfusion of 2 or more units of red blood cells, all within 7 days from the time of intervention.
Minor bleeding: Any bleeding episode within 7 days from the time of intervention that did not qualify for the major bleeding definition.
Escalation of treatment: Need for new or increased dose vasopressor, additional reperfusion procedure (CDI, SL, surgical embolectomy), or extracorporeal membrane oxygenation (ECMO) following SL or CDI.
Protocols
Baseline time period (2012-2013): Patient management was determined by the attending physician with subspecialty consultation as necessary.
Treatment algorithm period (2014-2015): A multidisciplinary team from cardiology, critical care, interventional radiology, hospital medicine, pulmonology, emergency medicine, vascular medicine, and clinical pharmacy developed and introduced the TA that was based on the existing guidelines, current publications, and expertise and resources of our treatment center. Treatment algorithm highlights (full TA is available as Supplemental Appendix 1): (1) Systemic anticoagulation in all patients without absolute contraindication; (2) RT in massive and consideration for RT in selected cases of high-risk submassive PE with moderate to severe RV dysfunction, positive biomarkers, worsening hemodynamic status, and simplified pulmonary embolism severity index (SPESI) 26 > 0; (3) SL (with consideration for reduced-dose SL in patients without hypotension) as the primary RT; (4) CDI for RT-eligible patients with increased bleeding risk but without absolute contraindication; (5) SL regimens: (a) full dose: 100 mg tissue plasminogen activator (TPA) intravenous infusion over 2 hours, 27 (b) reduced dose: 10 mg bolus + 40 mg TPA infusion over 2 hours, or 0.5 mg/kg TPA infusion over 2 hours for patients < 50 kg 11 ; (6) systemic anticoagulation: Intravenous heparin infusion stopped during SL administration and resumed when post-TPA partial thromboplastin time was less than 2 times of the upper limit of normal; (7) immediate availability of the SL safety checklist (contraindications, dosing recommendations), and a reference to the dedicated order set.
Treatment algorithm +PERT period (2016-2017): The attending provider was able to activate PERT via a dedicated hospital-wide phone number or direct contact of its members. The conference call involved an intensivist, cardiologist, and interventional radiologist who had access to the TA and patient’s clinical data and imaging. Initial management recommendations were provided at the conclusion of the call. Updates were provided via a follow-up conference call or by direct provider communication as necessary.
Catheter-directed intervention: The interventional radiologist, in consultation with the treatment team selected treatment modalities based on clinical situation, imaging, and risk of complications. They included TPA bolus + pigtail catheter fragmentation, low-dose local TPA infusion for 12 to 24 hours at 0.5 to 2 mg/h via unilateral or bilateral multisidehole infusion catheters, either EKOS (BTG) or Cragg McNamara (Medtronic), and/or mechanical thrombectomy using the Indigo CAT 8 (Penumbra) system.
The TA remained unchanged during the entire study time frame, with the exception of the added PERT consult recommendation during the TA+PERT period. The PERT leadership that included representatives from all specialties involved in the TA development held bimonthly meetings to monitor practice patterns and complications but had no influence on clinical decisions made by the treatment teams. The patient’s attending physician remained the ultimate decision maker on the treatment modality selection over the entire study period.
Statistical Analysis
Analysis of variance and Mann-Whitney tests were used to compare continuous variables, and χ2 and Fisher exact tests were used to compare categorical variables as appropriate. Analyses were performed globally across the 3 time periods where relevant. The proportion of RT across the 3 time periods were further examined with post hoc tests for pairwise comparisons with Bonferroni corrections. Values of P <.05 were considered significant. Statistical analyses were conducted using R, version 3.5.1 (the R project for statistical computing).
Results
Study Demographics and RT Choices
Between 2012 and 2017, 1105 PE patients were admitted and one-third received intensive care unit (ICU) care (Table 1). There were no clinically significant differences in patient demographics, comorbidities (except for a higher prevalence of hypertension during baseline period), or proportion of SPESI > 0. While the overall hemodynamic parameters and prevalence of abnormal biomarkers were similar, there was an increase in the total ICU admissions and a nonsignificant but notable increase in the number of massive PE with cardiac arrest cases in patients treated with RT during the TA+PERT period. The frequency of RT increased from 4.7% at baseline to 8.2% after the introduction of the TA (adjusted P = .27) and to 16.1% during the TA+PERT period (adjusted P = .004, and P < .001 for all). Use of SL as the primary reperfusion mode increased from 40% at baseline to 83.9% during the TA period (adjusted P = .015) and 81.8% in the TA+PERT period, with the respective decrease in CDI utilization (P = .003 for all). The frequency of reduced-dose SL administration increased from 13.3% in the initial period to 64.5% and 54.5% of all RT cases in the 2 subsequent periods (P = .003 for all).
