Abstract
Intracranial hemorrhage due to vitamin K deficiency is a serious disease that can lead to morbidity, mortality, and mental retardation. Our goal in this study is to determine the frequency of VKORC1-1639 G>A polymorphism in patients who have undergone intracranial hemorrhage due to vitamin K deficiency bleeding (VKDB). To study VKORC1-1639 G>A polymorphism, blood was drawn from patients (n = 51, age 8:0 ± 6:5 years) followed at the Pediatric Neurology and Hematology section, Faculty of Medicine, Erciyes University, between 1990 and 2016, diagnosed with VKDB as idiopathic or from patients diagnosed with intracranial hemorrhage due to secondary vitamin K deficiency and also from volunteers (n = 51, age 11 ± 4.5 years). Intensive care and nutrition needs of patients and the laboratory radiological imaging results and treatments that were applied were analyzed through scanning the files of the patients and information received from families. Through detailed physical examination, patients with neurologic sequelae and ongoing epilepsy were determined. The results were compared to clinical and laboratory results with control group. Eight (15.7%) of the patients were normal, 29 (56.9%) heterozygous carrier, and 14 (27.5%) homozygous mutants. In the control group, 19 (37.3%) were normal, 19 (37.3%) heterozygous carriers, and 13 (25.5%) homozygous mutants. The VKOR1-1639>A (SNP:rs9923231) mutant positivity (homozygous plus heterozygous mutant) was significantly higher in the patient group when compared to controls. There were no significant differences between patient and control groups in terms of the prognosis.
Introduction
Vitamin K deficiency bleeding (VKDB) is one of the most common hemostatic disorders at neonatal period and leading preventable cause of mental retardation, epilepsy, and cerebral palsy. 1,2 There are 3 types of VKDB. Early-onset VKDB is seen on the first day of life and associated with maternal use of drugs that disrupt vitamin K metabolism, while classical type results from insufficient vitamin K production that occurs between days 2 and 7 of life and has poor prognosis. Late-onset VKDB occurs due to insufficient vitamin K intake between 2 and 8 weeks of age. 3 The late-onset VKDB in newborns is characterized by intracranial bleeding that may occur between week 1 and month 6. 4 Vitamin K is a cofactor for gamma-glutamyl carboxylase (GGCX) that is involved in formation of tertiary structure and posttranslational modification of coagulation factors and some proteins. The reduced form of vitamin K (hydroquinone) is converted to vitamin K epoxide reductase (VKOR) and vitamin K epoxide (VKE), while glutamate (glu-) residues are converted to γ-carboxyglutamate in carboxylation process during hepatic synthesis of factors II, VII, IX, X and proteins S, C, and Z. To maintain this cycle, hydroquinone should be regenerated from VKE, which is controlled by an enzyme called vitamin K 2, 3 epoxide reductase complex (VKORC).
Bleeding predisposition develops due to inability to activate coagulation factors in case of vitamin K deficiency or VKOR polymorphism. γ-GK gene encoding VKOR was isolated on chromosome 16 in 2014. 5,6 Currently, it is known that single-nucleotide polymorphism 1639G>A at promoter region of VKOR gene causes up to 50% decrease in VKOR enzyme. 7 This is implied in the variation in doses of warfarin, a vitamin K antagonist. 8 Carboxylation is essential for normal activity of coagulation system. γ-carboxylation occurs in endoplasmic reticulum, and it is thought that carboxylation is catalyzed by GGCX. The carboxylated proteins are transferred to Golgi apparatus where they are secreted.
Vitamin K is the major cofactor for GGCX that adds carbon dioxide during conversion of glu-residues to gla-residues. 9,10 The GGCX enzyme was first isolated in 1991. The VCORK was first described in 1976; however, it was fully characterized in 2004 11 Gamma-glutamyl carboxylase and VKORC are involved in posttranslational modifications of proteins. In our study, it was aimed to investigate the molecular etiology in patients with late-onset VKDB who were followed in Pediatric Neurology and Pediatric Hematology departments of Erciyes University Medicine School and to identify VKOR1-1639G>A polymorphism encoding VKORC that is thought to cause VKDB in Turkish population.
Patients and Methods
All the participating children and parents were informed about the study, and informed consent was received. The study was approved by the Ethics Committee of Erciyes University Medical Faculty with no: TTU2016-6579 and was also supported by the Erciyes University Research Project Management Unit under the project designated as TTU2016-6579.
This study included 51 patients diagnosed with VKDB at Pediatric Neurology and Pediatric Hematology departments of Erciyes University Medicine School between 1990 and 2016 and 51 volunteers with normal platelet count. Patients with congenital malformation, chromosome anomaly, cholestatic liver disease, congenital valve defects, prenatal diagnosis, and periventricular leukomalacia were excluded. Neurological sequels and coagulation disorders were taken into account while selecting control group. The data regarding birth parameters such as clinical age, gender, form of feeding, and treatment were tabulated by reviewing medical charts. Psychomotor and neurological assessments were performed. In both patient and control groups, blood samples (4 mL) were drawn into EDTA tubes in order to study VKOR1-1639G>A polymorphism and stored at −80°C until DNA isolation. The DNA isolation, polymerase chain reaction (PCR) preparation, and restriction fragment length polymorphism (RFLP) procedures were performed at Genome Unite of Genome and Stem Cell Center of Erciyes University.
DNA Isolation
The EZ-10 Spin Column Genomic genomic DNA Minipreps isolation kits (Biobasic Inc, Markham, Ontario, Canada) were used to extract genomic DNA from blood samples. Overall, blood samples (0.4-5 mL) were drawn from 102 participants. For DNA extraction, 500 µL of complete blood sample was placed in the centrifugation tubes (2 mL) with addition of 800 µL tris borate phosphate buffer, which was then vortexed and kept for 1 minute at room temperature (The mixture should be bright red and red blood cells should be resolved). The mixture was centrifuged at 4000 rpm for 3 minutes, and the supernatant was removed. If the supernatant and blood pellet was still red, the second step and subsequent steps were repeated. Then, 500 µL Tris-borate magnesium buffer was added to tube, which was vortexed briefly. Again, 3 µL proteinase K was added to the tube, which was kept at 55°C for 30 minutes for incubation. If there was any unsolved material, the tube was centrifuged at 5000 rpm for 2 minutes. The supernatant was transferred to another tube (2 mL), and 260 µL absolute ethanol was added, which was then transferred to column and centrifuged at 10 000 rpm for 2 minutes. After adding 500 µL wash solution, the column was recentrifuged at 10 000 rpm for 1 minute. The collection tube was poured, and 500 µL wash solution was added for further centrifugation at 10 000 rpm for 1 minute. Then, to remove residual wash solution, tube was recentrifuged at 10 000 rpm for 1 minute. Spin was continued until purification was achieved. The column (1.5 mL) was placed to an Eppendorf tube, and 50 µL elution buffer was applied to center of column. After incubation at 37°C for 2 minutes, the tube was centrifuged at 10 000 rpm for 1 minute to extract DNA. The purified DNA was stored at −20°C until PCR analysis.
Polymerase Chain Reaction Preparation
The isolated DNA was used for PCR analysis. In each PCR tube, 22 µL of the mixture was placed and 3 µL DNA was added to this mixture, resulting in 25 µL of final volume. The samples were placed in the PCR device. Samples obtained from PCR device were run at 2% agarose gel at 90 V for 2 hours. Then, the PCR bands were imaged using Molecular Imager Gel Doc XR System device (Bio-Rad, Hercules, California). The PCR products with 290 base pair (bp) were assigned for PCR RFLP protocol. The PCR products were stored at 4°C until RFLP analysis.