Characteristics of Patients With Primary PE Diagnosis Admitted Between 2012 and 2017.
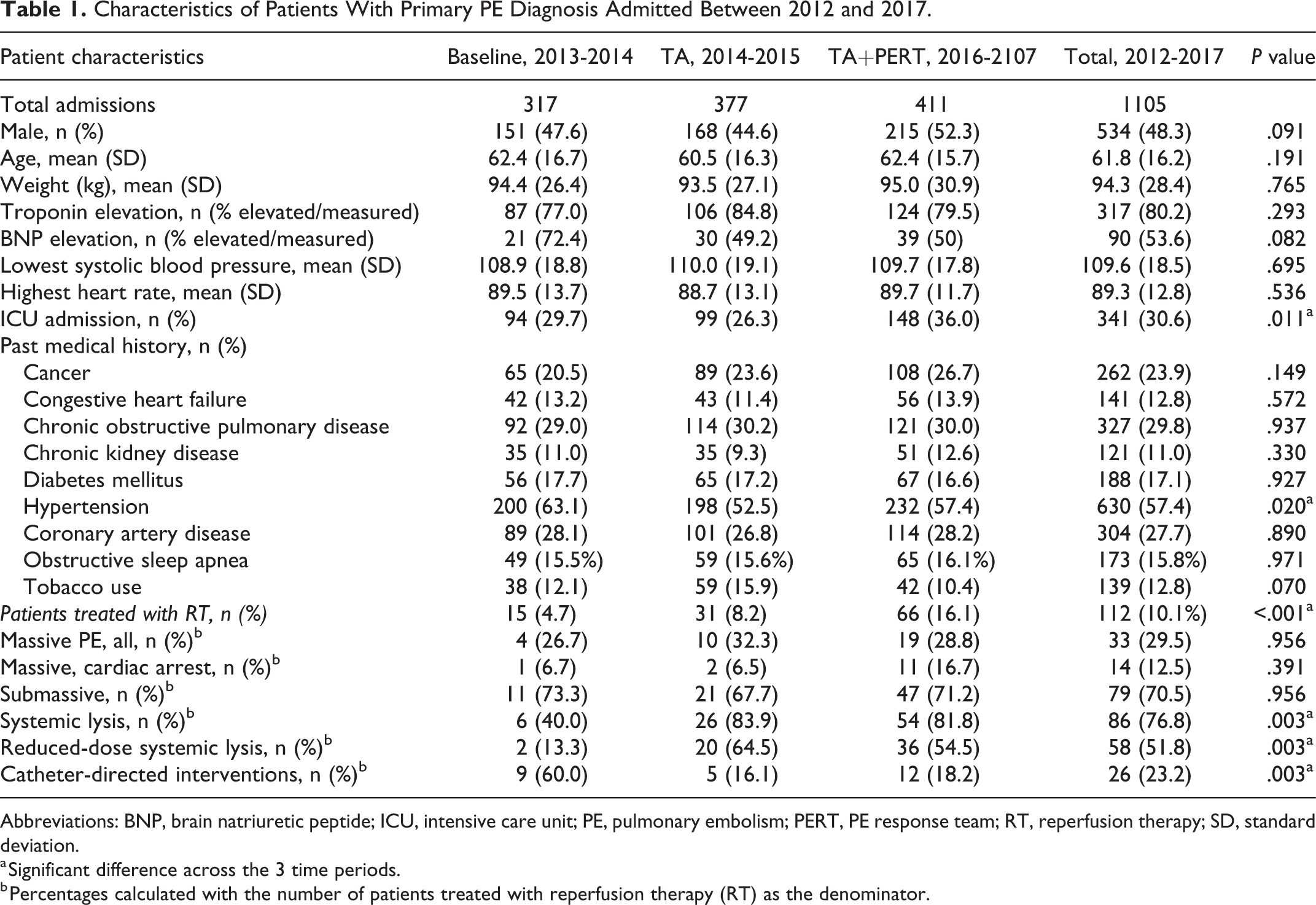
Abbreviations: BNP, brain natriuretic peptide; ICU, intensive care unit; PE, pulmonary embolism; PERT, PE response team; RT, reperfusion therapy; SD, standard deviation.
a Significant difference across the 3 time periods.
b Percentages calculated with the number of patients treated with reperfusion therapy (RT) as the denominator.
Upon implementation of the TA and PERT, CDI methods were transitioned from pigtail catheter fragmentation combined with TPA bolus (88.9% at baseline vs 16.7% in 2016-2017 period) to low-dose local TPA infusion via a multisidehole infusion catheter (0% at baseline vs 75% in 2016-2017; P < .01). The median total TPA dose was also significantly lower (45 mg vs 18 mg, P < .01) to minimize the risk of hemorrhagic complications.
Pulmonary Embolism Response Team
The PERT consultation was obtained on 87 (21.2%) PE patients during the TA+PERT period (Supplemental Appendix 2). The PERT patients had more frequent troponin and brain natriuretic peptide elevation, lower blood pressure, higher heart rate, and required ICU admission more frequently as compared to those not referred to PERT during the same time period (P < .05). Reperfusion therapy was used in 47 (54%) of PERT patients. Of them, SL was recommended in 80.9% and CDI in 19.1%. The reduced-dose regimen was used in 30 (78.9%) of 38 of the SL cases. The majority of PERT patients treated with RT had submassive PE with high-risk features, while 14.9% had massive PE. Hospitalist and emergency department physicians were the most common referring services (44% and 30%, respectively), and 17% of referrals originated at outside hospitals.
Overall Outcomes
Overall, there was a significant reduction in the hospital length of stay (LOS), from the baseline (median 4.78 days) to both subsequent periods (2.96 and 2.81, respectively, adjusted P < .001 for pairwise comparison between baseline and each of the subsequent periods), P < .001 for all (Table 2). A trend in reduction of ICU and hospital LOS from the TA to TA+PERT periods did not reach statistical significance. There was no significant difference in all-cause inpatient, 30-day, or 1-year mortality, or 30-day readmissions.
Overall Outcomes of Pulmonary Embolism Patients.

Abbreviations: ICU, intensive care unit; LOS, length of stay.
a Indicates significant difference across the 3 time periods.
Reperfusion Therapy Patient’s Outcomes and Complications
A total of 112 patients (29.5% massive, 70.5% submassive PE) received RT (Table 3). Systemic thrombolysis was administered in 86 (76.8%) cases, of which 58 received reduced-dose TPA; 26 (23.2%) received CDI. Patients treated with full-dose SL had higher frequency of massive PE and cardiac arrest, and a higher pulmonary embolism severity index 28 score (P < .05) than those treated with reduced-dose SL. In 17 (65.4%) CDI cases, patients had either relative or absolute contraindication to SL (Supplemental Appendix 3), while a relative contraindication was present in 10 (11.6%) of SL patients (P < .0001). The overall difference in comorbid conditions did not reach statistical significance. The majority of patients who received reduced-dose SL (79.3%) or CDI (80.8%) had submassive PE. Patients treated with SL had significantly shorter ICU and hospital LOS (P < .05) as compared to CDI. As expected, mechanical thrombectomy procedures requiring subsequent escalation of care appeared to be associated with the longer ICU and hospital LOS as compared to other CDIs but a small number of cases precludes definitive conclusions (Supplemental Appendix 3). In SL patients, 30-day all-cause mortality was 14% (n = 12). All but 1 death was in the massive PE group, and in 10 of 12 of the deaths, SL was administered in the setting of cardiac arrest due to suspected or confirmed PE. In the CDI patients, 30-day and 1-year mortality were 0% and 7.7%, respectively. Escalation of care did not significantly differ between patients receiving SL and CDI (P = .137) and included increase of SL intensity from reduced- to full-dose in a patient with submassive PE and severe RV dysfunction; 2 cases of full-dose SL administration in the setting of cardiac arrest followed by venoarterial ECMO as well as surgical embolectomy in 1 of them; and 3 CDI cases (vasopressor support after local thrombolysis, repeat intervention for local thrombolysis after CDI thrombectomy, and surgical embolectomy).
Characteristics and Outcomes of Patients Treated With Reperfusion Therapy.

Abbreviations: CDI, catheter-directed interventions; IQR, interquartile range; LOS, length of stay; NA, not applicable; PE, pulmonary embolism; PERT, PE response team; PESI, pulmonary embolism severity index; SL, systemic thrombolysis; SPESI, simplified pulmonary embolism severity index; TPA, tissue plasminogen activator.
a P values calculated for the difference between SL, total, and CDI.
b P value <.05 for the difference between TPA, reduced-dose and TPA, full-dose.