Restriction Fragment Length Polymorphism
HpaII restriction enzyme (New England Biolabs, Beverly, Massachusetts) was used for VKORC1-1639G>A polymorphism. RFLP was performed as follows. The PCR product (15 µL) was delivered to tubes according to patient number and then other substrates (10× Buffer Tango [33 mmol/L Tris-acetate, pH 7.9, 10 mmol/L magnesium acetate, 66 mmol/L potassium acetate, 0.1 mg/mL BSA], Hpa II restriction enzyme and water) were added at appropriate volumes. The mixture was incubated overnight at 37°C. The incubated samples were run on 3% agarose gel at 85 V for 2 hours. The PCR products and restriction enzymes were assessed by Molecular Imager Gel Doc XR System device. The products with 168 bp and 122 bp were considered as homozygous normal, while those with 290 to 168 bp and 122 bp were considered as heterozygous normal. The PCR products with 290 bp were considered as homozygous mutant.
Statistical Analysis
All statistical analyses were performed by using Statistical Package for Social Sciences (SPSS) for Windows version 11.0. In our study, we only compared categorical variables, so normality of the variables was not tested. In comparing categorical variables, the Pearson χ2 test was used. P value <.05 was considered statistically significant. Odds ratios are calculated with 95% confidence intervals.
Results
By reviewing history, data were tabulated for 51 patients (range: 1 month to 27 years) who were diagnosed with intracranial bleeding secondary to VKDB at neonatal period and 51 healthy volunteers (range: 1 month to 20 years). Median age was 8 years (1-27) in the patient group whereas 11 years (1-18) in the control group. Based on mutational analysis, normal GG was considered to have no mutation, while heterozygous GA and homozygous AA mutants were considered to have a mutation. There were 22 (43.1%) girls and 29 (56.9%) boys in the patient group, whereas 28 (54.9%) girls and 23 (45.1%) boys in the control group.