In total, 12 hemorrhagic complications occurred. The frequency of complications by treatment type (Table 4) was similar in the reduced-dose SL (10.3%, all minor), full-dose SL (10.7%, 1 minor and 2 major), and CDI groups (11.5%, 2 minor and 1 ICH). In the entire SL group, major bleeding occurred in 2.3% (2 of 86) and minor bleeding in 8.1% (7 of 86 patients). In patients with relative contraindications to SL, bleeding complications occurred in 3.5% of SL and 7.7% CDI patients. Minor complications included vascular access and recent surgical site bleeds, hematuria, and epistaxis, all either self-limited or requiring local control only. Major complications included retroperitoneal hematoma in a fully anticoagulated postcoronary angiogram patient and oropharyngeal bleeding during intubation in a patient receiving systemic TPA. The only ICH was observed in a patient with a history of syncope and falls who was treated with CDI involving bolus-dose TPA combined with pigtail-catheter thrombus fragmentation. The majority of complications occurred in the setting of recent invasive procedures or in emergency situations. There was no significant difference in the overall rate of hemorrhagic complications between the baseline, TA, or TA+PERT periods.
Hemorrhagic Complications by Treatment Type.
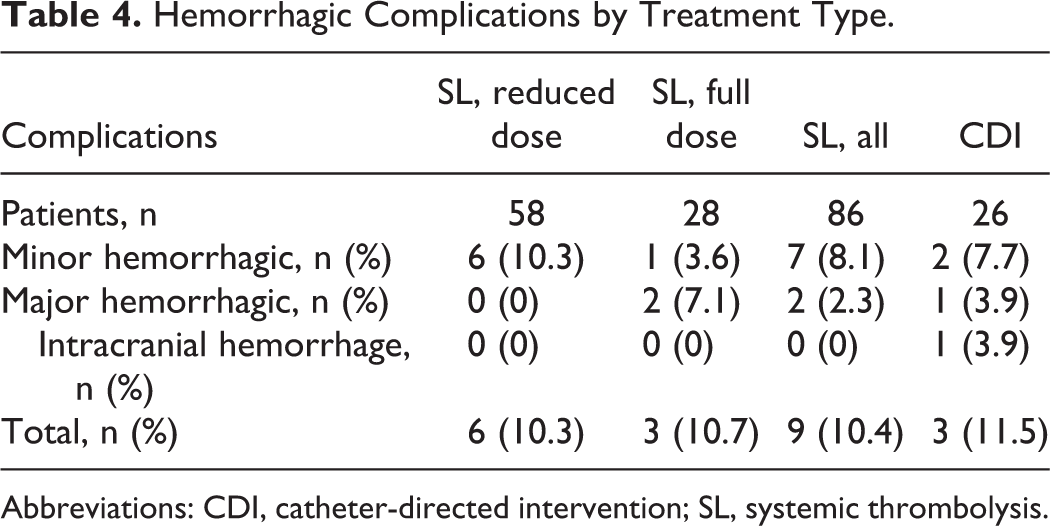
Abbreviations: CDI, catheter-directed intervention; SL, systemic thrombolysis.
Discussion
There is growing evidence documenting feasibility and benefits of a coordinated, multidisciplinary approach to the management of hospitalized PE patients. 18,19,23,29 Our study adds to the body of literature by analyzing the impact of a consensus-based TA and subsequent implementation of PERT as factors influencing treatment modality selection and patient outcomes and complications.
Introduction of the TA that took into account disease severity and risk of treatment complications was associated with an increase in RT utilization, transition of the primary RT mode from CDI to SL, more frequent use of reduced-dose TPA, and adjustments in CDI techniques. Patients treated with full-dose SL had higher disease severity including massive PE and cardiac arrest than those treated with reduced-dose SL regimen or CDI, while the majority of CDI patients had relative or absolute contraindications to SL. Trends toward using SL as the default RT modality while reserving CDI for patients with higher bleeding risk suggest overall compliance with TA and PERT recommendations. Consistent with prior experience, 19,24,30 PERT implementation was associated with further increases in RT use. The approach to the treatment modality selection remained unchanged in the TA+PERT period. The TA appeared to be the main factor influencing treatment decisions during both intervention periods, while the availability of PERT consult supplemented it by a multidisciplinary case review facilitating rapid decision-making. A significant portion of PERT consultation requests came from the emergency physicians and providers at outside facilities transferring patients to our center. The PERT was able to facilitate quick implementation of the treatment plan prior to the patient arrival at the hospital floor.
The PE program development was associated with hospital LOS reduction despite similar severity of illness across all 3 time periods. This finding may be indicative of the improved care efficiency in this patient population, also noted by other authors. 23,24 Intensive care unit and hospital LOS were longer in CDI patients as compared to those treated with SL, possibly due to the differences between the groups.
Consistent with other reports on PERT outcomes, 30 there was no significant change in mortality over the 3 time periods. In the RT group, it was almost exclusively limited to the patients presenting with massive PE and cardiac arrest. Mortality, 30-day readmission rates and hospital LOS in our younger patient population were lower than those reported in a retrospective analysis of Medicare beneficiaries 31 but comparable to other reports. 6
The overall complication rates were similar across all 3 time periods, as well as in all RT subgroups but the event rate was low. Two major bleeding episodes occurred in the full-dose SL group whereas no major hemorrhages were observed in the reduced-dose TPA patients; no ICH occurred in SL patients. The majority of hemorrhagic complications were minor and occurred in the setting of recent invasive procedures. Our SL complication rates were lower than those reported in recent publications. 6,19 An earlier randomized controlled study of SL in submassive PE also reported lower complication rates, with the incidence of major hemorrhage similar to anticoagulation alone. 32 The potential complication-reducing factors in our patients include the immediate availability of a SL safety checklist, a dedicated order set guiding SL dose regimens, cautious and standardized approach to systemic anticoagulation, and transition to CDI techniques with lower cumulative TPA dose. The CDI complication rates were consistent with previous publications. 33,34
Treatment selections in our patient cohort differed from other centers where the increase in RT was driven by either both SL and CDI23 or by CDI only. 30 In this observational study, direct comparisons of the treatments were not possible. However, the overall trends in outcomes and complications suggest feasibility of different center-specific algorithms and the need for the individualized RT modality selection as no intervention is risk-free.
The generalizability of the findings in this study is limited by the retrospective, observational single-center design with potential confounders affecting the outcomes, lack of RT to anticoagulation alone comparison, and a relatively small sample size.
Conclusion
Development of a structured approach to PE management was associated with increased use of SL, a reduction in hospital LOS, a trend toward the reduction in ICU LOS from TA to TA+PERT periods, but no change in mortality. The majority of hemorrhagic complications were minor and occurred in the setting of recent invasive procedures. No major hemorrhages occurred in patients treated with the reduced-dose SL regimen. The TA and PERT represent components of a decision support system facilitating treatment modality selection, contributing to improved outcomes, and limiting complications. Additional studies with larger number of patients are needed to directly compare outcomes of various approaches to the RT modality selection.
Supplemental Material
Supplemental Material, PE_manuscript_Supplemental_Appendix_2 - Effects of a Consensus-Based Pulmonary Embolism Treatment Algorithm and Response Team on Treatment Modality Choices, Outcomes, and Complications
Supplemental Material, PE_manuscript_Supplemental_Appendix_2 for Effects of a Consensus-Based Pulmonary Embolism Treatment Algorithm and Response Team on Treatment Modality Choices, Outcomes, and Complications by Roman Melamed, Catherine A. St. Hill, Bjorn I. Engstrom, David M. Tierney, Claire S. Smith, Vincent K. Agboto, Brynn E. Weise, Peter M. Eckman and Nedaa Skeik in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
Supplemental Material, Supplemental_Appendix_1_Treatment_Algorithm - Effects of a Consensus-Based Pulmonary Embolism Treatment Algorithm and Response Team on Treatment Modality Choices, Outcomes, and Complications
Supplemental Material, Supplemental_Appendix_1_Treatment_Algorithm for Effects of a Consensus-Based Pulmonary Embolism Treatment Algorithm and Response Team on Treatment Modality Choices, Outcomes, and Complications by Roman Melamed, Catherine A. St. Hill, Bjorn I. Engstrom, David M. Tierney, Claire S. Smith, Vincent K. Agboto, Brynn E. Weise, Peter M. Eckman and Nedaa Skeik in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
Supplemental Material, Supplemental_Appendix_3 - Effects of a Consensus-Based Pulmonary Embolism Treatment Algorithm and Response Team on Treatment Modality Choices, Outcomes, and Complications
Supplemental Material, Supplemental_Appendix_3 for Effects of a Consensus-Based Pulmonary Embolism Treatment Algorithm and Response Team on Treatment Modality Choices, Outcomes, and Complications by Roman Melamed, Catherine A. St. Hill, Bjorn I. Engstrom, David M. Tierney, Claire S. Smith, Vincent K. Agboto, Brynn E. Weise, Peter M. Eckman and Nedaa Skeik in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Note
All authors had access to the data and a role in writing this manuscript. We confirm that this work has not been previously published nor is it being considered for publication elsewhere.
Acknowledgments
The authors thank Dr Susan Olet, Department of Care Delivery Research, Allina Health, for the statistical review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Bjorn I. Engstrom, MD, has been a paid speaker for Penumbra Inc, Alameda, California, USA.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