When genetic mutations were assessed, it was found that 8 (15.7%) patients had no mutation, while 29 (56.9%) patients were heterozygous carrier and 14 (27.5%) patients were homozygous mutant in the patient group. It was found that 19 (37.3%) patients were normal, while 19 (37.3%) patients were heterozygous carrier and 13 (25.5%) patients were homozygous mutant in the control group (Table 1). These findings indicate significant differences between patient and control groups. The VKOR1-1639>A mutant positivity (homozygous plus heterozygous mutant) was significantly higher in the patient group compared to controls (P = .037). There was significant difference between patient and control groups regarding no mutation and heterozygous mutation (P < .05) but not homozygous mutation (P < .05).
Results Mutational Analysis in Patient and Control Groups.a
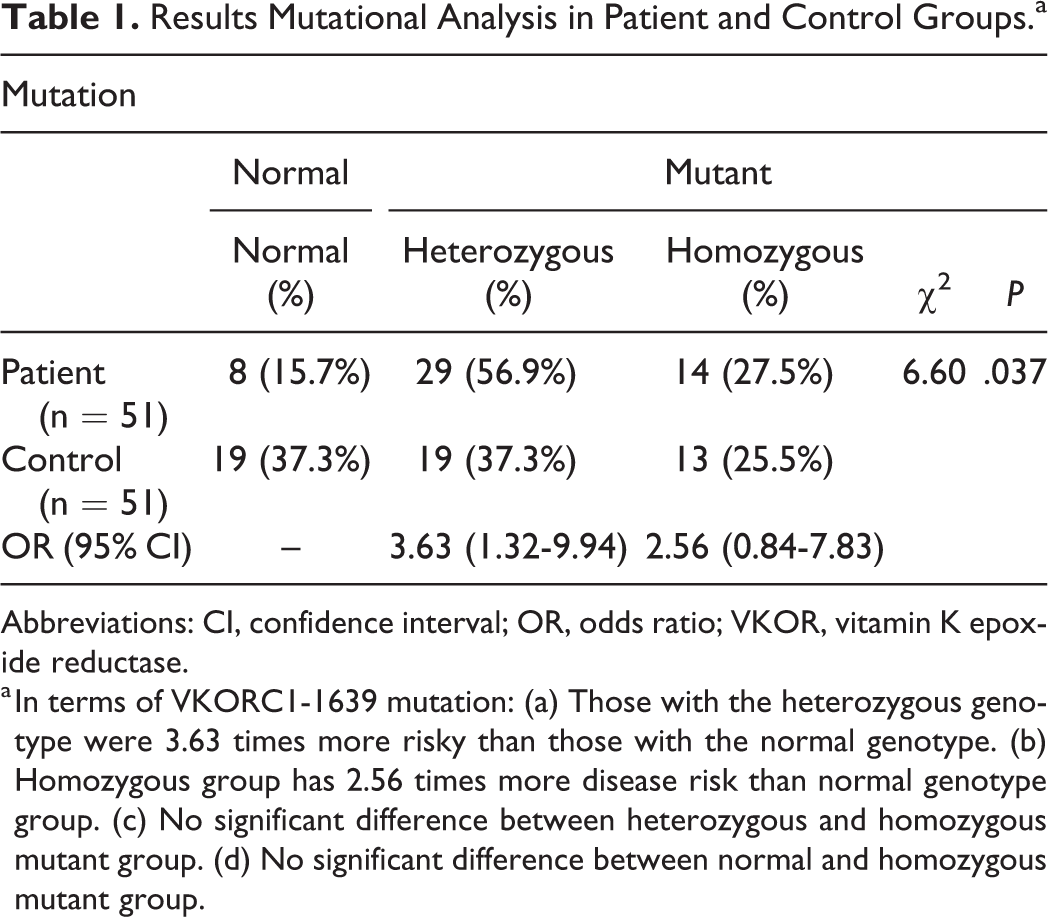
Abbreviations: CI, confidence interval; OR, odds ratio; VKOR, vitamin K epoxide reductase.
a In terms of VKORC1-1639 mutation: (a) Those with the heterozygous genotype were 3.63 times more risky than those with the normal genotype. (b) Homozygous group has 2.56 times more disease risk than normal genotype group. (c) No significant difference between heterozygous and homozygous mutant group. (d) No significant difference between normal and homozygous mutant group.
In our study, there was neurological sequel in 35 (68.6%) patients, whereas no neurological sequel in 16 (31.4%) patients. There was need for intensive care unit (ICU) care in 37 (72.5%) patients, while 14 (27.5%) patients did not require ICU care. Subsequent epilepsy was developed in 33 (64.7%) patients in the patient group. It was found out that 1 patient died due to convulsion after enrollment ito the study. The need for ICU care and epilepsy indicated that the disease has poor prognosis.
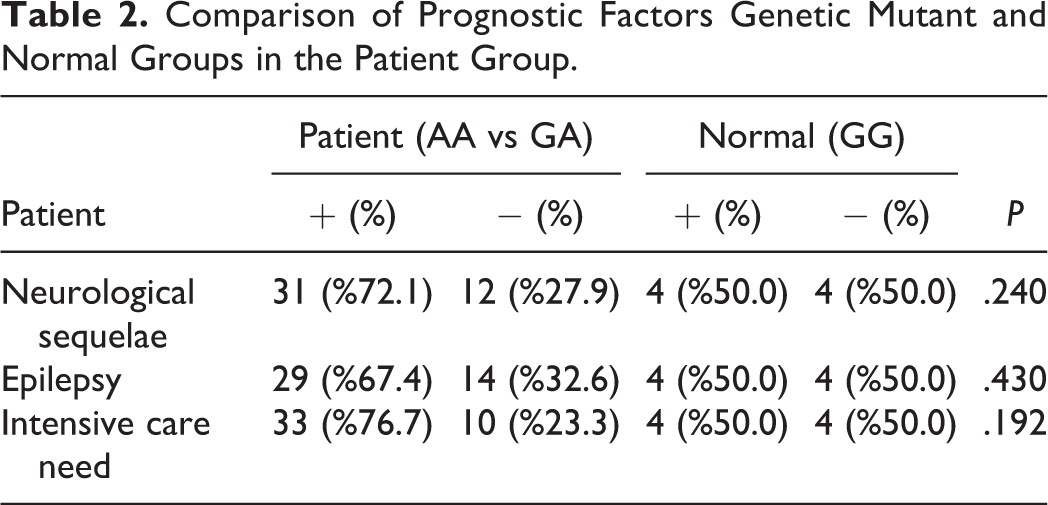
When effects of polymorphism on prognosis were assessed, no significant difference was detected between mutant group (AA and GA) and homozygous normal group (GG) regarding need for ICU need, neurological sequel, and epilepsy (Table 2).
Comparison of Prognostic Factors Genetic Mutant and Normal Groups in the Patient Group.
The results of imaging studies were available only for 29 patients. On imaging studies, there was intraventricular bleeding in 2 (6%), subdural hemorrhage in 6 (20%), subarachnoid hemorrhage in 3 (10.3%), and intraparenchymal hemorrhage in 12 (41.3%) patients.
Time of presentation, presenting complaint, platelet count and PT, and aPTT values were extracted from medical records. We failed to access imaging studies in all patients, since hospital database was replaced with a newer version. Time at first presentation to hospital was available in 25 patients. It was found that 13 patients presented to hospital at 15 to 30 days of life, while remaining 12 patients presented beyond first month of life. The patients were screened for presence of underlying etiology. No known reason was found in these patients. Presenting complaint was irritability and difficulty in feeding in 10, convulsion in 9, fontanel bulging in 2, fadedness in 2, and jaundice in 1 patient.
Discussion
There was significant difference in VKOR1-1639G>A polymorphism between patient and control groups. VKOR1-1639G>A mutant positivity was higher in the patient group when compared to controls. The rates of neurological sequels, epilepsy, and need for ICU care at neonatal period were higher than anticipated. No significant difference was found in the prognosis between mutant (heterozygous plus homozygous mutants) and normal group (no polymorphism).
Vitamin K deficiency is a major cause of morbidity and mortality at neonatal period. 12 Vitamin K deficiency bleeding is bleeding that occurs due to insufficiency of vitamin K-dependent coagulation factors and resolves by vitamin K replacement. In vitamin K-dependent coagulation factor deficiencies, onset and variation in findings and symptoms are associated with factor levels. Clinical presentation tends to progress with increased bleeding predisposition in vitamin K deficiencies. The addition of acquired reasons causing vitamin K deficiency such as drug use (antibiotics, anticonvulsants, etc), liver failure, or malabsorption may lead more severe and eventful course of hemorrhage than anticipated. In 2014 in a Turkish study by Unal et al, 1 it was suggested that reported VKDB rates didn’t reflect true incidence, resulting underestimation of VKDB incidence. It was reported that the incidence in Turkey was rather high, and lack of vitamin K prophylaxis in home-based deliveries or decreased activity of prophylactic vitamin K vials used in some hospitals were potential causes. 1 Thus, more comprehensive studies are needed in our country. In Turkey, vitamin K (1 mg, intramuscular [IM]) is routinely administered at time of delivery. In recent years, it is hospital-based delivery; thus, vitamin K prophylaxis has been increased as a result of increased cesarean deliveries. Solely breast-feeding, chronic diarrhea, or long-term antibiotic use predisposes infants to VKDB. Unal et al 1 published that the reporting system of the vitamin K prophylaxis is not solid; and prophylactic vitamin K injections were prepared in syringes, and we speculate that these previously prepared syringes may be a possible reason of decreased coverage of VK prophylaxis in Turkey.Therefore, we have reported that Turkish health-care providers, pediatricians, and the National Ministry of Health should be aware for the importance of prophylactic measures of VK, especially for infants born at home in rural areas. American Pediatrics Academy recommends IM injection of a single dose of vitamin K (0.5 to 1.0 mg) in all newborns. In this study, we retrospectively analyzed results of neuroimaging studies, blood tests, and PT and aPTT test performed at neonatal period. In VKDB, it is important to demonstrate prolonged PT and its normalization following vitamin K injection in confirming diagnosis. 13 Site of bleeding is one of the most important factors influencing on prognosis in VKDB. In newborns, bleeding due to vitamin K deficiency occurs in several sites; however, intracranial hemorrhages are those causing highest mortality. In previous studies, subdural hemorrhage was reported most frequently on CT scans. In 2012, Ozdemir et al 2 reported that subdural hemorrhage (28%) was most common intracranial bleeding followed by intracranial hemorrhage (23%), subarachnoid hemorrhage (14%), and intraventricular hemorrhage (8%). It was reported that fontanel bulging (70%), irritability (50%), convulsion (49%), bleeding and ecchymosis (47%), difficulty in feeding, vomiting, and feeding intolerance (46), diarrhea (34%), jaundice (11%), and fadedness (9%) were presented as complaints. 2 In our study, rates of subdural hemorrhage, intracranial hemorrhage, subarachnoid hemorrhage, and intraventricular hemorrhage were 20%, 41.3%, 10.3%, and 6%, respectively. The presenting complaints included irritability and difficulty in feeding (41.6%), convulsion (37.5%), fontanel bulging (8.3%), fadedness (8.3%), and jaundice (4.1%) in our study.
In recent years, after discovery of VKOR1-1639G>A involved in VKE gene system, several studies have been conducted about this gene. Most of these studies focused on investigation of anticoagulant activity in human. In the study by Nowak-Gottle et al, 14 it was shown that VKORC polymorphism accounts for dose variation in vitamin K antagonist anticoagulants. In the study by Schreiner et al, 15 it was shown that infants with VKORC 1639A allele are at high risk of intraventricular hemorrhage. In a rat study by Spohn et al, 16 VKORC homozygous rats which were healthy at birth died mainly due to intracerebral hemorrhage 2 to 20 days after birth. In our study, VKOR/1639G>A polymorphism was found to be significantly higher in the patient group than controls, and there was significant relationship between VKOR1-1639G>A and VKDB.
Although VKDB is a sporadic phenomenon in developed countries, it has considerable morbidity and mortality. It is more frequently seen in Turkey when compared to developed countries, and some studies were conducted to establish its mortality. In 2008, Cekinmez et al 17 reported that there were neurological findings in 21% to 73% of patients with mortality rate of 19% to 57%. In the literature, in a study by McNinch et al, 18 it was reported that 2 of 10 patients without vitamin K prophylaxis who received solely breast-feeding died in United Kingdom in 1991. When compared to the 1639 G-A distribution in the normal Turkish population. In our control group, we found the VKORC genotype as 19 (37.3%) for GG, 19 (37.3%) for GA, and 13 (25.5%) for AA alleles. Silan et al 19 found that the genotype of VKORC was detected as follows: 64 (21.9%) for GG, 220 (75.4%) for GA, and 8 (2.7%) for AA alleles 55% in the study conducted in 2012.
The implementation of routine prophylaxis program and lack of new cases due to paucity of delivery at home in Turkey are major causes for failure to include preplanned study population of 75 patients. Thus, majority of patients were old cases in this study. Some patients couldn’t be included due to replacement of hospital database and incomplete data gathered from parents. We think that limited number of patients precluded showing poorer prognosis in VKOCRC allele carriers than those without VKORC allele.
In conclusion, it has proven that VKDB is a cause of significant morbidity and mortality in previous studies. It was reported that survivors of intracranial bleeding secondary to VKBD displayed severe psychomotor retardation, cerebral palsy, hydrocephaly, microcephaly, and convulsions. Given the burden of VKDB which is a preventable disease with poor prognosis, it is apparent that etiology, prognosis, and outcomes should have to be established in our country. In this study, we attempted to demonstrate VKDB and its prognosis in our country. We think that an additional dose of vitamin K in first months of life will considerably decrease intracranial bleeding secondary to VKDB in infants receiving breast-feeding solely. 20 We think that the finding of higher rate of VKOR1-1639G>A polymorphism in the patient group will be guiding for future research. Higher incidence of VKOR1-1639G>A polymorphism in our society may imply additional dose of vitamin K.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
